Abstract
Primary and secondary nervous system involvement occurs in 4% and 5%–12%, respectively, of all canine non-Hodgkin lymphomas. The recent new classification of canine malignant lymphomas, based on the human World Health Organization classification, has been endorsed with international acceptance. This histological and immunocytochemical classification provides a unique opportunity to study the histologic anatomic distribution patterns in the central and peripheral nervous system of these defined lymphoma subtypes. In this study, we studied a cohort of 37 dogs with lymphoma, which at necropsy had either primary (n = 1, 2.7%) or secondary (n = 36; 97.3%) neural involvement. These T- (n = 16; 43.2%) or B-cell (n = 21; 56.8%) lymphomas were further classified into 12 lymphoma subtypes, with predominant subtypes including peripheral T-cell lymphoma (PTCL) or diffuse large B-cell lymphoma (DLBCL), respectively. This systematic study identified 6 different anatomically based histologically defined patterns of lymphoma infiltration in the nervous system of dogs. Different and distinct combinations of anatomical patterns correlated with specific lymphoma subtypes. Lymphoma infiltration within the meningeal, perivascular, and periventricular compartments were characteristic of DLBCL, whereas peripheral nerve involvement was a frequent feature of PTCL. Similarly cell counts above 64 cells/μL in cerebrospinal samples correlated best with marked meningeal and periventricular lymphoma infiltration histologically. Prospective studies are needed in order to confirm the hypothesis that these combinations of histological neuroanatomic patterns reflect targeting of receptors specific for the lymphoma subtypes at these various sites.
Keywords
In dogs, non-Hodgkin lymphomas account for about 10% of all tumors and 83% of all hematopoietic malignancies with central nervous system (CNS) involvement occurring in 5%–12% of all canine lymphomas. 8,24,33,35 Primary CNS lymphomas account for less than 4% of all lymphomas in dogs, 31 and most lymphomas affecting the CNS are secondary tumors associated with multisystemic disease. 30 Although an increased prevalence of CNS T-cell lymphoma (TCL) is reported in Rottweiler and Boxer breeds, 18,21,31 dogs with lymphoma are presumed to be immunocompetent in contrast to the well-documented relationship in humans between an underlying AIDS-associated immunodeficiency with Epstein-Barr virus infection and the striking susceptibility to some B-cell lymphoma (BCL) subtypes.
Recently, the new classification of canine malignant lymphomas, based on the human World Health Organization classification of non-Hodgkin lymphomas, has been endorsed with international acceptance. 36 This system can now be used to determine the histologic and immunophenotypic characteristics of the different lymphoma subtypes in dogs and to identify anatomically dependent patterns of nervous system lesions as has been recently demonstrated in some primary and secondary CNS lymphomas in humans. 5,19 Thus in a large case series of canine systemic extraneural lymphoma, 36 the most common subtypes of lymphoma in dogs from a complete list of about 40 different subtypes included diffuse large BCL (DLBCL; 40%), peripheral TCL not otherwise specified (PTCL; 15%), T-zone lymphoma (13%), T-cell lymphoblastic lymphoma (T-LBL; 4%), and marginal zone BCL (4%). Less common subtypes included hepatosplenic γδ TCL, 12 large granular TCL (LGTCL), and intravascular TCL (ITV-T). 13,36 This adapted classification includes emerging syndromes such as neurolymphomatosis, which is often systemic but targets peripheral nerves and is mostly of B-cell lineage. 15,20,27,34
The goals of this study were primarily to determine both histologically and immunophenotypically the subtypes of canine nervous system lymphoma using this scheme 36 and to establish the prevalence of primary versus secondary nervous system lymphomas in this population, and second, among the various subtypes of nervous system lymphoma, it is necessary to identify any characteristic patterns of microscopic lesions with respect to their neuroanatomical sites and cerebrospinal fluid (CSF) cytological findings.
