Abstract
In the past decade, NOD.Cg-Prkdcscid Il2rgtm1Wjl/SzJ (NSG, NOD scid gamma) mice have become a model of choice in several areas of biomedical research; however, comprehensive data on their spontaneous age-related pathology are not currently available in the literature. The prevalence of spontaneous morbidity affecting aged NSG female breeders enrolled in a parasitology study was documented with classification of neoplastic and non-neoplastic (inflammatory, metabolic, degenerative) lesions. Malignant mammary neoplasms were most commonly diagnosed, often accompanied by pulmonary metastases, while a low frequency of lymphoma and histiocytic sarcoma was documented. The major inflammatory conditions were suppurative pleuropneumonia and bronchopneumonia with abscess formation, from which Pasteurella pneumotropica was commonly isolated, followed by otitis media. Both inflammatory and degenerative lesions of the genital tract were identified, along with neoplasms such as endometrial yolk sac carcinomas and granulosa cell tumors. Novel conditions identified included renal tubular degeneration and necrosis associated with 2 concurrent types of intranuclear inclusions, focal or multifocal hyperostosis of the skull, and neuroendocrine tumors of the mesometrium. The majority of degenerative lesions that affected the genital tract, endocrine, and skeletal systems did not represent the actual underlying cause of death but rather were considered incidental findings. This study indicates that both inflammatory and neoplastic conditions contribute to morbidity and mortality in experimentally manipulated aged female NSG mice.
Keywords
NOD.Cg-Prkdcscid Il2rgtm1Wjl/SzJ (NSG, NOD scid gamma) mice carry several genetic defects primarily affecting the immune system and leading to a severe impairment of both the innate and adaptive immune responses. NSG mice are congenic on NOD/ShiLtJ (NOD, non-obese diabetic) background, whereas the contributions from other strains are minimal. The NOD background is characterized by a loss of function mutation in the C5 hemolytic complement gene, decreased (but not completely absent) natural killer (NK) cell activity, defects in antigen-presenting activity, and impaired macrophage function. 60 A defective somatic recombination at the level of T cell receptor and immunoglobulin chains loci derives from homozygosity for the Prkdcscid mutation, with consequent defective development and maturation of T and B cell clones, leading to an absence of functional lymphocytes and hypogammaglobulinemia. Furthermore, deficiencies in cytokine signaling and absence of NK cell development are the results of a targeted null mutation of the Il2rg gene coding for the gamma chain of the interleukin-2 receptor. 60
Because of its ability to support the engraftments of a wide range of human cell types, the NSG strain has become a model of choice in biomedical research in the past decade. 58 Furthermore, there is increased evidence that the NOD Sirpa allele confers support for the engraftment of human hematopoietic stem cells. 64 Common uses of this strain include the xenotransplantation of humanneoplastic cell lines and primary tumor samples (patient-derived xenografts) that are used as cancer models as well as the creation of humanized mice by implantation of hematopoietic stem cells (HSCs) and peripheral-blood mononuclear cells (PBMCs). 58,59 This has allowed the creation of new models in the fields of cancer, infectious disease, transplantation, and immunologic research. Despite its growth as one of the most commonly used strains in biomedical research, limited data have been published on the naturally occurring background pathology of NSG mice.
A low predisposition for the development of spontaneous tumors has been described. 60 While NOD.CB17-Pkrdcscid/J (NOD scid) mice have a high incidence of spontaneous thymic lymphoma limiting their life span (median survival time: 37 weeks), 61 thymic lymphoma has not been reported in experimentally naïve or irradiated NSG mice, and a median survival time of >89 weeks has been described for experimentally naïve NSG mice. 60 A low incidence of splenic lymphoma, mammary adenocarcinoma, and osteosarcoma has been reported. 28,60
Due to their high level of immunodeficiency, morbidity and mortality caused by opportunistic infections have been reported in NSG mice, even in breeding colonies in maximum level barrier facility by a commercial vendor. 12 In that study, naturally occurring lesions contributing to morbidity in male and female breeding NSG mice were described. The mice examined ranged from 17 to 42 weeks of age, and the cause of morbidity was predominantly opportunistic renal or multisystemic bacterial infections. Neoplasms including mammary adenocarcinomas, osteosarcomas, and lymphomas were mentioned, and the frequency and age of onset of these conditions were compatible with those in other NOD-derived strains (unpublished data). 12 Despite their reported life span of >89 weeks and increasing use in cancer and immune research, no comprehensive description of naturally occurring aged-related lesions of NSG mice has been published. Given the prominent use of this strain in biomedical research, a better understanding of naturally occurring pathology, including spontaneous age-related lesions and natural causes of morbidity and mortality, is required to ensure correct interpretation of results in experiments in which this strain is used.
Therefore, the aim of this study was to determine the major clinical manifestations, life span, spectrum of spontaneous neoplastic and non-neoplastic age-related lesions, and causes of death and contributory causes of death in this strain. To achieve this goal, a longitudinal survival study was performed on retired female NSG breeders, which were used for a parasitology study, during which the mice were allowed to survive their natural life span.
Materials and Methods
Female NSG mice were originally purchased from the Jackson Laboratory (Bar Harbor, ME) and housed at facility A at Memorial Sloan Kettering Cancer Center to establish a breeding colony. Breeders were composed of mice born within the aforementioned facility as well as mice received from the same vendor and introduced in the colony on a regular basis. Mice were experimentally naïve and kept in the aforementioned facility for breeding purposes either until 6 months of age or until decreased reproductive activity occurred (whichever occurred first), at which time they were retired from the breeding colony and transferred to facility B for the purpose of a parasitology study. For the purposes of the parasitology study, mice were transferred from facility A to facility B from April 2014 to April 2016. Mouse care and all procedures outlined in the parasitology study were approved by the Memorial Sloan Kettering Cancer Center’s Institutional Animal Care and Use Committee and maintained in accordance to the National Academy of Sciences’ Guide for the Care and Use of Laboratory Animals 44 in an AAALAC International-accredited facility.
Facility A was a barrier facility in which a rodent health monitoring program showed absence of the major murine viral and bacterial agents in surveillance sentinel mice throughout the study period (see Supplementary File 1 for additional information). Research facility B was a nonbarrier facility used for quarantine purposes and the parasitology studies involving ectoparasites. Although husbandry practices were similar to facility A, no routine microbial surveillance programs were performed. Also, other animals housed in facility B were of unknown health status. After being transferred, retired NSG breeders were assigned to one of the following experimental groups and were infected with parasites: G1 = Myocoptes musculinus and Demodex spp.; G2 = Myobia musculi and Radfordia affinis; G3 = Myocoptes musculinus and Myobia musculi; G4 = Aspiculuris tetraptera; G5 = Demodex spp.; G6 = Syphacia obvelata; G7 = Aspiculuris tetraptera and Syphacia obvelata. Mice belonging to these groups were infected by 1 of 3 methods: direct contact with infected animals, either NSG mice, Tac: SW mice (Swiss Webster), or mice belonging to the B6.Cg-Rag1tm1MomTyrp1B-wTg (Tcra, Tcrb)9Rest/M strain (TRP-1 TCR); contact with contaminated bedding; or through gavage of embryonated parasitic eggs (only for groups G4 and G6).
Female mice were group housed with 3 to 5 mice per cage and identified based on cage number and ear punches. With the purpose of identifying major behavioral and/or clinical abnormalities, all cages were observed daily by an animal caretaker, and each animal was individually checked on a weekly basis by a laboratory animal veterinarian.
All mice were sampled at the beginning of the experiment, monthly during the experiment, and at the end of the experiment (or at the time mice reached their humane endpoint and euthanasia was elected). Sampling was performed on live mice and included fecal pellet collection only (groups G4, G6, and G7), and fur pluck, deep skin scrape, PCR swabs, and fecal pellet collection (groups G1, G2, G3, and G5). No mice were removed at predefined time points. Complete pathologic examination and inclusion in the pathology study occurred when mice were submitted for euthanasia and/or necropsy in the event they became spontaneously ill, were in poor clinical condition, or were found dead. Humane endpoints included any of the following signs of advanced disease: diminished body condition score (average body condition score of 2/5, with evidence of minimal subcutaneous and visceral adipose tissues), 8 severely hunched posture, dehydration, ruffled hair coat, lethargy, lateral recumbency, head tilt, ocular or peri-ocular lesions, tumors greater than 1 cm3, and rectal or uterine prolapse. Mice that appeared healthy throughout the duration of the experiments were used for the purposes of the parasitology experiments.
