Abstract
Persistent bacterial infections of the gastrointestinal mucosa are causally linked to gastric carcinoma and mucosa-associated lymphoid tissue (MALT) lymphoma in people and laboratory animals. We examined the relationship of mucosa-associated bacteria to alimentary lymphoma in cats. Intestinal biopsies from 50 cats with alimentary lymphoma (small cell, n = 33; large cell, n = 17) and 38 controls without lymphoma (normal to minimal change on histopathology, n = 18; lymphocytic-plasmacytic enteritis, n = 20) were evaluated. The number and spatial distribution of bacteria (ie, in luminal cellular debris, villus-associated mucus, adherent to epithelium, mucosal invasion, intravascular, or serosal) were determined by fluorescence in situ hybridization with the eubacterial probe EUB-338. Mucosa-invasive bacteria were more frequently observed in cats with large cell lymphoma (82%, P ≤ .001) than in cats with small cell lymphoma (18%), normal to minimal change on histopathology, and lymphocytic-plasmacytic enteritis (3%). Intravascular bacteria were observed solely in large cell lymphoma (29%), and serosal colonization was more common in cats with large cell lymphoma (57%) than with small cell lymphoma (11%, P ≤ .01), normal to minimal change (8%, P ≤ .01), and lymphocytic-plasmacytic enteritis (6%, P ≤ .001). The high frequency of invasive bacteria within blood vessels and serosa of cats with large cell lymphoma may account for the sepsis-related complications associated with large cell lymphoma and inform clinical management. Further studies are required to determine the role of intramucosal bacteria in the etiopathogenesis of feline alimentary lymphoma.
Keywords
Lymphoma is one of the most frequently diagnosed neoplasms of domestic cats and represents up to 90% of hematopoietic tumors in the species. 31,38,44 Feline leukemia virus (FeLV) was the most common cause of cranial mediastinal, multicentric, renal, and central nervous lymphoma from the 1960s to the 1980s, but FeLV antigen is inconsistently associated with the gastrointestinal form of lymphoma. 11,16,23,31,38,39,44,45 Despite a general decline in FeLV-associated lymphoma, the overall prevalence of lymphoma in cats has been increasing, with alimentary lymphoma now considered the most common anatomic form. 23,24,31,38,44 The course of the disease and response to therapy has also changed, with overall median survival time reaching up to 704 days in low-grade lymphoma vs weeks to months in high-grade large cell lymphoma. 10,19 Until recently, the large B-cell phenotype predominated in Australia and the United Kingdom, but the small T-cell phenotype has recently emerged. 4,12,26,32,41 The sequential temporal emergence of low-grade alimentary lymphoma in the United States, United Kingdom, and Australia echoes the dramatic increase in the prevalence of feline hyperthyroidism since it was first described in the late 1970s and suggests that changes in the environment, diet, or exposure to infectious agents may be involved in etiopathogenesis. 29
Persistent bacterial infections are increasingly recognized as triggers for inflammation-associated neoplastic transformation. The concept that mucosa-associated bacteria can promote gastrointestinal carcinogenesis was initially established through study of Helicobacter pylori, which is now causally linked to the development of gastric adenocarcinoma and mucosa-associated lymphoid tissue (MALT) lymphoma in people. The pivotal importance of H. pylori in tumorigenesis is highlighted by the dramatic response of early stage mucosa-associated lymphoid tissue lymphoma to eradication of H. pylori with antibiotics, with up to 75% of patients going into long-term remission. 15,27 More recently, Campylobacter jejuni has been linked to the development of immunoproliferative small intestinal disease and lymphoma, and Borrelia and Chlamydia are associated with cutaneous and periocular B-cell MALT lymphoma, respectively. 1,8,9,22,28
It is emerging that chronic small intestinal inflammation in the absence of known pathogenic bacteria can also progress to enteropathy-associated T-cell lymphoma. 37 Whole-genome analyses and human leucocyte antigen genotyping of affected people have revealed 2 distinct subtypes of enteropathy-associated T-cell lymphoma, with type 1 developing as a complication of celiac disease and type 2 following nonceliac inflammation. 7 Lymphomatous transformation in celiac disease is associated with unresolved chronic lymphocytic inflammation, villus blunting, an interleukin (IL)–6– and IL-8–rich cytokine environment, and global shifts in the resident enteric microbiome toward proinflammatory proteobacteria and Escherichia coli, changes that parallel the microbial and mucosal abnormalities described in cats with lymphoplasmacytic enteritis (LPE). 17,18,33 –35
Taken as a whole, the factors responsible for the changes in prevalence, immunophenotype, and biology of feline alimentary lymphoma are not known. The evidence to date supports the possibility that mucosal bacteria, whether pathogens or resident species, might be involved in the etiopathogenesis of alimentary lymphoma. It is against this background that we sought to examine the relationship of mucosa-associated bacteria to alimentary lymphoma in cats. We hypothesized that the number and spatial distribution of mucosal bacteria differ between healthy cats, cats with LPE, and cats with alimentary lymphoma.