Materials and Methods
Selection of Cases
Relevant data from the 37 canine with lymphoma involving the CNS and peripheral nervous system (PNS) were obtained from their accessions to the medical records of the VMTH for the period 1983–2014. Inclusion criteria were (i) the availability of suitable formalin-fixed paraffin-embedded tissue including standardized transverse sections of the CNS, 38 specifically the brain and/or spinal cord, pituitary gland, and cranial and spinal nerves when clinically relevant and (ii) availability of formalin-fixed paraffin-embedded liver, kidney, lung, and at least 4 lymphoreticular tissues (ie, visceral and subcutaneous lymph nodes, spleen, gut-associated lymphoid tissue, tonsil, and bone marrow).
Analysis of CSF
The CSF was obtained from the cisterna magna for routine analysis. Erythrocyte and total nucleated cells counts were determined with a hemocytometer. Cytology preparations were prepared by centrifugation (Cytospin3; ThermoShandon, Pittsburgh, PA) of 500 μL of CSF for 5 minutes at 1000 rpm and routinely stained with Wright-Giemsa. A differential cell count was performed on 100 cells. Lymphoma was diagnosed if the cells consisted of a relatively monomorphic population of large (nuclei >2 times the diameter of a red blood cell) lymphocytes of immature appearance. In some instances, lymphoma was diagnosed on CSF examination, additional cytospin slides were used for immunophenotyping using the B-cell markers CD21 (CA2.1D6; P. F. Moore, UC Davis, CA) and CD79a (HM57; Dako Corp, Carpinteria, CA) and the T-cell markers CD3 (CD3-12; Serotec, Oxford, UK), CD4 (CA13.1E4; P. F. Moore), and CD8a (CA9.JD3; P. F. Moore).
Histopathology and Immunohistochemistry
A necropsy examination was performed in each dog and the brain and spinal cord were immersion fixed in 10% neutral-buffered formalin for 7–10 days. The brain was then trimmed in serial transverse 2- to 3-mm-thick sections corresponding to those from magnetic resonance imaging (MRI) sequences. 38 Subsequently, representative sections of the telencephalon (including rhinencephalon, hippocampus, neocortex, and basal nuclei), diencephalon (thalamus and hypothalamus), midbrain, medulla oblongata, cerebellum, and pituitary gland were routinely embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin. The spinal cord was available for 21 dogs and was similarly processed, including a minimum of 3 transverse sections from cervical, thoracic, and/or lumbar segments in addition to those sites with clinically localized lesions. A broad selection of extraneural tissues containing at least those previously cited as a requirement were similarly collected and processed.
Immunohistochemistry (IHC) was performed on all lymphomas with the aim of determining their B- or T- cell lineage and also their subtype. The B-cell-specific antibodies used were for CD20 (Neomarker RB-9031-P1, Fremont, CA), CD79a (HM57; AbDSero MCA2538H, Kidlington, UK), and Pax-5 (BD Bioscience, San Jose, CA), while an antibody to CD3 (CD3-12; P. F. Moore) was used as the T-cell marker. CD20 is a surface molecule expressed at all stages of B-cell differentiation except for mature plasma cells. CD79a is a single component of the B-cell antigen receptor expressed at all stages of B-cell differentiation. Pax-5 is a transcriptional factor expressed at early stages of B cell differentiation but not expressed in mature B cells. CD3 is a signaling component of the T cell antigen receptor. In addition, MUM1 (BC5; BioCare CRM352, Concord, CA), a transcriptional factor essential for plasma cell differentiation, was used to confirm plasmacytoid differentiation of BCL. Histiocytic and microglial cells were detected with antibodies to CD18 (CA18.3C10; P. F. Moore) or Iba-1 (Wako 19-19741, Richmond, VA USA), respectively. Antibodies to granzyme B (E2584; Spring Bioscience, Pleasanton, CA) were used to label cytotoxic T cells (CD8+) of LGTCL.