From 140 mice submitted for necropsy, 15 animals were noted grossly to be decomposed and were not evaluated further; therefore, a total of 125 animals were included in the present study. Animals belonged to the parasitology groups as follow: G1 (n = 20), G2 (n = 21), G3 (n = 17), G4 (n = 29), G5 (n = 23), G6 (n = 12), G7 (n = 3). Mice from G4, G6, and G7 groups entirely represented the colonies kept as infected source for the mice included in the parasitology study; mice belonging to the groups G1, G2, G3, and G5 either represented the infected source or the animals effectively enrolled in the parasitology study. No mice were removed at predefined time points for other purposes. Additionally, no experimentally naïve animals were available as the parasitology study did not require the inclusion of such animals.
Following euthanasia by carbon dioxide asphyxiation, 33 complete postmortem examination, including macroscopic organ inspection and dissection, was performed.
Microbiologic cultures were performed on selected tissues presenting gross lesions, and isolates were identified by a colorimetric biochemical testing (API Coryne, bioMerieux, Marcy l’Etoile, France); confirmation with MALDI-TOF mass spectrometry was achieved in selected cases (IDEXX BioResearch, Columbia, MO). In 2 selected cases, a PCR evaluation (IDEXX BioResearch, Columbia, MO) was performed on frozen renal samples for detection the following agents: mouse adenovirus 1, mouse adenovirus 2, murine cytomegalovirus, murine polyomavirus. Histologic examination of the following tissues was performed: heart, lung, lymph nodes (mediastinal, submandibular, mesenteric), thymus, kidney, liver, gallbladder, spleen, pancreas, salivary glands, stomach, duodenum, jejunum, ileum, cecum, colon, ovaries, oviducts, uterus, cervix, vagina, adrenal glands, trachea, esophagus, thyroid glands, bone and bone marrow (sternum, femur, tibia, vertebral column, maxillary and mandibular bones), stifle joint, skeletal muscles, skin, mammary tissue, and 4 cross-sections of the head including eyes, harderian gland, nasal cavity, brain, pituitary, ears, teeth, and tongue. All samples were fixed in 10% neutral buffered formalin (NBF), routinely processed in alcohol and xylene, embedded in paraffin, sectioned at 5-µm thickness, and stained with hematoxylin and eosin (HE). After fixation, bones were decalcified in a formic acid solution (Surgipath Decalcifier I, Leica Biosystems) for 48 hours. Routine spinal cord evaluation includes the cervical and thoracic regions (from C2 to T13) using longitudinal sections representing parasagittal plane taken lateral to the midline. Following our internal protocol, a total of 9 slides were produced and examined. In addition to the examination of these standard tissues, samples harvested at the time of necropsy from tumors or other lesions were collected, processed, and examined in an additional slide. Special stains, including Gram, periodic acid-Schiff reaction (PAS), Grocott’s methenamine silver (GMS), Masson’s trichrome, acid fast, and Congo red stains were performed on selected cases. Selected tissues were stained by immunohistochemistry for CD3 (primary antibody Vector Laboratories VP-RM01 applied at concentration of 1:100 following heat-induced epitope retrieval [HIER] in a pH 9.0 buffer), B220 (BD Pharmigen 550286, 1:200, HIER pH 6.0), MAC2 (Cedarlane CL8942B, 1:100, HIER pH 6.0), laminin (Abcam ab11575, 1:2,500, proteinase K epitope retrieval), chromogranin A (Epitomics 1782 -1, 1:500, HIER pH6.0), synaptophysin (BD Pharmingen 611880, 1:500, HIER pH6.0), myeloperoxidase (Dako A0398, 1:1000, HIER pH6.0), and CD45 (BD Pharmigen 550539, 1:250, HIER pH6.0). MAC2 and synaptophysin staining was performed manually, using a secondary antibody (for synaptophysin only, Vector Laboratories BA-2000) and an avidin-biotin detection system (Vectastain ABC Elite Kit, Vector Laboratories, PK-6100). All other stains were performed on a Leica Bond RX automated stainer using the Bond Polymer Refine detection system (Leica Biosystem DS9800). For both manual and automated methods, the chromogen was 3,3 diaminobenzidine tetrachloride (DAB), and sections were counterstained with hematoxylin. In each case, histologic slides were evaluated, and lesions were recorded by one of the multiple board-certified veterinary pathologists working at the institution.
For all animals, all lesions that were of significant severity, had likely contributed to the reported clinical signs, or were considered unusual were reviewed a second time by a single board certified pathologist (S.S.F.). Standardized diagnostic criteria and nomenclature from the International Harmonization of Nomenclature and Diagnostic Criteria 11,14,17,27,39,47,52,53,65 and the Mouse Models of Human Cancer Consortium 5,10,24,31,42,46 were used to diagnose and record lesions. The most likely causes of death (COD) and comorbidities were established for all animals according to recommended criteria. 62
Based on the histologic findings in the kidney of some mice, serum chemistry was subsequently performed on cagemates when they were submitted for necropsy. Blood was collected by terminal cardiocentesis and placed into 400 μL serum separator tubes. Serum concentrations of urea and creatinine were measured on a Beckman Coulter AU680 analyzer. For urea, results were compared to the internal reference ranges and the mean value available in the current literature for aged NSG mice. 30 For creatinine, results were compared to reference interval determined on this laboratory and instrument for experimentally naïve female BALB/c mice as no reference interval was available for the NSG strain.
For transmission electron microscopy (TEM), representative NBF-fixed tissue samples of the renal cortex collected from 2 animals with specific histopathologic changes were transferred to 2.5% glutaraldehyde (pH 7.2) for 3 hours and post-fixed in 1% OsO4 in 0.1 M cacodylate buffer for 60 minutes. The samples were subsequently dehydrated through an ethanol series, rinsed in propylene oxide, and embedded in epoxy resin. Sections of 0.5-μm thickness were placed on glass slides, stained with toluidine blue, and examined by light microscopy. Areas of interest in these sections were selected for ultrastructural examination, and 70-nm-thick sections were prepared from selected areas, cut, stained with lead citrate and uranyl acetate, and examined by TEM.
Results
Clinical Signs and Gross Abnormalities
For 68 mice, the median age at which they were submitted for necropsy was 52 weeks (range, 16–104 weeks). In the remaining animals (n = 57), the birth date was unavailable.
Based on the clinical history provided, poor body condition and/or decreased body condition score was reported in 43 of the 125 animals (34%). A hunched posture (43/125, 34%), lethargy (16/125, 13%), and distended abdomen (8/125, 6%) were also reported. Other clinical findings evidenced in this cohort included skin abnormalities such as chronic dermatitis with scaling and crusting (68/125, 54%), subcutaneous masses (45/125, 36%), cutaneous ulcers (18/125, 14%), and uterine prolapse (9/125, 7%). Given the nature of the experiment, skin abnormalities were most commonly detected in animals enrolled in groups G1, G2, G3, and G5; 1 G4 mouse had evidence of marked necrosuppurative dermatitis. Less common findings included ocular abnormalities such as exophthalmus (4/125, 3%), keratitis (2/125, 2%), and corneal ulcers (2/125, 2%). Ten mice (8%) were found dead in the cage.
At necropsy, gross examination confirmed the clinical findings; additionally, pulmonary abscesses (38/125, 30%) (Fig. 1) and mottling of the kidneys (16/125, 13%) were detected. For pulmonary lesions, a swab for microbiologic analysis was performed in 15 of 38 cases (39%). Tumors that were not evident clinically due to involvement of internal organs were observed grossly on necropsy after the animals were submitted for an alternate aforementioned clinical reason and are described in the following under histopathology results.
