Abstract
In horses, immune-mediated muscle disorders can arise from an overzealous immune response to concurrent infections or potentially from an inherent immune response to host muscle antigens. Streptococcus equi ss. equi infection or vaccination can result in infarctive purpura hemorrhagica (IPH) in which vascular deposition of IgA-streptococcal M protein complexes produces ischemia and complete focal infarction of skeletal muscle and internal organs. In Quarter Horse–related breeds with immune-mediated myositis, an apparent abnormal immune response to muscle antigens results in upregulation of major histocompatibility complex class (MHC) I and II on muscle cell membranes, lymphocytic infiltration of lumbar and gluteal myofibers, and subsequent gross muscle atrophy. Rarely, an inflammatory event results in myositis with subsequent systemic calcinosis characterized by a pathognomonic hyperphosphatemia and high fatality rate. This review presents an overview of these immune-mediated myopathies and highlights clinical and pathological features as well as the suspected pathophysiology.
Most forms of myodegeneration in horses, such as exertional myopathies, result in infiltration of muscle cells by macrophages. 52,54 Inflammatory cells such as polymorphonuclear cells (PMNs) and lymphocytes are rarely observed in common equine myopathies. The notable exception to this rule is inflammatory myopathies. Inflammatory myopathies can arise from infection or aberrant immune responses. In horses, Streptococcus equi is one of the primary infectious agents that incite acute rhabdomyolysis, 46 infarctive purpura hemorrhagica (IPH), 24 and immune-mediated myositis (IMM) 10,28 in horses. Other infectious agents, however, are capable of inducing an inflammatory response within equine skeletal muscle, but the specific agent or the immune or inflammatory basis for the myositis is not well characterized. 20,35,38 Myofibers are unique in that unlike most other tissues in the body, they do not express detectable major histocompatibility complex class (MHC) I or II on the sarcolemma. 2,31 In this review, those inflammatory muscle disorders best known to be a result of an abnormal immune response (IPH, IMM, and systemic calcinosis) will be described.
Infarctive Purpura Hemorrhagica
Infarctive purpura hemorrhagica is a form of vasculitis associated with purpura hemorrhagica that is so severe it results in muscle and other organ infarction. 24 Affected horses usually have a history of Streptococcus equi ss equi (S. equi) infection; however, other bacterial and viral agents have been implicated, as well as vaccination against S. equi. 38
Signalment and Clinical Features
There is no known sex or age predilection for IPH, but it has most frequently been reported in Quarter Horses. 24 Early clinical signs of infarction include focal firm painful swellings in those muscles that contact the ground when horses are lying down, particularly the pectoral, hind limb adductor, and gaskin muscles. 24 Painful infarctions produce severe lameness and muscle stiffness. 24 Variable signs of classic purpura hemorrhagica, including depression, petechiae and ecchymosis, and moderate to severe well-demarcated limb edema, may be present. 38 With progression of infarction and involvement of multiple organs, oral ulcerations, colic, and hemorrhagic gastric reflux often arise. Once colic signs occur, the presence of multiple necrotic gastrointestinal lesions is likely, and this has a strong negative impact on prognosis.
Diagnosis
Hematologic and biochemical parameters are often similar to cases of purpura hemorrhagica and include leukocytosis with a neutrophilia, left shift and toxic changes, hyperglobulinemia, and hypoalbuminemia. 24,38 Unlike horses with purpura hemorrhagica that typically have only mild elevations in creatine kinase (CK <3000 U/L), 38 horses with IPH typically have marked elevations in CK (>47,000 U/L) and aspartate aminotransferase (AST >960 U/L). 38 Similar to horses with purpura hemorrhagica, those with IPH usually have marked elevation in serum antibodies for S. equi M protein (SeM). 24
Antemortem diagnosis relies on clinical signs, hematology, biochemistry, serum antibody titers to SeM protein, and potentially ultrasound-guided muscle biopsies of infarcted muscle.