Briefly, for evaluation of IHC reactivity, 4- to 5-μm-thick sections were mounted on positively charged glass slides, air-dried overnight at 37°C, and deparaffinized and rehydrated. Endogenous peroxidase activity was blocked with 0.3% hydrogen peroxide in methanol, then the slides were rehydrated through graded alcohols to water. Antigen retrieval procedures included heat-induced epitope retrieval with Dako Target Retrieval Solution (S1699) or enzymatic digestion with Dako Proteinase K (S3020) following the manufacturer’s instructions. The antibody and blocking diluents and all the subsequent rinses were done with phosphate-buffered saline (PBS)-Tween 20 (0.02%). Sections were blocked with 10% normal horse serum for 20 minutes. Primary antibodies were applied for 1 hour. Granzyme B (1:80), CD3 (1:10), CD79a (1:100), CD20 (1:300), Pax-5 (1:100), and CD18 (1:100) were detected with Biocare Medical 4 + Detection System (anti-mouse link or anti-rabbit link and streptavidin-horseradish peroxidase (HRP) label HM606, GR 608, respectively, Biocare Medical, Concord, CA) for 10 minutes with each reagent. Multiple myeloma oncogene 1 (MUM-1) (1:100) and ionized calcium-binding adapted protein-1 (Iba-1) (1:600) were detected with Dako Envision (K4003, Dako, Carpinteria, CA). Double rinsing was done between each reagent application. Antigen detection was visualized using Vector NovaRed for peroxidase (SK-4800, Burlingame, CA, USA), following the manufacturer’s instructions. Sections were counterstained with Mayer’s hematoxylin. Sections in which either diluent or normal serum substituted the respective primary antibody were included as negative controls. Standardized positive control tissue was included for each antibody where appropriate.
Pathologic Evaluation
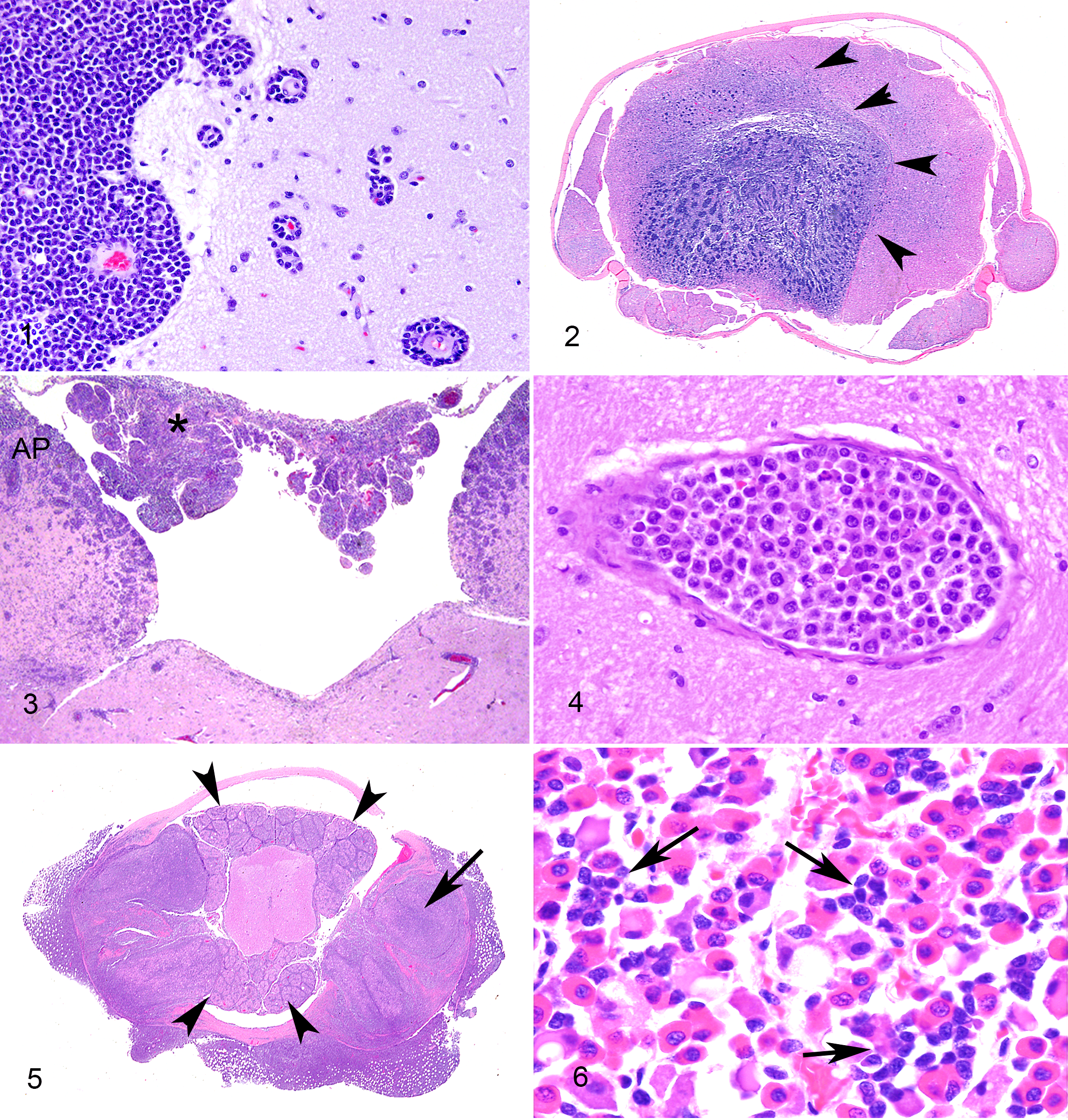
The gross location of each brain tumor was recorded as supratentorial, infratentorial, or both, whereas lesions were microscopically recorded as being focal, multifocal or disseminated in those sites (Supplemental Table 1). The specific topographical brain and/or spinal cord distribution of neoplastic infiltrates is shown in Supplemental Table 2. Localization of the neoplasms to the following neuroanatomical compartments (Figs. 1–6) was