Abstract
Hoof canker is described as progressive pododermatitis of the equine hoof with absent epidermal cornification and extensive proliferation of the dermal papillary body; however, in-depth research on the type of proliferative activity has not yet been reported. The aim of the present study was to determine cell-specific proliferation patterns together with morphological analysis of hoof canker tissue. Tissues removed during surgery from 19 horses presented for treatment of canker were compared with similar postmortem tissues of healthy hooves of 10 horses. Morphological alterations visible in light microscopy were assessed semiquantitatively and graded for severity. Proliferative activity was evaluated by means of anti-PCNA (proliferative cell nuclear antigen) and anti-Ki67 immunohistochemistry. Histologically, canker tissue showed 5 major morphological alterations–the presence of lacunae, vacuoles, giant cells, hemorrhage, and inflammation–not seen in control tissue. Also, there was a notable koilocytotic appearance of keratinocytes in canker tissue. Immunohistochemistry revealed increased levels of PCNA protein expression in keratinocytes and fibroblasts of canker tissue compared with control tissue. In control tissue, keratinocytes showed higher levels of Ki67 compared with canker tissue, while the dermal fibroblasts of both groups showed similar levels of Ki67, indicating similar proliferative activity of less than 3% of total dermal fibroblasts. These results demonstrate that, in contrast to previous reports, there is no evidence for increased proliferative activity of the dermal papillary body associated with hoof canker. Increased levels of PCNA protein expression and morphological alterations indicate that dysregulation of keratinocyte differentiation constitutes a key event in equine hoof canker development.
Keywords
Equine hoof canker (Pododermatitis chronica verrucosa sive migrans) is described as destructive and progressive pododermatitis with alterations in the cornification process of the keratinocytes (parakeratosis and hyperkeratosis). 15,24,26 Extensive proliferation of the dermal papillary body with subsequent absence of epidermal cornification is reported to be a major feature of disease. 23,24 Hoof canker can affect 1 or more hooves, and it compromises the use and welfare of horses due to ensuing instability of affected hoof capsules and lameness at late stages. The cause of this condition is unknown. Different theories for the development of hoof canker have been reported. Inadequate husbandry (eg, a lack of hoof hygiene and excessive moisture), genetic predisposition, (auto)immune reactions, microorganisms (such as fungi, spirochetes, and other anaerobic bacteria), and bovine papillomaviruses have been suggested to be involved in the development of canker. 3,9,19,20,28 However, for any of these, a causal association with the development of disease remains to be established. Morphological studies of hoof canker tissues are rare. 24,26 Ballooning of the keratinocytes, mainly in the stratum germinativum of the frog, has been reported together with the occurrence of perinuclear vacuoles in the cells of deep layers of the stratum spongiosum; the cells of the corium are covered by a thin layer of defective epidermis, leading to an increased risk of infection and bleeding. 26
To our knowledge, the proliferative character of canker has not been analyzed in detail. Immunohistochemistry allows characterization of tissue proliferation, as has been shown in a number of potentially metaplastic or neoplastic tissues. 4,13,14,27 During the proliferative phase of eukaryotic cells, different markers are used to determine the proliferative activity in healthy versus pathologically altered tissue. 4,13,27 In the present study, antibodies to detect the Ki67 protein and the proliferating cell nuclear antigen (PCNA) protein were selected to analyze the proliferative processes in canker tissue. Ki67 is a sensitive, well-established predictive marker for recurrence in human breast cancer; it is widely used in tumor diagnostic and cell proliferation research. 14,27 PCNA is a marker for proliferation; it indicates DNA damage and related repair mechanisms but can be induced also by growth factors independent of the cell cycle. 14 Based on the hypothesis that equine hoof canker is a proliferative disease, the aim of the present study was to investigate the proliferative status and morphological alterations in canker tissue compared with healthy hoof tissue.
Materials and Methods
Sample Collection
Tissues of 19 horses presented for treatment of hoof canker to the Equine University Clinic of the University of Veterinary Medicine, Vienna, Austria (Supplemental Table 1), were assessed. Hoof canker was diagnosed based on its typical clinical appearance. 1,23,24 Tissue samples were taken from superficial regions of canker-affected areas with a hoof knife on the first day of treatment and from deep regions including the dermoepithelial junction during surgery under regional anesthesia on the following day. Tissue sampling and evaluation, as well as anonymized publication of the data obtained, were carried out with the written consent of the horse owners, given on admission of the horse to the clinic. Sample collection was carried out during therapeutic trimming and surgery of affected hooves; all tissues used were removed based on clinical necessity.
