Abstract
Differentiating between inflammatory bowel disease (IBD) and small intestinal lymphoma in cats is often difficult, especially when only endoscopic biopsy specimens are available for evaluation. However, a correct diagnosis is imperative for proper treatment and prognosis. A retrospective study was performed using surgical and endoscopic intestinal biopsy specimens from 63 cats with a history of chronic diarrhea or vomiting or weight loss. A diagnosis of lymphoma or inflammation was based on microscopic examination of hematoxylin and eosin (HE)–stained sections alone, HE-stained sections plus results of immunohistochemical labeling (IHC) for CD3e and CD79a, and HE staining, immunophenotyping, and polymerase chain reaction (PCR) results for B and/or T cell clonality. In addition, various histomorphologic parameters were evaluated for significant differences between lymphoma and IBD using Fisher’s exact test. The sensitivity and specificity of each parameter in the diagnosis of lymphoma were also determined. Results of Bayesian statistical analysis demonstrated that combining histologic evaluation of small intestinal biopsy specimens with immunophenotyping and analysis of clonality of lymphoid infiltrates results in more accurate differentiation of neoplastic versus inflammatory lymphocytes. Important histologic features that differentiated intestinal lymphoma from IBD included lymphoid infiltration of the intestinal wall beyond the mucosa, epitheliotropism (especially intraepithelial nests and plaques), heterogeneity, and nuclear size of lymphocytes. Based on the results of this study, a stepwise diagnostic algorithm that first uses histologic assessment, followed by immunophenotyping and then PCR to determine clonality of the lymphocytes, was developed to more accurately differentiate between intestinal lymphoma and IBD.
Differentiating inflammatory bowel disease (IBD) from intestinal lymphoma in cats is a diagnostic challenge to both veterinary clinicians and pathologists. 30 Both conditions are most commonly diagnosed in middle-aged to older cats of any breed and sex. 3,27 The most common clinical signs include vomiting, diarrhea, weight loss, and changes in appetite. 18 In addition, food hypersensitivity and parasitism, both of which usually have an eosinophilic component, and endocrine, renal, or hepatic disease can cause similar signs. 3 Physical, ultrasonographic, and gross examination (endoscopic and surgical visualization) are of limited use in differentiating IBD from intestinal lymphoma. 3 However, an accurate diagnosis is critical because disease outcome and therapy depend on it.
The differentiation of intestinal lymphoma from IBD in live cats is usually based on the histologic evaluation of intestinal biopsy specimens. However, it can be extremely difficult to differentiate these two diseases by histomorphology alone. 2,14,20,28,30 Whereas B cell lymphomas have a distinct cellular morphology and are recognized fairly easily microscopically, intestinal T cell lymphomas and IBD are both characterized by marked infiltrates of small lymphocytes that cannot be distinguished by histology alone. 2,14 Unfortunately, primary feline intestinal lymphomas are most commonly of T cell origin. 2,14,18,20,33 Mucosa-associated lymphoid tissues (MALT) are normally populated primarily by T lymphocytes, and in both IBD and intestinal T cell lymphoma, there is expansion of T cells from the MALT. 14,22,26 In addition, lymphoma and inflammatory infiltrates frequently coexist. 14 However, in contrast to IBD, neoplastic T cells often infiltrate beyond the mucosa into the submucosa, tunica muscularis, and serosa, destroying normal tissue architecture. Because many routine intestinal biopsy specimens are collected endoscopically, histologic evaluation is often limited to the mucosa. Therefore, biopsy specimens of intestinal lymphomas in which neoplastic cells do not extend beyond the mucosa and endoscopic biopsy specimens are especially challenging for the pathologist, and there can be marked variation among pathologists in the histologic interpretations of intestinal samples. 31 Furthermore, the most common sites of feline intestinal lymphoma are the ileocecocolic junction and the jejunum, both of which are difficult to sample endoscopically. 3,13,14,31 In one study, the investigators concluded that endoscopic biopsy specimens were inadequate to distinguish between IBD and small intestinal lymphoma and recommended full-thickness specimens of the jejunum and ileum for accurate diagnosis. 3 Diagnosis is further complicated by the fact that clinical signs often do not correlate with the extent or severity of intestinal lesions. The difficulties in accurate diagnosis are exemplified by cats initially diagnosed with IBD that fail to respond to treatment and are subsequently diagnosed with intestinal lymphoma. 3
Immunophenotyping has become an important diagnostic tool in differentiation between IBD and intestinal lymphoma when histologic changes are ambiguous. 27 A monomorphic lymphocytic population supports a diagnosis of lymphoma, whereas a lymphoplasmacytic population supports a diagnosis of inflammation. Furthermore, increased numbers of intraepithelial lymphocytes also support a diagnosis of intestinal lymphoma. 2 Intraepithelial T cells are more easily identified in immunohistochemically labeled intestinal sections compared to hematoxylin and eosin (HE)–stained sections. 3,26
In human medicine, assessment for rearrangements of genes encoding both immunoglobulin heavy chain and T cell receptor gamma (TCRG) is an important diagnostic tool for lymphoproliferative disorders. 6,17 Moore et al 14 suggested assessment of TCRG V-J junctional diversity to detect T cell clonality as an adjunct test for the diagnosis of T cell lymphoma in cats. Similarly, a multiplex polymerase chain reaction (PCR) that targets the immunoglobulin heavy chain variable region genes has been useful in the diagnosis of feline B cell lymphoma. 15,29
The goal of this study was to develop a diagnostic algorithm for small intestinal surgical and endoscopic biopsy specimens from cats with chronic diarrhea. This algorithm is based on a stepwise strategy of histomorphologic assessment of biopsy specimens, followed by immunophenotyping and finally PCR to determine the clonality of the infiltrating T or B cells. Such a stepwise approach will assist pathologists in differentiating neoplastic from inflammatory lymphocytes in intestinal biopsy samples, thereby facilitating the prediction of clinical outcome and the selection of appropriate therapy.
Materials and Methods
Study Population
A total of 63 cats were included in this retrospective study. Each cat had a clinical history of chronic diarrhea, vomiting, or weight loss. Small intestinal biopsy specimens from all 63 cats had been submitted to the Diagnostic Center for Population and Animal Health between 2002 and 2005 from either the MSU Veterinary Teaching Hospital or two Michigan feline specialty practices. All cats had been diagnosed as either IBD (16 cases) or intestinal lymphoma (47 cases), based on histologic examination only.
Histopathology
Biopsy samples of ileum and jejunum were available from all cats. No gastric or large intestinal biopsies were examined. Fifty of the 63 cases were full-thickness biopsy samples; 13 were endoscopic specimens. The endoscopic tissues consisted of mucosa and submucosa. For all cases, sufficient formalin-fixed, paraffin-embedded samples were available for further testing. Serial sections from paraffin tissue blocks from each of the 63 cats were cut for routine HE staining, immunophenotyping, and PCR analysis.
Immunohistochemistry
Serial sections were deparaffinized and rehydrated for routine immunohistochemistry (IHC) 12 to detect expression of CD3e (1:20 dilution; P. Moore, UC Davis, Davis, CA) in T cells and CD79a (1:125 dilution; Clone HM57, Dako, Carpinteria, CA) in B cells. 12,14,15,27 Positive immunohistochemical controls included a normal feline lymph node to which the appropriate antisera were added. For negative controls, the primary antibodies were replaced with homologous nonimmune sera.