Pathophysiology
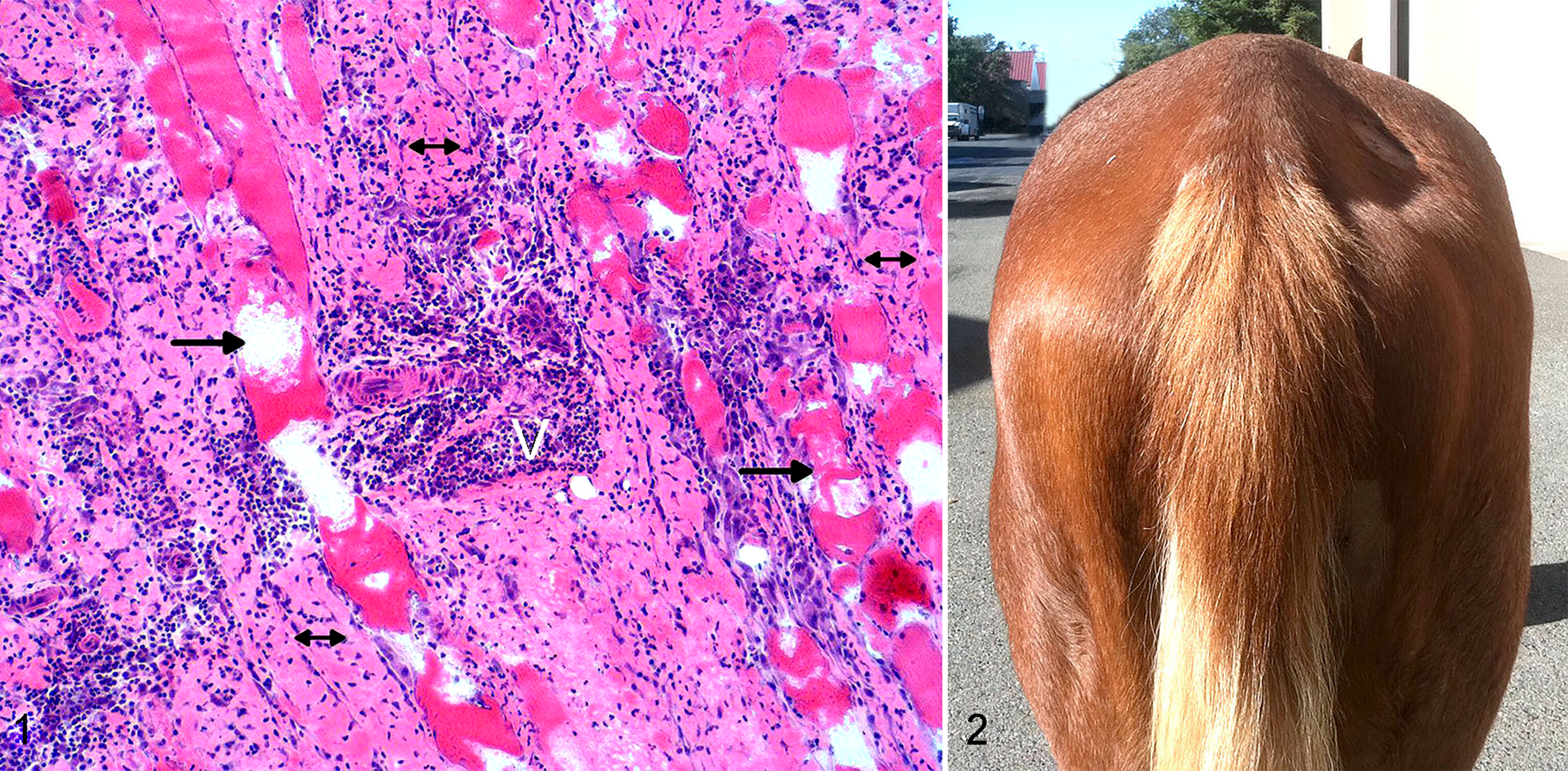
Horses with IPH usually have had an infection within the past month, often with S. equi or a strangles vaccination. 24 In susceptible horses, the immune response results in high antibody titers to the SeM protein (>1:1600) and SeM-IgA immune complexes. 15,47 It is thought that a type III hypersensitivity reaction leads to vasculitis, extravasation of albumin, and edema. 30 In horses with IPH, deposition of complement around immune complexes in vessel walls leads to profound leukocytoclastic vasculitis, vascular occlusion, tissue ischemia, and infarction (Fig. 1) in a fashion similar to Henoch-Schönlein purpura in humans. 15,24,41 Henoch-Schönlein purpura predominantly affects children 16 and is thought to be secondary to a bacterial infection, particularly streptococcal infection, viral infection, or a drug reaction. It is characterized by high levels of circulating IgA immune complexes and high C3d concentrations. 26 Patients with Henoch-Schönlein purpura develop skin rashes, nephritis, arthritis, abdominal pain, and gastrointestinal hemorrhage; however, infarction of skeletal muscle is rarely present. 41,45 Equine muscle infarction may be one of the first signs of IPH because of the high likelihood of ischemia in inflamed vessels that are being further compressed and occluded by the weight that muscles bear when horses are in recumbency.