Samples of 11 horses euthanized for reasons other than hoof or skin disease were used as control tissue (Supplemental Table 1). Prior to inclusion as controls, all hooves and areas of skin were examined macroscopically and histologically for absence of pathological alterations. Therefore, hoof tissues of corresponding regions of 10 of the 11 horses were sampled postmortem, and skin of 1 of the 11 horses was used for the establishment of immunohistochemical (IHC) protocols.
Histology and Immunohistochemistry
Tissues were cut into 1 × 2–cm pieces for histological embedding. Tissues for histological and IHC analysis were immersion-fixed in 4% buffered formaldehyde for a minimum of 72 hours and embedded in Paraplast (Vogel, Giessen, Germany). Histological sections of 3-μm thickness were cut and stained with hematoxylin and eosin (HE) for morphological analysis. 16 For IHC serial sections (3 μm) of skin and hoof, tissues were mounted on 3-aminopropyltriethoxysilane/glutaraldehyde-coated slides. Endogenous peroxidase activity was blocked by incubation in 0.6% H2O2 in methanol for 15 minutes at room temperature. A protein block (1.5% normal goat serum) was used to minimize unspecific binding of the primary antibody. The unlabeled monoclonal primary antibodies (anti-PCNA and anti-Ki67; for sources, pretreatments, and dilutions, see Supplemental Table 2) were detected with the ImmunoVision secondary system (ImmunoVision Technologies, Brisbane, California) using DAB (3, 3′diaminobenzidine-tetrahydrochloride substrate in Tris buffer pH 7.4 and 0.03% H2O2) as chromogen. Finally, slides were washed with distilled water, counterstained with haemalum, dehydrated, and mounted by use of xylene-soluble medium (DPX; Fluka, Buchs, Switzerland). Negative controls were obtained by substitution of the primary antibodies by PBS. Evaluation of the sections was performed using light microscopy (Polyvar; Reichert-Jung, Vienna, Austria) and a digital camera (Nikon, Austria). Sections of healthy equine skin were used as positive controls.
Morphological Analysis
Three HE-stained histological sections per tissue sample and 2 to 5 tissue samples per horse were evaluated. Five frequently occurring alterations of canker tissue were analyzed and evaluated in a semiquantitative setup (Table 1). The grading was performed in a blinded way by 1 person and combined frequency and intensity of the respective criteria. The alterations were graded for intensity as mild (1 point), moderate (2 points), or severe (3 points) and for frequency concerning the occurrence of the respective criterion in less than 20% of the examined area (1 point), between 20% and 50% (2 points), and more than 50% (3 points). The sum of the grades of all investigated sections per horse was calculated, and the mean value for each criterion was determined per horse.
Summary of the Semiquantitative Evaluation of Morphological Alterations Performed on Histological Sections of 19 Horses Affected with Hoof Canker. The grading was based on the combined frequency and intensity of the respective criterion.