DNA Preparation and PCR
DNA extraction was performed on three 20-μm-thick serial sections from the formalin-fixed paraffin-embedded blocks. Tissue sections were deparaffinized with 1 ml FisherBrand CitriSolv Clearing Agent (Fisher Scientific, Waltham, MA) and washed twice in 1 ml of 100% ethanol prior to DNA extraction using the DNeasy Tissue Kit (Qiagen, Valencia, CA) as per the manufacturer’s recommendations. Rearrangements of T and B cell variable regions were assessed by PCR amplification to determine clonality using 5 μl of the unquantified extracted product.
For T cell PCR, the rearrangement of the TCRG variable region was assessed by amplifying the complementarity determining region 3 (CDR3) as previously described in Moore et al. 14 The primers were derived from the TCRG V segment (5′-AAGAGCGAYGAGGGMGTGT-3′) and the TCRG J segment (5′-CTGAGCAGTGTGCCAGSACC-3′). PCR was performed using a 50-μl reaction containing both T cell primers at a final HotStar Taq concentration of 0.5 μm DNA. The polymerase (Qiagen) 2-step modified touchdown protocol described by Hecker and Roux 9 was used. It consists of an initial activation step of 95°C for 15 minutes, followed by 5 cycles of 94°C for 30 seconds and 70°C for 60 seconds, 5 cycles of 94°C for 30 seconds and 68°C for 60 seconds, and 35 cycles of 94°C for 30 seconds and 65°C for 60 seconds, followed by a final extension at 72°C for 10 minutes.
For B cell PCR, the primers designed by Werner et al 29 were used to amplify the CDR3 of the immunoglobulin heavy chain variable region (IGH V). Multiplex PCR was conducted using 1 of 2 sense primers (FR2: 5′-CCAGGCTCCAGGGAAGGG-3′ or FR3: 5′-TCCAGAGACAACGCCAAGAAC-3′) in combination with 2 antisense primers (J2: 5′-TGAGGACACTGTGACTATGGTTCC-3′ and JD: 5′-GGACACCGTCACYAKGVYTCC-3′) to detect the rearrangements. PCR was performed using a 50-μl reaction containing the varying combinations of B cell primers (Taq Master Mix Kit; Qiagen). In each reaction, 10 pmol of primers FR2, FR3, and J2 was used, but 100 pmol was used for primer JD. The amplification conditions were similar to the T cell protocol: an initial activation step of 95°C for 4 minutes, followed by 5 cycles of 94°C for 30 seconds and 70°C for 2 minutes, 5 cycles of 94°C for 30 seconds and 68°C for 2 minutes, and 35 cycles of 94°C for 30 seconds and 65°C for 2 minutes, followed by a final extension at 72°C for 10 minutes.
Both T and B cell PCR reactions were run in duplicate. Gel electrophoresis was performed using a 3% agarose gel for the T cell PCR and 4% NuSieve/agarose gel for the B cell PCR, running 17 μl of both native and denatured PCR products from each reaction. Native products were loaded directly onto the gel. For heteroduplex analysis, an aliquot of the PCR products was denatured at 95°C for 10 minutes and then allowed to reanneal on ice for 1 hour prior to gel electrophoresis. Gels containing both native and denatured PCR products were run in tandem in TAE buffer at 100 V for 1 to 2 hours.
As in Moore et al, 14 duplicate PCR samples were always run side by side. Gels were then visualized on an ultraviolet (UV) transilluminator. Clonality was determined by the number and size of the bands. Clonal samples, most consistent with a neoplastic cell population, were defined as 1 or 2 sharp bands of the same size in duplicate samples run side by side within a target range of 80 to 120 bp for T cell PCR, 250 to 300 bp for B cell PCR with FR2, and 130 to 180 bp for B cell PCR using FR3. 14,29 Oligoclonal samples exhibited 3 to 5 reproducible bands in duplicate analyses; up to 3 bands were considered to be most consistent with a neoplastic process. 14,29 Pseudoclonal samples contained 1 or 2 bands that are of different size or non-reproducible when run side by side. 14,29 Polyclonal samples, consistent with a non-neoplastic process, such as inflammation, were identified by the presence of a broad band, smear, or ladder of bands covering a range of product sizes in the target range. 14
Case Evaluation
The following parameters were recorded for all HE and IHC sections: location, distribution, and density of the lymphocytic population. The location was recorded as intravascular, serosal, tunica muscularis, submucosal, or mucosal in each section. Mucosal lymphoid infiltrates were classified as “diffuse” for cases with a relatively consistent infiltration of lymphocytes throughout the section, with or without epithelial involvement. “Diffuse” mucosal lymphocytic infiltrates were further divided into low-density (<2 cells), medium-density (3–6 cells), or high-density (>7 cells) infiltrates based on the average number of lymphoid cells across the width of the villous lamina propria. A classification of “single villus” was used for cases that had a high density of lymphocytes within a single villus, regardless of whether the rest of the section was classified as diffuse. Furthermore, infiltrates of T cells within the epithelium were recorded as either surface or crypt infiltrates, and intraepithelial infiltrates were divided into single cells, nests, or plaques (Figs. 1–3). Nests of lymphocytes were defined as ≥5 clustered intraepithelial lymphocytes (Fig. 2); plaques were defined as ≥5 adjacent epithelial cells overrun by lymphocytes (Fig. 3). Epitheliotropism in the entire sample was assessed, whether the lymphocytic infiltrates had been classified as “diffuse” or “single villus,” because the degree of intraepithelial infiltration from one villus to another varies. In addition, the lymphocytic infiltrates were evaluated for monomorphism versus polymorphism, number of mitotic figures per 400× field, and cell size (large vs small). Available nongastrointestinal tissues were analyzed for metastasis.