Materials and Methods
Case Selection
The feline database of the Section of Anatomic Pathology in the College of Veterinary Medicine, Cornell University was searched in June 2011 for small intestinal endoscopic and surgical biopsies and samples originating from necropsies collected during the years 2007 through 2011. Cats were considered for inclusion if their small intestinal sections were retrievable and previously diagnosed as normal small intestine, LPE/inflammatory bowel disease (IBD), or small intestinal lymphoma. Endoscopic biopsies, transmural surgical biopsies, and necropsy samples were accepted for the normal group. Because infiltration of deeper tissue layers is a major criterion to distinguish intestinal inflammation from lymphoma, only full-thickness surgical biopsies were included in the IBD group, and surgical samples were preferred over endoscopic samples in the lymphoma group. 20 IBD and lymphoma cases ultimately included in the study were chosen randomly to equally represent the years 2007 to 2011, as well as mild, moderate, and severe states of the LPE subtype of IBD. Samples taken at necropsy were included in the study only if the alignment and preservation of the sections allowed unhindered mucosal assessment. Surgically removed masses were consecutively sectioned, and the analyzed sections were obtained from within the mass rather than the surgical margins. Cases were excluded if specific causes for gastroenteritis could be demonstrated, neoplasia other than lymphoma was present, autolysis or freezing artifacts made mucosal assessment impossible, or they were known to have tested positive for FeLV or feline immunodeficiency virus.
Histopathology
Hematoxylin and eosin (HE)–stained slides of small intestine from the archival cases selected for inclusion in this study were reviewed by a board-certified pathologist (S.P.M.), who was blinded to their origin. Each case was assigned to the normal gastrointestinal tract, LPE, or lymphoma group, based on the diagnostic algorithm described by Kiupel et al. 20 Cases of normal small intestine and LPE were evaluated for morphological features and inflammation as described in the World Small Animal Veterinary Association (WSAVA) scheme for duodenal mucosa 6 with the addition of individual descriptors for villous fusion, epithelial erosion, and mucosal ulceration to the morphological features, as well as macrophages and mast cells to the inflammation category. We also enumerated plasma cells and lymphocytes separately. Separately graded features (scored as normal, mild, moderate, marked) were summed to yield a composite score and assign a final grade, with a total score of 0 to 2 being considered a normal small intestine (0), 3 to 7 mild LPE (1), 8 to 13 moderate LPE (2), and >13 severe LPE (3).
Features evaluated for cases of lymphoma included cell size, grade, and lineage by use of immunohistochemistry. Cases of lymphoma were subtyped according to the World Health Organization classification as detailed by Valli et al. 40 Nuclear size was compared with the diameter of an erythrocyte (small, <1.5×; intermediate, 1.5–2×; and large, >2× the diameter of an erythrocyte). The mitotic index was determined based on the average in 10 high-power fields (HPF; 400×) chosen at random. Low-, medium-, and high-grade lymphomas averaged ≤5, 6 to 10, and >10 mitoses per HPF, respectively. 40
After 30 days, all slides were randomized and assessed a second time. Discrepant results were resolved by a third round with the final grades determined by consensus. For the purpose of statistical analysis, sections of normal small intestine and mild LPE were collectively termed normal to minimal change (NMC), moderate to severe LPE was collectively termed LPE, and small cell lymphoma (SCL) was differentiated from large cell lymphoma (LCL).