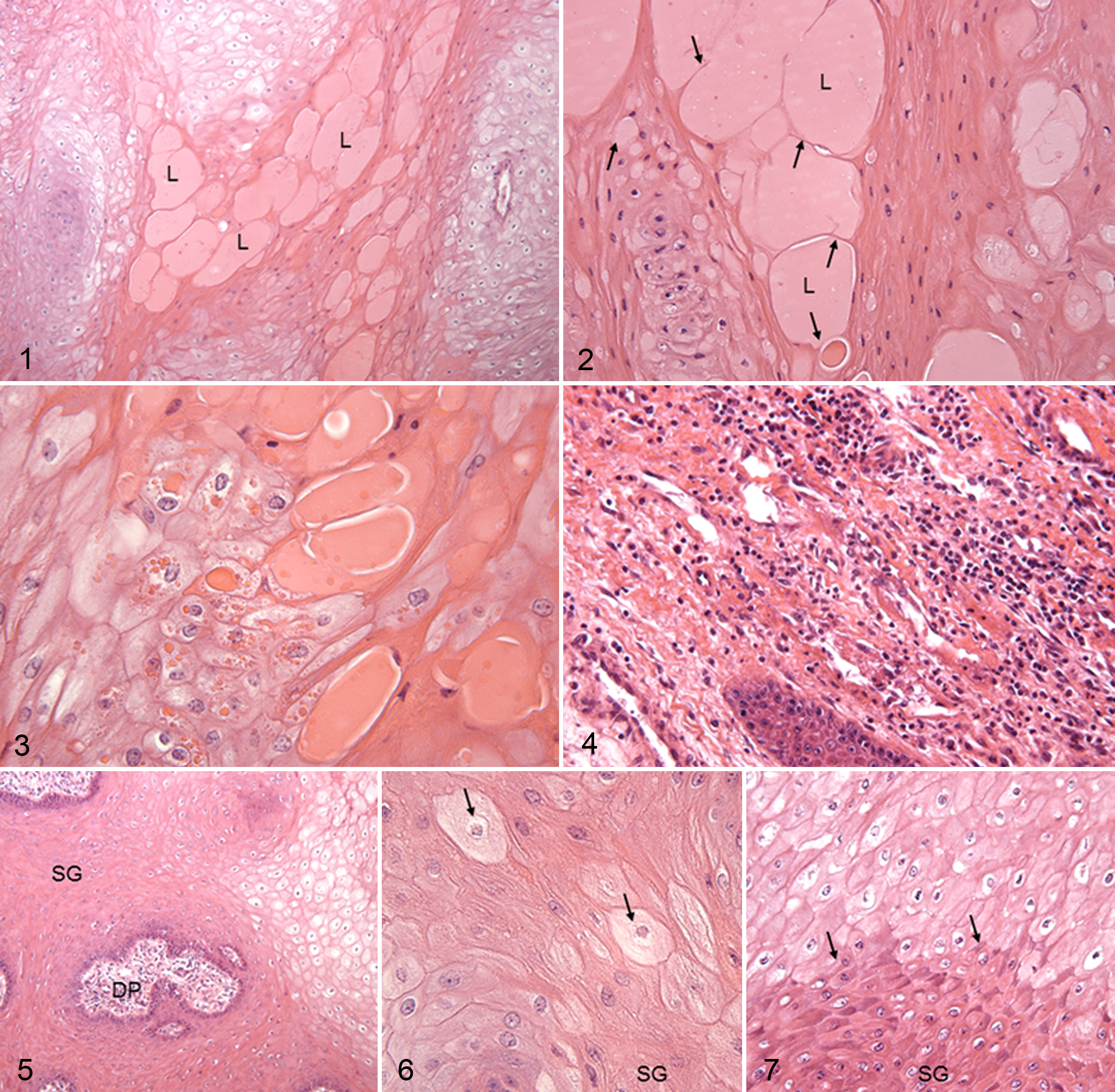
Lacunae were defined as fluid-filled cavities with eosinophilic content; vacuoles were defined as formations surrounding the nucleus of keratinocytes in different manifestations; giant cells presented as enlarged keratinocytes with morphologically intact cytoplasm and nucleus; hemorrhage was described as erythrocyte accumulation; and tissue-based neutrophils, lymphocytes, and monocytes indicated inflammation.
Scoring of Proliferative Activity
Proliferative activity was assessed using 2 antibodies (anti-Ki67, anti-PCNA). The nuclei positive for the respective protein were counted in 3 randomly selected fields featuring a minimum of 100 cells per field on 2 different slides for each tissue sample. In 3 different regions (stratum basale, stratum germinativum, and the dermal portion at the dermoepithelial transition zone in the papillae), proliferative activity was determined as the number of antibody-positive cells as a percentage of total cell number counted per field for each antibody (scoring index). In the present study, the stratum germinativum was defined as the entire proliferative zone of the hoof epidermis, combining the single-layered stratum basale and the proliferative cells of the stratum spinosum.
Statistical Analysis
A linear mixed model using SPSS statistics 20.0 software (IBM; Vienna, Austria) was applied to identify significant differences of proliferative activity (determined via PCNA and Ki67 protein expression, respectively) between healthy and hoof canker–affected cell types of the respective regions. Furthermore, a linear mixed model followed by Sidak correction was used to compare the values of proliferative activity between the different regions for each group (hoof canker tissue and control tissue). For all statistical tests, a P value ≤.05 was considered significant.
