Abstract
Following the performance of a superovulation protocol, multiple nodules were observed bilaterally in the uterine horns of 31 of 276 (11.2%) C57BL/6 J female mice aged 8.5 ± 0.6 (mean and standard error of mean) weeks. These lesions prevented embryo collection, and the uterine decidual reaction was suspected. Samples of pathological uteri (n = 20) and the normal genital tracts of donors treated with a similar superovulation protocol (control group, n = 10) were collected. Immunohistochemistry was performed to evaluate pancytokeratin, desmin, vimentin, progesterone receptor (PR), estrogen receptor α (ERα), Ki-67, cyclin D3 and c-Myc expression, as well as quantitative polymerase chain reaction to assess cyclin D3, Hoxa-10 and heparin-binding epidermal growth factor-like growth factor (HB-EGF) mRNA expression. The uterine decidual reaction presented a high degree of structural organization and specifically affected the antimesometrial region of the endometrium. The abnormal decidual cells were large polygonal cells that were frequently polyploid or binucleated and strongly positive for desmin. Immunohistochemistry showed higher Ki-67 proliferation index and higher expression of PR and cyclin D3 in decidual cells in the antimesometrial aspect of the endometrium, compared to nondecidualized endometrial stromal cells in the mesometrial aspect of affected uteri, and compared to endometrial stromal cells in healthy uteri. High expression of cyclin D3 and Hoxa-10 mRNA was also observed in uteri affected by the decidual reaction. These results suggest that PR overexpression in endometrial stromal cells, likely due to high progesterone levels, triggers cyclin D3 and Hoxa-10 overexpression, which may be involved in the pathological mechanisms of the mouse uterine decidual reaction.
Superovulation is a technique that is widely used in transgenic facilities in the production of genetically engineered mice. This method artificially induces the ovulation of large numbers of oocytes from a limited number of female mice, reducing the number of animals used. The technique of superovulation was described for the first time by Fowler and Edwards in 1957. 19 The standard protocol used a hormonal injection of pregnant mare serum gonadotropin (PMSG) and an injection of human chorionic gonadotropin (HCG) in immature female mice aged 4 to 5 weeks. Thereafter, the female mice were mated and checked the following morning for copulation plugs; the embryos were collected 2.5 days later.
Responses to the superovulation protocols varied depending on the mouse strain 5,28 and the female donor’s age. 25 When female mice exceeded the optimal age for superovulation, a lower ovulatory response was observed; the standard protocol requires improvement to maximize its productivity. The most common protocol consists of the administration of a higher hormone dosage than that normally used for younger mice. 25,28
In the physiological establishment of pregnancy, the uterine decidual reaction is critical in some species, such humans and rodents, and the stimuli that initiate the process are different among these species. In humans, decidualization begins near the end of the secretory phase of the menstrual cycle via progesterone (P4) effects. If pregnancy is unsuccessful, menstruation occurs; however, if pregnancy develops, the decidual reaction continues as the human conceptus is implanted. 14,21 In mice, the stimulus for decidualization is the implanting blastocyst on gestation day (gd) 4.5 (gd 0.5, vaginal plug positive commonly used to timed pregnancy in mice). 1,7,34 Decidualization consists of the extensive proliferation and differentiation of endometrial stromal cells into decidual cells, extracellular matrix remodeling, angiogenesis, and changes in immune cell populations at the site of embryo implantation. 1,11,14 Some decidual cells show characteristic polyploidy, caused by the transition from the mitotic cell cycle to an endoreduplication cycle (endocycle), in which cells undergo a repeated round of DNA replication without successive cell division. It has been speculated that genomic endoreduplication increases the number of gene copies for transcription, ensuring a higher protein synthesis than that needed by the growing embryo. 7,11,40