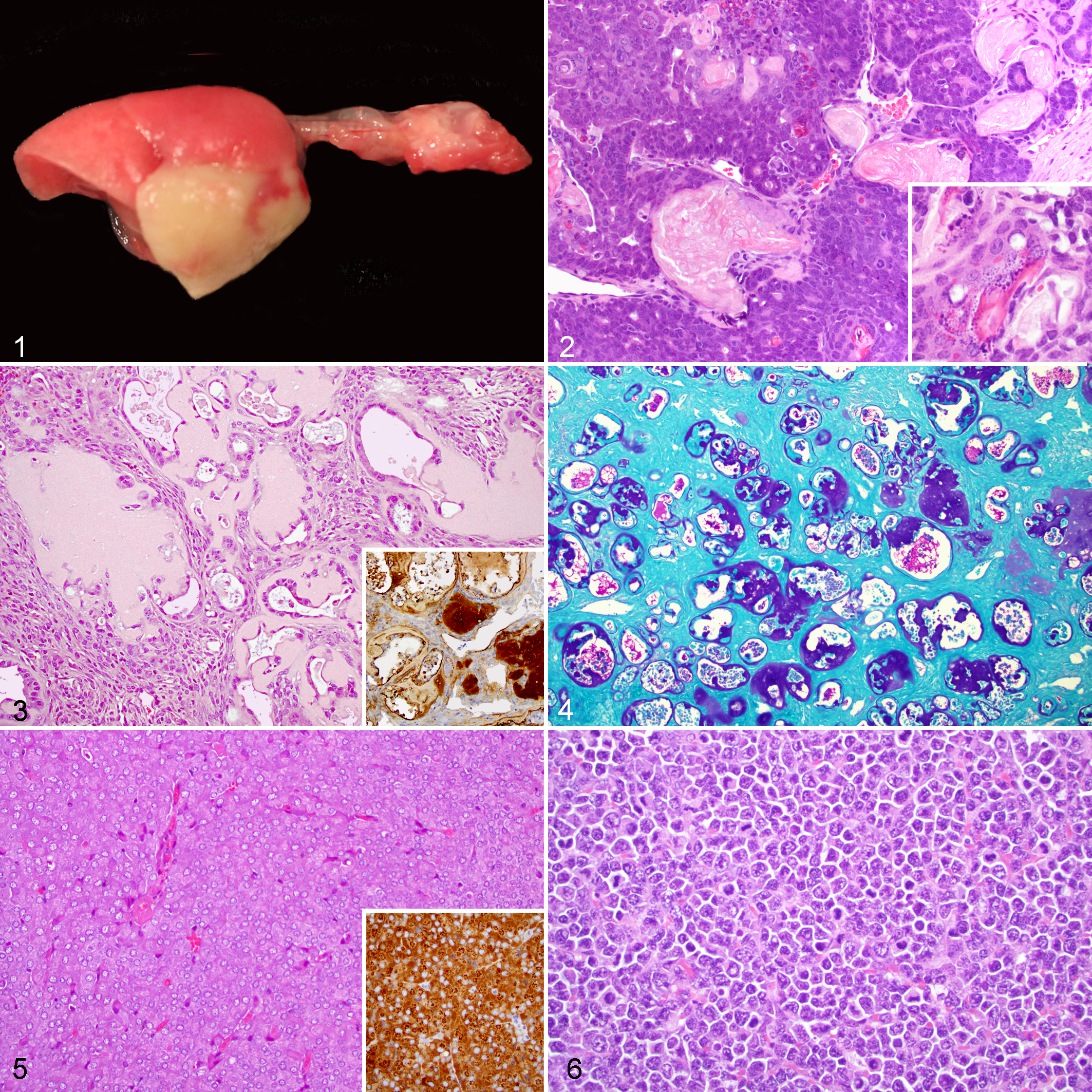
Histopathology of Neoplastic Lesions
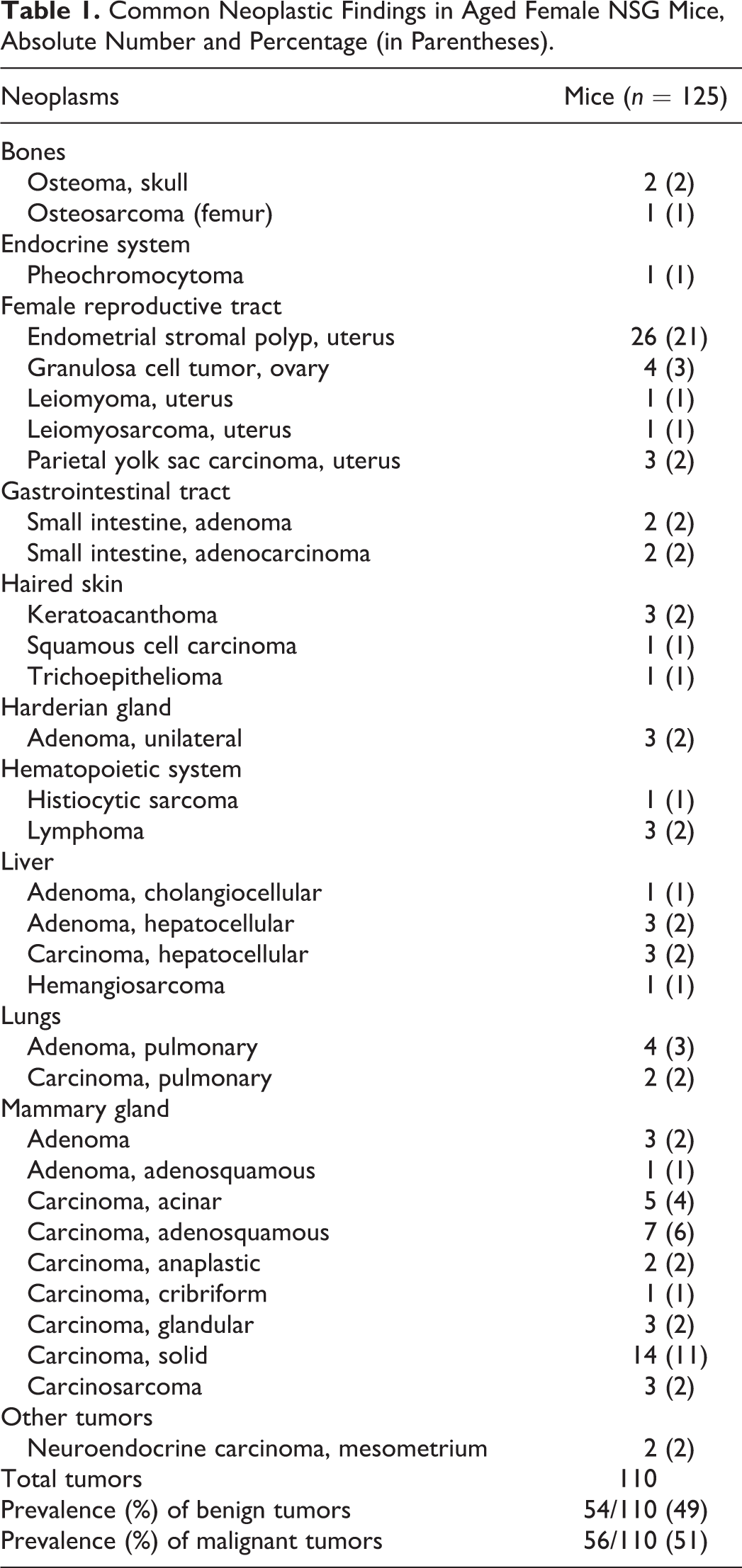
The major neoplasms are reported in Table 1.
Common Neoplastic Findings in Aged Female NSG Mice, Absolute Number and Percentage (in Parentheses).
The most common neoplastic lesion was mammary gland neoplasms (39/125, 31%), representing 37% of all tumors. According to the MMHCC and INHAND guidelines, 10,53 35 of 39 (90%) of mammary neoplasms were malignant and were classified either as solid, acinar, glandular, or cribriform carcinomas (25/39, 64%); adenosquamous carcinomas (7/39, 18%); or carcinosarcomas (3/39, 8%). Adenosquamous carcinomas were composed of epithelial cells arranged in well-differentiated microacinar structures intermingled with central cavities lined by well-differentiated stratified squamous epithelium producing keratin and were characterized by local invasion of the subcutaneous tissues and subjacent skeletal muscles. Squamous differentiation was also commonly characterized by the presence of trichohyalin granules and/or keratohyalin granules (Fig. 2).