Results
Clinical Findings
The study included 10 geldings and 9 mares (Supplemental Table 1) presenting for hoof canker at the Equine University Clinic of the University of Veterinary Medicine, Vienna, Austria, from 2011 to 2014. The average age was 15 years (7–24 years). In 10 of the 19 horses only 1 hoof showed canker, while in 6 of the 19 horses 2 hooves were affected and in 3 of the 19 horses 3 hooves were affected. Hoof canker was diagnosed in hind limbs in 9 of the 31 hooves, whereas front limbs were affected in 22 of 31 hooves (Supplemental Table 1).
Morphological Analysis
Results of the morphological evaluation of histological sections obtained from hoof canker tissue are listed in Table 1. The presence of lacunae was mainly observed at the end of papillae (Figures 1–3). In mildly affected tissue, vacuoles were detected as slim white halos surrounding intact nuclei as revealed by HE staining. In severely affected tissue, nuclei were pyknotic and cells were enlarged and filled with perinuclear vacuoles. These cells were referred to as ballooning keratinocytes or koilocytotic keratinocytes (Figures 1, 3, 5, and 7). Giant cells (Figure 6) were analyzed by frequency only. Hemorrhage in the epidermal tissue was identified on basis of erythrocyte accumulation. In all canker tissues, signs of inflammation including tissue-based neutrophils, lymphocytes, and monocytes were present in varying severity (Figures 4 and 6).
Proliferative Activity
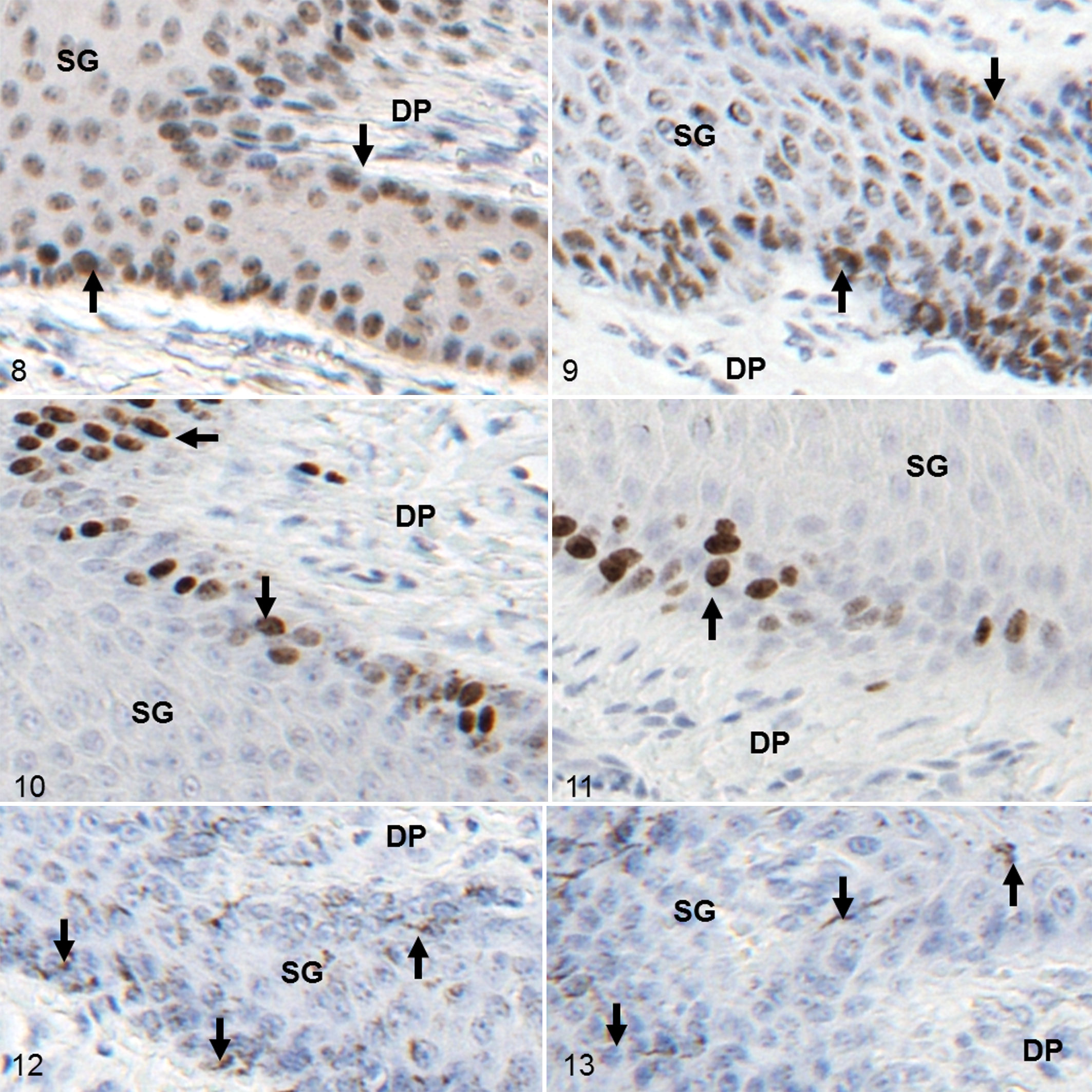
Immunohistochemical analyses of PCNA and Ki67 protein expression showed layer-specific differences (Figures 8–11). In control tissue, epidermal PCNA protein expression was of lower intensity and frequency in the stratum basale and the stratum spinosum compared with corresponding canker tissue (Figures 8 and 9). In the hoof canker tissue, a high number of PCNA-expressing keratinocyte nuclei were located in the stratum spinosum. In the affected