A similar, but not identical, pattern of uterine decidual reaction can be initiated by artificial stimuli mimicking embryo implantation, triggering the decidual reaction in the endometrium. Early studies have indicated that this event can occur in mice on day 3 or 4 of pseudopregnancy by exposure to progesterone at least for 48 hours, followed by a small amount of estrogen to stimulate uterine progesterone receptor expression, which is required for decidualization. 11,12,27 Traumatic deciduogenic stimuli using a needle scratch were used in the past to induce the decidual reaction in mice; it appears that the physical stimulus induces prostaglandin (PG) secretion in the endometrium that mimics conceptus implantation. 24 Presently, the infusion of sesame oil or blastocyst-sized ConA-coated sepharose beads, placed into the uterus on day 2.5 of pseudopregnancy, is employed to induce the decidual reaction in mice. 2,20,23,31 In contrast, the rodent spontaneous endometrial decidual reaction is an infrequent pathological process that has been reported in rats 13,16,42 and in 134-week-old female mice. 10 Cases of the pathological decidual reaction have also been reported in other mammals, such as humans 18 and monkeys. 15
Grossly, the decidual reaction has been described in rats as unilateral or bilateral solitary or multiple uterine nodules and, occasionally, as diffuse horn enlargement that collapses the uterine lumen. 12,16 Histopathologically, the decidual reaction is characterized by stromal cell proliferation and the transition to decidual cells and presents a high degree of structural organization showing distinct areas. 16,42 There is a paucity of information related to the immunohistochemical characterization of the rodent decidual reaction; only 1 study 42 has described the immunohistochemical patterns in 2 spontaneous uterine deciduomas in rats.
The pathogenesis of the uterine decidual reaction remains elusive. Genetic and molecular studies have noted the activation of diverse cell cycle mechanisms in the decidualization process of stromal cells in mice. 11,17,20,31 Previous investigations have demonstrated that cyclin D3, a G1-phase cell cycle regulator, is upregulated at the site of implantation in the mouse uterus 8,37,39 and plays an important role in functional interactions with cyclin-dependent kinases (cdks), such as cdk4 and cdk6, to induce cell proliferation, differentiation and the development of polyploidy during stromal cell decidualization in mice. 7,8 Heparin-binding epidermal growth factor-like growth factor (HB-EGF), a member of the EGF superfamily, is one of the earliest known molecular mediators of implantation in mice and humans, 9,35 which promotes stromal cell polyploidy via the upregulation of cyclin D3. Likewise, Hoxa 10, a member of the Hox gene family, is highly expressed in the mouse uterine decidualization of stromal cells and is controlled by progesterone (P4) and progesterone receptor (PR) expression. 4,7,26,30 The null mutation of Hoxa-10 in mice causes female infertility by severe decidualization defects, mainly due to the reduced stromal cell responsiveness to P4, 3 which may be partially rescued by the overexpression of cyclin D3, triggering other genes involved in mice decidualization. 37
Therefore, the main goal of this study was to analyze the histopathological and molecular expression patterns, using RT-qPCR and immunohistochemical studies, of the uterine decidual reaction observed in a superovulation protocol treatment in young-adult female donors. Second, we sought to elucidate the possible pathogenic mechanisms of this process, which, to our knowledge, have not been described.
Materials and Methods
Animals
Two hundred seventy-six female mice in a C57BL/6 J homogeneous genetic background, aged 8.5 ± 0.6 (mean and standard error of mean) weeks, were superovulated between 2013 and 2014. In 31 of 276 (11.2%) donors, bilateral multifocal thickening of the uterine horns was observed. This thickening obliterated the endometrial lumen and prevented embryo extraction. The mice were housed in a specific pathogen-free (SPF) barrier zone at the Barcelona Biomedical Research Park (PRBB), an AAALAC International Accredited Unit, under a 12:12-h light:dark cycle at temperatures of 21–24°C. The animal care and experimental protocols were approved by the Institutional Animal Use and Care Committee (IACUC) of PRBB.
Superovulation and Mating
Female mice were injected with 7.5 IU of PMSG (MSD Animal Health, Salamanca, Spain) and 7.5 IU of HCG (DFV; Divasa Farmavic Group; Barcelona, Spain) 47 hours later (at 13 hours, 5 hours during the light cycle). Immediately after HCG administration, the donor mice were mated 1:1 to male mice of the same strain. We checked female mice for copulation plugs at 08:00 the following morning. Positive female mice were considered pregnant at post copula day 0.5. The female donors were euthanized by cervical dislocation 2.5 days later, and their complete genital tract was removed. The oviducts were placed into a culture dish, and embryos (E3) were collected by oviduct perfusion using sterile-filtered M2 medium (M7167 Sigma-Aldrich, Madrid, Spain) suitable for mouse embryos.