microscopically assessed in all 37 canine patients: meningeal (Fig. 1), perivascular (Fig. 1 inset), intraparenchymal (Fig. 2), periventricular (Fig. 3), intravascular (Fig. 4), and PNS (cranial and spinal nerves; Fig. 5). Additionally, localization to choroid plexus (Fig. 3) or pituitary gland (Fig. 6) was evaluated in 34 and 23 cases, respectively. The total score was defined as the sum of all neural compartments involved.
Results
Signalment
Of the 37 cases of nervous system lymphoma, various breeds were represented, with 18 female (16 spayed) and 19 male (16 castrated) dogs. The ages ranged from 1 to 15 years (7.2 ± 3.2 years, mean ± SD; Supplemental Table 1). Neurologic signs, seen on presentation for 29/37 cases, will be described in detail in a companion clinical paper (Sturges BK et al, in preparation). No significant correlations were found between lymphoma subtype and breed, sex, age, or clinical neurological findings.
Analysis of CSF
Lymphoma was diagnosed by analysis of CSF in 13/19 (68%) cases in which the samples were assessed, including 5/7 (71%) and 8/12 (67%) cases of TCL and BCL, respectively (Supplemental Table 3). Grouping of cases by CSF cell count and histologic pattern of meningeal involvement of lymphoma showed the following patterns: (i) <65 cells/μL in CSF with no or minimal meningeal involvement (n = 9), (ii) 102–257 cells/μL in CSF with variable meningeal involvement (n = 4), and (iii) 485–15,600 cells/μL in CSF with prominent meningeal infiltration with lymphoma (n = 6; Supplemental Table 3). Of the 6 dogs in which lymphoma was not diagnosed by CSF analysis, 2 cases were suggestive of lymphoma with low numbers of large immature lymphocytes accompanied by either neutrophilic inflammation and hemorrhage or mixed mononuclear inflammation, and 4 cases had no evidence of lymphoma in CSF (of which 3 had mixed histiocytic and/or lymphocytic inflammation). Of the latter 4 dogs, 2 had intravascular lymphoma and 2 had minimal focal meningeal involvement (histologically), likely explaining the absence of neoplastic lymphocytes in the CSF. Of the 3 cases with immunocytochemical analysis of CSF, 2 were CD3+ (T cell) and 1 was CD79a+ (B cell), in concordance with subsequent immunophenotyping of postmortem samples.
