Abstract
Molecular clonality analysis of T-cell receptor (TCR) genes for diagnosing T-cell lymphoma is widely used in veterinary medicine. However, differentiating chronic enteritis (CE) from intestinal lymphoma is challenging because of the incompatibility between histopathologic and clonality analysis results. On the basis of findings that canine intestinal T-cell lymphoma and celiac disease share some common features, we conducted serologic examinations in combination with histopathologic and T-cell receptor clonality analyses in 48 dogs diagnosed with either CE or intestinal lymphoma. Immunoglobulin A (IgA) and immunoglobulin G (IgG) antibodies against gliadin and tissue transglutaminase (tTG) were quantitatively measured using ELISA. The conditions were classified according to the histopathologic diagnosis, clonality analysis, and combined histopathologic/clonality analysis. Histopathologic analysis showed that dogs with intestinal lymphoma were likely to have high levels of serum IgA antibodies against gliadin and tTG, and serum IgG antibodies against tTG. No correlation between the diagnosed groups and control group was observed in the results of the clonality analysis and histopathologic/clonality analysis. It is interesting that dogs with intestinal lymphoma had a higher serum IgA titer against gliadin and tTG than did dogs with CE. These results suggest an association between repetitive inflammatory stimulation by gliadin peptides and subsequent intestinal lymphoma in dogs.
Keywords
Chronic enteritis (CE) and intestinal lymphoma (IL) are common gastrointestinal disorders in dogs that often result in protein-losing enteropathy (PLE). The etiology of these diseases is largely unknown and likely to be multifactorial. Differentiating CE and IL based solely on histopathologic analysis can be challenging for veterinary pathologists. 11 Compared to full-thickness biopsies, endoscopic biopsies are less invasive and are thus commonly used in the diagnosis of CE and IL. 37 However, lesions may be overlooked because of limited areas of sample collection (eg, sample collection only in the duodenum and distal ileum) and inability to obtain tissues below the muscularis mucosae. 19 The introduction of polymerase chain reaction (PCR) for detecting antigen receptor gene rearrangement (PARR) was expected to aid the accurate detection of IL. 7 However, the specificity of PARR is questionable because of the discrepancy between its results and the actual histopathologic diagnosis and clinical outcome. One study found a comparatively low sensitivity (66.7%) of PARR in detecting IL, and another study showed that clonal lymphocytes were frequently detected in dogs with inflammatory bowel disease. 13,26 Although the mechanism remains unclear, transformation of CE to IL in dogs has been suggested in several studies. 10,14 On the basis of these findings, it can be presumed that canine intestinal lymphomagenesis is a continuous process from inflammatory conditions that makes differentiation between CE and IL difficult.
Celiac disease (CD) is an autoimmune inflammatory enteropathy in humans that results from the intake of gliadin and other related prolamins in genetically predisposed individuals. 32 Immunogenic peptides, which are produced via the deamidation of gliadin peptides by tissue transglutaminase (tTG), are presented by antigen-presenting cells, including dendritic cells that express HLA-DQ2 and DQ8 molecules, to CD4 T-cells. 4,18 Celiac disease is histologically characterized not only by the atrophy of the intestinal mucosa, reduced height of villi, increased number of intraepithelial lymphocytes (IELs), and inflammatory cell infiltration in the lamina propria but also by changes in enterocyte appearance including cuboidal morphology and cytoplasmic vacuoles. 25 Intestinal T-cell lymphoma is known to be a complication of CD.
Irish setter dogs are predisposed to gluten-sensitive enteropathy exhibiting gluten-dependent diarrhea and histopathologic changes. 2,3,16 However, previous studies failed to demonstrate the presence of antigliadin and anti-tTG antibodies as well as MHC class II linkage with disease in these animals. 28,29 One study described antigliadin and anti-tTG immunoglobulin G (IgG) in the sera of dogs with CE; however, the association between canine CE and human CD is largely unknown. 36
We therefore hypothesized that CE develops into IL due to continuous antigenic stimulation by dietary proteins and subsequent autoimmune pathogenic reactions. This study was designed to investigate the role of gliadin peptides and a probable autoimmune involvement in canine CE and IL by serologic evaluation of antibody against gliadin and tTG combined with histologic and clonality analyses.
Materials and Methods
Cases and Sample Collection
All endoscopic intestinal tissue samples and sera were collected from dogs that presented with alimentary signs such as vomiting, diarrhea, and low serum albumin levels at the Japan Small Animal Medical Center (JSAMC) between 2014 and 2015. All dogs had a chronic history of gastrointestinal signs for more than 3 weeks or a sign of PLE with low serum albumin levels below 2.7 g/dl. Complete blood count, serum biochemistry, fecal examination, urinalysis, and abdominal ultrasonography were performed to rule out infectious causes, pancreatic insufficiency, and other causes of hypoalbuminemia such as hepatic and renal diseases. Histopathologic diagnosis and clonality analysis were performed at the Laboratories of Veterinary Pathology and Veterinary Internal Medicine, respectively, at the Graduate School of Agricultural and Life Sciences, The University of Tokyo. Forty-eight dogs diagnosed with either CE (n = 30) or IL (n = 18) were selected for this study. Of the 48 dogs, sera were not available for 2 dogs (dog Nos. 33 and 34); thus, they were excluded from the ELISA and western blot (WB) analyses. Immunofluorescence histochemical analysis was performed using sera from representative cases. The dog breeds included miniature Dachshund (n = 7), Chihuahua (n = 6), Toy Poodle (n = 5), Pomeranian (n = 4), mixed breed (n = 4), Maltese (n = 3), Shiba Inu (n = 5), Boston Terrier (n = 2), French Bulldog (n = 2), Welsh Corgi (n = 2), American Cocker Spaniel (n = 1), Beagle (n = 1), Border Collie (n = 1), English Setter (n = 1), Great Pyrenees (n = 1), Shetland Sheepdog (n = 1), Weimaraner (n = 1), and Yorkshire Terrier (n = 1). There were 19 females (spayed, 15) and 29 males (castrated, 16), with a mean age of 7 years 5 months. The majority of the dogs in this study had already been introduced to a gluten-free or lowfat diet at the time of endoscopic examination. The details of the dogs are summarized in Supplemental Table S1.
Histopathologic and Immunohistochemical Analyses
Mucosal samples of 46 out of the 48 dogs were obtained from the stomach, proximal duodenum, distal duodenum, ileum, and colon. A minimum of 6 tissue samples was collected per region, and each tissue sample included at least 3 villi that were considered suitable for evaluation. 38 The collected tissue samples were fixed in 10% buffered formalin and processed for routine histopathologic analysis after paraffin embedding. Deparaffinized sections (thickness, 4 µm) were rehydrated through graded alcohols and stained with hematoxylin and eosin (HE). Immunohistochemical analyses using CD3 (DAKO-Japan, Tokyo, Japan) and CD79α (Dako-Japan) were done with procedures described in a previous study with minor modifications. 27 The details of the antibodies were summarized in Supplemental Table S2. All intestinal biopsies were evaluated by the same pathologist. Diagnostic criteria for histopathologic evaluation were set on the basis of the histopathologic standards for the intestines established by the World Small Animal Veterinary Association Gastrointestinal Standardization Group. 11,24,34 Diagnosis of IL was performed on the basis of the World Health Organization classification. 34 Intestinal lymphomas were further subclassified into small cell lymphoma (SCL) or large cell lymphoma (LCL) depending on nuclear sizes. 21 A cell nuclear size less than 2 red blood cells in diameter was defined as SCL, whereas those of greater than 2 red blood cells were considered LCL. Lesions with more than 40 IELs per 100 villous enterocytes were defined as epitheliotropic.