dermis, fibroblasts frequently showed PCNA expression in the nuclei; this was a less common observation in control tissue. The Ki67 protein expression patterns were distinct from PCNA expression patterns in both sets of tissues in regard to reactivity and localization (Figures 10 and 11). Keratinocyte nuclei expressing Ki67 were predominantly found in the stratum basale and the deep layers of the stratum spinosum. Ki67-positive fibroblasts were rare in both sets of tissues. Negative controls showed no staining in all IHC reactions (Figures 12 and 13), but melanin granules were occasionally observed in the stratum basale and the stratum spinosum.
Results of quantitative analysis are summarized in Supplemental Table 3 and presented in Figures 14 and 15. The number of PCNA-positive keratinocytes in the stratum basale was significantly higher in canker tissue than in control tissue. A similar scenario was observed for the entire stratum germinativum. In canker tissue, the numbers of PCNA-positive keratinocytes in the stratum basale were significantly lower compared with the stratum germinativum. This significant difference was not evident in the control tissue. The number of PCNA-positive dermal fibroblasts was significantly smaller than the number of PCNA-positive epidermal cells in canker tissue; there was no significant difference in the control tissue.
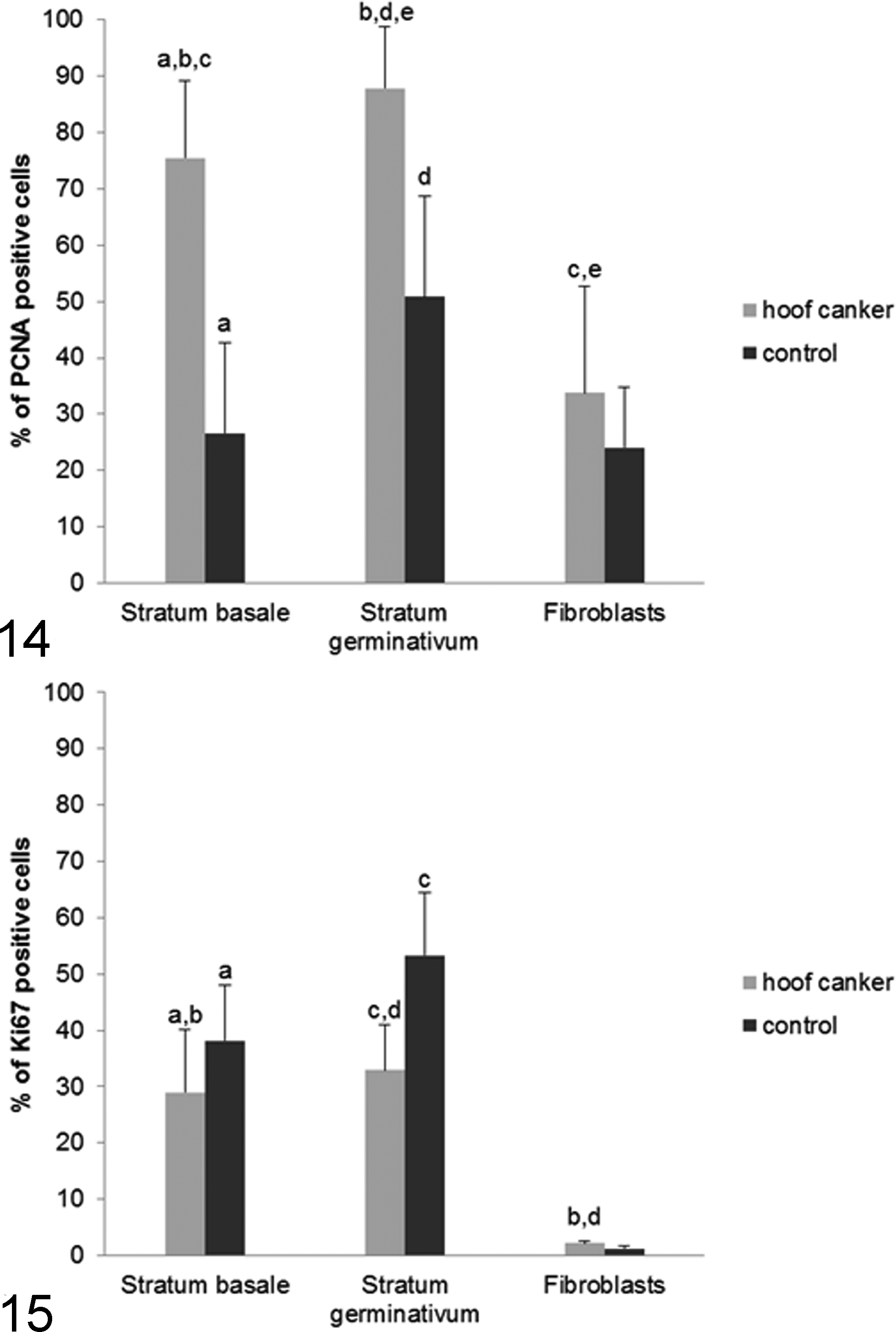
The Ki67 protein staining revealed only a small population of positive cells in canker and control tissue. In control tissue, proliferative activity of the keratinocytes, as determined by Ki67 staining, was higher than in canker tissue in the stratum basale as well as in the entire stratum germinativum. In both sets of tissues, the scoring index for Ki67 in dermal fibroblasts was similar (<3%).
Discussion