In 4 cases, pulmonary metastases of primary mammary neoplasms were detected, most commonly of adenosquamous type (2/4, 50%), followed by 1 acinar carcinoma (25%) and 1 carcinosarcoma (25%).
In the female reproductive tract, neoplastic lesions were detected in 35 of 125 cases (28%). Specifically, primary ovarian neoplasms (4/35, 11%) were represented by 4 granulosa cell tumors, whereas primary uterine neoplasms (31/35, 89%) included single or multiple endometrial stromal polyps (26/35, 74%), 3 parietal yolk sac carcinomas (9%) (Fig. 3), 1 leiomyosarcoma, and 1 leiomyoma (3%, respectively). Parietal yolk sac carcinomas were composed of nests and cords of polygonal or cuboidal neoplastic cells embedded in an abundant eosinophilic, PAS-positive, trichrome-positive, and Congo red-negative hyaline extracellular matrix (Fig. 4). Intracytoplasmic PAS-positive granules were occasionally present within the neoplastic cells. The hyaline matrix was positive for laminin (Fig. 3, inset); additionally, the neoplastic cells exhibited strong cytoplasmic positivity for the same marker. Adjacent to the uterus within the mesometrium, 2 neuroendocrine carcinomas composed of packets of cells with granular basophilic cytoplasm were identified (Fig. 5). These neoplasms stained positively for synaptophysin and chromogranin A (Fig. 5, inset).
Liver tumors were identified in 8 of 125 mice (6%); of note, 3 mice had hepatocellular adenomas, and 3 had hepatocellular carcinomas. Pulmonary epithelial neoplasms were identified in 6 of 125 mice (5%). Four mice had pulmonary adenomas, whereas 2 mice had pulmonary carcinoma.
Primary cutaneous neoplasms were detected in 5 cases (4%), including keratoacanthoma (3/5, 60%) and single cases of trichoepithelioma and squamous cell carcinoma.
The prevalence of hematopoietic malignancies was also relatively low (4/125, 3%). Three cases (2%) morphologically consistent with lymphoma were diagnosed. Immunohistochemistry revealed that all lymphomas were positive for CD45 and negative for myeloperoxidase and MAC2. Furthermore, 2 cases were positive for B220 (B-cell origin), while 1 case was positive for CD3 (T-cell origin); these were further classified as small B-cell lymphoma, diffuse large B-cell lymphoma (DLBCL), and precursor T-cell lymphoblastic lymphoma, respectively, according to the MMHCC classification of lymphoid neoplasms in mice. 42 Lymphoid tumors exhibited variable degree of pleomorphism (Fig. 6) and high mitotic rate. Diffusely, dense sheets of round neoplastic lymphoid cells extensively infiltrated and partially effaced multiple organs, with disruption of tissue architecture in lungs, liver, kidneys, spleen, lymph nodes, thymus, gastrointestinal tract, bone marrow, ovaries, and brain. Given the advanced stage and concurrent lymphoid neoplastic infiltration in multiple organs, the site of lymphoid tumor origin was not established.
Only 1 multiorgan histiocytic sarcoma was diagnosed. Specifically, a focal large, oval, pale tan, and firm mass of 18 × 14 × 12 mm was detected at the level of the right thoracic region. Histologically, poorly demarcated sheets of polygonal cells with moderate amount of cytoplasm, round to oval nuclei, and moderate cellular and nuclear pleomorphism were infiltrating the dermis, subcutis and subjacent skeletal muscle, and elevating the overlying epidermis. Infiltration of visceral organs (kidneys, lungs, and mediastinal lymph node) was also noted. Immunohistochemistry revealed that the neoplastic cells were positive for MAC2. Given the remarkable size of the cutaneous tumor and the minor infiltration of the other organs (lung, kidney, mesenteric lymph node), the skin likely represented the primary site of origin.
Within the gastrointestinal tract and specifically at the level of duodenum or ileum, 4 tumors were diagnosed (4/125, 3%), including 2 adenomas and 2 adenocarcinomas.
A low prevalence was documented for Harderian papillary cystadenomas (3/125, 2%; classified according to 32 ), adrenal neoplasms (1/125, 1%; diagnosed as pheochromocytoma), and neoplasms arising from bones (osteoma: 2/125, 2%; osteosarcoma: 1/125, 1%).
Non-Neoplastic Inflammatory and Infectious Lesions With Confirmed Cause
Inflammatory and degenerative lesions are summarized in Table 2. Suppurative pleuropneumonia and bronchopneumonia (38/125, 30%) emerged as the most prevalent non-neoplastic conditions in this cohort, accounting for almost one-third of all cases. In a large proportion of animals (33/38, 87%), numerous aggregates of gram-negative coccobacilli were noted histologically. In all cases in which microbiologic investigation was carried out, Pasteurella pneumotropica was isolated and confirmed as the causative bacterial agent in all these cases. Cases in which no culture was performed had similar findings on HE and Gram stains.
Common Inflammatory and Degenerative Lesions in Aged Female NSG Mice, Absolute Number and Percentage (in Parentheses).a
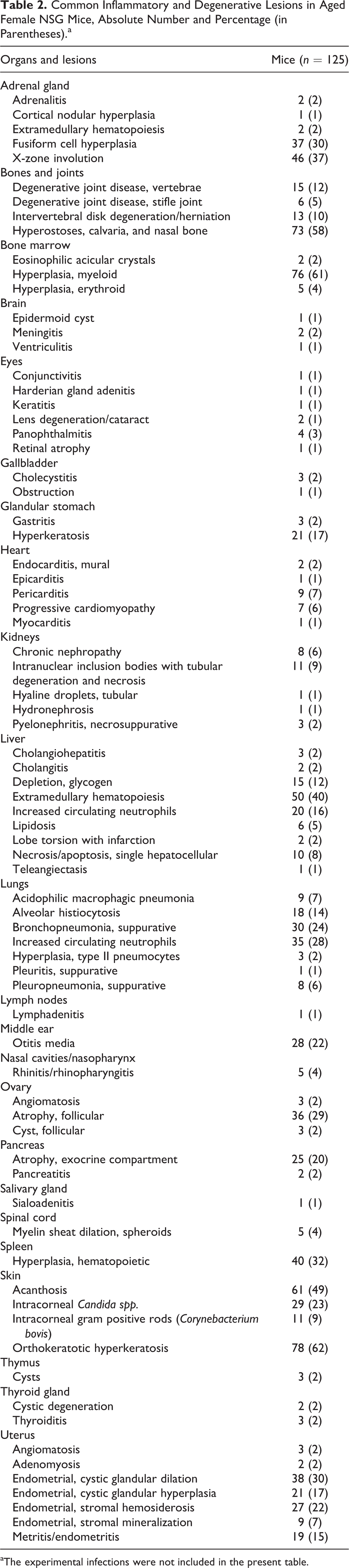
aThe experimental infections were not included in the present table.
Acidophilic macrophage pneumonia (AMP) was identified in 9 of 125 cases (7%). Specifically, AMP was found in association with pulmonary adenomas or carcinomas in 3 cases and with metastases of mammary carcinomas in 2 cases, while it was the only pulmonary finding in the remaining 4 cases. Suppurative otitis media was commonly encountered in our caseload (28/125, 22%) and was reported to be unilateral in the majority of cases (20/28). In some cases (6/28), otitis media was diagnosed in conjunction with multisystemic inflammatory lesions. Based on the finding of multiple inflammatory processes involving different anatomical compartments and the characterization of morphologically similar bacterial populations in each diseased organ, a diagnosis of sepsis was made. 25,45 Aggregates of gram-negative coccobacilli interspersed between the inflammatory cells were occasionally noted, along with sloughed epithelial cells with loss of cilia; however, no culture of the ears was done at the time of necropsy, and it was not possible to confirm the etiology of these lesions. Concurrent involvement of the cardiovascular system was detected in 11 of 125 mice (9%), including 9 of 11 cases (82%) with suppurative pericarditis accompanied by mural endocarditis in 2 cases, and less common findings of myocarditis and epicarditis (1 case each; 9%).
Inflammatory lesions affecting the reproductive system were also highly represented. Particularly, necrosuppurative endometritis and metritis with a mixed population of gram-negative and gram-positive bacteria were recorded in 19 of 125 cases (15%), accompanied by occasional thrombosis of myometrial vessels.
