Abstract
A fatal epizootic of salmonellosis occurred in farmed juvenile American alligators in Louisiana. Six animals were examined. Gross lesions included severe fibrinonecrotizing enterocolitis, necrotizing splenitis, coelomic effusion, and perivisceral and pulmonary edema. Microscopic examination revealed severe necrotizing enterocolitis and splenitis with intralesional bacteria and pneumocyte necrosis with fibrin thrombi. Salmonella enterica serovar Pomona was isolated from intestine and lung. Clinical salmonellosis is a rare finding in reptiles and salmonellosis caused by S. Pomona is not previously reported in American alligators. Since S. Pomona is a commonly isolated Salmonella serotype from patients with reptile-associated salmonellosis in the United States, and since alligator meat is consumed and the skin is exported to numerous countries, risk of human and animal infection should be considered.
Salmonella enterica serovar Pomona (S. Pomona), a highly pathogenic serotype, can cause bloody diarrhea, cramps, fever, and occasionally death in humans, especially in children. S. Pomona has been reported as a cause of human reptile-associated salmonellosis in the United States and elsewhere. Turtles have been identified as a major reservoir of S. Pomona in these outbreaks in the United States. 1
Salmonella spp were more commonly isolated from cloacal swabs of indoor farmed alligators (20%) than from wild alligators (2.8%). 10 Prevalence of Salmonella spp in captive crocodiles (Crocodylus porosus and Crocodylus johnstoni) has also been investigated at crocodile farms in Australia. Salmonella Singapore, Salmonella Enteritidis, Salmonella Cerro, Salmonella Poona, and S. enterica subsp arizonae were most commonly isolated from cloacal and fecal swabs from crocodiles. 8 Here, we describe the pathologic findings of S. Pomona infection in farmed juvenile American alligators.
In July 2015, 6 juvenile 11-month-old American alligators were presented with a history of decreased appetite and 4 of these animals died within 48 hours. The remaining 2 animals were euthanized. Decreased appetite was observed throughout the affected building. All animals were approximately 11 months old. Overall, mortality occurred in about 50 out of approximately 5000 animals housed in a single indoor building. Of the approximately 50 000 animals housed at the time, no other buildings at the farm had reported morbidity or mortality. Animals in the affected building had been handled to grade the quality of their skins 48 hours prior to the onset of clinical signs. Alligators were fed a dry, commercially available alligator feed.