Immunohistochemistry
Immunohistochemical analysis on formalin-fixed, paraffin-embedded tissue from cases with intestinal lymphoma was performed at the Animal Health Diagnostic Center at Cornell University (Ithaca, NY). The detection of CD3+ and Pax5 as markers for T- and B-lymphocytes, respectively, was considered sufficient to yield a lineage diagnosis as previously described and validated. 17,43 Staining was performed using the Dakocytomation Auto Stainer plus according to the manufacturer’s instruction (Dako, Carpinteria, CA). Polyclonal rabbit anti-CD3+ (cat. A0452, 1:100; Dako) and monoclonal mouse anti-Pax5 (cat. 08-1411, prediluted; Invitrogen, Carlsbad, CA) primary antibodies were detected with streptavidin-biotin-peroxidase (LAB-SA) detection systems (Histostain SP Bulk Kit for Rabbit polyclonal antibodies [cat. 956143B] and Histostain SP Bulk Kit for Mouse monoclonal antibodies [cat. 956543B]; Invitrogen) and the chromogen 3,3′-diaminobenzidine-tetra hydrochloride (DAB; Dakocytomation, Carpinteria, CA). Antigen retrieval was done by digestion with pepsin for 20 to 45 min for CD3+ and by microwaving in EDTA (pH 8.0) for 20 minutes for Pax5. Slides were counterstained with hematoxylin (Dakocytomation). Negative controls were performed by substituting the primary antibody with nonspecific mouse anti–cat or nonspecific rabbit anti–cat antibodies, respectively (Negative mouse cat [N1698] and Negative rabbit cat [N1699]; Dako). Slides were read and lineage diagnosis achieved by a board-certified pathologist (S.P.M.).
Fluorescence In Situ Hybridization
Fluorescence in situ hybridization (FISH) of formalin-fixed, paraffin-embedded sections was conducted as described previously, with minor adjustments to optimize deparaffinization and staining of the tissue. 17,30 Briefly, histological sections of 4 μm were mounted on Probe-On Plus slides (Fisher Scientific, Pittsburgh, PA). Slides were heat-pretreated in an incubator (20 minutes, 60°C) and sections subsequently deparaffinized and rehydrated by passage through xylene (3×, 30 minutes), 100% alcohol (2×, 7 minutes), 95% ethanol (1×, 7 minutes), and 70% ethanol (1×, 7 minutes) prior to being mounted with DNA probe mix for hybridization (Integrated DNA Technologies, Coralville, IA). For assessment of bacterial numbers and spatial distribution, the eubacterial probe EUB-338 (GCT GCC TCC CGT AGG AGT) 5′-labeled with Cy3 was applied simultaneously with the irrelevant probe non-EUB-338 (ACT CCT ACG GGA GGC AGC) 5′-labeled with 6-FAM to control for nonspecific hybridization. Probe specificity was controlled by including positive control slides prepared from cultured E. coli DH5α and Streptococcus bovis in each assay.
The spatial distribution of bacteria was determined at 40× magnification according to the presence or absence of bacteria in the following regions: luminal (in the luminal cellular debris), mucus (in the villus-associated mucus), adherent (adherent to the apical surface of villous enterocytes), invasive (in the lamina propria), intravascular (in the lumen of mucosal blood vessels), or serosal (adherent to the serosal surface). Furthermore, the density of colonization was measured by semiquantitative evaluation of bacterial number in each area of colonization: no bacteria (0), 1 to 3 bacteria (1), 4 to 6 bacteria (2), and >6 bacteria (3) per microscopic field.
Statistical Analysis
The validity of the histopathological evaluation of biopsies assigned to the normal and LPE groups (mild, moderate, and severe) was performed by determining the agreement (κ) between evaluations performed at the separate time points. 21 Correlation of histopathology and FISH was subsequently performed for the 4 groups: NMC, LPE, SCL, and LCL.
Qualitative differences in the spatial distribution of bacteria between study groups were compared by use of a 2-tailed Fisher exact test, and results were considered significant when at P < .05. For analysis of bacterial numbers at the different locations (debris, mucus, adherent, invasive, serosal, vascular), the same semiquantitative numerical FISH scores were used (no bacteria [0], 1–3 bacteria [1], 4–6 bacteria [2], and >6 bacteria [3]). One-way analysis of variance (ANOVA) was used to compare median values of the surrogate categories of the different groups at the same location. If the Kruskal-Wallis test was significant, all of the groups were compared with Dunn’s test to see where the differences between groups lie. Results were considered significant when at P < .05. Statistical analyses were performed using GraphPad Prism version 4.00c for Macintosh (GraphPad Software, San Diego, CA).