Inflammatory renal lesions were of low frequency in our study, with 3 of 125 (2%) cases of unilateral necrosuppurative pyelonephritis with intralesional colonies of gram-positive coccoid bacteria.
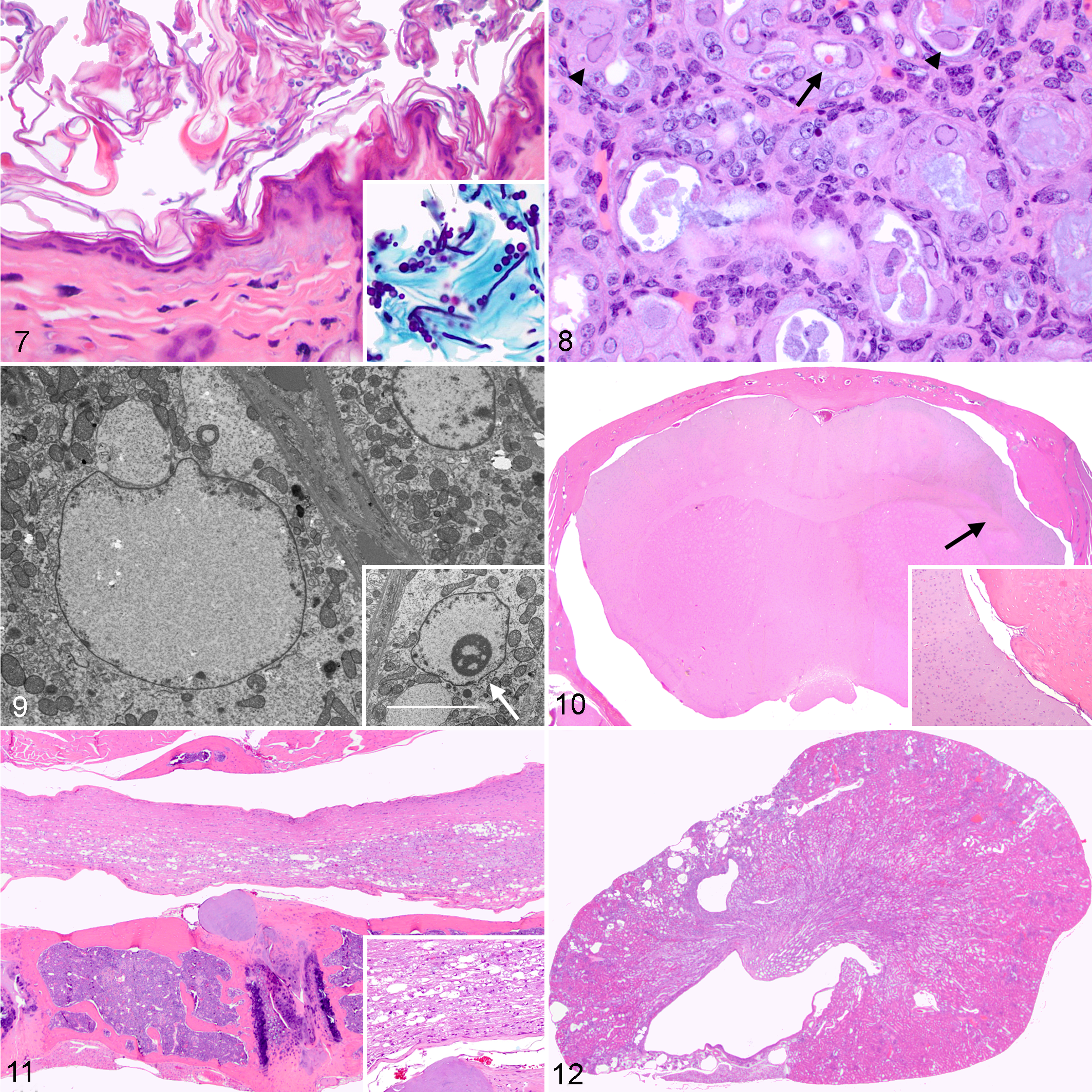
Cutaneous orthokeratotic hyperkeratosis was noted in 78 of 125 cases (62%) in which fur mites and/or Demodex spp. (66/78, 85%) were present. More severe cases exhibited multifocal epidermal acanthosis and spongiosis (61/78, 78%), along with occasional minimal mixed inflammatory infiltrates in the subjacent dermis. Furthermore, enmeshed within the keratin lamellae, numerous basophilic, PAS- and GMS-positive, oval to elongate, 3- to 5-μm-diameter yeasts, together with pale basophilic pseudohyphae, 3 μm in thickness, composed of chains of elongate yeast-like cells, and occasional septate hyphae were noted within the stratum corneum in 29 of 78 cases (37%) (Fig. 7). These organisms were morphologically compatible with Candida spp., although further characterization and definitive culture was not carried out and were observed on the head mostly at the level of the mandibular region. Animals displaying cutaneous candidiasis belonged to the following groups: G3 (n = 10, infected with Myocoptes musculinus and Myobia musculi), G2 (n = 8, infected with Myobia musculi and Radfordia affinis), G4 (n = 7, infected with Aspiculuris tetraptera), G5 (n = 2, infected with Demodex musculi and kept on oral amoxicillin); G1 (n = 2, infected with Myocoptes musculinus and Demodex spp.). In 11 of 78 animals (14%), aggregates of basophilic 1- to 2-μm-diameter, gram-positive short rods morphologically compatible with Corynebacterium bovis were detected within the stratum corneum. Specifically, C. bovis was noted in animals infected with fur mites (7 cases) and less commonly in animals with concurrent Demodex spp. infection (4 cases). The infection with C. bovis was confirmed in 5 of 11 cases (45%) by culture.
Three cases of cholecystitis (2%) were diagnosed and were accompanied by cholangiohepatitis in 2 cases. One of those cases had gallbladder obstruction secondary to a necrosuppurative cholecystitis with intralesional rod-shaped bacteria (1/125, 1%), and Hafnia alvei was isolated on culture. Also, 2 cases (2%) of cholangitis were observed.
Other less common findings included panophthalmitis (4/125, 3%), keratitis (1/125, 1%), conjunctivitis (1/125, 1%), and rhinitis (5/125, 4%).
Secondary histologic changes most consistent with ongoing inflammation included myeloid hyperplasia in the bone marrow of 76 of 125 animals (61%), increased circulating neutrophils within pulmonary capillaries and/or hepatic sinusoids in 55 of 125 cases (44%), and splenic hematopoietic cell hyperplasia with marked increase in the myeloid cell lineage in 40 of 125 cases (32%). Also, pancreatic atrophy of the exocrine compartment with zymogen granules depletion, consistent with decreased food intake, was noted in 25 of 125 animals (20%).
Variable numbers of intestinal flagellates were noted within the lumen of the large intestine, as previously described for immunodeficient mice. 66 These intraluminal protozoa were not associated with any clinical signs or evidence of host response or tissue damage.
Non-Neoplastic Inflammatory/Infectious Lesions Without Confirmed Cause
In 11 cases (9%), 2 types of inclusion bodies were noted within renal cortical and, to a lesser extent, medullary tubular epithelial cells. Large amphophilic inclusion bodies measuring approximately 15 to 20 μ in diameter were detected within renal tubular nuclei and were accompanied by margination of the chromatin, anisocytosis, anisokaryosis, and karyomegaly (Fig. 8). Smaller, 3 to 5 μ in diameter, round bright eosinophilic intranuclear inclusions, surrounded by a clear halo and margination of the chromatin, were also noted (Fig. 8). Both types of inclusions were negative on acid fast stain. All cases had renal tubular degeneration and necrosis, characterized by loss of cellular details, flattening of remaining tubular epithelial cells, and intraluminal sloughed necrotic cells and amphophilic casts. These changes were marked in all cases except 2 that were moderate. Additionally, papillary necrosis was evidenced in 5 mice (45%) with severe tubular changes, while papillary fibrosis was noted in 1 mouse with moderate changes.