Histopathology and Immunohistochemical Findings
Primary CNS lymphoma, identified in 1/37 (2.7%) cases, was restricted to the cervical spinal cord (Fig. 2 and Table 1 [Case 37]) and was a CD79a+ centroblastic DLBCL. Of the 36 metastatic lymphomas, 16 were immunoreactive only to CD3 (T-cell lineage) and 20 were immunoreactive to CD20, CD79a, and/or Pax5 (B-cell lineage).
The 20 metastatic CNS BCL (Supplemental Table 1) included DLBCL (18/37, 49%), 1 intravascular BCL (1/37, 3%) and 1 lymphoblastic lymphoma (precursor B-LBL; 1/37, 3%; Fig. 1). The 18 DLBCLs were further subtyped as centroblastic/immunoblastic (12/18, 67%; Figs. 3 and 5), T-cell/histiocytic rich (2/18, 11%), plasmacytoid (3/18, 17%; Fig. 6), or anaplastic (1/18, 6%). Most BCL was immunoreactive for CD20+ and CD79a+ or Pax5+. However, the single precursor B-LBL was CD20+ CD79a−, and the single anaplastic DLBCL was CD20− CD79+. The 2 T-cell/ histiocytic-rich DLBCLs had many reactive CD3+ T lymphocytes and CD18+ Iba-1+ histiocytes admixed with the neoplastic B-cell population. Neoplastic B cells in the plasmacytoid DLBCL resembled plasma cells and up to 50% of the cells expressed MUM1.
The 16 metastatic CNS TCL (Supplemental Table 1) included nodal PTCL not otherwise specified (7/37, 19%), ITV-T (4/37, 11%; Fig. 4), LGTCL (3/37, 8%), 1 lymphoblastic lymphoma of T-cell origin (precursor T-LBL) and one hepatosplenic lymphoma. All TCL were CD3+ and CD79a−/Pax5−. In LGTCL, frequent CD3+ T-cells contained eosinophilic intracytoplasmic granules, which were granzyme B immunoreactive, whereas neoplastic cells of the single hepatosplenic lymphoma were CD11c+ but granzyme B−.
Neuroanatomic Compartmentalization of Lymphomas
As shown in Supplemental Table 1, the meningeal pattern was the most frequent CNS compartment infiltrated by both TCL and BCL (29/37, 78%; Fig. 1). Neoplastic cells variably filled the subpial and/or subarachnoidal spaces in a random distribution or formed perivascular infiltrates around large and medium subarachnoidal vessels and/or smaller subpial vessels. The meningeal pattern was most commonly found within the DLBCL subtype (16/18, 89%) but, as shown in Fig. 7, absent from the 5 ITV-T and BCL, the single metastatic anaplastic DLBCL, and from 1 of the plasmacytoid DLBCL.