Treatment and Prognosis
Without early recognition and treatment with high-dose corticosteroids, IPH cases are invariably fatal. 17,24,38 However, high doses of dexamethasone (0.04–0.2 mg/kg) have been used successfully to treat IPH. 38 Horses with IPH that develop colic due to gastrointestinal infarction have a particularly poor prognosis. 24 By comparison, severely affected patients with Henoch-Schönlein purpura, even those with gastrointestinal infarctions, can be successfully treated with immunosuppressive doses of methylprednisolone, followed by cyclophosphamide and azathioprine. 33,55 If concurrent active S. equi infection is suspected, then appropriate antimicrobials should be administered. 38
Pathology
Gross postmortem evaluation reveals multifocal well-demarcated areas of hemorrhage in skeletal muscle as well as petechial and ecchymotic hemorrhages of the oral mucosa and gastrointestinal serosa and mucosa. 24 Gross evidence of infarction in numerous other organs as well as abscessation of retropharyngeal and/or submandibular lymph nodes caused by S. equi is usually evident. 24
Leukocytoclastic vasculitis is the primary histopathologic feature of purpura hemorrhagica in the dermis. 24,38 IPH is further typified by leukocytoclastic vasculitis of multiple internal organs and by characteristics of muscle infarction, including vacuolation, loss of cross-striations, cytoplasmic eosinophilia, mineralization, proliferation of satellite cells, nuclear karyolysis or pyknosis, and necrosis of whole muscle fascicles (Fig. 1). 24 Necrotic regions contain perivascular inflammatory cells, primarily degenerate neutrophils, lymphocytes, plasma cells, and macrophages. 24
Immune-Mediated Myositis
Signalment and Clinical Features
Equine immune-mediated myositis is an inflammatory myositis of predominantly Quarter Horses and related breeds (Paints, Appendix horses, and Appaloosas), 28 although it has been reported in other breeds. 3 In Quarter Horses, there is a predilection for certain bloodlines, 28 and the suspected genetic basis is supported by the identification of a genetic locus associated with the phenotype through a genome-wide association study. 12 Affected horses are usually 8 years and younger or 17 years and older; there is no sex predilection. 28 Of affected horses, 39% have a history of being recently exposed to a “triggering factor” such as S. equi infection, a respiratory virus, 28 or vaccination (S. J. Valberg, 2015, unpublished observation).
Dramatic muscle atrophy of predominantly the epaxial and gluteal muscles is the most prominent clinical sign (Fig. 2). 28 In 1 pony with IMM, focal and symmetrical atrophy of the cervical muscles was observed. 3 The muscle atrophy is often accompanied by stiffness and nonspecific malaise. The atrophy is rapid and can involve 40% of the gluteal and epaxial muscles within 48 hours and usually persists for months. 11 Severely affected horses may develop generalized weakness and frequent episodes of recumbency.