In 2 cases, TEM was performed to better characterize the inclusions. Ultrastructurally, they consisted of flocculent electron-lucent material that was either filling the nucleus with displacement of chromatin (large inclusions) (Fig. 9) or partially replacing and disrupting the nucleolus (small inclusions) (Fig. 9, inset). Mitochondrial swelling and disruption of cristae (cristolysis) was often evident in the cytoplasm of affected epithelial cells. No structures consistent with viral particles were observed. Additionally, by PCR, renal samples collected from 2 animals were negative for mouse adenovirus 1, mouse adenovirus 2, murine cytomegalovirus, and murine polyomavirus. In 2 cases where serum chemistry was performed, both animals exhibited elevated serum urea (261 mg/dL and 364 mg/dL, respectively; internal reference interval: 20–32 mg/dL; mean value: 24.8 mg/dL according to 30 ) and creatinine (1.9 mg/dL and 2.31 mg/dL, respectively; internal reference interval: 0.14–0.23 mg/dL).
Histopathology of Non-Neoplastic Noninflammatory Degenerative Lesions
Severe lymphoid hypoplasia affecting all lymphoid tissues (thymus, lymph nodes, spleen, mucosal-associated lymphoid tissues) was observed in these NSG mice. These abnormalities were not classified as lesions as they represent an expected phenotype in this strain.
Degenerative changes are reported in Table 2 and most commonly involved the reproductive tract, the endocrine, and the skeletal systems. Within the uterus, either focal or diffuse endometrial cystic glandular dilation or cystic glandular hyperplasia were detected in 59 of 125 cases (47%). Other findings in the reproductive tract of older mice included uterine stromal hemosiderosis (27/125, 22%) and decreased ovarian follicles and corpora lutea in 36 of 125 cases (29%). Senescent changes in the adrenal glands consisted of intracytoplasmic accumulation of ceroid within macrophages at the level of the X zone (X zone involution) in 46 of 125 cases (37%) and marked subcapsular fusiform cell hyperplasia (37/125, 30%).
In the skeletal system, focal or multifocal hyperostoses at the level of the calvaria and/or nasal bone, protruding within the nasal or cranial cavities, were detected in 73 of 125 cases (58%). Histologically, this consisted of well-organized and locally extensive formation of woven and lamellar bone beyond preexisting periosteum (proliferative hyperostosis). In most cases, this encompassed the dorsal portion of the skull at the level of hippocampus and cerebellum (Fig. 10); however, bony proliferation along the lateral sides of the skull was also evidenced. Occasionally, the proliferative bone displayed variable subperiosteal extensions, forming a bony fold entirely covered by the periostium (Fig. 10, inset). In 23 cases, the bony protrusions were gradually compressing the brain; however, there was no evidence of encephalomalacia.
Moderate signs of degenerative joint disease (osteoarthritis) consisting of cartilage disorganization, chondrone formation, and surface deposition of amorphous eosinophilic material were evidenced at the vertebral end plates (15/125, 12%) or stifle joint (6/125, 5%). Intervertebral disc protrusion characterized by rupture of the inner layers of the annulus fibrosus, partial displacement of the nucleus into the disrupted annulus, and subsequent annular hypertrophy was noted in 13 cases, mostly located at the level of the thoracic segment (Fig. 11). Consequent locally extensive spinal cord compression with myelin sheath dilation (Wallerian degeneration) and spheroid formation (Fig. 11 inset) was evident in 5 of these cases. Despite the changes described at the level of the spinal cord, vertebral bodies, or intervertebral discs, none of them was associated with specific neurologic signs.
Organs with few pathologic degenerative changes included the kidneys and heart. In 8 cases (6%), kidneys displayed large multifocal and occasionally wedge-shaped cortical areas characterized by severe cystic dilation of glomeruli, glomerular atrophy, tubular loss, and interstitial (replacement) fibrosis (Fig. 12). In some cases, these lesions extended in the subjacent medulla. These changes were unilateral in all cases; the percentage of affected parenchyma varied between 20% (2 cases), 30% to 40% (4 cases), 70% (1 case), and 100% (1 case) of the organ.
Mild progressive cardiomyopathy was observed in 7 mice. Lesions consisted mainly of floccular degeneration of cardiomyocytes (5/125, 4%) and cardiomyocyte necrosis (2/125, 2%), accompanied by interstitial fibrosis in some cases.
Causes of Death
The major COD are summarized in Table 3. Mammary tumors represented the most common neoplastic COD and were accompanied by pulmonary metastases in 5 cases. Other tumors identified as single major COD in NSG female mice included 3 multisystemic lymphomas, 1 histiocytic sarcoma with multisystemic dissemination, 2 hepatocellular carcinoma, 2 cutaneous malignant neoplasms, and 1 uterine parietal yolk sac carcinoma.
Relevant Causes of Death (CODs) in Aging Female NSG Mice, Absolute Number and Percentage (in Parentheses).
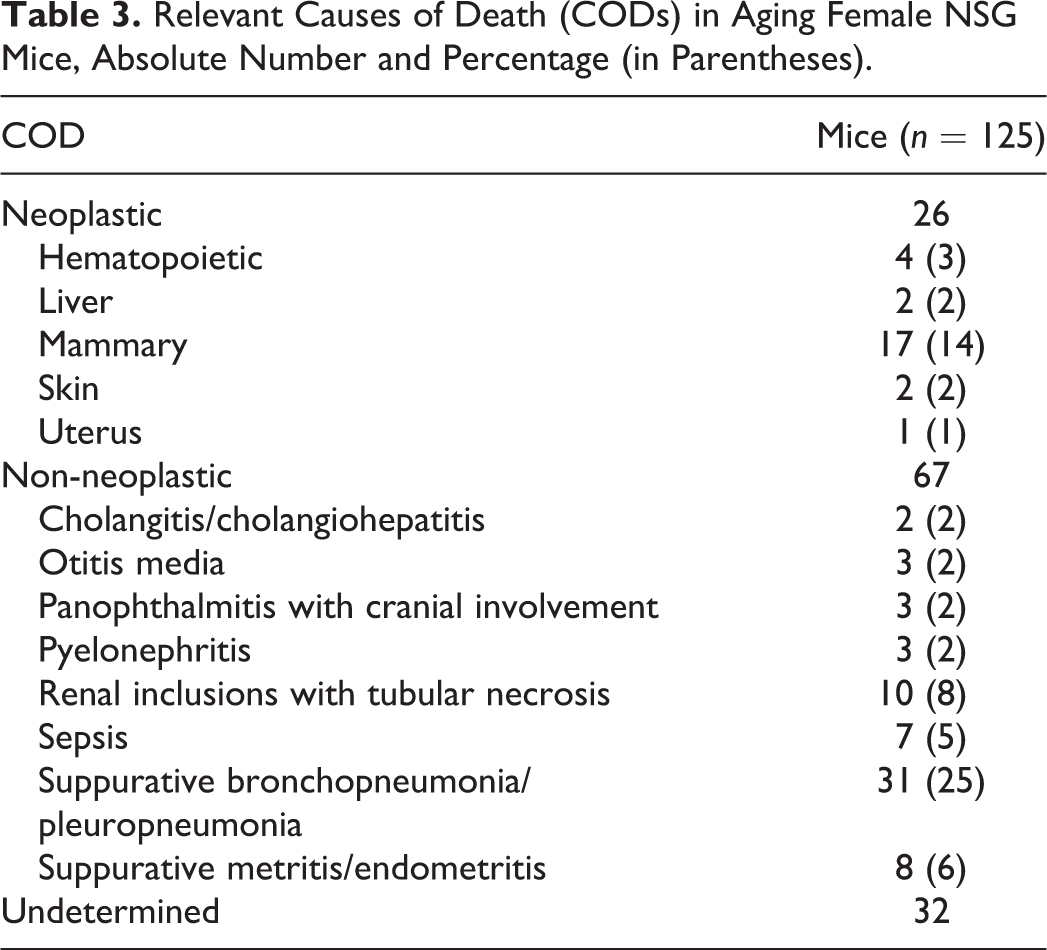
Non-neoplastic COD included suppurative bronchopneumonia/pleuropneumonia, sepsis, suppurative metritis/endometritis, renal intranuclear inclusions with tubular degeneration and necrosis, and otitis media.