PCR for Antigen Receptor Gene Rearrangement
Tissue samples were endoscopically collected from the duodenum and ileum of 45 out of the 48 dogs and stored at –20°C until use. Clonality analysis was not performed in 3 dogs (dog Nos. 5, 41, and 42). The 45 dogs included in this analysis were diagnosed with either CE (n = 29) or IL (n = 16). Genomic DNA was extracted from frozen sample tissues by using the QIAamp DNA Blood Mini Kit (Qiagen, Valencia, CA, USA), according to the manufacturer’s instructions. Rearrangements of the IgH/TCRγ genes were analyzed by amplifying CDR3 to determine lymphocyte clonality using primers designed for the V and J segments. 15 Two genes (IgM C region and HGF) were also amplified as positive controls. All of the forward primers were labeled with fluorescent dyes at the 5′ end. Polymerase chain reactions were carried out in a final volume of 25 μL of the PCR mixture composed of 1× Amplitaq Gold 360 Master Mix (Applied Biosystems, Foster City, CA, USA), forward primer (200 nM), reverse primer (200 nM), and 100 ng of DNA template. The following thermal profile was used: an initial denaturation and enzyme activation was performed at 95°C for 5 minutes; this was followed by 40 cycles of denaturation at 94°C for 15 seconds, annealing at 62°C or 56°C for 30 seconds, and extension at 72°C for 30 seconds; and a final extension was performed at 72°C for 30 minutes. All PCRs were conducted in duplicates. Then, GeneScan analysis was performed on the ABI 3130xl Genetic Analyzer (Applied Biosystems) as a multiplex run. One microliter of diluted PCR product mixture (1:40) was placed together with 8.5 μl of Hi-Di formamide (Applied Biosystems) and 0.5 μl of 600 LIZ size standard (Applied Biosystems) onto an optical 96-well plate. Prior to the analysis, the samples were denatured at 95°C for 5 minutes and then cooled on ice for 15 minutes. Data were analyzed using the Peak scanner software (Applied Biosystems). A clonal T-cell population was defined by the presence of distinct peaks of fluorescence intensity at least 2-fold higher than other background peaks. Monoclonal or biclonal peaks were considered positive results.
ELISA Testing for Serum IgG and IgA
Canine sera had been collected at the time of presentation at JSAMC and were stored at –20°C until use. Sera were not available for 2 of the 48 dogs (dog Nos. 33 and 34); they were excluded from ELISA and WB studies. Sera collected from 18 clinically healthy dogs were used as negative controls. IgG and immunoglobulin A (IgA) antibodies to gliadin and tTG were quantitatively measured by an ELISA technique. Gliadin from wheat (Sigma-Aldrich, St Louis, MO, USA) was mixed with 70% ethanol, and then the mixture was centrifuged at 3000 × g at 4°C for 30 minutes. The supernatant was retrieved to a fresh tube and protein concentration was determined using the Lowry method. Gliadin (0.25 μg/well) and human recombinant tTG (0.3 μg/well; Immundiagnostik AG, Bensheim, Germany) were immobilized on immunoassay plates (Thermo Scientific, Tokyo, Japan) at 4°C overnight. The plates were then washed 3 times with 0.02% phosphate buffered saline supplemented with Tween 20 (PBST) and blocked (Pierce Protein-Free [PBS] Blocking Buffer; Thermo Scientific) at room temperature for 2 hours. Canine sera were then diluted 1:100 with 0.2% PBST and applied to duplicate wells, followed by the reaction with HRP-conjugated goat polyclonal antidog IgG (Bethyl, Montgomery, TX, USA) or IgA (Bethyl). After 3 washes with PBST, the plates were incubated with 2,2′-azinobis 3-ethylbenzothiazolin-6-sulfonic acid (Sigma-Aldrich). The optical density was measured at a wavelength of 405 nm with a microplate reader (iMark™; Bio-Rad, Tokyo, Japan). All dogs were screened for IgA deficiency by using the Dog IgA ELISA Quantitation Set (Bethyl). Total serum IgA was measured as recommended by the manufacturer except for the blocking solution, washing buffer, and antibody diluents described above. The optical density was measured at a wavelength of 450 nm.
Statistical Analysis
ELISA data were analyzed using SPSS software (IBM Corporation, Armonk, NY, USA). Differences in IgG and IgA values between the control and experimental groups were considered statistically significant at P < .05.
Western Blot Analysis for Antigliadin Antibody in Canine Sera
Western blot (WB) analysis was performed to confirm the presence of antigliadin IgG and IgA in canine sera. Gliadin-2% SDS mixture (1 μg/lane) was loaded on a 10% SDS-PAGE gel. Following electrophoresis at 600 V and 20 mA/gel for 80 minutes, gels were blotted on nitrocellulose membranes and the membranes were washed with 1% TBST for 5 minutes, followed by blocking with 5% skimmed milk in TBST at room temperature for 1 hour with gentle agitation. The sera diluted with TBST (1:5000) were incubated with the membranes at 4°C overnight, washed 3 times for 10 minutes each in TBST, and then incubated with HRP-conjugated goat anticanine IgA (1:5000) (Bethyl) with gentle agitation at room temperature for 1 hour. Following 3 washes in TBST for 10 minutes each, the membranes were incubated with the Amersham ECL Prime Western Blotting Detection reagent at room temperature for 5 minutes. Image analysis was conducted using the ChemiDoc XRS plus (Bio-Rad) with an exposure time of 90 seconds.
Double Immunofluorescence Microscopy for Anti-tTG IgA Antibody
An immunofluorescence study was performed to qualitatively demonstrate anti-tTG IgA antibodies in canine sera. Since a threshold of antibody titer was not clearly defined, the experiment was done with representative cases. Fresh rat esophagus was frozen in the Tissue-Tek compound (Sakura-Finetek, Tokyo, Japan) and cut into 8-μm-thick slices by using a cryostat microtome. The sections were air-dried at room temperature for 3 hours, rehydrated in DW, and blocked with 8% skimmed milk in TBST at 37°C for 60 minutes. The sections were incubated at 4°C overnight, with representative canine sera. After washing with TBST, goat anticanine IgA (Bethyl) and mouse antitransglutaminase 2 antibody (Abcam, Cambridge, UK) were applied on the sections and incubated at 37°C for 1 hour. After 3 washes with TBST, the sections were incubated with Alexa 488-conjugated donkey antigoat IgG (1:200; Invitrogen, Eugene, OR, USA) and Texas red-conjugated horse antimouse IgG (1:200; Vector Laboratories, Burlingame, CA, USA) at room temperature for 1 hour. The colocalization of canine IgA and tTG binding pattern was evaluated on the muscular layer of the rat esophagus by using a laser scanning confocal microscope (LSM700; Zeiss, Tokyo, Japan).