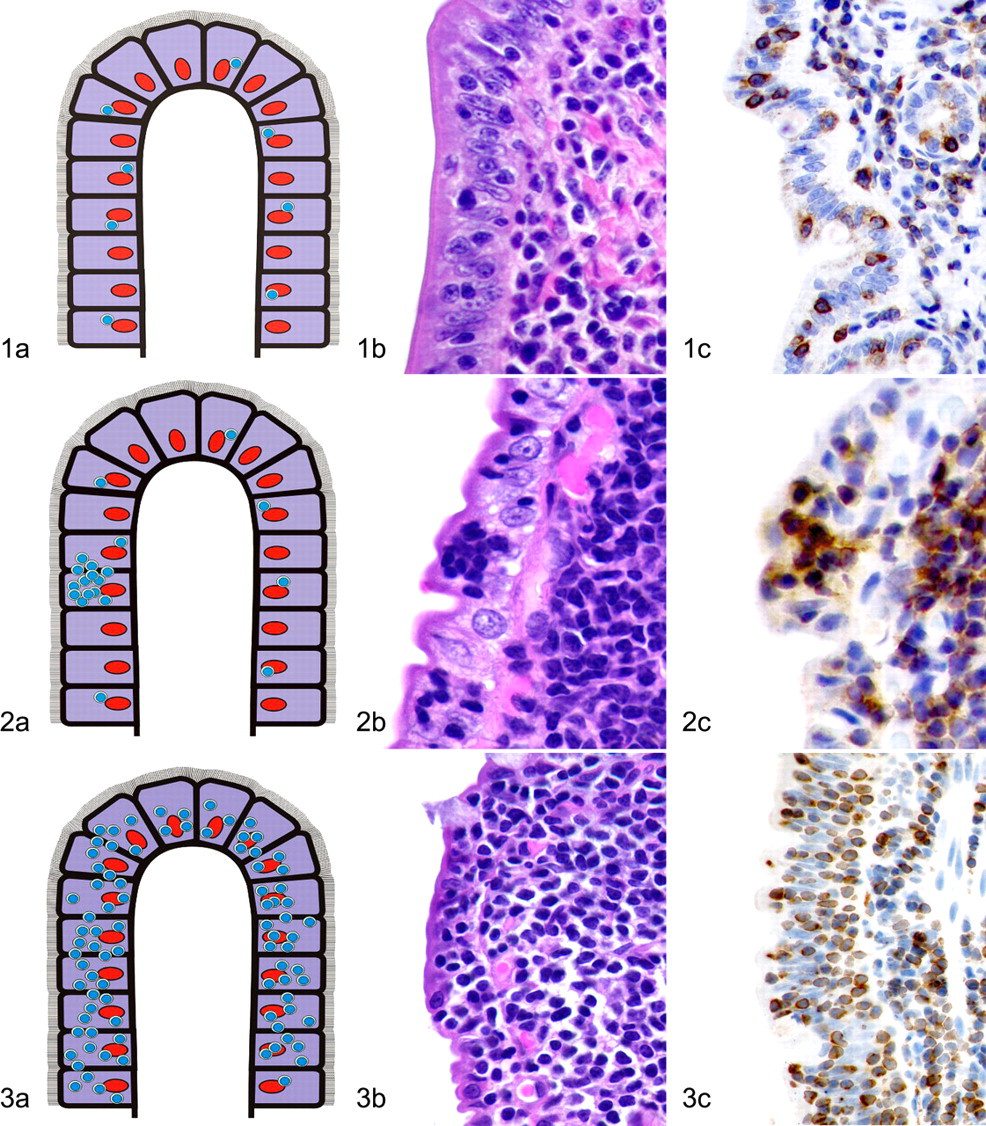
Histologic sections of small intestine from each cat were evaluated blindly by one pathologist (MK), and solely on the microscopic appearance of the most severely affected section, a diagnosis of IBD, T cell lymphoma, or B cell lymphoma was based on published criteria. 3,4 Without knowledge of these results, HE-stained sections were then analyzed a second time by the same pathologist (MK) in parallel with the serial immunohistochemistry preparations for CD3e and CD79a. Based on histologic features and immunohistochemistry for CD3e and CD79a, a diagnosis of IBD, T cell lymphoma, or B cell lymphoma was made using published criteria. 3,4 The original HE-based diagnoses were compared to those made in conjunction with immunophenotyping. The number of cases that were reclassified after immunophenotyping was determined. Each case was then evaluated a third time, without knowledge of the previous diagnoses, by combining histomorphology (HE) with immunophenotyping and PCR results for lymphocyte clonality. The differences between the HE diagnosis and the final diagnosis following IHC and PCR, as well as the differences between the HE plus IHC diagnosis and the final diagnosis, were compared.
Clinical Case Data
Available information regarding clinical history, including number of days of clinical signs prior to biopsy, treatment regimen, and survival data, including survival in days from biopsy until the end of the study (31 December 2005) and total survival times (survival time from detection of clinical signs until the end of the study), was obtained for each cat.
Statistical Analysis
Fisher’s exact test was performed to determine the significance of each histologic parameter in terms of predicting a diagnosis of lymphoma (T cell or B cell) versus IBD as well as in predicting IBD versus T cell lymphoma. The sensitivity and specificity of each parameter in predicting lymphoma (T cell or B cell) versus IBD and in predicting T cell lymphoma versus IBD were also determined.
Data were analyzed with Bayesian confidence profile models by using WinBUGS statistical software. 32 The estimated mean and 95% probability intervals for the sensitivity and specificity of diagnosing lymphoma by using 3 separate methods (HE only, HE combined with IHC, and HE combined with IHC and PCR) were determined. Prior distributions were specified by using beta distributions to model subjective probability about the unknown parameters. An appropriate beta (α, β) distribution for sensitivity can be constructed using data from previous studies with the following relationship: α = number of positives in n trials +1, and β = number of negatives in n trials +1. 7 We used a beta (9999, 1) for specificity to allow for false-positive results about 1 time in 10,000, and a noninformative prior, beta (1, 1), for prevalence. Inferences were based on 90,000 iterations after discarding an initial 10,000 samples as burn-in (i.e., the number of samples needed to reach convergence on the target distribution).
A Kaplan-Meier survival graphic was created to visualize the difference between the survival times of cats with lymphoma and those with IBD. Uni- and multivariate survival statistics were performed.
Results
The ages of the 63 cats ranged from 9 months to 19 years; the mean was 11 years. Thirty-six cats (57%) were neutered males; 27 cats (43%) were spayed females. Cats were domestic short- or long-haired breeds; more than 50% were of mixed breeding. All cats were negative for feline leukemia virus by PCR and IHC.
Clonality
PCR for T cell clonality was performed on all 63 cases (Fig. 4 ). PCR for B cell clonality was performed on each sample that was determined to have a polyclonal or oligoclonal population of T cells and all cases with a CD79a-positive lymphocytic infiltration suggestive of a B cell lymphoma. Thirty-four samples were determined to have a clonal population of T cells; 10 samples were oligoclonal. When HE and IHC results were considered, the final diagnosis was T cell lymphoma in 6 of these 10 cases, B cell lymphoma in 3 cases, and IBD in 1 case. Three samples initially had pseudoclonal results, but upon repeat testing, 2 were determined to have clonal populations of T cells and 1 to have a polyclonal population of T cells. In addition, 1 case with dual clonality for T and B cells was given a final diagnosis of B cell lymphoma based on the IHC results. Of 16 samples with polyclonal T cell populations, the final diagnosis was IBD in 11 cases and B cell lymphoma (based on B cell clonality by PCR) in 4 cases. For 1 case that had a clonal B cell population and a polyclonal T cell population, the final diagnosis was T cell lymphoma, based on histomorphology and immunophenotyping. Five cases had clonal populations of B cells, including 1 case that also tested positive for T cell clonality. For 4 of these cases, final diagnoses of B cell lymphoma were made; 1 case was given a final diagnosis of IBD. Twenty-two cases were polyclonal for B cells. Thirteen of these 22 cases had a polyclonal population of B and T cells; 10 of the 13 were diagnosed as IBD, 2 as B cell lymphoma, and 1 as T cell lymphoma based on HE and IHC results. Nine of these 22 cases had a polyclonal population of B cells and an oligoclonal population of T cells; the final diagnosis was B cell lymphoma for 3 of the 9, T cell lymphoma for 5 of the 9, and IBD for 1, based on the combined results.