Despite the reported higher incidence of hoof canker in hind feet, more than two-thirds of the affected feet were front feet in this study. 20,25 The main limitation of this study was that canker was diagnosed based on its macroscopic appearance in the absence of any other gold standard of diagnosis; main outcomes were microscopic features, therefore introducing a potential risk of circular argumentation.
In the present study, the monoclonal anti-Ki67 antibody detected fewer positive keratinocytes in canker tissue compared with control tissue. The protein Ki67 is a highly sensitive marker of proliferating cells in the G1, S, and G2 phase as well as during mitosis, but it cannot be detected in cells in G0 phase. 27 In contrast, PCNA IHC staining resulted in significantly more positive keratinocytes in canker tissue compared with control tissue, illustrating either a higher proliferative activity or DNA damage-related repair mechanisms in canker tissue. The protein PCNA is more stably expressed and can also be detected at the end of the cell cycle; in its function as a nuclear ring clamp protein, PCNA is also involved in DNA repair mechanisms and can therefore be induced also by growth factors independently from the cell cycle. 14,33 The detection of elevated numbers of PCNA-positive cells and reduced numbers of Ki67-positive cells in the same samples, as found in the present study, has already been described in a variety of tumors and normal tissues. 2,6,10,17
Possible scenarios leading to such results include the detection of PCNA protein at the end of the cell cycle as well as in nonproliferating cells during DNA repair or growth factor activation. Therefore, it is reasonable to find more PCNA-positive cells than Ki67-positive cells. This also explains why Ki67 protein is the more sensitive and specific marker for proliferative events in tumor diagnosis. 6,10,17 Based on this knowledge, the Ki67 protein expression data in the present study indicate that no increase of proliferative activity occurs in epidermis and dermis of canker tissue.