Diagnosis
Hematologic parameters are usually normal, and mild to severe elevations in serum CK and AST activities are present in most affected horses. 10
Transcutaneous biopsy of affected muscles is considered the most useful diagnostic test for horses with active IMM. 28 Formalin-fixed Trucut samples or fresh-frozen Bergstrom needle biopsies of the epaxial and/or gluteal muscles are usually sufficient to establish a clinical diagnosis. The classic diagnostic finding is varying degrees of lymphocytic infiltration into myocytes, and in 76% of cases, there are lymphocytic cuffs surrounding vessels (Figs. 3, 4). 10 Scattered MHC I (Figs. 5, 6 and Suppl. Fig. S1a) and II staining is present in 81% and 71% of horses with active IMM, respectively. 10 The inflammatory infiltrate in 48% of horses is predominantly CD4+ T lymphocytes and in 28% of cases predominantly CD8+ T lymphocytes (Suppl. Fig. S1b,c). 10 Other inflammatory cell types present include CD20+ B cells (Suppl. Fig. S1d) and macrophages. 10 Major histocompatibility complex class I and CD8+ lymphocyte scores are positively correlated. 10 Consistent IgG staining on the epimysium/sarcolemma is not a common feature in the muscle of IMM horses. 28 Other muscle biopsy findings include myogenic atrophy characterized by anguloid atrophy and evidence of regenerating myofibers characterized by basophilic staining of the cytoplasm and central nuclei. 28 Findings in the semimembranosus or semitendinosus muscles can be absent; if present, they are usually similar to but less severe than the gluteal or epaxial muscle lesions, and in several cases, no abnormalities are present. 28
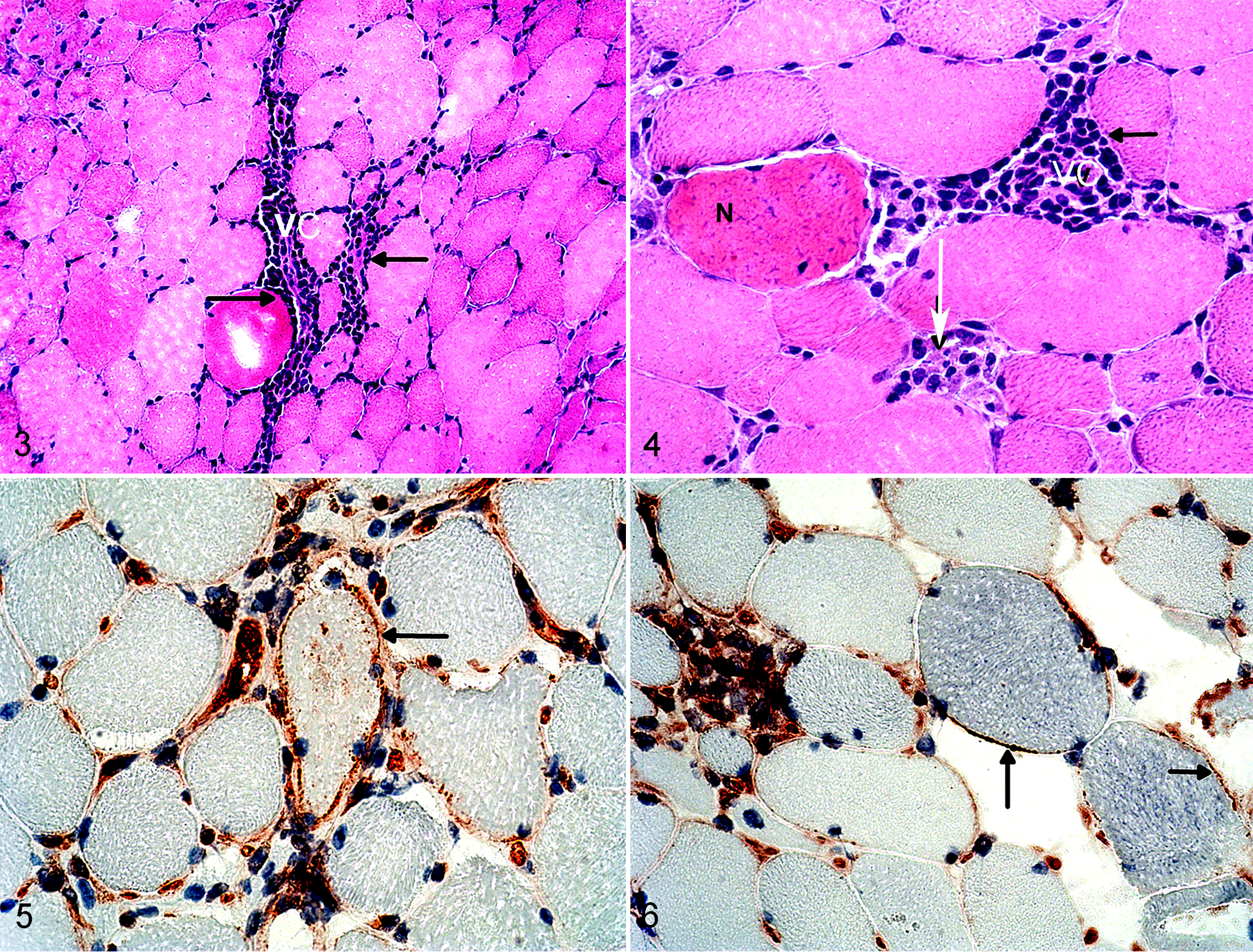
Muscle biopsies of horses in later stages of IMM, when profound atrophy has been present for weeks, may lack diagnostic inflammatory infiltrates and present only with evidence of myogenic atrophy and regeneration, making a specific diagnosis difficult.