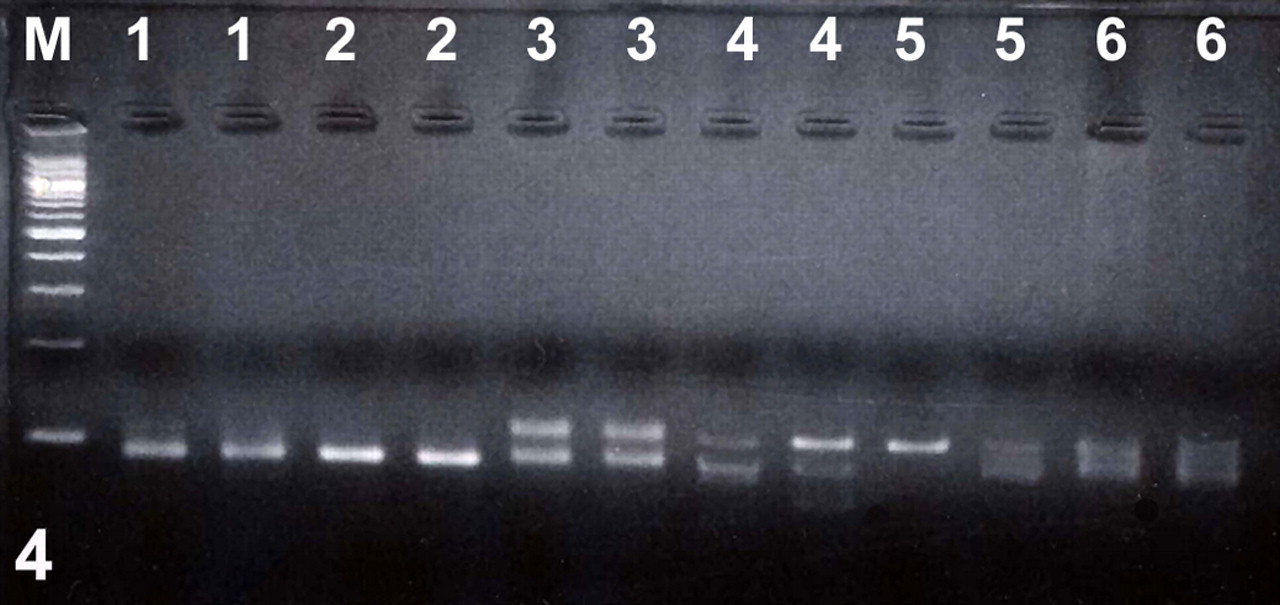
T cell receptor gamma (TCRG) rearrangements in intestinal biopsy samples from cats with intestinal lymphoma or inflammatory bowel disease. All samples were run in duplicate. Lane M: 100-bp ladder; lanes 1 and 2: clonal; lane 3: biclonal; lanes 4 and 5: pseudoclonal; lane 6: polyclonal.
Case Evaluation
All 63 samples were diagnosed by histopathology, immunophenotyping, and PCR. Based on morphologic evaluation alone, 37 cases (59%) were diagnosed as T cell lymphoma, 7 cases (11%) were diagnosed as B cell lymphoma, and 19 cases (30%) were diagnosed as IBD. When immunohistochemical findings were used in conjunction with the microscopic findings, 40 cases (63%) were diagnosed as T cell lymphoma, 8 cases (13%) as B cell lymphoma, and 15 cases (23%) as IBD (Table 1 ). Five of 19 cases (26%) that had been originally diagnosed as IBD by histopathologic examination alone were reclassified as T cell lymphomas based on the combined HE microscopic and immunohistochemical results. When PCR results were used in conjunction with HE and immunophenotyping results, 43 cases (68%) were diagnosed as T cell lymphoma, 8 (12%) as B cell lymphoma, and 12 (19%) as IBD (Table 1). Ten of 19 cases (53%) that originally had been diagnosed as IBD by histopathologic examination alone were reclassified as T cell lymphoma when IHC and PCR results were evaluated in conjunction with the HE findings (Table 1). Three of 37 cases (8%) that originally had been diagnosed as T cell lymphoma by histopathologic examination alone were reclassified as IBD when IHC and PCR results were evaluated in conjunction with the HE findings (Table 1).
Comparison of Diagnoses Based on Morphologic Evaluation Alone, in Combination With Immunophenotyping and in Combination With Immunophenotyping and PCR for Clonality
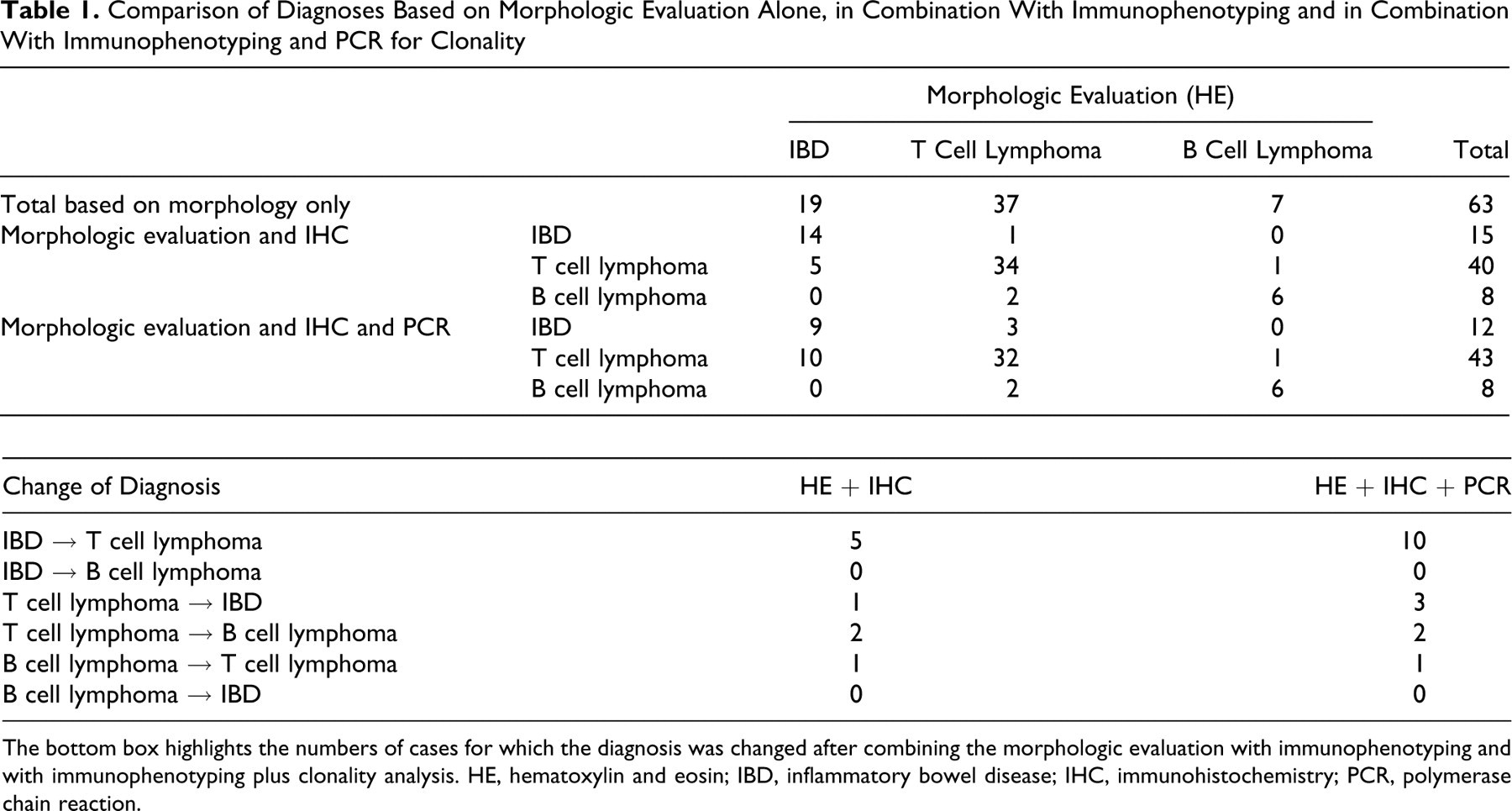

The bottom box highlights the numbers of cases for which the diagnosis was changed after combining the morphologic evaluation with immunophenotyping and with immunophenotyping plus clonality analysis. HE, hematoxylin and eosin; IBD, inflammatory bowel disease; IHC, immunohistochemistry; PCR, polymerase chain reaction.