Results
Characterization of the Study Group
Histopathology reports from 551 cases of gastrointestinal biopsies were reviewed for inclusion in the study, of which 198 cases were excluded. Twenty-two cases of NMC, 179 cases of LPE, and 152 cases of alimentary lymphoma (136 SCL and 16 LCL) were eligible for inclusion in this study. Of these, 25 cases of LPE and 32 cases of low-grade SCL were randomly chosen for inclusion. Cases of LPE were considered only if they were surgical full-thickness biopsies. Samples from this group were subselected to ensure an even distribution of mild, moderate, and severe LPE grades for each year between 2007 through 2011. Cases of low-grade SCL were subselected to ensure an equal number of cases each year from 2007 through 2011. All NMC and high-grade LCL cases were included in the study.
Three cases originally diagnosed as LPE moved to the SCL group after reevaluating HE-stained and immunohistochemistry sections. One case originally considered an early lymphoma lesion was reclassified as severe LPE upon review and was excluded from subsequent analysis as the sample was acquired endoscopically and did not meet the inclusion criteria for the LPE group. Of the LCL group, 15 were characterized as high-grade lymphoma, 1 as low-grade lymphoma, and 1 as a mixed large and small cell lymphoma. One case of lymphoma with an ambiguous pathology report that was selected as a presumptive SCL was recategorized as LCL. Two cases included in the LCL group were reclassified as low-grade LCL and mixed small and large cell lymphoma, respectively.
Blinded histopathological reevaluation of archival samples selected for inclusion in this study revealed that assigning cases to the normal or mild LPE groups on the basis of repeated histopathology was not reliable (κ = 0.6508). As 50% of the variation was due to inconsistency in differentiating mild LPE from normal, we combined these 2 groups into a group termed NMC. Within the LPE group, there were 14 samples categorized as moderate and 6 as severe, but these were analyzed together because of limited numbers in the severe category. Consequently, following histopathological review, the study groups were NMC (n = 18), LPE (n = 20), SCL (n = 33), and LCL (n = 17). The demographic, histopathological, and immunohistochemical features of the study groups are shown in Supplemental Table S1.
Histopathology and Immunohistochemistry
All 33 cases of SCL were low-grade T-cell epitheliotrophic lymphomas. Of the 17 LCLs, 15 (88%) were high grade and 1 was low grade. Seven LCLs (44%) stained positive for Pax5 and 8 (50%) for CD3; neither antigen was detected in 1 case. The 1 case of mixed LCL-SCL had 2 distinct appearances. A population of small CD3+ T cells populated the villi and effaced the villous lamina propria, compatible with low-grade SCL T-cell SCL. A second, distinct population of large CD3+ T cells with a high mitotic rate infiltrated the lamina propria between crypts, compatible with a high-grade T-cell LCL. Pax5-positive cells were clustered between the crypts and compatible with the normal B-cell population in this portion of the intestinal mucosa. Mucosal ulceration was present in 2 cases of SCL (6%) and 15 LCL (88%).
Presence and Spatial Distribution of Mucosal Bacteria Determined by FISH
This study employed culture-independent eubacterial FISH to determine the presence, abundance, and spatial distribution of bacteria in archived formalin-fixed, paraffin-embedded tissue using previously validated methods. 17 Slides of cultured Streptococcus and E. coli were included with every batch of tissue sections to control for permeabilization and hybridization. Biopsy specimens were examined for red fluorescent structures with bacterial morphology (EUB-338; 5′-labeled with Cy3) that did not exhibit green fluorescence (irrelevant probe non-EUB-338, 5′-labeled with 6-FAM) and were DAPI positive (DNA stained blue). EUB-338–Cy3 bacteria were visualized in formalin-fixed, paraffin-embedded sections from all samples.
The presence of bacteria in luminal cellular debris, in the mucosa, and in blood vessels could be assessed in every case (NMC, n = 18; LPE, n = 20; SCL, n = 33; LCL, n = 17). In 2 cases of SCL and 8 cases of LCL, bacteria in villus-associated mucus and adherent to the epithelium could not be assessed due to mucosal ulceration (SCL, n = 31; LCL, n = 9). Serosa was present and could be assessed as a spatial category in most surgical samples and samples originating from necropsies (NMC, n = 13; LPE, n = 17; SCL, n = 19; LCL, n = 14).