Results
Histopathologic Analysis
Histopathologic evaluation of the 48 mucosal specimens revealed CE in 30 (Supplemental Table S1). Chronic enteritis lesions were characterized by an increased number of lymphocytes and plasma cells, predominantly in the lamina propria with various degrees of destruction of histologic architecture (Figs. 1, 2, 3a).
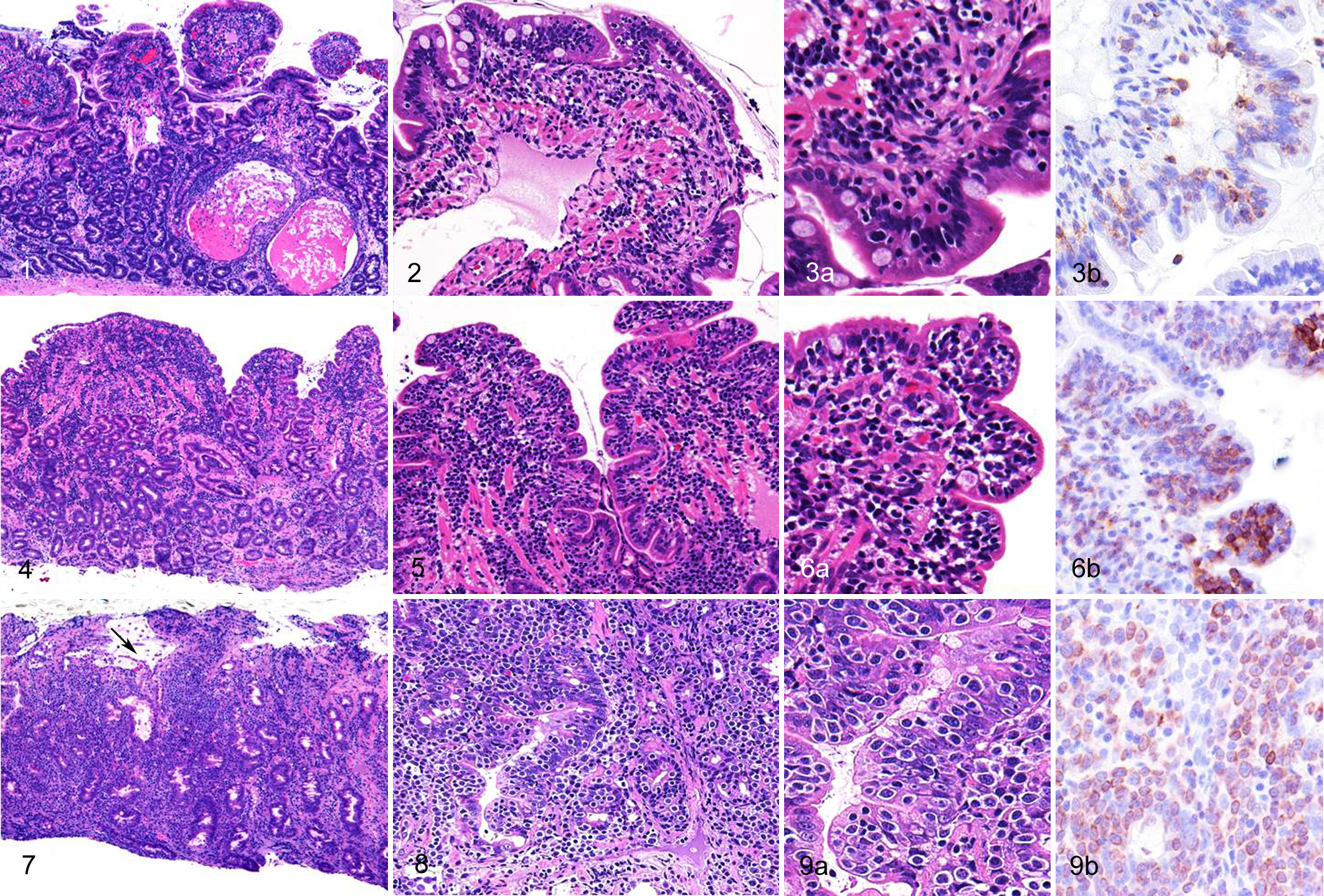
Eighteen cases of IL were histopathologically subclassified into 11 cases of SCL and 7 cases of LCL (Supplemental Table S1). The duodenum and ileum were the intestinal regions most commonly affected by lymphoma. The lesions were also found in the stomach, colon, or rectum in a few dogs.
Lesions of lymphoma were characterized by diffuse proliferation of neoplastic lymphocytes in the lamina propria with various degrees of invasion to the epithelial layer (Figs. 4, 5, 6a). Small nests of IELs within the epithelial layer were frequently observed. Neoplastic cells of SCL were characterized by small, round, and monomorphic cells with a round and monomorphic nucleus containing dense chromatin (Fig. 6a). In contrast, LCL were characterized by moderate-to-large pleomorphic cells with a round-to-ovoid nucleus containing scattered or dense chromatin and distinct nucleoli (Figs. 7–9). The neoplastic cells had slightly eosinophilic pale granules in the cytoplasm in 3 dogs with LCL. Immunohistochemically, all neoplastic cells showed a T-cell phenotype (CD3+, CD79α–) (Figs. 3b, 6b, 9b).
An increase in the number of IELs was observed in 6 of 30 dogs with CE, 11 of 11 dogs with SCL, and 6 of 7 dogs with LCL. The lesions were found as clusters or diffuse infiltrates of cells in the epithelial layer. Marked villous atrophy was observed in 8 dogs with CE, 7 with SCL, and 5 with LCL. Moderate-to-marked lacteal dilation was evident in 18 dogs with CE, 7 with SCL, and 2 with LCL. Crypt distention was observed in 17 dogs with CE, 4 with SCL, and 4 with LCL. Changes in enterocyte appearance, including cuboidal morphology and cytoplasmic vacuoles, were evident in 11 dogs with CE, 10 with SCL, and 7 with LCL. Increased fibrous stroma was observed in 23 dogs with CE, 9 with SCL, and 4 with LCL. Infiltration of eosinophils was prominent in 2 dogs with CE. Histopathologic evaluation of the duodenum and ileum was not available in 1 dog with LCL.
PARR
A summary of clonality analysis is shown in Table 1. Clonal TCR gene rearrangements were demonstrated in 21 (72%) of 29 dogs with CE, 9 (90%) of 10 dogs with SCL, and 5 (83%) of 6 dogs with LCL. Biclonal rearrangements (TCR and IgH) were detected in 3 (10%) of 29 dogs with CE, 3 (30%) of 10 dogs with SCL, and 1 (17%) of 6 dogs with LCL.