The intestinal mucosa was infiltrated by lymphocytes in all 63 cases. In 24 cases, lymphocytic infiltration was limited to the mucosa; 7 of these cases were diagnosed as lymphoma (T cell, in each case). In 39 cases, the lymphocytic infiltration extended into the submucosa; all but 2 of these were diagnosed as lymphoma (30 T cell, 7 B cell lymphomas). In 28 of the 50 cases in which tunica muscularis was in the biopsy specimen, it was also infiltrated by lymphocytes; 27 of these cases were diagnosed as lymphoma (20 T cell, 7 B cell lymphomas). The remaining case with tunica muscularis infiltration was diagnosed as IBD. However, only 20 of 34 T cell lymphomas that were diagnosed in full-thickness biopsy specimens had intramuscular infiltrates. Serosal lymphocytic infiltration was detected in 22 of the 49 cases in which serosa was included in the specimen; each of these 22 cases was diagnosed as lymphoma (16 T cell, 6 B cell lymphomas). All cases with serosal infiltration also had mucosal, submucosal, and tunica muscularis infiltration. Of the 59 cases that could be evaluated for intravascular infiltration, 8 had intravascular lymphocytic infiltration, and all were diagnosed as lymphoma (5 T cell, 3 B cell lymphomas). None of the IBD cases had intravascular lymphocytic infiltrates.
In all but one T cell lymphoma and one IBD, lymphocytic infiltration was diffuse throughout the section. Single villus involvement was identified in 6 cases: 3 T cell lymphomas, 1 B cell lymphoma, and 2 cases of IBD. In cases of diffuse infiltration, the average cellular density across the width of the villous lamina propria was low (≤2 cells) for 5 cases (4 IBD, 1 T cell lymphoma), medium (3–6 cells) in 21 cases (15 T cell lymphomas, 2 B cell lymphomas, 4 IBD), and high (≥7 cells) in 35 cases (26 T cell lymphomas, 6 B cell lymphomas, 3 IBD). One case (T cell lymphoma) without diffuse involvement of the section did have a focally high density of lymphocytes.
Epitheliotropism was evaluated in CD3e-labeled slides (Figs. 1–3). Intraepithelial lymphocytes were always CD3e positive and CD79a negative. Fifty-five cases (39 T cell lymphomas, 5 B cell lymphomas, 11 IBD) had increased intraepithelial infiltrates. Of these 55 cases, 53 had epitheliotropism of the surface epithelium; 22 of these cases (10 T cell lymphomas, 3 B cell lymphomas, 9 IBD) only had single intraepithelial lymphocytes, whereas 19 (16 T cell lymphomas, 1 B cell lymphoma, 2 IBD) had nests, and 12 cases (11 T cell lymphomas, 1 B cell lymphoma) had plaques. Of the 55 cases with intraepithelial infiltrates, 44 had epitheliotropism of the crypts; 37 cases (25 T cell lymphomas, 4 B cell lymphomas, 8 IBD) only had scattered single lymphocytes within the crypt epithelium, 3 cases (all T cell lymphomas) had intraepithelial nests, and 4 cases (all T cell lymphomas) had plaques.
In 50 cases (39 T cell lymphomas, 8 B cell lymphomas, 3 IBD), the lymphocytic infiltration was monomorphic. Polymorphic infiltrates were identified in 13 cases (4 T cell lymphomas, 9 IBD). Forty-five cases had an average of 0 mitoses per 400× field, seven cases had a range of 0-1 mitotic figures per 400× field, three cases had an average of 1 mitotic figure per 400× field, and eight cases (6 B-cell lymphomas, 2 T-cell lymphomas) had an average of two mitoses per 400× field. The lymphocytic infiltrate in 42 cases (36 T cell lymphomas, 2 B cell lymphomas, 4 IBD) was classified as small cell only, in 7 cases (1 T cell lymphoma, 6 B cell lymphomas) as large cell only, and in 14 cases (6 T cell lymphomas, 8 IBD) as both small and large cell. Seventeen cases (11 T cell lymphomas, 6 B cell lymphomas) had evidence of metastasis within nongastrointestinal tissues.
Clinical Case Data
Most of the cats originally diagnosed with IBD were treated with a diet change, with or without corticosteroid therapy. Of the 48 cats originally diagnosed with intestinal lymphoma, 37 underwent chemotherapy, with chlorambucil being the most commonly used drug. The owners of 9 cats refused treatment, usually because of the severity of the disease. Fourteen cats died or were euthanized within 1 month of diagnosis.
Twenty-four cats with T cell lymphoma had died or had been euthanized; 19 cats with T cell lymphoma were still alive at the conclusion of the study (31 December 2005). The survival times after biopsy ranged from 1 to 1,183 days with an average of 316 days. All cats with B cell lymphoma had died or had been euthanized by the end of the study; postbiopsy survival ranged from 11 to 182 days with a mean of 66.8 days (Fig. 5 ). Five cats with IBD had died or were euthanized; 7 were alive at the end of the study period. Postbiopsy survival ranged from 0 to 1395 days with a mean of 748 days (Fig. 5).