At necropsy, all 6 alligators had similar gross lesions. There was severe perivisceral edema and clear effusion in the coelomic cavity. The small and large intestinal mucosa had multifocal to coalescing or diffuse necrosis and appeared thickened (up to 4 mm), opaque, brown, and gray (Fig. 1). The intestinal content was diffusely scant, malodorous, and watery. The spleen was enlarged (2 × 1.5 × 1 cm) and soft; on the cut surface, there were coalescing pale areas of necrosis affecting 80% of the parenchyma (Fig. 2). The lungs were heavy and full of clear fluid, an indication of severe edema.
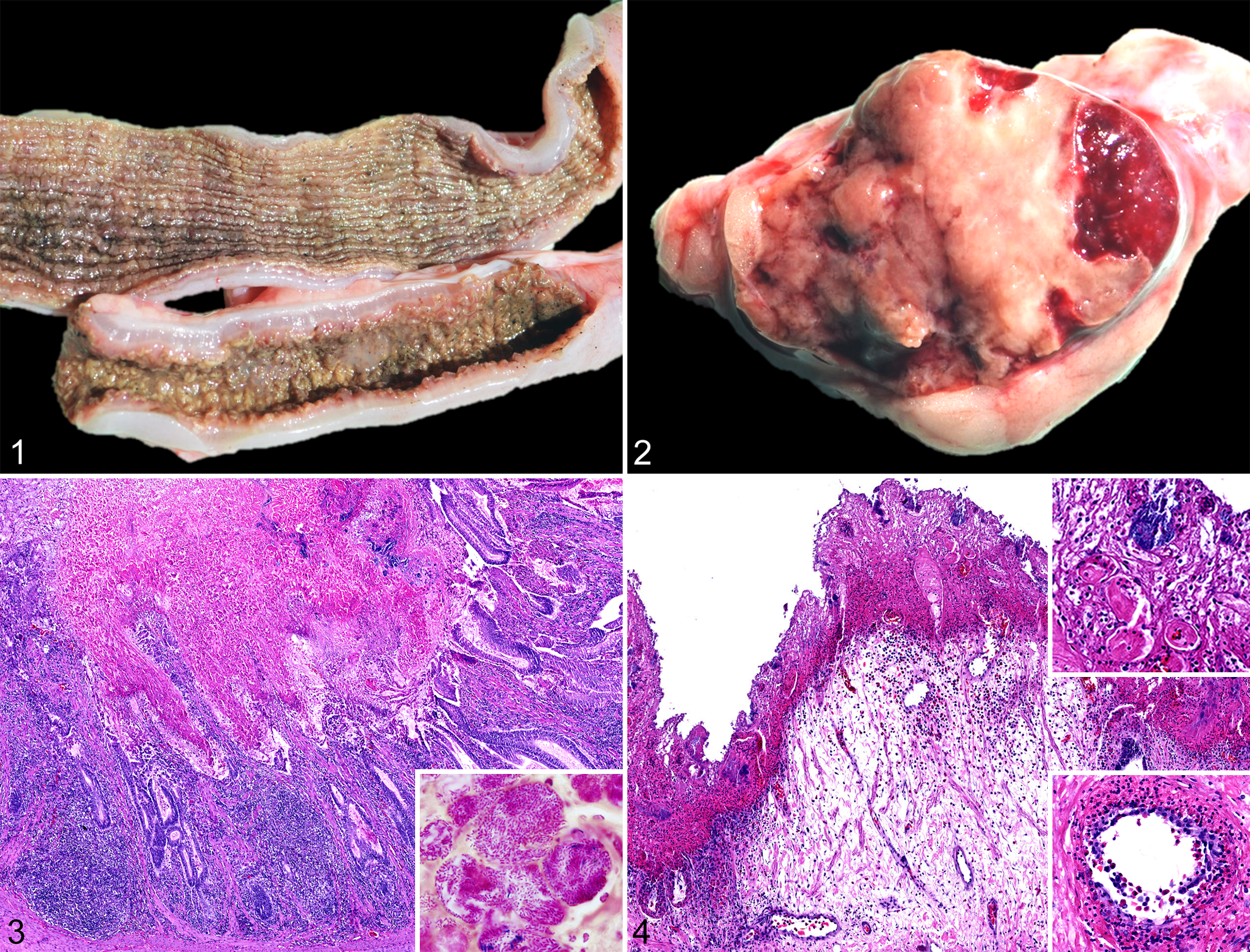
Tissue samples from all organs were collected from the 2 euthanized animals (case Nos. 1 and 2) and fixed in 10% neutral buffered formalin, and histologic sections were prepared routinely and stained with hematoxylin and eosin. Gram, acid-fast, and Gömöri methenamine silver stains were also used. Samples examined included encephalon, spinal cord, nerves, eyes, skin, skeletal muscle, tonsil, tongue, heart, lung, trachea, esophagus, liver, pancreas, spleen, stomach, small and large intestines, cloaca, adrenal glands, kidneys, bones, and tail.
Histologically, there was severe diffuse coagulative necrosis in the small and large intestine mucosa (Figs. 3, 4). The necrotic mucosa was multifocally effaced by degenerate and necrotic heterophils and colonies of short gram-negative rods (Fig. 3 inset). There were also scattered gram-negative and gram-positive rods, some of them forming spores, and a minority of gram-positive cocci. The remaining lamina propria was edematous and mildly infiltrated by heterophils, lymphocytes, and plasma cells; heterophils extended into the underlying tunica muscularis. In the large intestine, numerous vessels were infiltrated and effaced by large numbers of heterophils (Fig. 4). Multiple intravascular fibrin thrombi were also noted in the affected superficial mucosa of the large intestine (Fig. 4). The lymphoid follicles of the lamina propria and submucosa contained abundant pyknotic and karyorrhectic cellular debris, macrophages, and heterophils. Approximately 80% of the splenic parenchyma was affected by extensive coagulative necrosis (Supplemental Fig. 1). The necrotic areas were demarcated by hemorrhage and moderate infiltration of degenerate and necrotic heterophils with intralesional predominantly gram-negative and fewer gram-positive rods. The pulmonary faveolar spaces were filled with eosinophilic amorphous fluid, fibrin, and hemorrhage with moderate infiltration of heterophils and macrophages (Supplemental Fig. 2). There was multifocal faveolar epithelial necrosis and loss, with multiple fibrin thrombi within the subepithelial capillaries (Supplemental Fig. 2). The liver had minimal to mild infiltration of heterophils and lymphocytes and rare plasma cells in the portal areas in both alligators. No acid fast positive bacteria or mycetes were noted histologically in any of the examined tissues.