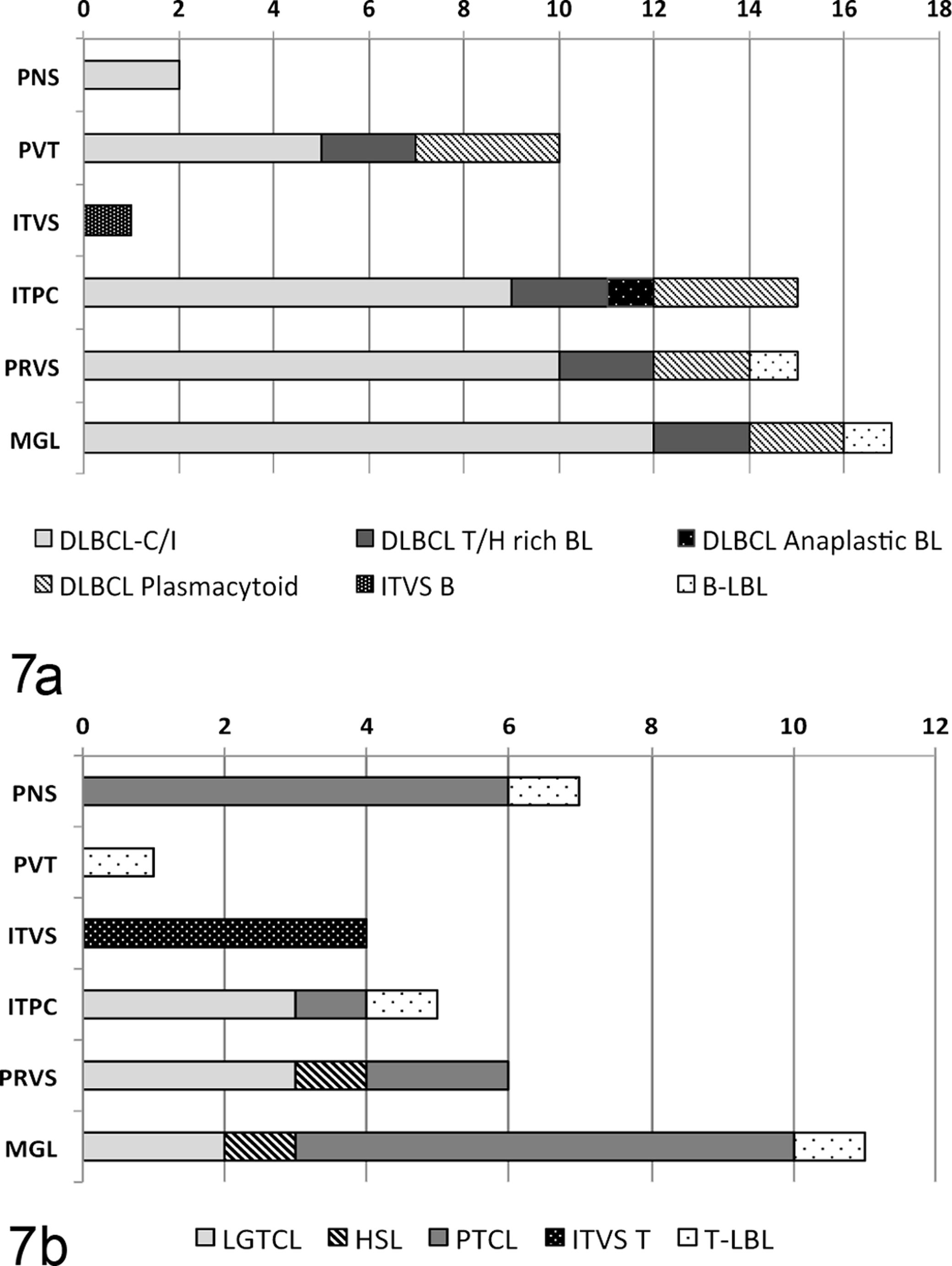
Histologic neuroanatomic compartments involved in metastatic nervous system lymphoma subtypes of B-cell (Figure 7a, n = 20) or T-cell (Figure 7b, n = 16) lineage. The number of cases of each lymphoma subtype is shown, for each histologic neuroanatomical pattern. Histological patterns: PNS, peripheral nervous system; PVT, periventricular; ITVS, intravascular; ITPC, intraparenchymal; PRVS, perivascular; MGL, meningeal pattern; B-LBL, B-cell lymphoblastic lymphoma; DLBCL, diffuse large B-cell lymphoma; DLBCL subtypes: C/I (centroblastic/immunoblastic), T/H rich BL (T-cell histiocytic rich lymphoma), anapl. (anaplastic) and plasma. (plasmacytoid); ITVS B, B-cell intravascular lymphoma.