In the present study the protruding tissue, which is so typical for hoof canker, lacked keratinization and related desquamation, whereas hypertrophic events abounded, as revealed by lacunae formation, ballooning keratinocytes, and giant cell formation. These hypertrophic events in combination with loss of tissue integrity might explain the typical filamentous or cauliflower-like appearance of hoof canker, as the dermal parts start to prolapse due to lack of pressure of the horn capsule; this is an effect often seen after the creation of hoof horn defects, if too little pressure is exerted onto the granulation tissue. 36 Lack of keratinization becomes apparent through epidermal cells, which remain spherical, thus creating a larger tissue volume without hyperproliferating. In addition, the production of abnormal quantities or types of cell proteins possibly contributes to the clinical picture of extensive growth and white cheesy exudate. 5,21
In contrast to other descriptions of canker, the present study provides no evidence of increased proliferative activity. 19,30 Yet, hypertrophy and DNA damage with subsequent disturbance of keratinization of epidermal cells were frequently encountered.
Not surprisingly, hemorrhage was observed in 18 of 19 surgical hoof canker samples. The hemorrhage was potentially enhanced by detected inflammatory processes also present in all canker tissues. Inflamed regions were commonly found in and adjacent to the dermal papillae, potentially induced by reduced tissue cohesion between maldifferentiated, noncornified epidermis and corium. A high number of free connective tissue cells were observed in the dermal portion of the dermoepithelial zone and in the dermal papillae, serving as an additional sign of inflammation. Some samples contained bacteria, as the barrier function of the epidermis is impaired with the cessation of keratinocyte differentiation. This is similar to previous reports indicating that bacterial infection is an inconsistent finding in hoof canker. 31
In most hoof canker samples, formation of large epidermal lacunae was observed; these lacunae were filled with eosinophilic fluid and might result from accumulation or increased production of this fluid (Figure 2). Lacunae formation is suggested to be related to instability of the epidermal tissue. 18,32 The instability of the hoof capsule is a common cause for canker-associated lameness in late stages of the disease. 30
Hoof canker–associated keratinocytes are commonly described as “ballooning keratinocytes” because of higher cell volumes, pyknotic nuclei, and perinuclear vacuoles, which were also found in the present study. 19,24 In human keratinocytes, changes induced by human papillomavirus type 16 (HPV-16) include comparable alterations of the keratinocytes (and human cervical cells in vitro) described as koilocytotic cells. These cells feature perinuclear vacuoles appearing as white halos and mostly acentric, hyperchromatic nuclei. 11,12 The same cellular appearance is reported for other virus-infected cells, including molluscum contagiosum, a poxvirus infection in humans that leads to cutaneous lesions with acanthosis, hyperkeratosis, and nuclear atypia including koilocytotic keratinocytes. 29 Therefore, koilocytosis observed in keratinocytes of equine hoof canker tissue may indicate a potential viral involvement in etiology. The success of local cisplatin chemotherapy for equine hoof canker is comparable to the success of intralesional cisplatin chemotherapy for papillomavirus-induced equine sarcoids, which is a well-documented therapy for this disease. 1,7,34,35 The increased expression of PCNA protein in hoof canker–associated keratinocytes is another parallel with symptoms of viral infection, given that in human keratinocytes in vitro, PCNA expression is down-regulated by growth inhibition via transforming growth factor β or calcium-induced differentiation, while in HPV-transformed keratinocytes this effect was reduced and PCNA expression was still high. 8 These observations support the concept of papillomaviruses being involved in the development of equine hoof canker, although the causal association with onset and progression remains to be established. 3 Still, neither morphological analyses nor detected proliferative activity of hoof canker tissue revealed evidence of neoplastic transformation of cells of the papillary body. Reduced PCNA levels were found in differentiated keratinocytes, which might also explain the significantly higher PCNA levels in the poorly differentiated keratinocytes in hoof canker. 8,22 Together with the acanthosis seen in most samples, this is additional evidence that hoof canker is related to disturbances in differentiation of the keratinocytes rather than a proliferative dysfunction.
The results of the present study suggest that hoof canker is not a hyperproliferative process of the dermal papillary body shown by immunohistochemical anti-Ki67 detection; rather, increased levels of PCNA protein expression and observed morphological alterations indicate that dysregulation of keratinocyte differentiation constitutes a key event in the development of equine hoof canker.
Footnotes
Acknowledgements
The authors thank Claudia Höchsmann and Stefanie Burger for establishing the immunohistochemical staining protocols and Birgit Machac and Anne Flemming for assisting in production of the histological sections.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article. The study was funded by the intra-university research funding program ‘Profillinien’ of the University of Veterinary Medicine, Vienna, Austria, Profile line 2 – Infection and Prevention.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