Fresh samples of lung, small intestine, and spleen from 2 euthanized animals (case Nos. 1 and 2) were submitted for Salmonella culture. In alligator No. 1, the lung, liver, kidney, and small intestine were submitted for aerobic culture, and small intestine for anaerobic culture. For Salmonella spp isolation, approximately 1 gram of tissue was prepared and inoculated into 9 to 10 ml of primary enrichment broth, Selenite broth, or Tetrathionate broth/Iodine-iodide solution. Following the primary enrichment, the media was swabbed onto xylose lysine deoxycholate agar (XDL, Hardy Diagnostics) plates and then incubated at 35°C to 37°C overnight. The plates were examined for the presence of Salmonella: black or black-centered colonies were selected for testing and then identified as Salmonella using polyvalent agglutination serum Salmonella O Antiserum Poly A-1 and Vi (Becton Dickinson). If Salmonella was present, the organisms were subcultured to nutrient agar and referred to the National Veterinary Services Laboratory for serotyping. Salmonella spp were isolated from the intestine of both alligators and from the lung of animal No. 1. No suspect Salmonella colonies were identified in the spleen of either alligator or the lung of alligator No. 2. Serotyping of the isolate identified Salmonella enterica serovar Pomona. Enterococcus sp and Escherichia coli were also isolated from intestine. Aerobic and anaerobic cultures were otherwise negative. Tests of liver, lung, small intestine, and conjunctiva for West Nile virus and Chlamydiaceae via polymerase chain reaction 2,5 were negative.
Asymptomatic infection with Salmonella is common in reptiles, and several serotypes of Salmonella have been isolated from many reptilian species. Clinical salmonellosis is rare. Salmonella enterica subsp enterica (serogroup I), salamae (II), arizonae (IIIa), diarizonae (IIIb), and houtenae (IV) have been isolated from various species of snakes, chelonians, and lizards kept as pets and from zoological gardens in Croatia. 7 Reptiles are known to be asymptomatic carriers. Nonetheless, salmonellosis has been reported in various reptiles. In crocodilians, an outbreak of salmonellosis was associated with mortalities in hatching Nile crocodiles (Crocodylus niloticus) and a single case of hemorrhagic ulcerative colitis caused by S. choleraesuis in a Nigerian captive Nile crocodile. 4,9 Pathological descriptions are very limited in these articles.
As observed in cases of Salmonella enteritis in mammals including pigs, horses, and calves, the alligators in this case had severe necrotizing enterocolitis with numerous intralesional gram-negative bacilli, edema, hemorrhage, vasculitis, and lymphoid necrosis. There was extensive necrosis of the spleen as well as pulmonary thrombosis with pneumocyte necrosis. S. Pomona was isolated from the intestine and in pure culture from the lung, indicating systemic distribution. Microbacterium (gram-positive bacilli), Enterococcus faecalis (gram-positive cocci), Aeromonas hydrophila (gram-negative rods), and E. coli (gram-negative rods) have been most commonly isolated from cloacal swabs collected from 29 randomly captured wild Nile crocodiles in Botswana. 6 Therefore, the Enterococcus sp and E. coli isolated from intestine and the gram-positive cocci and rods microscopically observed in the present cases were considered innocent or opportunistic bacteria.
Animals in intensely managed production settings can be more susceptible to stressors such as cold, excessive heat, sudden temperature changes, and high stocking density. 3 In this case, mortality was approximately 1% of those in the group. The fact that no other building on the farm had affected animals suggests that this was an isolated incident. Management practices were the same for all buildings. The only event specific to the animals in the affected building is that they were handled to grade the quality of their skins 48 hours prior to the onset of clinical signs. It is not uncommon for alligators to have decreased appetite for a few days after being handled but mortality events after handling are rare.
To the authors’ knowledge, there have been no previous reports of salmonellosis caused by S. Pomona in American alligators and this is the first clinical report of S. Pomona infection in any species of crocodilian. In the United States, S. Pomona is a commonly isolated Salmonella serotype from patients with reptile-associated salmonellosis. Although prevalence of S. Pomona among American alligators is uncertain, alligator meat and skin is a potential source of S. Pomona infection in humans.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