Collection of Samples and Pathological Analysis
Samples of ovaries, oviducts and uterine horns from pathological females (n = 20), aged 8.4 ± 0.7 weeks, and from the normal female genital tracts of donors treated with a similar superovulation protocol (control group, n = 10), aged 8.7 ± 0.5 weeks, were collected and fixed in 4% buffered formalin, dehydrated in increasing concentrations of ethanol, embedded in paraffin, sectioned at 4-µm thickness and stained with hematoxylin and eosin and periodic acid-Schiff (PAS). Uterine samples (mouse pathological group: n = 9; mouse control group: n = 10) were immediately placed in RNAlater® tissue collection (Applied Biosystems, Foster City, CA, USA) and were stored at –80°C for later RNA extraction.
Immunohistochemistry
Immunohistochemistry staining was performed using the streptavidin-biotin peroxidase complex method. Tissue sections were dewaxed, rehydrated through a series of graded volumes of ethanol and washed in distilled water (5 minutes). Sections were pretreated with 10 mM citrate buffer (pH 6.0) in a pressure cooker for antigen retrieval, cooled for 30 minutes at room temperature (RT) and washed twice in distilled water. The protocol was carried out as recommended by the manufacturer (NovoLink Polymer Detection System, Leica Microsystems, Newcastle Ltd, UK). Endogenous peroxidase activity was blocked (Peroxidase Block, 5 minutes, RT), and nonspecific binding was blocked by incubation with Protein Block for 5 minutes (RT). The protocols followed for each analysis are summarized in Supplemental Table 1. Thereafter, the sections were incubated (Novolink Post-Primary Block for 30 minutes, RT) and were developed with 3-3’ diaminobenzidine tetrachloride (DAB) for 5 minutes at RT. The sections were washed twice (5 minutes each) with TBS Tween, with gentle rocking to remove excess reagents from the specimens after each incubation step in the assay protocol. After a 10-minute wash in distilled water, the tissue sections were counterstained with hematoxylin, dehydrated in alcohol, cleared and mounted. For the negative control sections, an equivalent concentration of nonimmune mouse IgG or rabbit IgG antibody (Dakocytomation, BD Diagnostics, Sparks, MD, USA) was used instead of the primary antibody.
The glands in the antimesometrial and mesometrial aspects of the endometrium were considered different compartments and were analyzed separately. To evaluate pancytokeratin, desmin and vimentin expression, semiquantitative analysis was performed using the mean staining intensity score (0 = negative, 1 = weak, 2 = moderate, 3 = strong positive). The mean and standard errors of ERα, PR, Ki-67, c-Myc, and cyclin D3 immunopositive cells were determined by evaluating 500 cells in 5 randomly selected high-power fields (400× magnification) of each sample section. Images were obtained using a Vanox AHBS Olympus microscope with a DP75 camera.
Reverse Transcription Quantitative Polymerase Chain Reaction
Total RNA was extracted from tissue samples using the RNeasy mini kit (Qiagen, Venlo, Netherlands) following the manufacturer’s instructions. The samples were reverse transcribed in a GenAmp® PCR System 9700 (Applied Biosystems, Foster City, CA, USA) using an iScript™ cDNA synthesis kit (Bio-Rad Laboratories Inc, Hercules, CA, USA) following the manufacturer’s instructions. The resulting cDNA was stored at –20°C.
RT-qPCR was performed using the cyclin D3 (Assay ID: Mm01612362_m1), Hoxa 10 (Assay ID: Mm00433966_m1), HB-EGF (Assay ID: Mm00439306_m1), and GAPDH (Assay ID: Mm99999915_g1) genes in the uterine samples and primer sequences supplied by Applied Biosystems (Foster City, CA, USA). Each sample was analyzed 3 times, and the mean was calculated.