The presence or absence of bacteria in different spatial compartments is summarized in Table 1. Bacteria were more frequent within luminal debris in SCL (P < .01) and LCL (P < .001) than NMC and more frequent within luminal debris in LCL (P < .01) than LPE (Figs. 1, 2). Luminal cellular debris was more distant from the epithelium of cases of NMC and LPE with intact epithelium and mucus layer than cases of LPE, SCL, and LCL with inflamed, eroded, or ulcerated mucosa. Bacteria in mucus were found significantly more often in LCL than LPE (P < .01). The frequency of bacteria adherent to the villus epithelium did not differ between groups (Fig. 1). Mucosal-invasive bacteria were most frequently observed in cats with LCL (Figs. 3, 4). Intravascular bacteria were observed solely in LCL (Fig. 5), and serosal colonization was more common in LCL than SCL, NMC, and LPE (Fig. 6, Table 1).
Spatial Distribution of Bacteria in Small Intestinal Samples of Cats.

Fisher’s exact test vs NMC: *P ≤ .05, **P ≤ .01, ***P ≤ .001; vs LPE: # P ≤ .05, ## P ≤ .01, ### P ≤ .001; vs SCL: ^ P ≤ .05, ^^ P ≤ .01, ^^^ P ≤ .001. Luminal: in the luminal cellular debris; Mucus: in the villus-associated mucus; Adherent: adherent to the villous enterocytes; Invasive: in the lamina propria; Vascular: in mucosal blood vessels; Serosal: adherent to the serosal surface. LCL, large cell lymphoma; LPE, lymphoplasmacytic enteritis; NMC, normal to minimal change; SCL, small cell lymphoma.
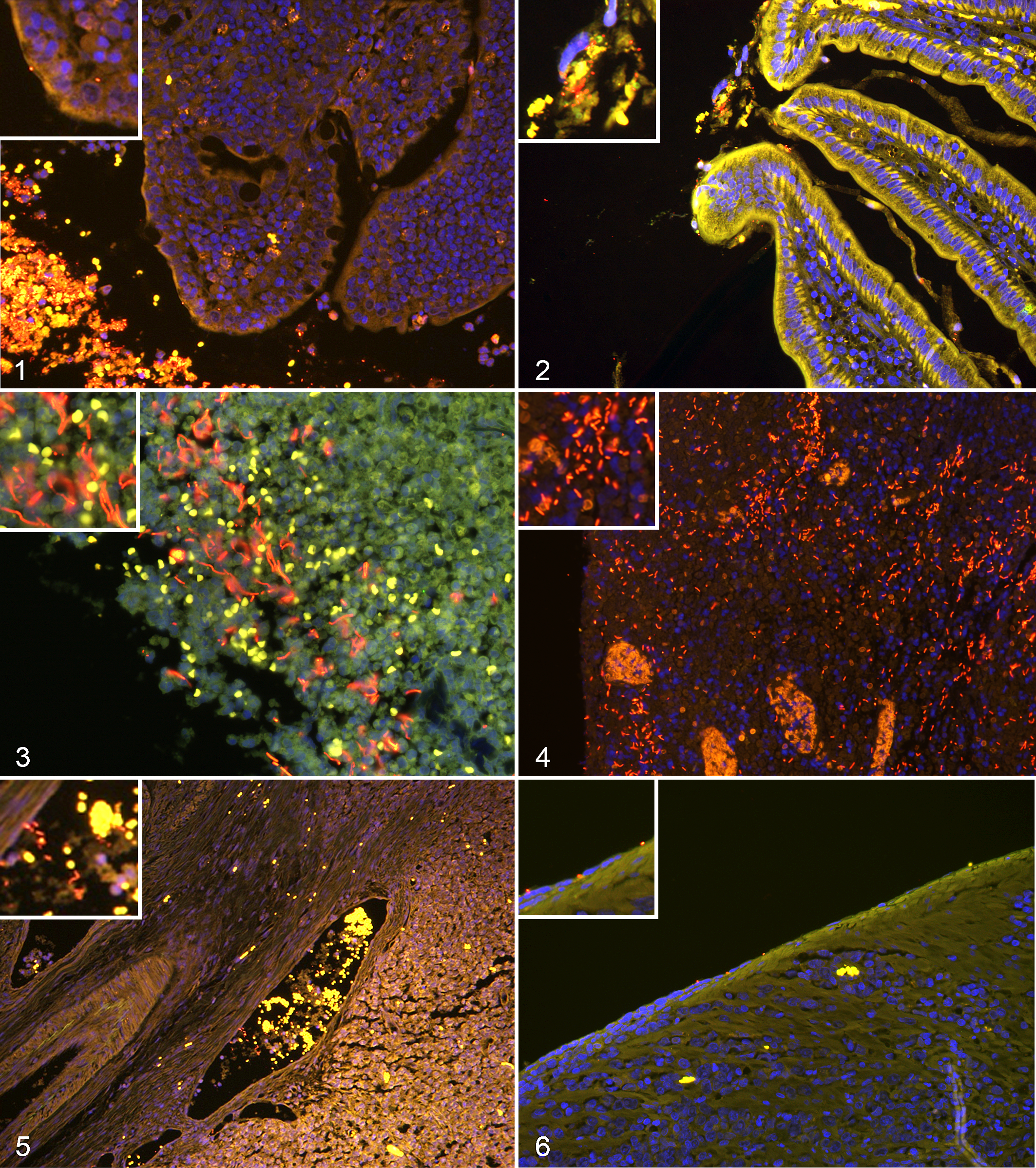