Discussion
This study aimed to characterize the spectrum of spontaneous neoplastic and non-neoplastic lesions in aged NSG female mice. Several genetic backgrounds have contributed to the NSG strain, including the NOD, BALB/c, C57BL/6, and 129 strains, with NOD (a Swiss-derived strain) 34 being the predominant genetic background, and a relatively minor contribution from the 3 others; 60 therefore, the NOD background is expected to significantly contribute to spontaneous pathology of the NSG strain. While the background pathology of BALB/c, C57BL/6, and 129 has been extensively described in the literature, information on naturally occurring lesions of NOD mice is relatively limited, except for its well-characterized immune mediated diabetes mellitus phenotype (which is not present in NOD-derived immunodeficient strains). Two reports describing spontaneous neoplasia in the NOD and NOD-derived substrain listed lymphomas, osteosarcomas, and mammary carcinomas as most common tumors. 28,34
Major clinical manifestation and cause of morbidity were recorded, and mice exhibiting moderate to severe signs of systemic illness were included in end-of-life analysis and the calculation of the median survival time.
Previous reports of NSG mice documented a median survival time of 89 weeks (range, 59–95 weeks), 60 which was significantly higher than the median life span of NOD scid mice (37 weeks). 61 A shorter survival time was reported for the NSG mice described herein (median age: 52 weeks; range, 16–104 weeks). Potential contributors to the shorter life span observed in this study include: husbandry conditions that exposed the mice to opportunistic pathogens, absence of a prophylactic antibiotic treatment in the majority of mice, and parity. Parasites (from experimental infections in Facility B) did not overtly contribute to death in this study.
Mammary neoplasms, along with lymphoma, pulmonary, and hepatic neoplasms, have long been recognized as some of the most prevalent naturally occurring neoplasms in the most commonly used laboratory mouse strains, although tumor spectrum is highly influenced by genetic background. 6 In this study, mammary tumors were the most common type of neoplasm and a common COD. While we are aware of few previous report of mammary neoplasms in NSG mice, 12,61 they were described as a common finding in NOD and NOD-derived strains. 28 Mammary tumors were often accompanied by pulmonary metastases. A common subtype was adenosquamous carcinomas, which were previously reported to occur occasionally in non–MMTV-infected mouse strains and more common in carcinogen-treated mice. 10 The presence of both trichohyalin and keratohyalin granules within the lining squamous epithelium and the abundant keratin support squamous differentiation. It is notable that the presence of trichohyalin granules is generally interpreted as representing follicular inner root sheath differentiation in cutaneous epithelial tumors; it is not a common feature of mammary tumors and is not mentioned in the MMHCC and INHAND classifications; however, it has been described in some murine mammary neoplasms. 40,55
Endometrial yolk sac carcinomas were identified in this study. In rodents, both spontaneous and induced yolk sac tumors have histologic patterns mimicking 1 or 2 layers of fetal membranes, which are represented by the parietal and the visceral yolk sac. 37 In our cases, yolk sac tumor cells produced an abundant, eosinophilic, PAS-positive matrix in which nests and cords of neoplastic cells were embedded, mimicking the parietal layer of the fetal membranes. The hyaline ground substance characteristic of parietal yolk sac tumors is reminiscent of Reichert’s membrane from normal embryos, a basement membrane that separates the visceral and parietal layers of the rodent yolk sac from the maternal trophoblast. 37 Several histochemical methods have been reported to better characterize this hyaline substance, including silver impregnation, mucicarmine, PAS, PTAH, and staining for collagen. 63 Both the neoplastic cells and the matrix stain positive for laminin, 11,54,63 as documented here. Spontaneous yolk sac carcinomas of the reproductive tract are considered rare in both rats and mice 37,54 and were reported both in breeding animals and animals who had never been mated. 63 Metastatic spread has been reported in both species, 54,63 although in our cases, metastases were not detected, and the tumors were only locally invasive.
Two cases of neuroendocrine carcinoma were observed in the mesometrium, without evidence of involvement of other organs. We are not aware of prior reports of neuroendocrine carcinoma arising in mesometrium in rodents. These 2 tumors appeared to represent paragangliomas occurring at an atypical site.
Lymphomas were uncommon in this study. Spleen, intestinal mucosal-associated lymphoid tissue (MALT), and lymph nodes were most commonly involved. All lymphomas were positive for CD45 and positive for either B220 or CD3, indicating B or T cell origin, respectively. Furthermore, thymic involvement was only sporadic. This finding is not surprising; although thymic lymphomas occur early with high incidence in NOD scid mice 50 and has been described in NOG mice, 26 it has been reported that NSG mice do not develop thymic lymphomas. 60 This finding contrasts with the higher incidence of hematopoietic neoplasms in the strain that contributed to the NSG genetic background, including NOD, NOD scid and BALB/c mice, 7 B6;129, 20 and FVB (another Swiss-derived strain). 36
Osteosarcomas are the most commonly reported bone neoplasms in laboratory mice. 1,41 Specifically, a high frequency (11%) has been reported for NOD mice, with lower frequency in NOD-derived strains, including a 3% incidence in NSG mice. 28,29 In agreement with these findings, only 1 case of osteosarcoma (1%) was diagnosed in our cohort of NSG mice, and only 2 osteomas arising from the periosteal surface of the skull were noted.
Pulmonary epithelial neoplasms and adrenal pheochromocytoma were uncommon in our caseload, in contrast with the prevalence reported for old FVB females. 36
Major infectious conditions evidenced herein and classified as common causes of death were represented by suppurative pleuropneumonia and bronchopneumonia with abscess formation. All the animals with evidence of suppurative lesions in the lungs were kept in ventilated racks without autoclaved feed and bedding and did not receive amoxicillin-supplemented diet, confirming the importance of these special husbandry practices in preventing opportunistic infections in highly immunodeficient mouse strains. P. pneumotropica was the only bacterium isolated from pulmonary lesions, and it may have been the underlying cause of other commonly encountered lesions, including otitis media, pericarditis, endometritis, and myocarditis, although microbiology was not performed on these lesions. Also, in those mice exhibiting suppurative pulmonary lesions in which no culture was performed, P. pneumotropica was suspected to be the cause due to similarities on HE and Gram stains. P. pneumotropica is a gram-negative, non–spore-forming opportunistic pathogen with high prevalence in murine research colonies. 66 Transmission occurs through direct contact, although its true nature as a primary pathogen is questionable. Outbreaks of P. pneumotropica have been reported in numerous multiple immunodeficient strains. 13
Otitis media caused by various opportunistic bacteria was also relatively common, with more severe lesions considered a COD. Common organisms implicated in the onset of otitis in immunocompromised strains include Mycoplasma pulmonis, Pseudomonas aeruginosa, Klebsiella oxytoca, Streptococcus sp, reovirus, and Sendai virus. 2 In some animals in our study, similar to previous findings in mice with B6;129 background, 20 no bacteria were observed within the middle ear; however, the lack of a causative organisms in these animals cannot completely rule out the presence of an initiating infectious agent.
Necrosuppurative lesions of the genital tract were also considered a common cause of morbidity and death in our NSG mice and were often secondary to uterine prolapse. A variety of opportunistic bacteria, including P. pneumotropica, K. oxytoca, and staphylococci, are normally responsible for the development suppurative lesions in the urogenital tract. 2 The cases reported herein were not investigated microbiologically.
AMP was identified in a few animals. AMP has been originally described in Swiss mice 18 and in mice with a Swiss background; 43 it has been reported as a major cause of disease and death in 129S4/SvJae 23 and 129S6/SvEvTac mice, 51 while it has been less commonly observed in B6;129 mice 7,20 and C57BL/6 J. 7 In our study, this finding was only sporadic, focal, and minimal to mild in severity. Therefore, it was considered only an incidental finding that did not appear to contribute to death.
Inflammatory renal lesions caused by ascending urinary tract infections were sporadically reported in our study. Previous studies conducted on NSG male and female breeders identified Enterococcus sp. and K. oxytoca as the major opportunistic bacteria causing acute and chronic renal lesions following ascending infections from the lower urinary tract. 12 K. oxytoca and Enterococcus organisms are not considered primary mouse pathogens and are often isolated from the gastrointestinal tract of immunocompetent mice without evidence of disease. 12 In our NSG cohort, intralesional gram-positive coccoid bacteria compatible with Enterococcus sp. were seen histologically; however, microbiology was not performed at the time of necropsy, and the etiology cannot be confirmed with certainty.