Histopathologic Diagnosis and Clonality Analysis in Dogs With Chronic Enteritis or Intestinal Lymphoma.a
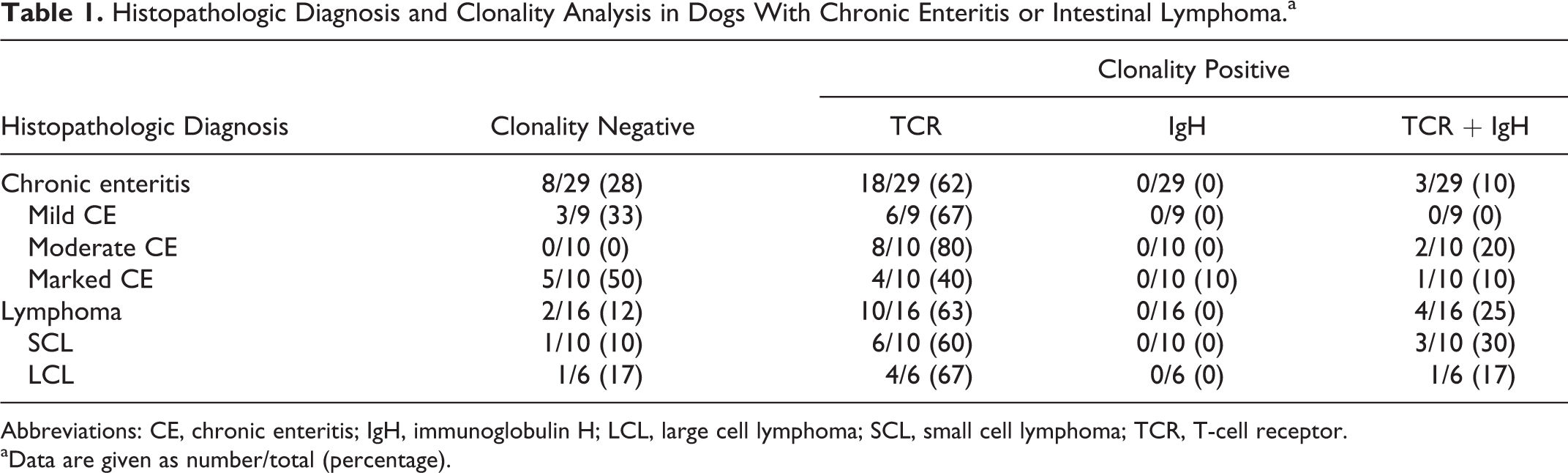
Abbreviations: CE, chronic enteritis; IgH, immunoglobulin H; LCL, large cell lymphoma; SCL, small cell lymphoma; TCR, T-cell receptor.
aData are given as number/total (percentage).
ELISA
Levels of IgA antibodies against gliadin and tTG in the serum were higher in CE and IL than in control dogs (Fig. 10). Two dogs with SCL (dog Nos. 35 and 37) showed an increase in antigliadin IgA and anti-tTG IgA levels (Figs. 10a, 10b). It is interesting that 4 CE dogs (dog Nos. 17, 18, 21, and 29) high in antigliadin IgA levels did not show higher anti-tTG IgA levels.
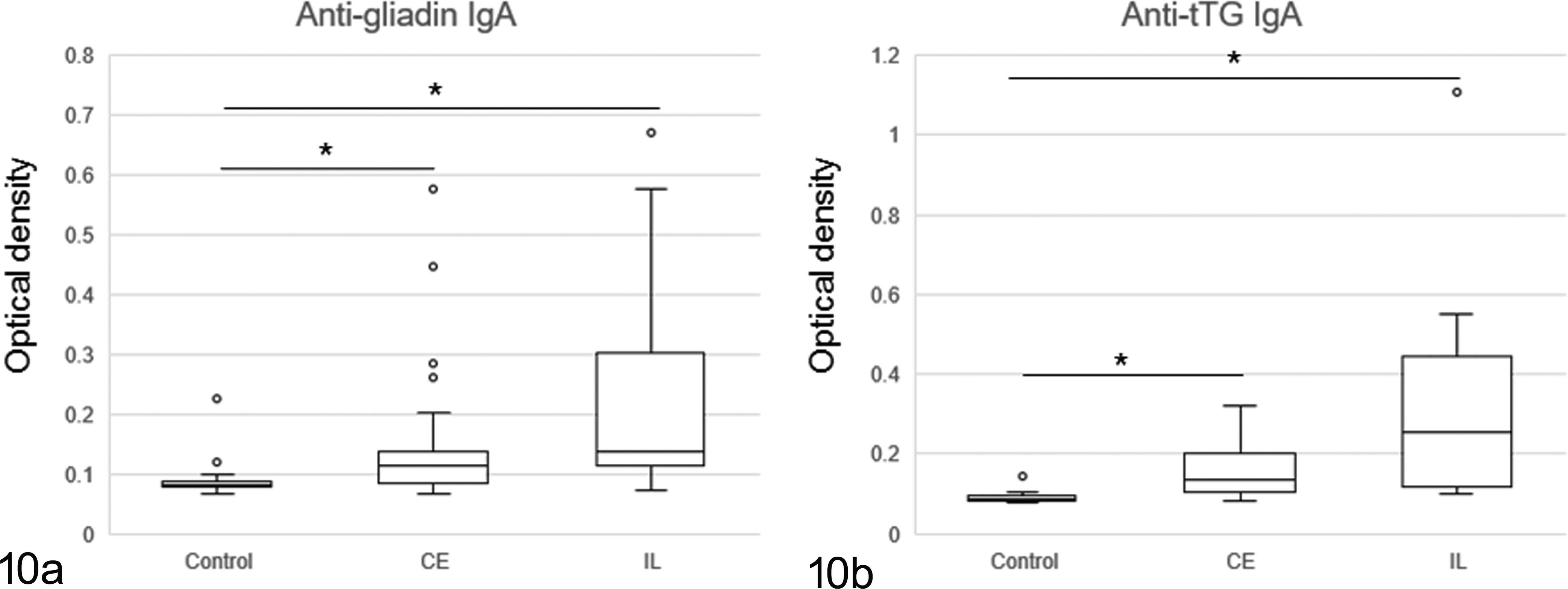
The levels of antigliadin (a) and anti-tTG (b) immunoglobulin A (IgA) antibodies in dogs with intestinal lymphoma (IL) and chronic enteritis (CE), and the control dogs. Levels of antigliadin IgA and anti-tTG IgA are higher in dogs with IL and CE than in controls. *P < .05.