The distribution of mucosal bacteria according to lymphoma cell lineage yielded similar results to the distribution according to grade because the T-cell lymphoma group was dominated by SCL (33/41, 80%). Invasive bacteria were observed in only 1 case of LPE, which had severe disease (grade 3). Invasive bacteria were present in 6 nonulcerated SCLs (19%), nonulcerated SCLs, 2 nonulcerated LCLs (100%), and 12 ulcerated LCLs (80%).
Differences in Number of Mucosal Bacteria Determined by FISH
Quantitative analysis of mucosa-associated bacteria yielded similar results to the presence or absence of results presented above (Table 2). More bacteria were present in luminal debris of LCL than any other group; SCL had significantly more bacteria in luminal debris than NMC. Significantly more bacteria were found in villus-associated mucus of LCL than LPE. Mucosa-invasive bacteria, intravascular bacteria, and serosa-adherent bacteria were more numerous in LCL than in any other group. Intravascular bacteria were more frequent in B- than T-cell LCL with an average of 1.7 vs 0.3 bacteria, respectively, but the small number of cases per group precluded statistical analysis of this relationship.
Median Numbers of Bacteria Within Different Spatial Compartments in Small Intestinal Samples of Cats.
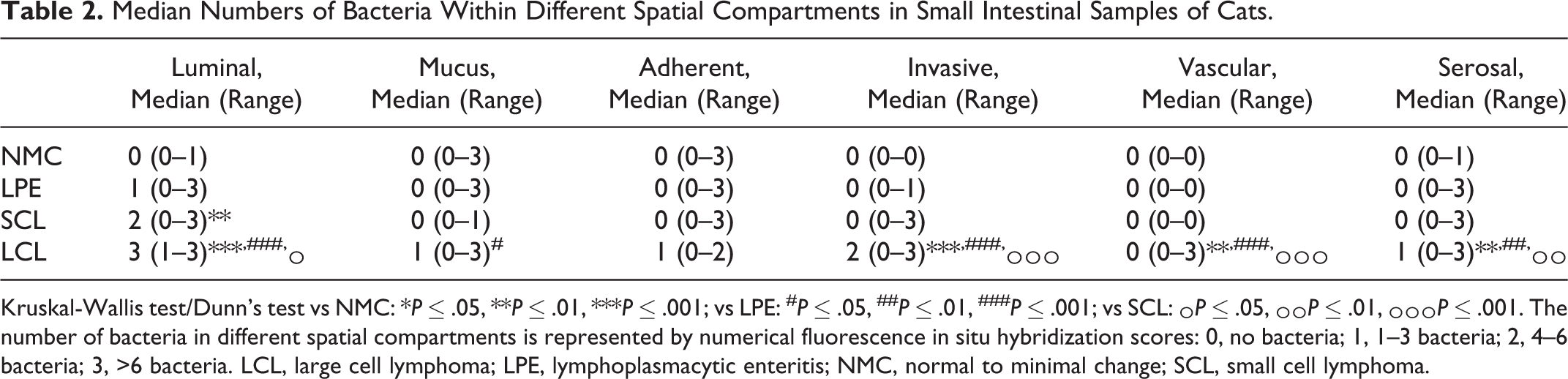
Kruskal-Wallis test/Dunn’s test vs NMC: *P ≤ .05, **P ≤ .01, ***P ≤ .001; vs LPE: # P ≤ .05, ## P ≤ .01, ### P ≤ .001; vs SCL: ^ P ≤ .05, ^^ P ≤ .01, ^^^ P ≤ .001. The number of bacteria in different spatial compartments is represented by numerical fluorescence in situ hybridization scores: 0, no bacteria; 1, 1–3 bacteria; 2, 4–6 bacteria; 3, >6 bacteria. LCL, large cell lymphoma; LPE, lymphoplasmacytic enteritis; NMC, normal to minimal change; SCL, small cell lymphoma.