Chronic dermatitis characterized by orthokeratotic hyperkeratosis, epidermal acanthosis, and crusts was commonly encountered in this strain of mice. Certainly, the experimental infestation of these animals with fur mites and Demodex spp. contributed to increase the incidence. However, other infectious agents such as fungal element morphologically compatible with Candida spp. (but not confirmed by culture) and C. bovis were noted. Candida spp. are trimorphic fungi 3,22 commonly isolated from mucosal surfaces of humans and animals. 48 The normal bacterial flora usually restricts their growth in the alimentary tract of immunocompetent animals. 48 In fact, upper gastrointestinal tract and vagina represent the most common sites involved in candidiasis models. Initiation of invasive infections usually requires immune suppression of the host; however, C. albicans can infect common inbred strains of mice such as DBA, BALB/c, A/JCr, or C57BL/6 without the need for immune suppression. 16 To our knowledge, there are no previous reports of naturally occurring cutaneous candidiasis in NSG mice. Proliferation of these organisms along the epidermal surface could be related to disruption of the cutaneous barrier by the ectoparasitic burden. Further investigation would be needed to fully characterize the cause and significance of this cutaneous fungal infection in NSG mice.
C. bovis is a gram-positive coryneform that has been associated with skin disease in mice carrying deficits for T-cell (Foxn1nu athymic nude mice) and T- and B-cells (Pkrdcscid). 9,56,66 Morbidity can be high, as transmission and persistence in the environment are associated with shedding of the infected keratin flakes. 66 The underlying mechanism responsible for the epidermal proliferation is unknown; however, mice deficient in host defense components are likely to have increased disease. 66
A case of gallbladder obstruction secondary to a marked, chronic, fibrosing, and abscessing bacterial cholecystitis was described. Hafnia alvei, a member of the Enterobacteriaceae family of bacteria, was isolated from this lesion. This genus of bacteria is considered normal flora in the human gastrointestinal tract and has been shown to cause opportunistic infections, particularly in immunocompromised individuals. To our knowledge, there are no previous reports of Hafnia alvei infections in mice.
Complete depletion of lymphoid tissues was seen as a normal feature in all mice, and there was no evidence of functional activation and expansion of the lymphoid component.
In 11 mice, examination of the kidneys revealed 2 types of intranuclear inclusions in tubular cells, associated with karyomegaly and tubular degeneration and necrosis, and TEM and PCR did not reveal any evidence of viral infection. Large amphophilic intranuclear inclusions causing margination of chromatin and karyomegaly, which closely resemble 1 type of inclusion observed in the present study, have been reported in immunocompetent (ICR) 4 and immunodeficient mice (scid, NOD scid, and B6.129S7-Rag1tm1Mom/J). 38 In these cases, TEM showed that the inclusions were composed of electron lucent material of undetermined nature, without evidence of viral particles. Serology, PCR, and IHC were also negative for viruses, with the exception of positive IHC for a bovine papillomavirus antigen in 1 study, a finding of unclear significance. 38 Other authors have anecdotally reported similar inclusions, with unsuccessful attempts at identifying a viral etiology. 2 While the large amphophilic inclusions reported here in NSG mice correspond to these previous reports on light microscopy and TEM, 2 previously unreported findings were observed: the concurrent presence of a second type of intranuclear inclusions, smaller and bright eosinophilic, and the occurrence of moderate to marked tubular degeneration and necrosis, in contrast to the rare pyknotic cells previously described. 4 Additionally, the epidemiology of our findings suggested a transmissible opportunistic pathogen affecting preferentially immunodeficient mice: several female mice belonging to another immunodeficient strain (TRP-1 TCR) exhibited similar renal pathology after being housed with these NSG mice while similarly co-housed Swiss Webster mice were not affected (results not shown). However, the result of our investigation supports the conclusion of other authors: given the lack of evidence of a viral etiology, these inclusions appear to represent a noninfectious degenerative process of undetermined mechanism.
While several types of nonviral intracytoplasmic inclusions have been report in rodent kidneys, such as myeloid bodies, protein precipitates, giant mitochondria, or proliferating endoplasmic reticulum, 14 the only well-characterized mechanism for intranuclear inclusions is accumulation of viral matrix proteins 14 and lead toxicity. 19 However, in our study, inclusions were not composed of a discrete electron dense central core surrounded by fibrillar structures, as described for lead, 19 and were acid fast negative. Additionally, no known source of lead was present within the environment in which these mice were housed.
Multiparous or breeder mice have substantially different incidences of certain lesions or phenotypes compared to nulliparous mice. 15 The non-neoplastic noninflammatory degenerative lesions evidenced in multiple organs but most commonly encountered in the reproductive tract, endocrine, and skeletal systems were not identified as significant comorbidities in these mice and were most likely incidental findings. Endometrial stromal polyps represent a common finding in the uterus and cervix of aged mice of most strains, 11 including BALB/c; 7 our findings in NSG likely support this statement. Additionally, several cases of uterine prolapse were diagnosed herein, and a link has been made previously between prolapse and other uterine lesions like severe cystic changes and/or tumors in aged mice. 11 Age-related atrophy of the ovaries is another common outcome of reproductive senescence in the female, 11 with high incidence in C57BL/6 mice 7 and FVB mice, 36 and it is characterized by depletion of primordial follicles and changes in endocrine responsiveness of the hypothalamic-pituitary-ovarian axis.
Aging female mice of A, C57BL/6, BALB/c DBA/2, 6 CD-1 (Crl: CD 1(ICR)BR), 49 and FVB 36 strains have a higher incidence of adrenal subcapsular fusiform cell hyperplasia compared to males, although these changes were minimally present in female NSG mice.
Common age-related lesions of the bones and joints in mice include osteoporosis, intervertebral disc disease, and degenerative joint disease (DJD). 21 DJD occurs with both strain and sex differences, with males reported to be more affected than females. 35 In the present study, the stifle joint and the vertebral end plates appeared to be most commonly affected by chronic changes, indicating that mechanical trauma may be a factor in the etiopathogenesis of this disease.
We identified hyperostotic lesions at the level of the calvaria and/or nasal bone. Previous reports of spontaneous periosteal hyperostosis have been described for DBA/1 male mice in hind limbs and forelimbs 57 and for aging B6C3F1 and ICR mice, 35,67 especially at the level of sternum and tibia in both sexes. However, to our knowledge, there are no previous reports either for NSG mice or the strains contributing to the NSG genetic background.
Less common conditions in these animals include unilateral chronic nephropathy. Given the morphology and the distribution of the lesions, 2 etiopathogenetic mechanisms are most probable: these areas may represent a sequela of chronic pyelonephritis (as NSG mice are predisposed to ascending bacterial infection as previously mentioned), or they could potentially represent chronic renal infarcts.
A major pitfall of the present study included the lack of data on aging male NSG mice and the subsequent absence of comparison between sexes. We cannot exclude that the morphology and incidence of some lesions may be related to hormonal or metabolic characteristics of females and that major sex-related variations may exist for the most common neoplastic categories. Some age-related lesions in other strains exhibit a clear sexual dimorphism, and it is likely the same is true in NSG mice. Furthermore, it is worth noting that the mice considered in the present study were not experimentally naïve, and a proper experimentally naïve control group was lacking. While intestinal nematodes were not associated with any overt lesions, ectoparasites clearly induced some cutaneous lesions. The vast majority of the age-related lesions described here did not show any clear causative link to the parasites, but it is possible that these infestations may have contributed to the occurrence of these lesions in subtle ways, and age experimentally naïve NSG mice may show a different spectrum of lesions. However, despite the limitations of this study (examination of females only, presence of different parasites, non-homogeneous husbandry/treatment conditions, and complete lack of health monitoring surveillance in facility B), the comprehensive pathologic examination of large numbers of aged NSG mice allowed to live their life span offered a unique opportunity to study age-related pathology in this strain.
Footnotes
Acknowledgements
We thank Leona Cohen-Gould and Juan Pablo Jimenez from the Electron Microscopy and Histology Service of the Imaging Core Facility of the WCM Core Laboratories Center for their technical contributions with electron microscopy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by NIH/NCI Cancer Center Support Grant P30 CA008748.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