There was no significant association in levels of antigliadin IgG between tested and control groups. In contrast, levels of IgG antibodies to tTG were higher in IL than in control dogs (Figs. 11a, 11b). Review of those cases that had high titers of antibodies to gliadin and tTG did not reveal unique or consistent clinical and histologic features (Supplemental Figs. S1–S3). Moreover, there was no significant difference in the levels of antigliadin and anti-tTG IgA between the clonality-positive and clonality-negative groups (Supplemental Fig. S4). The levels of antigliadin IgA were higher in IL dogs showing a negative clonal TCR gene rearrangement than in the other 3 groups (Supplemental Fig. S5a). No significant differences were evident between the 4 groups with respect to the levels of anti-tTG IgA (Supplemental Fig. S5b). Serum IgA levels were higher in IL than in CE and control dogs (Fig. 12).
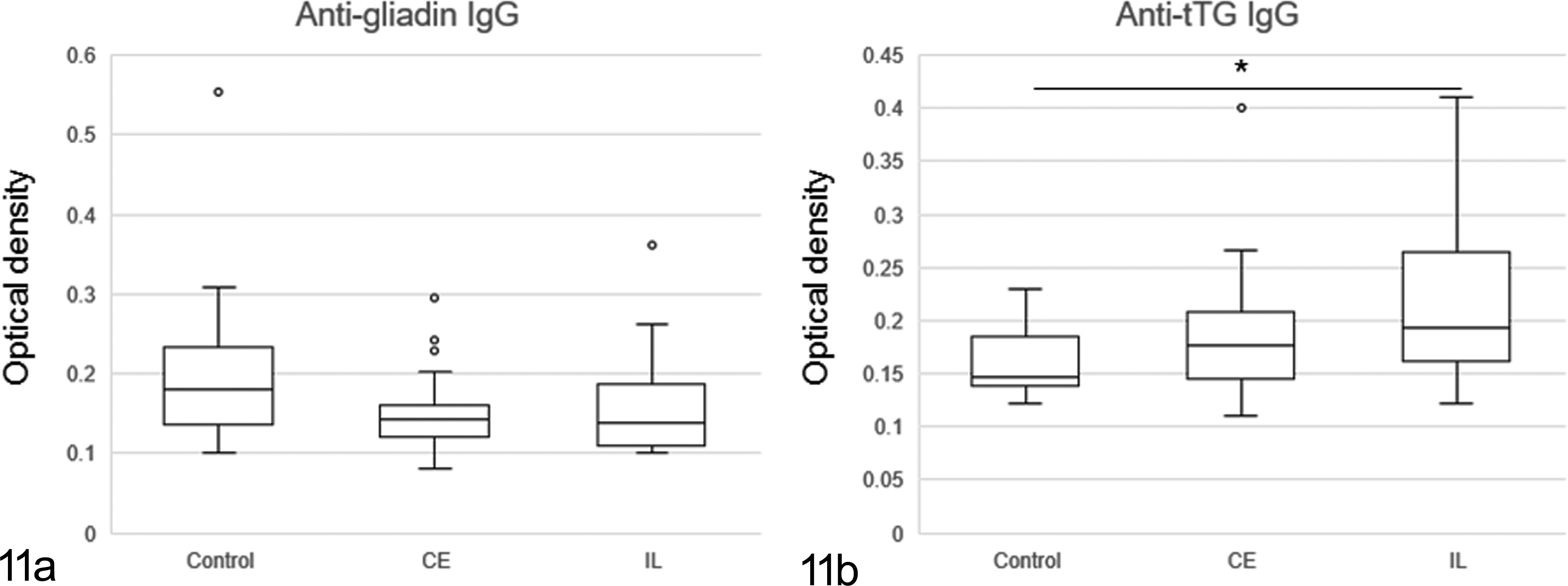
The levels of antigliadin (a) and anti-tTG (b) immunoglobulin G (IgG) antibodies in dogs with lymphoma and chronic enteritis, and the control dogs. There is a significant difference between control dogs and dogs with intestinal lymphoma in levels of anti-tTG IgG. *P < .05.
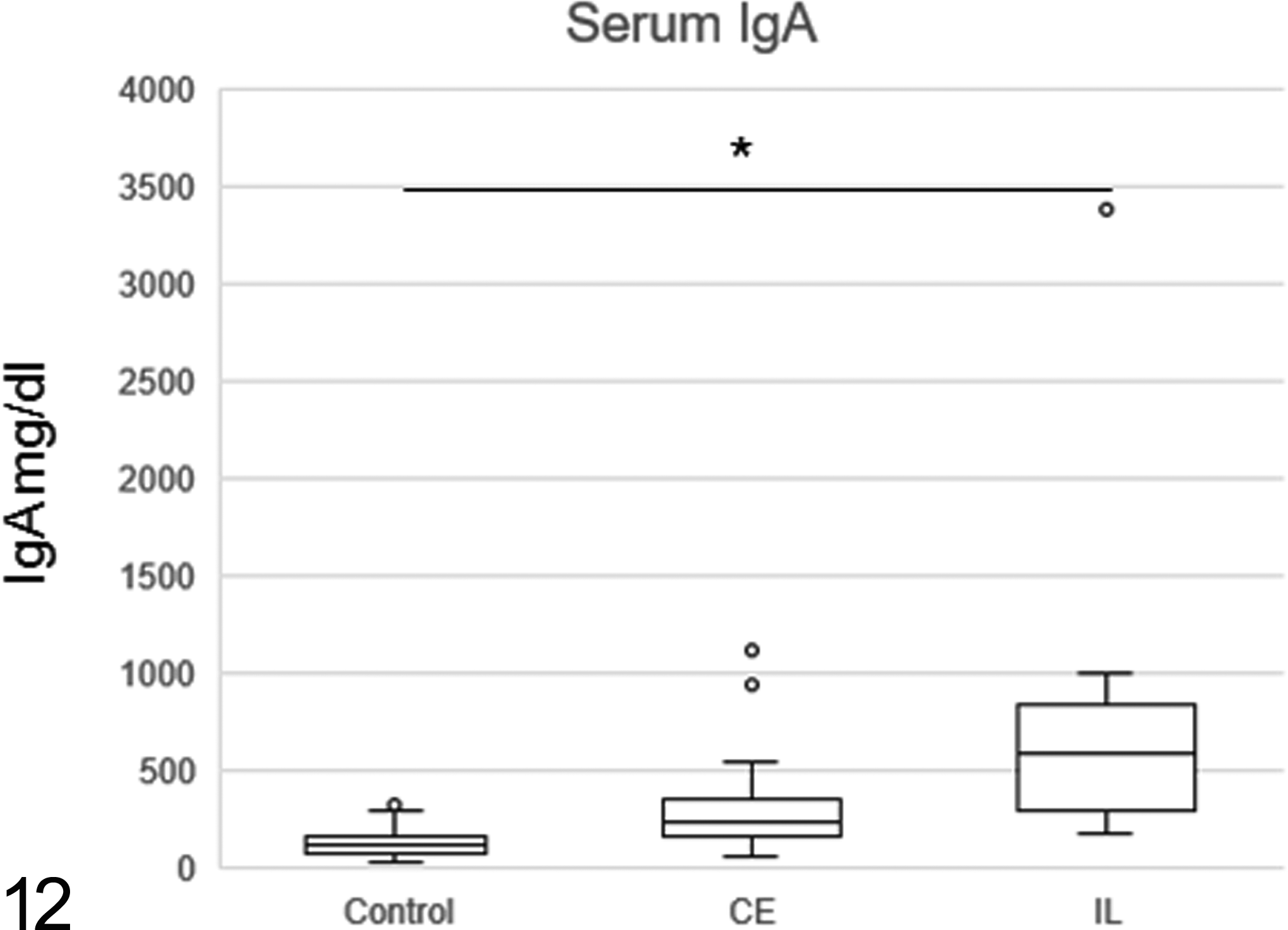
The levels of total serum immunoglobulin A (IgA). Level of total serum IgA was higher in intestinal lymphoma than in control dogs. *P < .05.