Pathophysiology
Equine IMM has several features in common with human and canine immune-mediated myopathies. 37,44 Equine IMM is rarely generalized, and there is a predilection for the gluteal and epaxial muscles while other muscles such as the semimembranosus and semitendinosus muscles are relatively unaffected. 10,28 This is similar to canine masticatory myositis (CMM), which only affects the masticatory muscles. Autoantibodies to a unique myosin heavy chain isoform only located in the masticatory muscles have been identified in dogs with CMM, and the inflammation in affected muscle biopsies is predominantly localized to the type 2 M fibers. 13,58 Quarter Horses have a high proportion (>80%) of myosin heavy chain type 2A and 2X fibers in their gluteal muscle compared with other muscles such as the semimembranosus muscle. 53 However, type 2A and 2X fibers are not unique to the gluteal muscle, and this therefore does not completely explain the predilection for the gluteal and epaxial muscles. Specific binding of IgG to myofibers has not been identified in affected epaxial or gluteal muscles of horses with IMM. 28 Thus, the reason for the localized atrophy is unknown.
Muscle is unusual in that it does not express MHC I or II when healthy. 2,21,25 Major histocompatibility complex class II expression has been shown to be highly specific for immune-mediated myopathies. 23 The expression of MHC I or II on the sarcolemma of humans 4 and dogs 32,34 with immune-mediated myopathies is typical of but not exclusive to an immune-mediated muscle disorder. Selected muscle fibers of horses with IMM have MHC expression, supporting an immune-mediated basis for the muscle atrophy. 10 Upregulation of myofiber MHC is sometimes present in dystrophin-deficient myopathy, 2 some limb-girdle muscular dystrophies, 20 dysferlin deficient myopathy, 8 and fibers of horses that contain abnormal polysaccharide. 10 Horses with active IMM are neither dystrophin nor dysferlin deficient, and they are not consistently affected by polysaccharide storage myopathy. 10 Major histocompatibility complex expression in horses with IMM is presumed to be due to immune activation. The cause of the MHC upregulation in immune-mediated myopathies is not fully understood but has been shown to occur in the presence of cytokines, 57 particularly interferon-γ.
The immune-mediated basis for equine IMM could be due to changes in the normal state of T-cell tolerance or a loss of self-tolerance and activation of autoreactive T cells. Loss of self-tolerance has been shown to occur due to (1) “molecular mimicry,” where epitopes of an infectious agent are highly similar to self-peptide, (2) the release of superantigens by an infectious agent, or (3) high concentrations of cytokines, which can also contribute to proteolysis and muscle catabolism. 9 Almost 40% of horses with IMM have a history of recent respiratory disease 28 or vaccination (S. J. Valberg, 2015, unpublished observation), and although no research has been published on the underlying mechanism, it is likely that exposure of genetically susceptible horses to appropriate environmental conditions could lead to an episode of IMM. The high prevalence of IMM in Quarter Horses of particular bloodlines 28 and the results of a genome-wide association study support an underlying genetic basis for IMM in Quarter Horses. 12
Treatment and Prognosis
Anti-inflammatory doses of corticosteroids 3 for approximately 1 month combined with antibiotics if infection is present are often successful in halting muscle atrophy. 54 Muscle mass will regenerate over weeks to months, and horses can make a full recovery. Almost 50% of horses will have recurrent episodes of muscle atrophy. 28
Pathology
There are no descriptions of gross lesions in horses with IMM. 3,28
Systemic Calcinosis
Systemic calcinosis is a fatal myositis that likely has an immune-mediated component. 14,49 This syndrome could well be underdiagnosed because muscle biopsies are often not a diagnostic priority due to the presence of a myriad of other serious systemic perturbations.