RT-qPCR was performed with a CFX96 Touch real-time PCR detection system using a C1000 Touch™ thermal cycler (Bio-Rad Laboratories Inc, Hercules, CA, USA) and the iQ™ SYBR® green supermix kit (Bio-Rad Laboratories Inc, Hercules, CA, USA). The cycling profile for each gene included denaturation (95°C, 3 minutes) followed by PCR amplification and quantification (95°C, 10 s; 60°C, 30 s), with the fluorescence measurement at specific acquisition temperatures for 5 s, repeated for 40 cycles. The melting program was 65–95°C at a rate of 0.5°C/s, with continuous fluorescence measurement. Relative mRNA expression, normalized to the levels of GAPDH, was quantified using Bio-Rad CFX Manager software (Bio-Rad Laboratories Inc, Hercules, CA, USA), with the results calculated using the difference in threshold cycle (2-ddCt) method.
Statistical Analysis
Statistical analysis was conducted using SAS® 9.4 (Statistical Unit of Complutense University, Spain). The Wilcoxon test was used to assess the difference between groups. Spearman correlation coefficient determination was carried out to measure the linear correlations between variables. The results were expressed as the mean ± standard error of mean (SEM), and statistical significance was accepted as a P value less than .05.
Results
Gross and Histological Findings
Grossly, pathological uteri of superovulated mice with the uterine decidual reaction were characterized by bilateral multifocal enlargement of the uterine horns, with multiple round nodules (Fig. 1) that collapsed the uterine lumen. The perimetrium and broad ligament were congested. The ovarian cortex had corpora lutea and follicles.
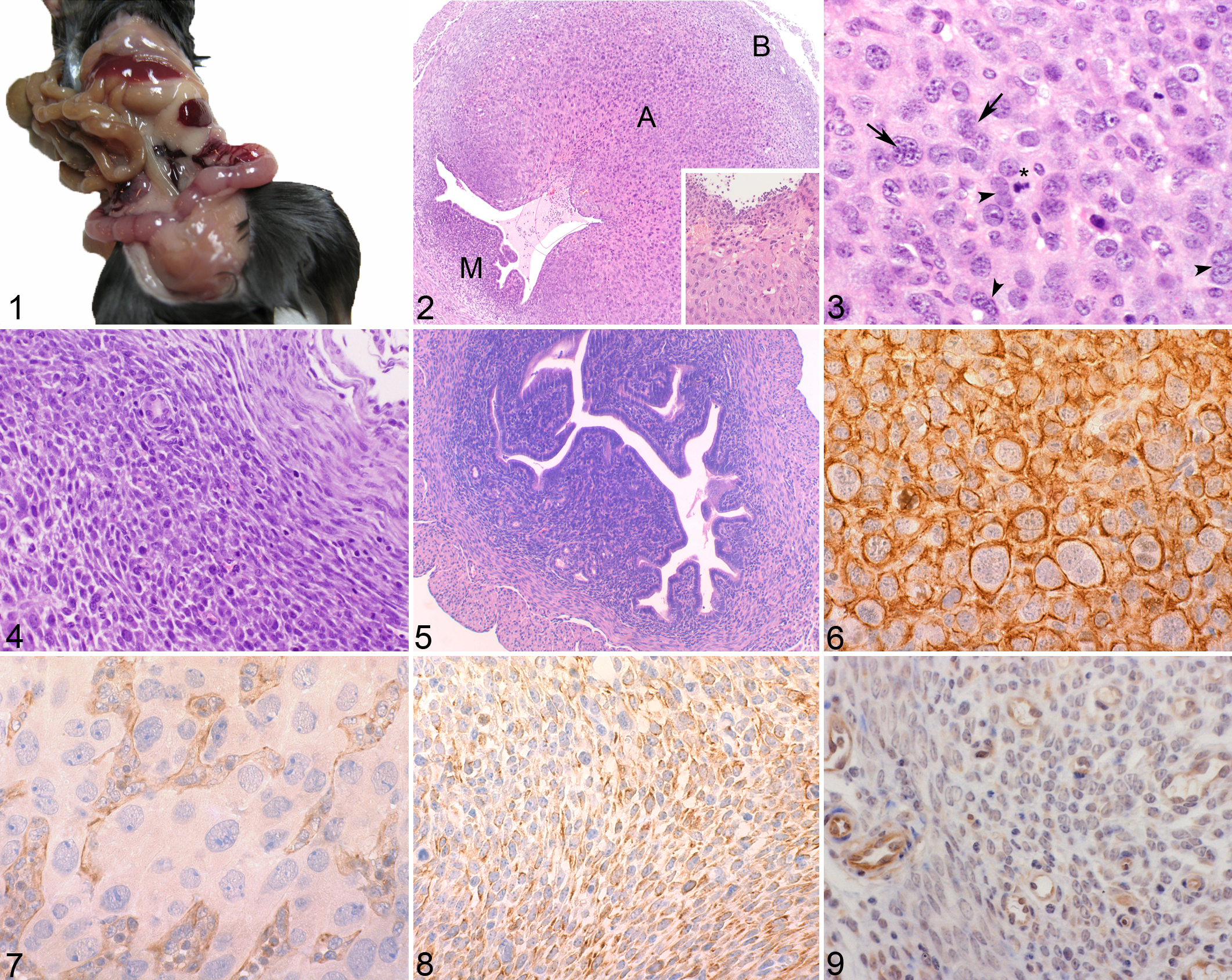
Histopathologically, the enlarged endometrium obliterated the uterine lumen, which contained cellular debris, and focal superficial epithelial erosion was frequently present (Fig. 2). Immediately under the superficial epithelium, a moderate acute diffuse inflammatory reaction with numerous viable and degenerate neutrophils was present (Fig. 2 inset). The endometrium had an intense stromal proliferative reaction. Three different areas of endometrium were distinguished: the basal, antimesometrial and mesometrial regions (Fig. 2). The outer area was the basal zone, which contacts the myometrium and comprised a narrow band of collagen fibers and fibroblasts, together with the endometrial gland remnants. The antimesometrial region comprised densely packed