WB Analysis
The results of antigliadin IgA detection by WB are summarized in Table 2. Antigliadin IgA was detected in 12 (40%) of 30 dogs with CE and in 8 (50%) of 16 dogs with lymphoma. Serum samples from dogs with lymphoma tended to include antibodies binding to larger molecular weights of gliadin, whereas samples from dogs with CE tended to include antibodies binding to smaller gliadin fractions (Fig. 13). One of the 18 control dogs tested positive for antigliadin IgA.
Western Blot Banding Patterns in Chronic Enteritis, Intestinal Lymphoma, and Control Dogs.
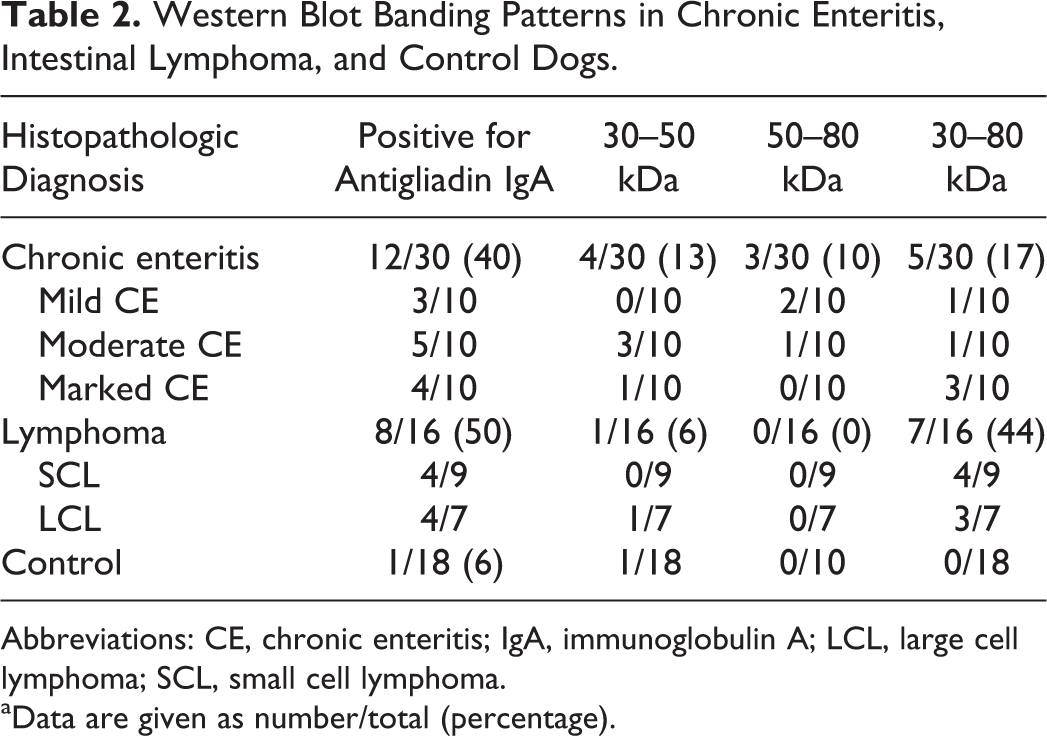
Abbreviations: CE, chronic enteritis; IgA, immunoglobulin A; LCL, large cell lymphoma; SCL, small cell lymphoma.
aData are given as number/total (percentage).
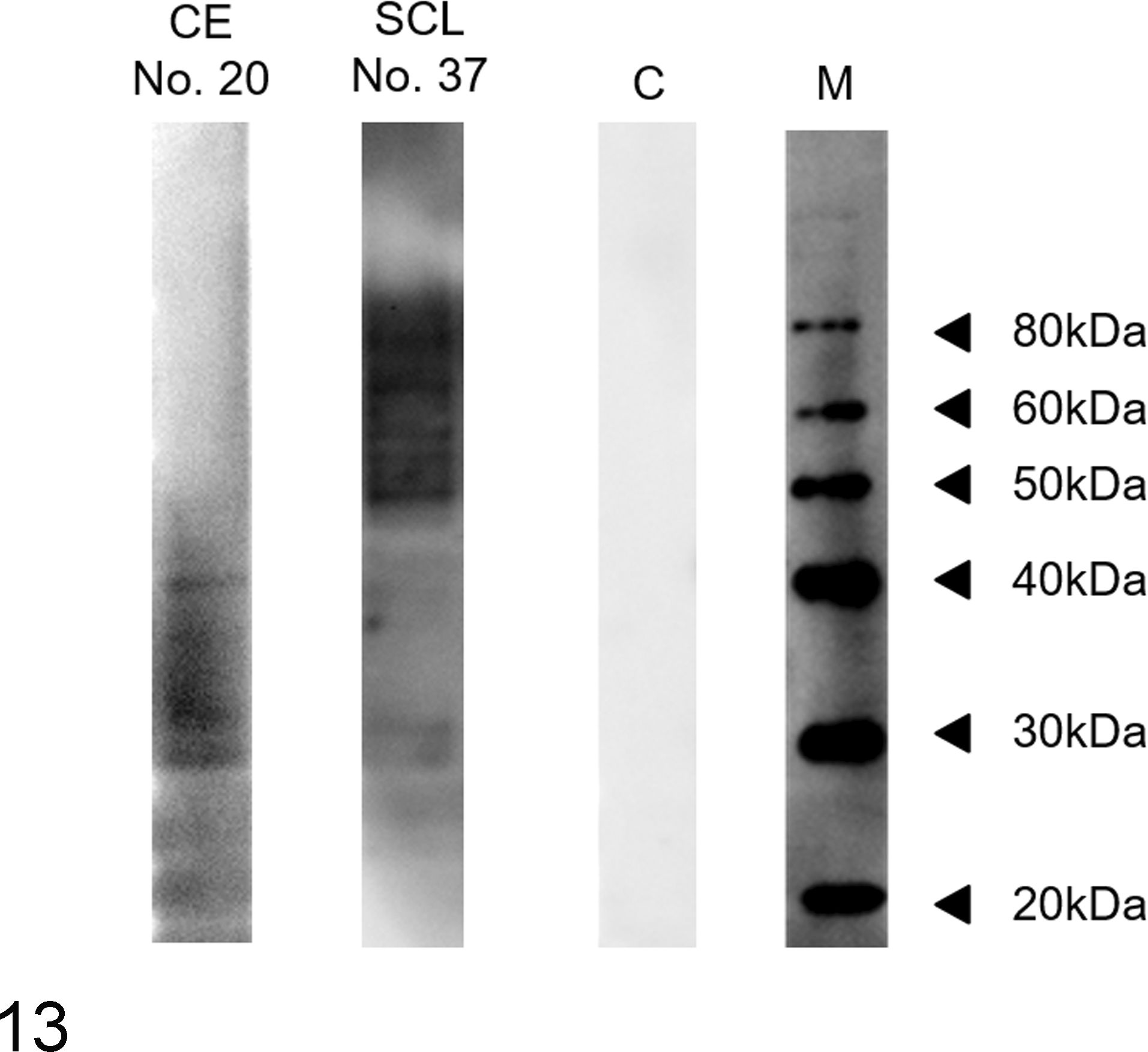
Western blot analysis. Dogs with moderate chronic enteritis (CE) (dog No. 20), small cell lymphoma (SCL) (dog No. 37), and a control (C). Compared to serum from a dog with moderate CE (dog No. 20), the serum from a dog with SCL (dog No. 37) binds to a larger set of gliadin fractions between 30 and 80 kDa. M, molecular weight markers.