Clinical Features
The initial clinical presentation is similar to IMM with signs of malaise, mild pyrexia, stiffness, and muscle atrophy, particularly of the gluteal and epaxial muscles. 49 Horses with systemic calcinosis, however, develop further signs of mild ventral edema and respiratory distress, intestinal infarction, laminitis, renal disease, or progressive weakness depending on which tissue has the most severe calcification. 14,49 There does not appear to be a sex predilection, but all reported horses have been young (9 years or younger) Quarter Horses or related breeds. 14,49
Diagnosis
Hematologic parameters vary, but most affected horses have an inflammatory leukogram, characterized by a leukocytosis with a neutrophilia and hyperfibrinogenemia. 14,49 The pathognomonic finding on serum biochemistry is hyperphosphatemia with a product of the serum calcium (Ca) concentration (mg/dl) multiplied by the serum phosphorous (P) concentration (mg/dl) of >65. 11,49 Other biochemistry findings depend on the affected calcified tissues but include azotemia, hypoalbuminemia, and substantial elevations in serum CK and AST activities. 14,49
Pathology
The most common gross pathology findings are regions of pallor in the skeletal muscle, kidneys, heart, and lungs. A smaller number of affected horses had regions of pallor of the liver and foci of elastin fiber calcification of the tunica media of arteries. 14,49
Skeletal muscle histopathology is characterized by diffuse anguloid atrophy, centrally located nuclei, loss of striation, acute necrosis of scattered myofibers, and marked dystrophic calcification of myofibers (Fig. 7) particularly evident on staining with Von Kossa. 49 Gluteal muscle is more severely and diffusely affected than semimembranosus muscle from the same horse. 49 Marked macrophage infiltration of myofibers, regenerative fibers, multinucleated giant fibers, and a mononuclear vasculitis are more variable features of muscle tissue (Fig. 7b). 49
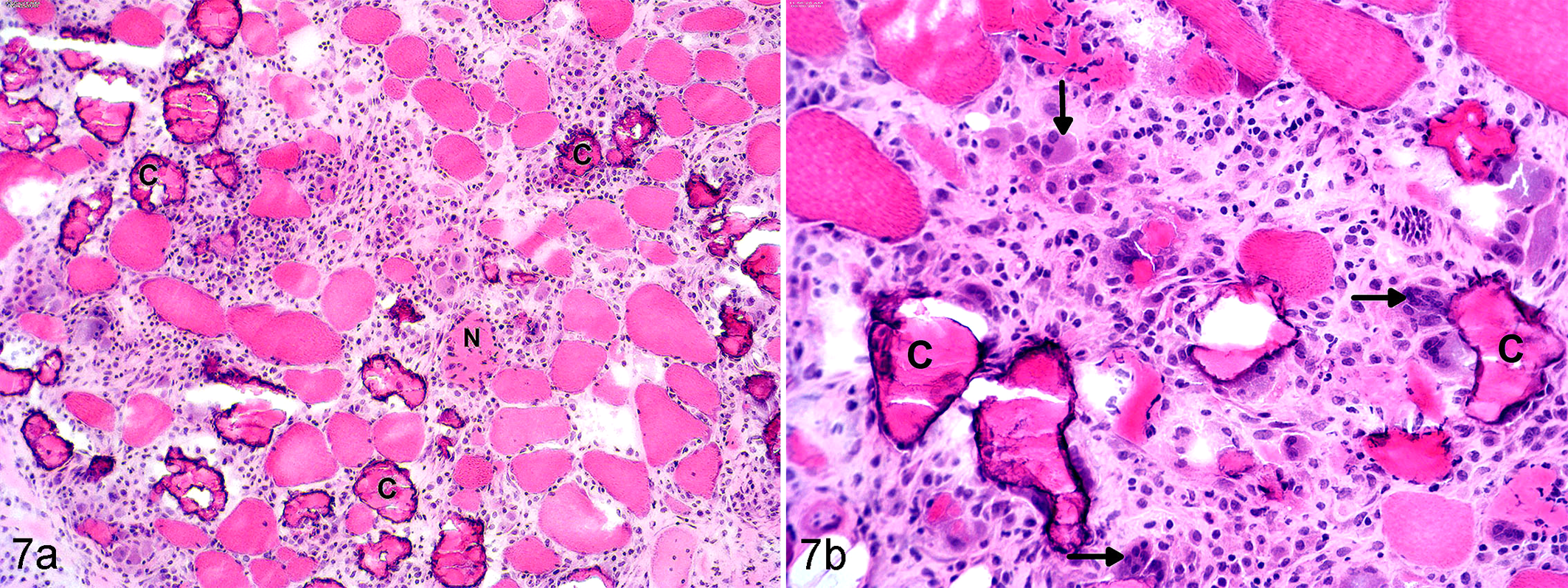
Systemic calcinosis, gluteal muscle, horse. (a) Marked dystrophic calcification of myofibers (C), acute necrosis (N), and fibrosis. Hematoxylin and eosin (HE). (b) Higher power view shows calcified fibers (C) surrounded by fibroblasts, macrophages, a few polymorphonuclear cells, regenerative basophilic myotubes (vertical arrow), and multinucleated giant cells (horizontal arrow). HE.
Cardiac muscle is characterized by swollen, pale myocytes with reduced cross-striations and evidence of fragmentation with accumulation of basophilic calcified debris. 49 Dystrophic calcification of the endothelium of the aorta and aortic sinuses, as well as fibrous connective tissue surrounding arteries, can be present. 14,49 In the lung, calcification of the primary bronchioles, alveoli, bronchiolar musculature and cartilage, and alveolar walls has been identified in some horses. 14,49 All horses with systemic calcinosis have been reported to have mineralization of the kidney (glomeruli, renal tubules, and/or collecting ducts). 14,49 Less common findings included renal tubular necrosis and coagulative necrosis of the renal cortex and medulla, vessel thrombosis, and renal interstitial infiltration of macrophages, lymphocytes, and plasma cells. 49 Other tissues affected by dystrophic calcification included the intestine and liver. 14,49
Pathophysiology