The perivascular pattern (21/37, 57%; Fig. 1) consisted of 1 or more cell layers filling the Virchow-Robin spaces of medium- and small-caliber arterioles in both the white and gray matter. In the contiguous capillaries, around which no Virchow-Robin space exists, tumor cells had infiltrated the adjacent parenchyma. The perivascular compartment (Fig. 7) was most commonly involved in DLBCL (14/18, 78%) and LGTCL (3/3) but at lower frequency in PTCL (2/7, 28%). This pattern was predominant in the cerebrocortical white matter in the 2 T-cell/ histiocytic-rich DLBCL.
In the periventricular pattern (11/37, 30%; Fig. 3), lymphoma cells were arranged focally, segmentally, or circumferentially in the subependymal tissue around the ventricles. This was frequently represented in the DLBCL (10/15, 67%) but absent in mature TCL (0/17; Fig. 7). Of the DLBCL, the periventricular pattern was present in 2/2 T-cell/histiocytic-rich, 3/3 plasmacytoid, and 5/12 centroblastic/immunoblastic subtypes (Fig. 7).
The intraparenchymal pattern (23/37, 62%) was characterized by either a diffuse infiltration of neoplastic cells within the neuropil (21 cases) or by neoplastic cells forming a single well-demarcated non-encapsulated mass (2 cases). This diffuse intraparenchymal pattern was most prominent in those lymphomas with a meningeal and perivascular component (Fig. 7), including DLBCL (16/18, 89%) and LGTCL (3/3, 100%), but these patterns were less prominent in the case of hepatosplenic lymphoma. However the intraparenchymal pattern forming a space-occupying mass was the only pattern identified in the single cases of anaplastic DLBCL and primary BCL (Fig. 2).
The PNS pattern (7/37, 19%), characteristic of neurolymphomatosis, predominantly involved spinal nerves and their roots and less often cranial nerves. This pattern was identified in 4/7 (60%) cases of PTCL, only 2/18 (11%) DLBCL (in which it was restricted to the centroblastic/immunoblastic subtype; Fig. 5), and none of the other subtypes (Fig. 7).
The intravascular pattern, identified in 5/37 (13%) cases, was defined by neoplastic cells restricted to the lumina of blood vessels (Fig. 4). This pattern, which predominantly involved neoplastic cells of T-cell lineage, was often associated with multifocal areas of ischemia and/or large infarcts as a result of intraluminal thrombosis.
The pituitary gland was infiltrated in 14/23 (61%) cases, of which 12/15 were B-cell and only 2/8 were TCL (Supplemental Table 1). The pituitary gland targeting was most consistent in centroblastic/immunoblastic (7/9) and plasmacytoid (3/3; Fig. 6) subtypes of DLBCL, and in the single cases of intravascular BCL and hepatosplenic lymphoma. Conversely there was no pituitary gland involvement in the anaplastic (0/1) or the T-cell/histiocytic-rich DLBCL (0/1), LGTCL (0/3), or ITV-T (0/4).
Lymphoma infiltrated the choroid plexus (Fig. 3) in 13/34 (35%) cases. There was no clear association with the periventricular pattern (Supplemental Table 1). The choroid plexus was infiltrated in 7/16 BCL cases (intravascular and centroblastic/immunoblastic subtypes), only in 5/14 (36%) TCL cases, and in none of the primary CNS BCL, ITV-T, or the anaplastic, plasmacytoid, or T-cell/histiocyte-rich DLBCL (Supplemental Table 1).
The involvement of each neuroanatomical compartment in the 12 canine lymphoma subtypes is shown in Supplemental Fig. 1.
Systemic Distribution of Lesions in Metastatic CNS Lymphomas
Microscopic evaluation of the cases with secondary nervous system lymphoma revealed the involvement of lymph nodes (21/31 cases, 68%), liver (16/34, 47%), spleen (14/33, 42%), kidney (14/35, 40%), and intestine (11/28, 39%). Generally, neoplastic lymphocytes diffusely infiltrated the paracortical and medullary sinuses of lymph nodes and spleen, hepatic sinusoids, renal interstitium, and intestinal lamina propria and submucosa. No differences in neoplastic cell distribution were appreciated between different lymphoma subtypes.