Double Immunofluorescence Microscopy
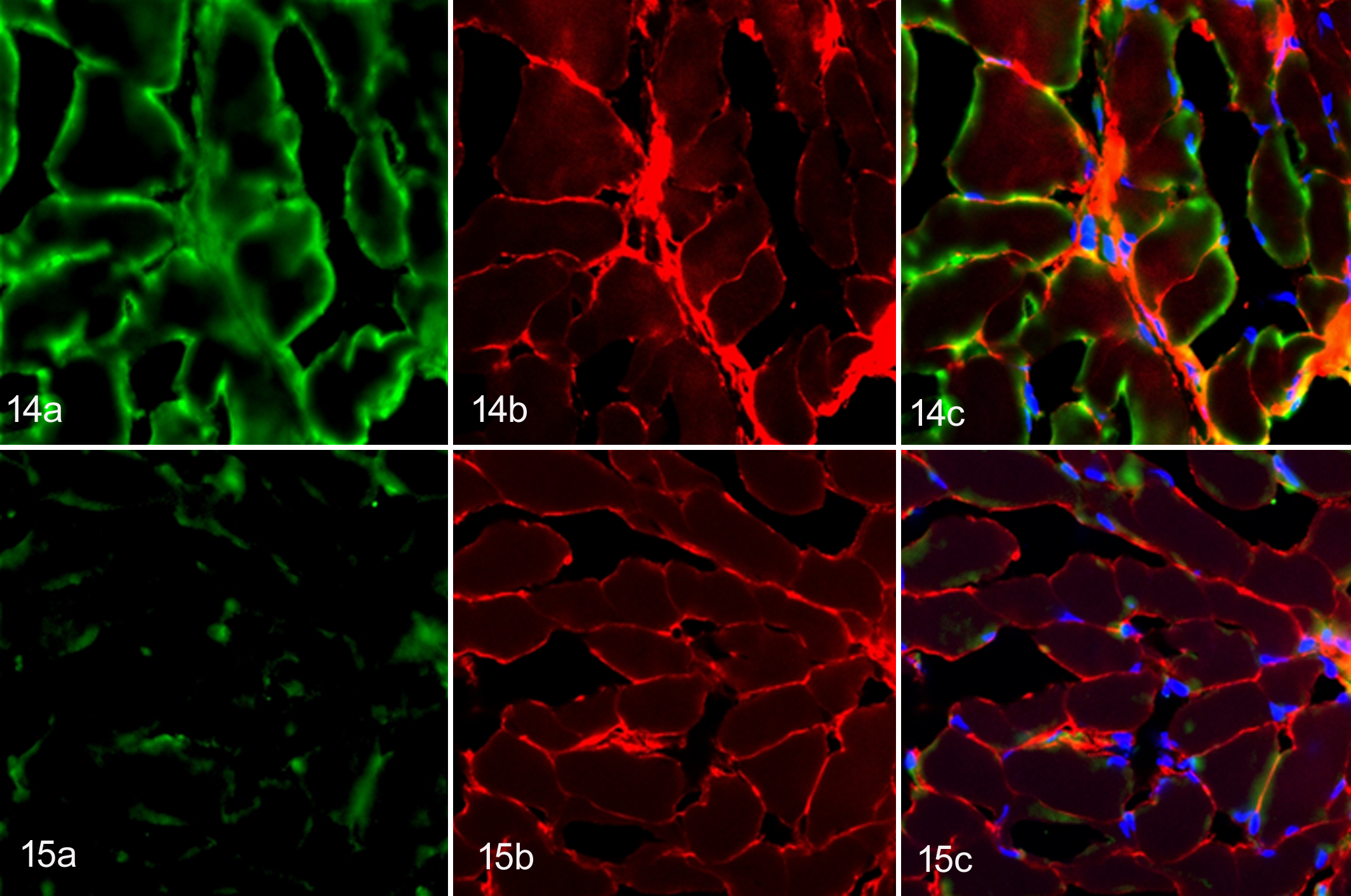
Representative canine sera, which included high levels of anti-tTG IgA, bound to the endomysium of the rat esophagus (Fig. 14a), whereas no distinctive binding was noted when control sera were used (Fig. 15a). The endomysium was also immunostained with mouse anti-tTG antibody (Figs. 14b, 15b). The serum from dog No. 37 showed a reticular binding pattern of positive canine serum colocalizing with tTG (Fig. 14c). In contrast, there was no colocalization between the control sera and mouse tTG antibodies (Fig. 15c). These results supported the ELISA findings.
Discussion
Despite the wide availability of clonality analysis for diagnosing lymphoma, the diagnosis of IL by using endoscopically collected samples can still be challenging in the field of veterinary medicine. Lymphoma can be detected on the basis of a monomorphic lymphocytic proliferation. However, positive clonality results are commonly obtained even in inflammatory lesions because a clonal population of lymphocytes can be seen in IL and in some cases of CE. Some populations of canine lymphoma are expected to develop from CE in response to repetitive stimulation with dietary protein antigens. This study was conducted to identify the role of gliadin peptides and a possible autoimmune involvement in canine CE and IL by serologic evaluation combined with histopathologic evaluation and clonality analysis of small intestinal biopsy samples.