Discussion
The causal association of persistent bacterial infections of the gastrointestinal mucosa with gastric carcinoma and mucosa-associated lymphoid tissue lymphoma in people and laboratory animals prompted us to examine the relationship of mucosa-associated bacteria to alimentary lymphoma in cats. We detected mucosa-invasive bacteria in 82% of cats with LCL and 18% with SCL relative to 3% of cats with NMC and LPE. The high frequency of bacteria within blood vessels (29%) and serosa (57%) of cats with LCL suggests these cats are at risk for bacterial translocation, septicemia, and peritonitis and should be considered in the clinical management.
Our finding that bacteria were more frequently present and more abundant in luminal debris in LCL and SCL likely relates to the extent of mucosal abnormality and enterocyte and tissue sloughing in these patients. Within the lymphoma group, there was a clear distinction between SCL and LCL in the presence of invasive bacteria and their localization to the vascular and serosal compartments. To our knowledge, the association of LCL with bacterial invasion has not been previously reported. The presence of bacteria in the vascular and serosal compartments of LCL is highly relevant to septicemia and septic peritonitis that have been reported in feline alimentary lymphoma before and after chemotherapy. 31,38,44 Sepsis in cats is difficult to diagnose antemortem, and our findings suggest that cats with LCL should be considered at risk of sepsis, and prophylaxis with antibiotics should be used prior to immunosuppressive chemotherapy. Studies in people with Crohn disease and rodent models of ileitis have shown that changes in the spatial distribution of bacteria within the intestine are related to the severity of inflammation and ulceration. 3,5 Unfortunately, case numbers for nonulcerated LCL and ulcerated SCL were insufficient for us to determine if the difference in invasive bacteria in LCL and SCL was due to the extent of mucosal damage or the cell phenotype. Thus, it remains unclear if invasive bacteria are a potential driving factor in the development of LCL or an opportunistic colonizer of damaged mucosa. The evaluation of intact intestinal segments from adjacent nonulcerated lymphomatous and nonlymphomatous intestine could potentially help to address this question. However, adjacent intestinal segments were not available for evaluation in this study due to its retrospective nature.
The similar number and distribution of mucosal bacteria in NMC (control) cats and those with LPE are consistent with the results of a previous FISH study of cats with gastrointestinal disease. 17 In this previous study, the numbers of Enterobacteriaceae, E. coli, and Clostridium spp correlated with upregulation of inflammatory cytokines and abnormalities in mucosal architecture, implicating mucosal bacteria in the etiopathogenesis of LPE. 17 The present study focused on the detection of eubacteria in general and did not evaluate the presence or distribution of different bacterial species, and thus it is not possible for us to comment on the presence or absence of dysbiosis as previously described in cats with LPE. 17 Future studies combining broad-range sequence-based analysis and FISH with specific probes would help to determine if distinct bacterial signatures are associated with alimentary lymphoma.