large polygonal vacuolated cells with round vesicular nuclei. Frequently, binucleate cells and bizarre polyploid nuclei were observed. These cells were identified as deciduated endometrial cells (Fig. 3). A rich network of capillaries was arranged among the decidual cells. The mesometrial region comprised 2 cell populations—loosely packed spindle cells with prominent cytoplasmic processes and eosinophilic cytoplasm and scattered round cells with central oval nuclei (Fig. 4). In addition, a few large lymphocytes with prominent PAS-positive cytoplasmic granules and round nuclei were present (uterine natural killer cells). Histological analysis of the ovaries and oviducts of pathological female donors showed no lesions.
In the control group (n = 10) of superovulated mice that did not develop the uterine decidual reaction, the uteri were lined by a simple columnar epithelium. The endometrium comprised spindle and round stromal cells (Fig. 5), vascular tissue, endometrial glands, and some migratory lymphocytes. However, neutrophils were more numerous in control uteri that had a mild to moderate multifocal acute inflammatory reaction.
Immunohistochemical Findings
The immunohistochemical characterization of uterine cells is summarized in Table 1. In the antimesometrial area, deciduated cells were strongly positive for desmin (Fig. 6) and negative for pancytokeratins and vimentin (Fig. 7). Endothelial cells of capillaries had intense vimentin (Fig. 7) and mild pancytokeratin immunostaining. In the mesometrial area, mild to moderate vimentin staining was present in stromal (Fig. 8) and endothelial cells, and the latter also had mild pancytokeratin staining. The gland cells, in the basal area, were moderately to intensely positive for pancytokeratins.
Immunochemical Characterization of Uterine Cells in Superovulated Mice With the Uterine Decidual Reaction (Pathological Group) and Control Mice Without the Uterine Decidual Reaction (Control Group).

aPan-CK, pan-cytokeratins 4/5/6/8/10/13/18.
In the control group (superovulated mice without the decidual reaction), there was mild vimentin immunostaining in endometrial stromal cells (Fig. 9), which were negative for desmin and pancytokeratins. Capillary endothelial cells had intense immunoreaction to vimentin (Fig. 9); mild reaction to pancytokeratins, and negative reaction to desmin. Finally, the gland cells, in the basal area, had only moderate immunopositivity for pancytokeratins.