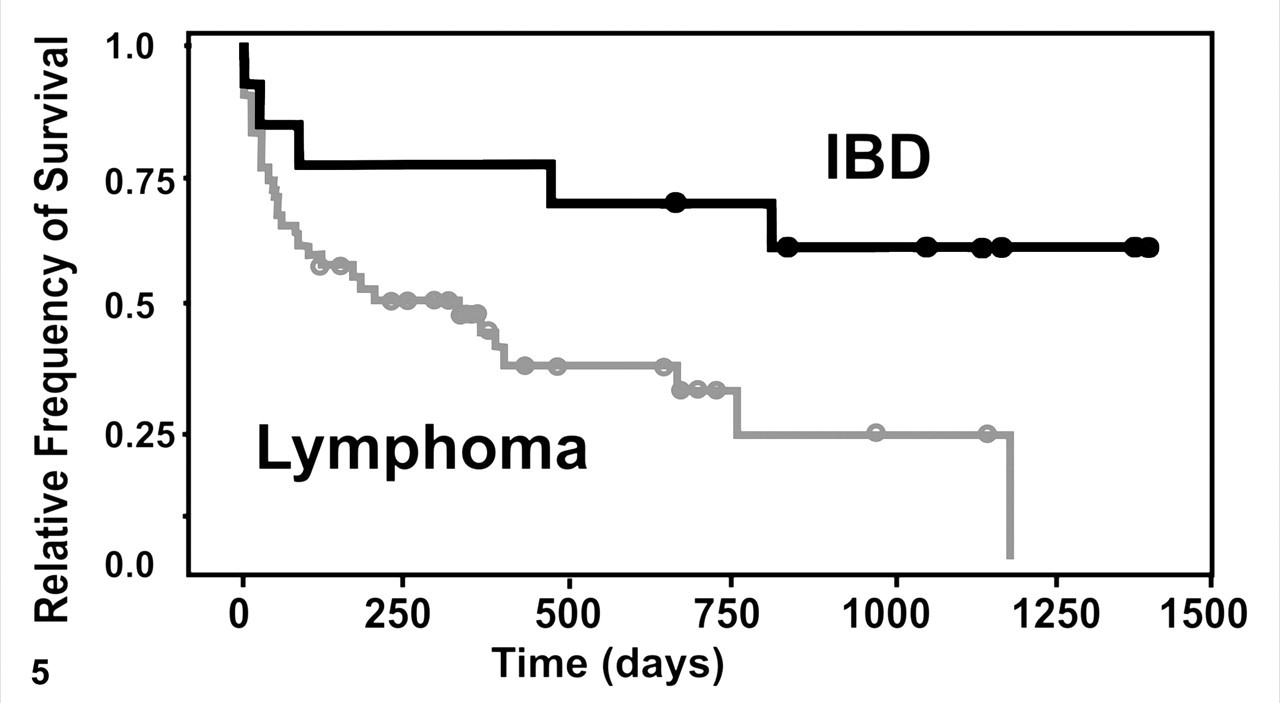
Kaplan-Meier survival graph for cats with inflammatory bowel disease (IBD) and intestinal lymphoma. Cats with lymphoma have shorter survival times than those with IBD; >50% of cats with lymphoma were dead within a year.
The total survival time after detection of clinical signs was known for 23 cats. The 12 cats with a final diagnosis of T cell lymphoma survived from 3 to 3,870 days with a mean of 756 days. The 2 cats with B cell lymphoma lived 115 and 547 days, respectively. Total survival times for the 9 cats with IBD ranged from 5 to 2,515 days with a mean of 941 days.
Statistical Analysis
Based on Fisher’s exact tests, the following histologic parameters differed significantly between IBD and lymphoma (T cell or B cell): submucosal infiltration (P = .0065), tunica muscularis infiltration (P = .0345), serosal infiltration (P = .0123), diffuse infiltration (P = .0019), intraepithelial surface nests (P = .0077), monomorphism (P = 5.385E-06), polymorphism (P = 5.385E-06), and large cell infiltrates (P = .0141). Although the presence of lymphocytes in the submucosa, tunica muscularis, and serosa was each significant in differentiating between IBD and lymphoma, the specificity increased from the submucosa (75%) to the tunica muscularis (86%) to the serosa (100%). Intravascular infiltration, single villus infiltration, single-cell intraepithelial surface infiltration, surface intraepithelial plaques, intraepithelial crypt infiltration (single cells, nests, and plaques), mitotic index, and small cell infiltrates were parameters that were not significantly different between IBD and lymphoma (T cell or B cell). However, additional parameters were statistically significant in differentiating between IBD versus T cell lymphoma: intravascular infiltration, mitotic index, and small cell infiltrates. Fisher’s exact test could not be performed for the parameter “mucosal infiltration” because all cases had mucosal infiltration. Parameters that had 100% specificity for lymphoma versus IBD were intravascular infiltrates, serosal infiltration, surface intraepithelial plaques, crypt intraepithelial nests and plaques, and metastasis. Parameters that had 100% specificity in diagnosing T cell lymphoma were crypt intraepithelial nests and plaques. Although the finding of intraepithelial surface plaques was 100% specific for a diagnosis of lymphoma, only 12 cases exhibited this parameter. This small number resulted in a Fisher’s exact test P value (P = .1004) that was not statistically significant. The sensitivity and specificity could not be determined for the parameters mucosal infiltration, diffuse infiltration, and mitotic index. The estimated mean and 95% probability intervals for the sensitivity and specificity of diagnosing lymphoma by using 3 separate methods (HE only [HE diagnosis], HE plus immunohistochemistry [IHC diagnosis], and HE, IHC, and PCR [final diagnosis]) were determined using Bayesian analysis (Table 2 ). The mean percent sensitivity was highest (0.826), intermediate (0.7814), and lowest (0.7217) for the final diagnosis method, IHC diagnosis, and HE diagnosis, respectively. More than 50% of cats with lymphoma were dead within a year (Fig. 5). When performing uni- and multivariate survival statistics, there was no significant difference in survival times between cats with IBD and cats with lymphoma.
Sensitivity and Specificity for Diagnosis of Feline Intestinal Lymphoma by 3 Methods
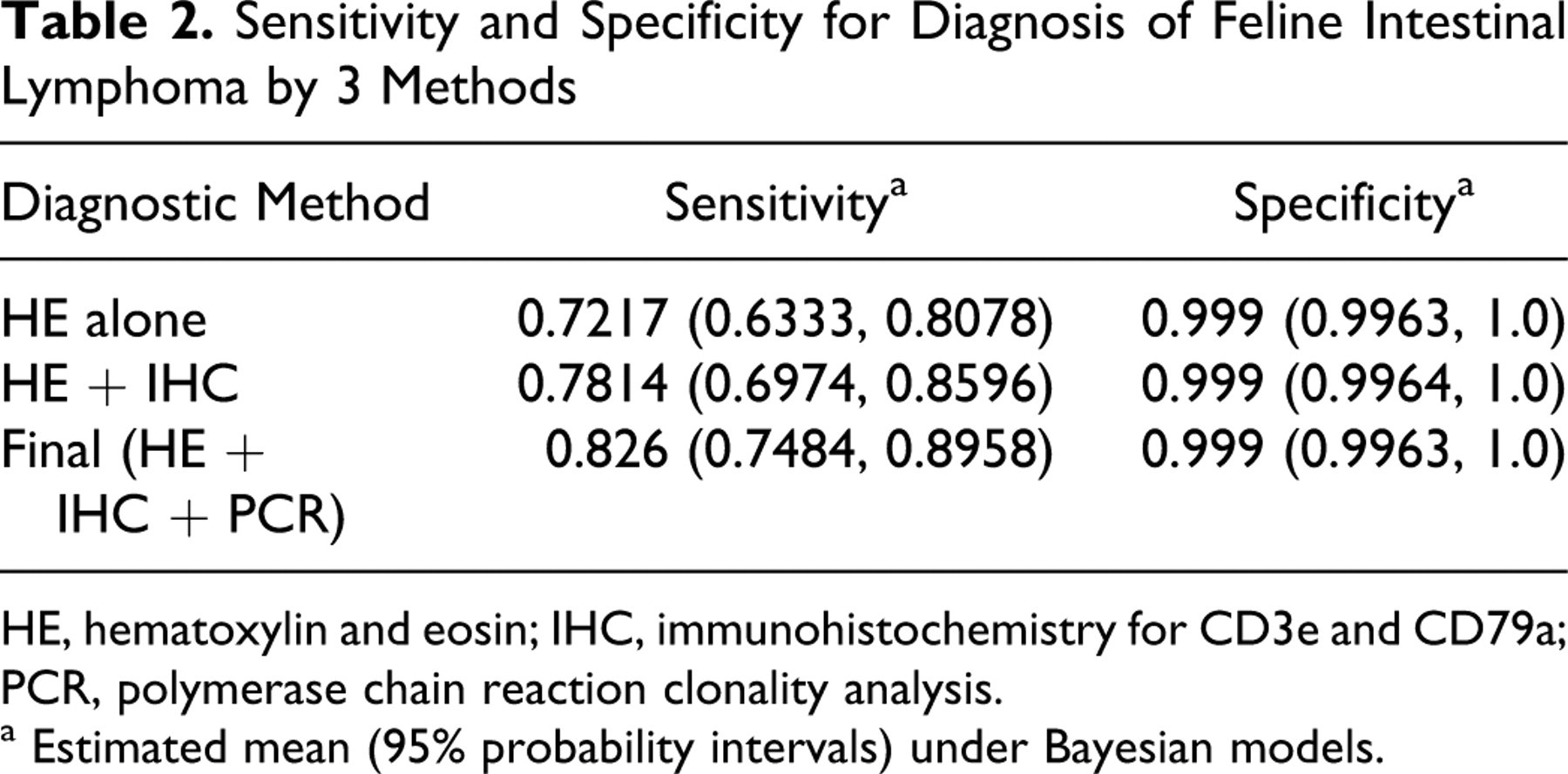
HE, hematoxylin and eosin; IHC, immunohistochemistry for CD3e and CD79a; PCR, polymerase chain reaction clonality analysis.