Lymphomas of B- and T-cell lineage have been associated with chronic gastrointestinal inflammation in response to infectious, chemical, and unknown stimuli, with bacteria such as H. pylori considered directly mutagenic. 2 The absence of invasive bacteria in LPE and in the majority of SCL cases does not preclude the involvement of bacteria in the inflammatory process or tumorigenesis. For example H. pylori associated MALT lymphoma and enteropathy-associated lymphoma in people with celiac disease are independent of bacterial invasion. 15,34,35
Allocation of patients into study groups (NMC, LPE, SCL, LCL) for FISH analysis was based on histopathologic evaluation of small intestinal biopsies, which is currently considered the gold standard to distinguish these entities. To standardize evaluation of biopsies from nonlymphoma cases, we employed an extended grading scheme based on the 10-category WSAVA scheme and evaluated each biopsy on 2 occasions. The histopathological severity of intestinal inflammation is used to inform therapy, with mild disease treated with a stepwise approach and more severe disease with multimodal therapy. 36 Because the WSAVA scheme provides no clear guidelines on how to determine severity, 6 we summed the scores for morphological features and inflammation to give an indication of severity, with a total score of 0 to 2 being considered a normal small intestine (0), 3 to 7 mild LPE (1), 8 to 13 moderate LPE (2), and >13 to 45 severe LPE (3). Using this approach, we could not reliably assign cases to the normal or LPE categories (κ = 0.6508). 14 As 50% of the variation was due to inconsistency in differentiating mild LPE from normal, we combined these 2 groups into a group termed NMC. Due to small sample size, we elected to combine cases with moderate or severe LPE. Because interobserver agreement is better when there are fewer categories, our strategy of collapsing 4 categories (normal, mild, moderate, and severe LPE) to 2 (NMC and LPE) may help to improve agreement between pathologists. 42 The use of summed WSAVA scores has previously been shown to underestimate the severity of granulomatous colitis in dogs. 25 Previous studies in cats with chronic enteropathies indicate that certain histopathological features correlate better with clinical signs than others; specifically, atrophy and fusion of villi correlate with clinical signs. 17 Thus, the development of standardized scoring schemes that differentially weight histopathological criteria based on their correlation with clinical severity and outcome may improve the utility of such schemes.
The ability of histological phenotype (small, large) to predict cellular lineage (T- and B-cell) in lymphoma is controversial. While it has been suggested that low-grade, small cell lymphomas might be mostly the T-cell immunophenotype and high-grade, large cell lymphomas more likely to be B-cell lymphomas, this correlation has been questioned, and to date it has not been proven that the histologic phenotype can predict lineage diagnosis in intestinal lymphoma. 13,31,44 We found that all cases with a small cell phenotype had a low mitotic index and T-cell lineage. In contrast, the large cell, high-grade lymphoma phenotype was split between the B- and T-cell lineage, with 44% positive for Pax5 and 50% positive for CD3. This observation raises the possibility that high-grade T- and B-cell LCL may be different diseases and differ in their etiopathogenesis. We were not able to dissect the relationships of immunophenotype and lymphoma cell phenotype to bacterial colonization because of co-segregation on the basis of cell morphology—that is, the T-cell group was dominated by SCL (33 of 41, 80%).
This study has several limitations. The search of our histopathological database during the years 2007 to 2011 yielded a total of 136 SCL and only 16 LCL cases. This distribution, along with the T-cell immunophenotype of SCL, parallels studies documenting a change in the anatomic distribution grade and immunophenotype of lymphoma in the post-FeLV age. 10,23,24,31,45 It is important to address the potential impact of selection bias on our study. Only the NMC and LCL groups represent the full spectrum of cases seen at the Section of Anatomic Pathology during the 2007 to 2011 period; the cases constituting the LPE and SCL groups were more numerous and subselected with regard to year sampled, method of sampling, and extent of histological lesions. Due to that selection bias, we decided not to formally compare demographic information between groups. While FISH using EUB-338 consistently enabled bacteria to be visualized in formalin-fixed, paraffin-embedded samples from all samples, this technique may have failed to detect bacteria with impermeant cells walls (eg, some Gram-positive and acid-fast bacteria that require specialized permeabilization for detection).
In conclusion, the present study shows that the spatial distribution of mucosa-associated bacteria varies depending on the type of alimentary lymphoma, with mucosa-invasive bacteria observed more often in cats with large cell lymphoma of any immunophenotype. An unexpected finding of this study was the similar prevalence of B- and T-cell immunophenotypes in high-grade LCL. Our finding that mucosa-invasive bacteria are common in cats with alimentary large cell lymphoma has potential implications for the pathogenesis of complications associated with LCL, particularly septicemia and septic peritonitis. Further studies are required to determine the role of mucosal bacteria in the etiopathogenesis of feline alimentary lymphoma.
Footnotes
Authors’ Note
The results of this study were presented in the form of a dissertation text to the Dissertation Committee of the Vetsuisse Faculty, University of Zurich, Switzerland, in order for S.N.H. to attain the title of Dr med vet.
Partial results of this study were presented as an oral abstract at the 2012 ACVIM Forum, New Orleans, Louisiana, and the 2012 Comparative Gastroenterology Society Meeting, Winter Park, Colorado.
Acknowledgements
We thank Francis Davis for technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