Ki-67 immunoreactivity was detected in the nuclei of decidual and stromal cells in both affected and unaffected mice (Fig. 10). The number of Ki-67 positive cells was significantly higher in the deciduated cells in the antimesometrial aspect of affected uteri (8.5 ± 2.2), compared to the endometrial stromal cells in the mesometrial aspect of pathological uteri (1.3 ± 1.1) (P < .0001) and in the endometrial stromal cells in the control group (1.2 ± 0.5) (P < .0001).
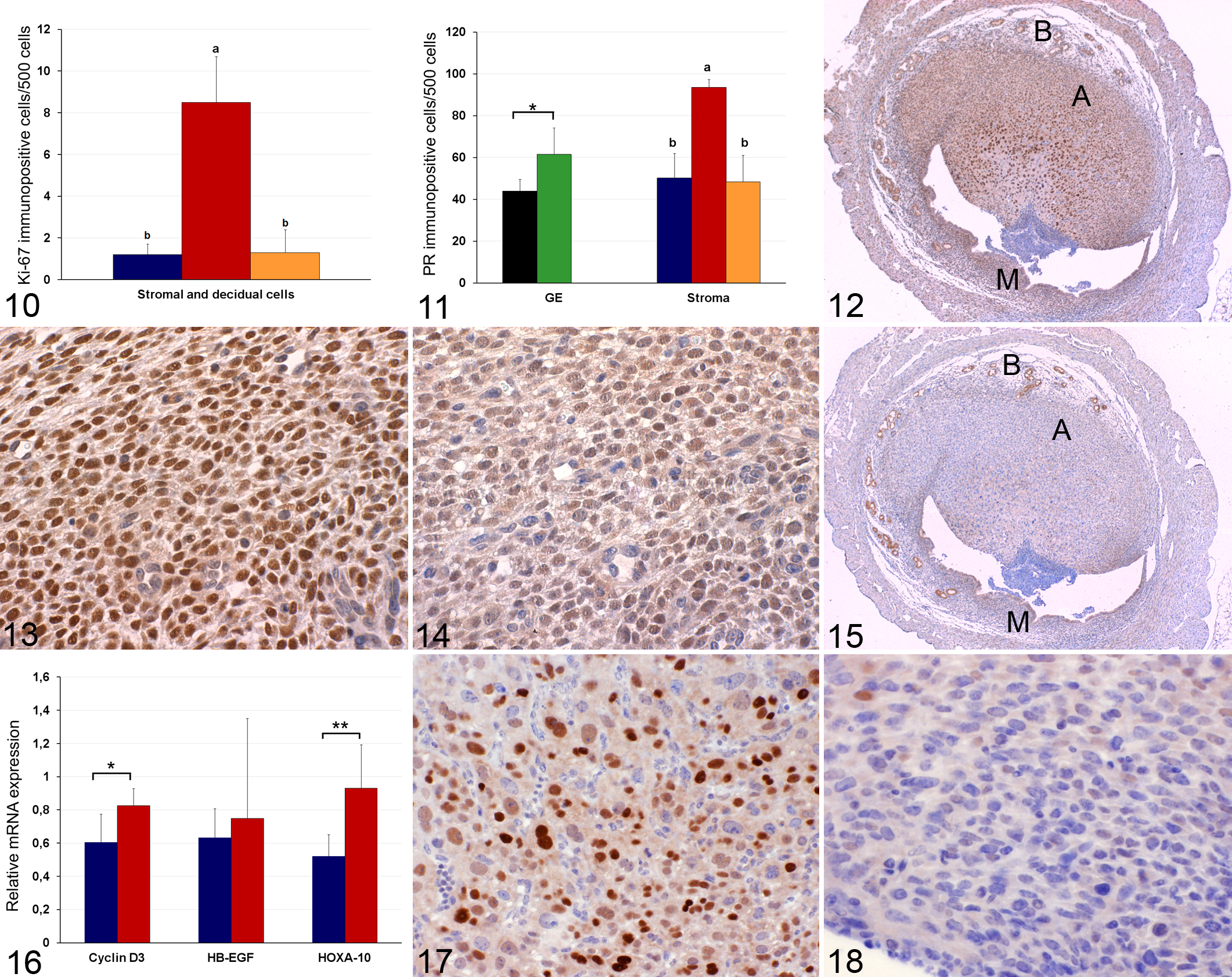
PR was expressed in the nucleus of glandular, decidual and stromal endometrial cells in both groups of mice (Fig. 11). The highest positivity for PR was observed in the deciduated cells in the antimesometrial aspect of pathological uteri (93.6 ± 3.7) (Figs. 12 and 13) and was significantly greater (P < .0001) than that in the endometrial stromal cells in the mesometrial aspect of pathological uteri (48.4 ± 12.7) (Fig. 14) and with the stromal cells in control mice (50.3 ± 1.6). Similarly, PR positivity was significantly higher (P < .001) in the glandular cells of pathological uteri (61.6 ± 12.6) compared with those of control uteri (44.1 ± 5.4) (Fig. 12).