a Estimated mean (95% probability intervals) under Bayesian models.
Discussion
Based on the data of this study, primary small intestinal T cell lymphomas are far more common in cats than primary small intestinal B cell lymphomas, which support the results of some studies 2,15,18,33 but refute the results of others. 4,5,16,23,27 In one study, 52% of small intestinal lymphomas were of T cell lineage, 38% were of B cell lineage, and 8% were nonreactive for B or T cell markers. 18 When the tumors that infiltrated only the small intestine in that study were analyzed separately from tumors that infiltrated multiple gastrointestinal sites, the percentage of small intestinal T cell lymphomas increased to 60%. However, when the numbers of gastric and large intestinal lymphomas were combined with those of small intestinal lymphomas, B cell lymphomas predominated with 54% compared to 38% T cell lymphomas. 18 All gastric lymphomas in that study were of B cell lineage. 18 In one study with data contrary to ours, most alimentary lymphomas were not immunoreactive for CD3; however, that study did not distinguish between gastric and intestinal lymphomas, and no immunohistochemistry for B cells was performed. 23 Most alimentary lymphomas in yet another study were classified as B cell lymphomas; however, intestinal lymphomas again were not separated from lymphomas at other alimentary sites. 16 Furthermore, in some studies, the localization of CD79a immunolabeling is not discussed. 16,23 Because CD79a commonly aberrantly labels the nuclei of both lymphocytes and epithelial cells, 21 true cytoplasmic labeling must be distinguished from aberrant nuclear labeling. Based on our data and critical interpretation of the literature, primary small intestinal lymphomas in cats are more likely to be of T cell origin.
All PCR clonality tests were performed in duplicate since single bands representing pseudoclones may occur in single columns and could be misinterpreted as a clonal cell population. 14 Furthermore, we also ran one gel of native PCR product and one that was denatured (heteroduplex analysis). The denaturing will not affect single bands representing clonality but will lead to a smear for polyclonal cell populations. 14 T cell pseudoclonality was initially identified in 3 cases, but when PCR was repeated, 2 of these cases had a clonal population of T cells, and 1 had a polyclonal population of T cells. However, all 3 cases were diagnosed as T cell lymphomas when histomorphology and immunophenotyping were also considered, which emphasizes the need to combine histomorphology, immunophenotyping, and PCR to make a final diagnosis. Ten cats had an oligoclonal population of T cells. When histomorphology and immunophenotyping results were taken into consideration, 6 of the 10 cases were diagnosed as T cell lymphomas, 3 as B cell lymphomas, and 1 as IBD. Oligoclonality can occur in situations of markedly restricted clonal diversity, as observed with chronic inflammatory disease or emerging neoplasia, in neoplasms involving more than a single clone (biclonal T cell lymphoma) and in response to viral and other pathogens or other types of neoplasia. 1,10,14,15,19,24
One case had dual clonality for both T and B cell populations. This is not an unusual finding in a percentage of lymphoid neoplasms 17,25 and can occur when a B or T cell lymphoma carries a dual genotype (lineage infidelity) or, less commonly, when there is “concurrence of 2 monoclonal malignant B- and T-cell populations (composite lymphoma).” 6 In one study, 11% of 63 human lymphoid neoplasms were bigenotypic, with rearrangements of both Ig and Tβ genes. 17 Clonal rearrangement of the B cell Ig heavy chain as well as the gamma chain of the T cell receptor has also been reported in human cutaneous lymphomas. 6,8,11 Importantly, lineage specificity of T and B cell rearrangements is not absolute, 14,15,17 and bigenotypic lymphomas maintain their immunophenotype. 17 Laser capture microdissection can separate a population of cells and improve genotypic analysis of that population; however, this is a costly test that is not readily available at most veterinary laboratories. 6,25 In the absence of this procedure, molecular clonality determination should not be the sole basis for lymphocyte lineage assignment and must be evaluated in conjunction with immunophenotyping. 14,17 Histomorphology and immunophenotyping should always precede assessment of clonality, and immunophenotypic assessment should take precedence over results of molecular clonality when determining lymphocyte lineage. 14,15
In this study, we examined several morphologic parameters for their usefulness to differentiate between IBD and intestinal lymphoma. Infiltration of lymphocytes below the submucosa, intraepithelial surface nests, a monomorphic and small lymphoid cell population, intravascular infiltration, and a high mitotic index were the parameters that were statistically significant in determining a diagnosis of T cell lymphoma versus IBD using Fisher’s exact test. Although the finding of intraepithelial surface plaques was not a statistically significant parameter, it was 100% specific for a diagnosis of lymphoma. Additional parameters with 100% specificity for lymphoma were intravascular infiltrates, serosal infiltration, crypt intraepithelial nests and plaques, and metastasis.