The elevated Ca × P product of >65 has been used in humans as an indicator of the risk for developing dystrophic calcification. 6 Calciphylaxis occurs in humans, most frequently in patients on renal dialysis for end-stage renal disease. 40 Horses with systemic calcinosis are typically healthy prior to the onset of clinical signs, and without a history of lethargy, weight loss, and polydipsia, it is unlikely that they have preexisting chronic renal failure. 43 Notably, almost half of horses with chronic renal failure typically develop hypophosphatemia 42 rather than the hyperphosphatemia that develops in human patients with calciphylaxis. 40
The reason for the elevated Ca × P calculation in horses with systemic calcinosis is unknown but is thought to occur due to the presence of inflammation. 11 Although the presence of tumor necrosis factor (TNF)–α alone is insufficient to promote osteoclastogenesis, the addition of the receptor activator of nuclear factor kappa B ligand (RANKL) leads to synergistic effects with TNF-α and production of multinucleated osteoclasts, leading to the syndrome known as inflammatory osteolysis. 27 The synergistic effects of TNF-α and RANKL and subsequent bone resorption may be sufficient to lead to the hyperphosphatemia seen in patients with systemic calcinosis. Of the 6 horses reported to have systemic calcinosis, 3 had a recent history of respiratory disease, 1 horse had suspected anaplasmosis, 1 horse had concurrent salmonellosis, 1 horse reacted to an immunostimulant, and 1 horse had a 6-month history of muscle atrophy and was being treated for IMM prior to the development of systemic calcinosis. 14,49 It is possible that these concurrent diseases may have been trigger factors that led to inflammation and the development of hyperphosphatemia. 49
Hyperphosphatemia can lead to dystrophic calcification through 4 different mechanisms 29,49,56 : (1) supersaturation of the blood, leading to passive calcium phosphate deposition; (2) conversion of smooth muscle cells to osteogenic cell types through an active process; (3) elevated phosphorous concentration, leading to increased parathyroid hormone secretion; and (4) high phosphorous concentrations interfering with renal production of 1,25-dihydroxyvitamin D. Ultimately, the dystrophic calcification leads to multiorgan failure. 29
Another cause of hyperphosphatemia is vitamin D toxicosis, but animals with vitamin D toxicosis typically have concurrent hypercalcemia and gastrointestinal hemorrhage. 36 None of the reports of horses with systemic calcinosis had consistent hypercalcemia or a history of exposure to excessive vitamin D. 14,49
Treatment and Prognosis
The prognosis is poor based on the available literature, with no reports of affected horses surviving. 14,49 Attempted treatments include supportive care, particularly with fluid therapy, antimicrobial therapy for any primary bacterial infections, and anti-inflammatory medications, including corticosteroids. Corticosteroids are contraindicated in humans with hyperphosphatemia due to upregulation of RANKL, leading to increased bone resorption and potential worsening of the hyperphosphatemia. 22
Uncharacterized Immune-Mediated and Inflammatory Myopathies