By contrast, ERα was expressed only in the nuclei of endometrial gland cells in the basal endometrial region of both groups of mice. ERα expression was not significantly different between the pathological (50.1 ± 10.3) (Fig. 15) and control animals (48.9 ± 11.7).
No positive immunostaining for c-Myc was observed in the uterine samples in either mouse group. Cyclin D3 positivity was detected only in the cell nuclei of the endometrial decidual and stromal cells in both mouse groups. When assessing cyclin D3 immunostaining in the pathological group, the highest values were observed in the deciduated cells of the antimesometrial region (43.5 ± 5.1) (Fig. 17), which was significantly greater (P < .0001) than that in the endometrial stromal cells in the mesometrial aspect of the uteri (2.5 ± 1.7) (Fig. 18) and stromal cells in the control uteri (1.9 ± 0.8).
RT-qPCR
RT-qPCR detected measurable amounts of cyclin D-3, Hoxa-10 and HB-EGF mRNA in all uterine samples of both groups (Fig. 16).
Cyclin D3 mRNA expression was significantly higher (P < .01) in the pathological (deciduated) uteri (0.8 ± 0.1) than in the control group (0.5 ± 0.1).
Although HB-EGF mRNA expression was slightly higher in the pathological uteri (0.7 ± 0.6) than in the control uteri (0.6 ± 0.1), the difference was not significant (Fig. 16). Finally, Hoxa-10 transcription levels were significantly (P < .001) higher in the pathological uteri (0.9 ± 0,1) than in the control donor mice group (0.5 ± 0.1) (Fig. 16).
Discussion
In the present study, the bilateral multifocal uterine decidual reaction was diagnosed in 11.2% of donor female C57BL/6 J mice, aged 8.5 ± 0.6 weeks, treated with a superovulation protocol recommended in young adult female mice. Uterine deciduated cells showed PR overexpression and an elevated cell proliferative index, together with high levels of desmin and cyclin D3 immunostaining. Moreover, the mRNA expression levels of cyclin D3, HB-EGF and Hoxa-10 were upregulated in the decidual reaction. To our knowledge, this is the first pathological and molecular characterization of the mouse decidual reaction provoked by a superovulation protocol as a possible artificial stimulus to induce these lesions in mice. The induced uterine decidual reaction has been previously reported in rats and mice by exposure to different hormonal treatments 12,15,21 or provocation by multiple artificial stimuli.2, 6,32,36 Grossly, in the present study, the mouse decidual reaction associated with the superovulation protocol showed a multifocal nodular pattern with uterine horn enlargement, which is similar to that observed in other induced uterine decidual reactions in mice, 12,32,37 while spontaneous uterine decidual reactions in mice 10 and rats 42 have been described as solitary nodules. Histologically, in the current study, the uterine decidual reaction showed structural organization in 3 different regions, in accordance with previously described spontaneous decidual reactions in older rats 16,42 and in arachis oil-induced deciduomas in mice. 32 By contrast, no transition regions were noted between the mesometrial and antimesometrial areas of the endometrium, as previously described in rat spontaneous deciduomas. 42
Our results showed that uterine decidual cells strongly expressed desmin, which is considered one of the characteristic markers of deciduated cells, 14,17,22 but negative staining for vimentin, which is in contrast to the findings described by other authors. 42 Similar to previous descriptions regarding mouse physiological decidual changes 33 or rat spontaneous deciduomas, 42 we observed uterine natural killer cells in the decidual reaction, which may contribute to immune tolerance at the maternal-fetal interface regulating the penetration of trophoblast cells during implantation. 17 Another hallmark of the decidualization process is the high grade of stromal cell proliferation. 7,17 In the current study, the Ki-67 index confirmed the high proliferative capacity of uterine deciduated cells, which is in agreement with findings previously reported in rat deciduomas using proliferative cell nuclear antigen. 42