Based on the data presented here, full-thickness surgical biopsy specimens of the small intestine are superior to endoscopic biopsy specimens for differentiating small intestinal lymphoma from IBD because infiltration of lymphocytes deeper than the mucosa is a major criterion. However, a diagnostic approach that combines morphologic with immunohistochemical and clonality analysis will dramatically improve the pathologist’s ability to differentiate between IBD and lymphoma in endoscopic biopsy sections. Morphologically, the pathologist should evaluate whether the mucosal lymphocytic infiltration is diffuse and/or involves single villi, with particular attention to villous and crypt epithelium for intraepithelial nests or plaques. Differentiating feline intestinal T cell lymphoma from IBD is critical for the selection of appropriate therapy and the accurate prediction of clinical outcome. This study demonstrates that a significant number of feline intestinal T cell lymphomas are incorrectly diagnosed as inflammation using histomorphology alone or histomorphology in conjunction with immunohistochemistry. Combining the morphologic evaluation of small intestinal biopsy specimens with immunophenotyping of the lymphocytic infiltration and analysis of clonality will result in a more accurate differentiation of neoplastic from inflammatory lymphoid cells. We propose a novel diagnostic algorithm for small intestinal biopsy specimens from cats with a history of chronic diarrhea that uses a stepwise testing strategy composed of the histomorphologic assessment, followed by immunophenotyping and finally PCR to determine the clonality of infiltrating T and B cells (Fig. 6 ). It is important that histologic examination, immunophenotyping, and PCR are always performed in this sequence and that the results are only interpreted in context. If, after a purely morphologic diagnosis of IBD, a cat does not respond appropriately to therapy, the histologic sections should be reviewed in combination with immunophenotyping, and re-biopsy should be considered. Immunohistochemistry should be performed in any case with a morphologic diagnosis of lymphoma or lymphoma-suspect. In many cases, IHC will confirm a diagnosis of lymphoma by identifying a monomorphic population of B or T lymphocytes. When the diagnosis remains uncertain, the appropriate PCR test, based on IHC results, can be used to detect clonality of B or T lymphocytes to aid in a diagnosis of lymphoma. If no clonal cell population is detected, an inflammatory process should be considered most likely. This systematic assessment of feline intestinal biopsy specimens will decrease the likelihood of misdiagnosis and will facilitate both prognosis and selection of appropriate therapy. Additional biopsy specimens, ideally full thickness, may be required for cats that present with early lymphoma. 3
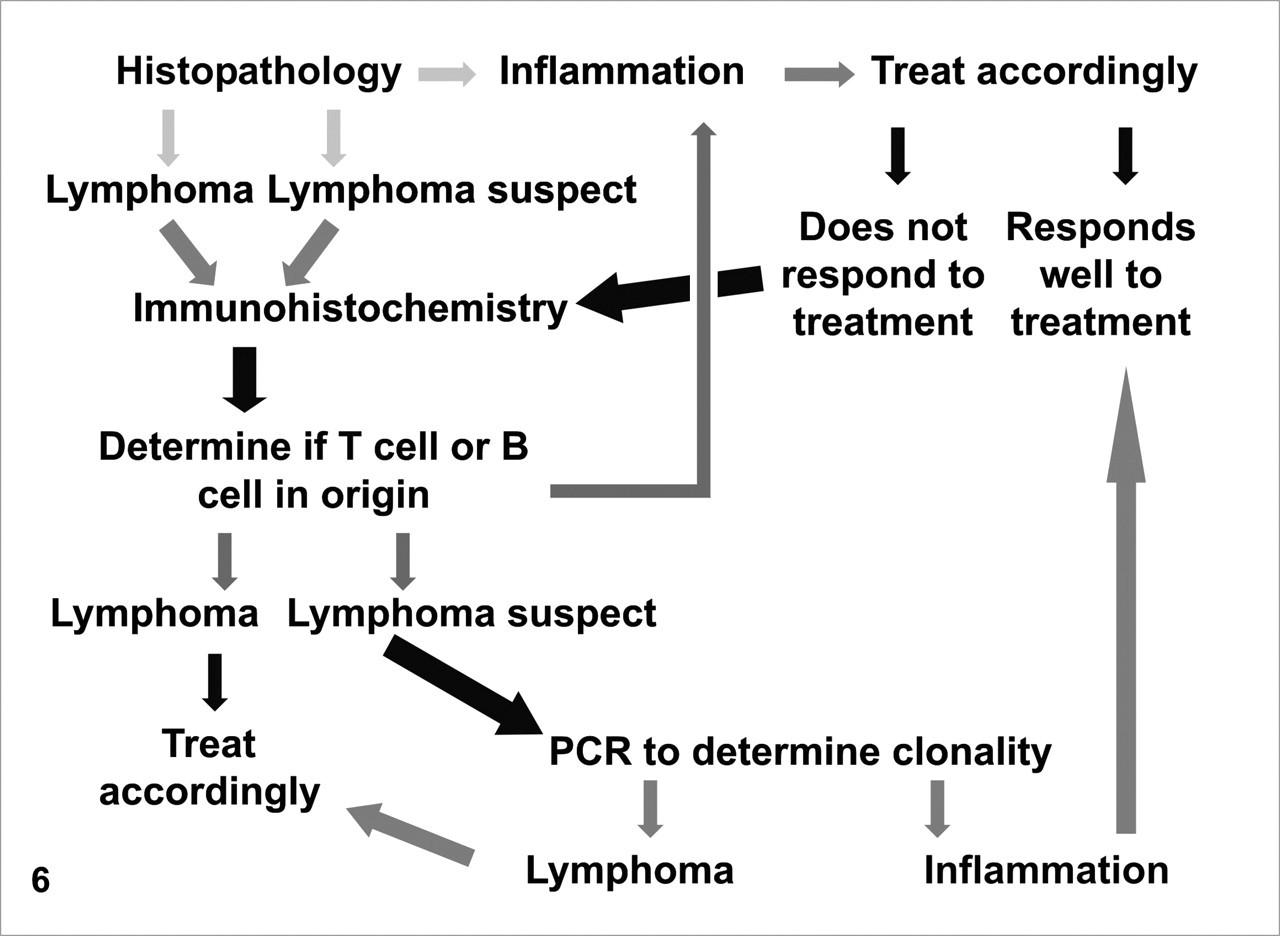
Diagnostic algorithm to differentiate feline intestinal lymphoma from inflammatory bowel disease in small intestinal surgical biopsy specimens. Polymerase chain reaction (PCR) clonality analysis should be guided by immunophenotype; however, lineage specificity of T and B cell rearrangements is not absolute, and additional PCR reactions may be required.
Cats with lymphoma died sooner (more than 50% within 1 year) than those with IBD. However, these survival data should be interpreted with caution because owners may have elected euthanasia based on the poor prognosis of lymphoma and/or an unwillingness to manage a diarrheic cat. The lack of information of the actual cause of death, as well as insufficient data about the length and severity of clinical signs before biopsy, hampers our ability to analyze total survival times. With uni- and multivariate statistics, there was only a trend but no significant difference in survival times between cats with IBD and cats with lymphoma. A prospective study using the proposed diagnostic algorithm and appropriate therapeutic protocols would determine the prognostic value of accurately differentiating IBD from small intestinal lymphoma.
Footnotes
Acknowledgements
We thank Tom Wood and the DCPAH Histology and Immunohistochemistry Laboratory for technical support, Dan Marshall and John Hoschek for providing biopsy material and clinical information, MingFei Li and Li Wang for help with statistics, and the MSU Companion Animal Fund for the financial support for this study.
The author(s) declared that they had no conflicts of interests with respect to their authorship or the publication of this article.
The author(s) declared that they received no financial support for their research and/or authorship of this article.