There are several reports of inflammatory myopathies secondary to infectious agents or unknown causes that are not well characterized. Quarter Horses can develop acute onset of rhabdomyolysis concurrent with S. equi infection. 39,46 A brief mention of a horse with multifocal muscle atrophy with histologic changes in muscle biopsies consistent with IMM was reported in a review article. 5 A small number of horses from non–Quarter Horse breeds have been evaluated with a history of acute or chronic muscle atrophy and lymphocytic infiltrates in affected muscle biopsy specimens. 11 The gluteal and epaxial muscles were predominantly affected, but other muscle groups were reported to be involved. 11 The underlying pathophysiology of disease in these horses is not understood.
Infectious agents such as Toxoplasma and Neospora are important causes of inflammatory myopathy in humans 19 and dogs. 13 The most common infectious agents identified in the muscle of horses are Sarcocystis bertrami, Sarcocystis equicanis, and Sarcocystis fayeri, with dogs as a definitive host. 50 The sarcocysts are typically considered incidental findings and are not associated with inflammatory infiltrates. 11 However, there are reports of myositis secondary to Sarcocystis infection. 1,18,51 Clinical signs include general malaise, anorexia, weakness, asymmetric atrophy of some muscles, and symmetric atrophy of gluteal, epaxial, and quadriceps muscles. 1,18,51 Hematologic findings and serum biochemistry profiles can be normal, 51 whereas other cases have had a systemic eosinophilia. 18 Diagnosis is based on muscle biopsy findings of sarcocysts within myofibers with concurrent evidence of an inflammatory reaction into myofibers, specifically infiltration of lymphocytes, macrophages, and in some cases eosinophils. 18,51 Although MHC staining has been reported on the surface of sarcocysts in dogs, no MHC class I or II sarcolemmal staining has been identified on myofibers in affected muscle biopsies. 48 The reason that some horses develop a profound inflammatory response and atrophy in response to infection by Sarcocystis sp. is not known, although immunosuppression, exposure to large numbers of sporozoan parasites in canine feces, 7 concurrent disease, 1 and/or a genetic predisposition, particularly when multiple individuals in a family are affected, 18 are suspected. Treatment with antiprotozoal drugs, including pyremethamine and trimethoprim-sulfa, was successful in 1 case, 51 and treatment with ponazuril improved clinical signs in another case. 18 However, a third case did not respond to aggressive treatment with sulfamethoxazole and trimethoprim and anti-inflammatory drugs. 18
Conclusions and Further Directions
Inflammatory myopathies cause morbidity and in some cases mortality in horses. Abnormal or excessive immune system activation is important in the pathophysiology of all equine inflammatory myopathies. A combination of individual, environmental, and genetic factors contributes to the development of clinical disease. A recent history of respiratory disease, other bacterial or viral disease, and vaccination are clearly important risk factors for all of the inflammatory myopathies described here. Early and accurate diagnosis through muscle biopsy evaluation allows for the initiation of appropriate treatment and the best clinical outcome in cases of IPH and IMM. Systemic calcinosis carries a grave prognosis. Overall, there is considerable potential for further investigation into these important diseases, including exploration of autoantibodies in horses with IMM and investigating any potential genetic association in horses with IMM.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