Previous descriptions in rat superovulation treatment 38 showed that the large number of follicles induced by the PMSG/hCG protocol provoked a higher ovarian androgen, estradiol or progesterone concentration for a prolonged period. In our study, the high hormone dosage used in young adult mouse donors may be related to higher or persistently elevated steroid levels and the overexpression of hormonal receptors observed in pathological uteri. In the present study, decidual cells showed the highest number of PR receptors, corroborating previous studies regarding the relevant role played by PR overexpression in deciduated cells of mice. 6,29,35,36 However, another study of rat spontaneous deciduomas found similar PR expression in decidual and stromal cells, downplaying the role of PR in decidualization. 42 It is known that P4 plays a pivotal role in the physiological onset of mouse embryo implantation on day 4, increasing the proliferation and differentiation of uterine stromal cells into decidual cells. 7,11,29,36 Our results are in agreement with previous descriptions of delayed embryo implantation in P4-treated mouse pseudopregnant uteri. 11,17 Moreover, in PR (−/−) mice, the stimuli failed to induce uterine decidualization. 29 However, in ERα(−/−) mice, the uteri exhibited decidualization only in the presence of P4. 6,36 Steroid hormones, through their binding to receptors, induce the transcription of factors that can modulate the expression of many genes related to cell proliferation in embryo implantation. 4,17,30 Hoxa-10 is regulated by P4 in the mouse 26 and human endometrium, 30,41 and cyclin D3 leads to Hoxa-10 expression in the decidual cells of the pregnant mouse uterus. 3,7,8 This is in agreement with our study, in which cyclin D3 and Hoxa-10 mRNA expression was significantly greater in the uterine decidual reaction. Therefore, multiple regulatory factors involved in uterine mouse decidualization, such as PR, cyclin D3, and Hoxa-10, were overexpressed in the present study, suggesting that their overexpression may be implicated in the decidualization of stromal cells observed in the decidual reaction in superovulated female donors.
However, PG signaling participates in endometrial decidualization, 11,24,27 and the uterine PGE2 receptors EP3 and EP4, modulated by P4 and PR expression in stromal cells, may play an essential role in implantation and decidualization via their vasoactive and mitogenic properties. 11,27 In the present study, the decidual reaction was associated with a diffuse moderate acute inflammatory reaction. We speculate that inflammatory mediators, such as PG resulting from COX-2 activation, may be involved in the pathogenesis of the observed uterine lesion. Further studies are necessary to clarify this possible pathogenic mechanism.
The mild increase in HB-EGF mRNA expression in pathological uteri in the current study was expected because of the lack of embryo implantation in the decidual reaction, because HB-EGF is expressed in uterine superficial epithelial cells at the site of blastocyst apposition at the maternal-fetal interface. 9,17,35
Collectively, our results suggest that the increase of PR signaling which is likely due to elevated or persistently abnormal P4 levels in mice treated with a superovulation protocol when female mice exceed the optimal age, triggers cyclin D3 and Hoxa-10 overexpression, which may be involved in the pathogenesis of the decidual reaction.
Footnotes
Acknowledgements
We are grateful to C. Martí and M. Margalef from Barcelona Biomedical Research Park; P. Aranda and V. Mesa from Histopathology Laboratory of Veterinary Medicine of Complutense University for their technical support; and Dr L. Peña and the Statistical Unit of Complutense University for statistical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclose receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Santander Central Hispano Bank-Complutense University of Madrid research grant (GR3/14).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