The present study confirmed the previous finding of an increased number of IELs in dogs with intestinal T-cell lymphoma. 8 Lymphocytes with epitheliotropic behavior were more commonly seen in small cell lymphoma but also seen in large cell lymphoma as described in a previous study. 27 Large cell lymphoma was often accompanied by a change in enterocyte appearance, including cuboidal morphology and cytoplasmic vacuoles. Canine intestinal T-cell lymphomas observed in this study were morphologically classified into SCL and LCL. Small cell lymphoma was characterized by the proliferation of small, round, and monomorphic lymphocytes with scant, pale cytoplasm. In contrast, LCL was characterized by moderate-to-large pleomorphic cells with a round-to-ovoid nucleus containing scattered or dense chromatin. Proliferation of monotonous, intermediate-size lymphocytes described in human enteropathy-associated T-cell lymphoma (EATL) type II was rarely observed in dogs. On the basis of these findings, further investigations are required to determine whether EATL classification criteria are applicable to canine intestinal lymphomas.
Clonality analysis revealed that 72% (21/29) of CE and 88% (14/16) of IL showed either monoclonal or biclonal proliferation of lymphocytes. Such a high rate of positive clonality in dogs with CE may be associated with the small amount of DNA isolated from biopsy samples, which resulted in the detection of only a dominant clone. 39 Alternatively, the detection of monoclonal gene rearrangements in CE can be explained by the chronic inflammation elicited by stimulants other than gluten peptides. 1,5 The negative results of TCR and IgH assays in 2 dogs with high levels of antigliadin and anti-tTG IgA (dog Nos. 35 and 37) can be attributed to inconsistencies resulting from differences in biopsy sampling sites between PARR and histopathologic evaluation, or the limited coverage of the primers used in this study. 17
We found that the levels of antigliadin IgA and anti-tTG IgA were higher in dogs with lymphoma and CE than in control dogs. A subset of dogs with increased antigliadin IgA may share a common mechanism by which gluten peptides induce inflammatory responses. It is interesting to note that 2 SCL dogs (dog Nos. 35 and 37) showed significant increases in both antigliadin IgA and anti-tTG IgA levels, whereas CE dogs (dog Nos. 17, 18, 21, and 29) with higher anti-IgA levels showed variable degrees of anti-tTG IgA. This might be explained by an alteration in anti-tTG IgA levels resulting from a gluten-free diet (GFD), because a group of dogs had already been introduced to the gluten-free prescription diet by the time of presentation. In human patients with CD, the levels of anti-tTG antibodies are known to decrease rapidly following the introduction of a GFD, whereas histologic changes such as increased IEL and villous atrophy remain persistent. 33 Alternatively, the variation in levels of anti-tTG IgA in a group of those with CE may suggest that CE and IL are potentially heterogeneous diseases. However, the fact that a clonal population of lymphocytes was equally present in CE and IL and that there were no statistical differences in antigliadin IgA and anti-tTG IgA between CE and IL suggests a continuum between CE and IL.
Impaired tissue integrity of the intestinal barrier may have resulted in increased intestinal permeability and a subsequent increase in IgA in tested groups. However, there was no statistical significance in serum antiovomucoid IgA levels between controls and tested groups (Supplemental Fig. S5). The levels of antigliadin and anti-tTG IgA were associated with neither histologic changes in the mucous membrane nor the canine IBD activity index (CIBDAI). These findings reinforce our idea that gliadin peptides are primarily involved in a pathogenic process rather than the result of mucosal damage.
A previous study demonstrated no significant association in serum IgA levels between CE, IL, and control dogs. 22 However, our study has shown elevated serum IgA levels in IL, possibly due to chronic inflammation associated with antigliadin and anti-tTG production. Taken together, these findings suggest that elevated serum IgA levels may be involved in tumorigenesis in intestinal T-cell lymphoma in only a subset of dogs.
Antigliadin IgG, anti-tTG IgG, or antiendomysial IgG can alternatively be used in human patients with CD and IgA deficiency. 9,20,35 A previous study showed the presence of antigliadin IgG and anti-tTG IgG in dogs with CE. 36 However, the significance of IgG antibodies in dogs with CE and IL was questionable in this study. In addition, the correlation between histopathologic diagnosis and antigliadin IgG was inconsistent. In a previous study, elevated titers of antigliadin IgG were observed in rabbits fed gluten as a part of their regular diet. 23 High levels of antigliadin IgG may have nonspecifically resulted from regular gluten consumption and are not necessarily indicative of a pathogenic state. 23 Although we found that the levels of anti-tTG IgG were significantly higher in dogs with IL than in the controls, elevated anti-tTG antibody should be interpreted carefully because it is known to be increased in non-CD enteropathies in humans. 12
Different types of gluten peptides have been demonstrated to induce the production of cytokines and subsequent proliferation of T-cells in the intestinal mucosa. 6 Antigliadin IgA was detected in 40% (12/30) of dogs with CE and 50% (8/16) of dogs with IL in the present study. Our WB results revealed that IgA antibody binding patterns distributed in the molecular range of 50 to 80 kDa of gliadin fractions in dogs with mild or moderate CE and in the range of 30 to 80 kDa in dogs with marked CE or IL. No bands were detected in control sera except for 1 case. The difference in the detected molecular fractions between dogs with CE and IL in the present study may suggest the different pathologic roles of gliadin fractions in these 2 pathologic conditions. 31 A previous study demonstrated preferential reactivity toward different gluten peptides in patients with schizophrenia and CD. 31 A difference in antigenic specificity suggests different antigliadin immune responses in patients with schizophrenia and CD. 31
The results of the double immunofluorescence assay also revealed anti-tTG IgA in sera from representative dogs (dog Nos. 35 and 37), whereas no anti-tTG IgA was detected in the controls. A reticular binding pattern was observed in the canine sera colocalizing with mouse anti-tTG antibodies on the rat esophagus. The endomysial antibody primarily targets the tTG, and a significant correlation between anti-tTG and endomysial antibody has been established in CD. 30 Although an exact role of anti-tTG IgA production is not clearly understood, the presence of tTG IgA together with gliadin IgA in a subset of IL cases may indicate a possible involvement of an autoimmune process in lymphomagenesis of canine intestinal lymphomas.
In conclusion, the present study demonstrated the potential pathogenic roles of IgA antibodies against gliadin and tTG in canine CE and IL. Although altered integrity of the mucous membrane may have contributed to increased IgA levels, the changes in IgA levels are poorly associated with histologic changes of the mucous membrane or clinical conditions. IgA to another dietary protein failed to demonstrate statistical significance. For these reasons, it can be presumed that elevated antigliadin IgA are more likely to be associated with transformation of CE to IL in some individuals. The variable levels of anti-tTG IgA in a group of those with CE may indicate that CE and IL are potentially heterogeneous diseases. However, it is difficult to conclude that this is due to an insufficient sample size. Considering the fact that a clonal population of lymphocytes was frequently present not only in IL but also in CE, it may be more reasonable to consider these 2 diseases, CE and IL, as a continuum. Additional cases and follow-up studies of dogs with CE would be required for understanding the pathogenesis in those individuals with IgA changes. These future studies may eventually provide supportive information to characterize the subcategory of CE cases at risk of developing intestinal T-cell lymphoma.
Footnotes
Acknowledgement
This study was supported by a Grant-in-Aid for Challenging Exploratory Research (KEKENHI No. 26660235).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