Centroblastic/immunoblastic DLBCL and PTCL displayed the most widespread organ dissemination (Supplemental Fig. 2, whereas plasmacytoid and anaplastic DLBCL and the intravascular lymphomas had the least systemic dissemination. Tissue targeting varied substantially in LGTCL: 2 cases involved only a single nonneural site (kidney or nasal cavity), whereas all examined tissues contained neoplastic cells in the third case.
Discussion
This retrospective study of 37 lymphoid tumors involving the nervous system identified that primary lymphoma of the nervous system is uncommon in dogs, representing only a single case of DLBCL, while the remaining 36 tumors were secondary TCL or BCL. Twelve major lymphoma subtypes were identified, 36 and many of these specific lymphoma subtypes targeted 1 or a selected number of neuroanatomical compartments. There were no similar targeting patterns found in the extraneural organs. The findings suggest that these anatomic patterns may reflect differing tropism of neoplastic cells for cellular receptors in various parts of the CNS and PNS.
The CSF findings indicate that when there is meningeal and/or periventricular infiltration of neoplastic lymphocytes, as confirmed by histologic examination of postmortem samples, a clinical antemortem diagnosis of CNS lymphoma can often be made due to the presence of these cells in the CSF. The 13 dogs with a diagnosis of lymphoma based on CSF analysis had nucleated cell counts that ranged from 4 to 15,600 cells/μL. Not surprisingly, greater histologically evident degrees of meningeal involvement corresponded to higher total nucleated cell counts in CSF. Dogs with nucleated cell counts <65 cells/μL had mild to occasionally moderate focal or multifocal meningeal involvement, while those with counts >484 cells/μL had marked diffuse or marked multifocal to segmental meningeal involvement. Similarly, dogs with marked periventricular involvement tended to have higher nucleated cell counts than those with mild periventricular involvement, regardless of the degree of meningeal involvement. The lymphoma lineage (B cell vs T cell) was also determined immunocytochemically in the CSF of 3 dogs antemortem, and all 3 results were concordant with subsequent immunohistochemical immunophenotyping. These results suggest that CSF assessment can clearly be useful in the antemortem diagnosis and lineage determination of CNS lymphoma in dogs, in particular lymphoma subtypes with meningeal and/or periventricular involvement.
An unexpected finding in this study was that >70% of metastatic DLBCL infiltrate the pituitary gland, resulting in gross enlargement of the gland, with dorsal midline extension into the ventral aspect of the third ventricle and into the hypothalamus. These gross and histological features, initially recognized by MR imaging in 2/7 patients with DLBCL, were not seen in any other lymphoma subtype.
There was a highly consistent arrangement of neoplastic cells in various neuroanatomic compartments in most of the 12 morphologic subtypes of lymphoma. A single neuroanatomic compartment was involved in 7 cases, while the remaining 30 cases had a consistent combination of 2 or more compartments. In all 5 intravascular lymphoma cases, neoplastic cells were present—by definition—only within the blood/intravascular compartment regardless of the cell lineage within both meningeal and intraparenchymal blood vessels. Meningeal, perivascular, intraparenchymal, periventricular, pituitary gland, and choroid plexus involvement were overrepresented in BCL, in particular the DLBCL group, when compared to TCL. In contrast, the PNS compartment was overrepresented in TCL, in particular the PTCL. Neoplastic cells restricted to the CNS in cases of metastatic anaplastic DLBCL and the primary DLBCL resulted in a space-occupying mass consisting of a single focal intraparenchymal pattern.
We suspect that these specific lesional distributional patterns may reflect differing homing mechanisms of neoplastic lymphocyte subpopulations for these distinct compartments of the nervous system. This concept of specific targeting by T and B cells is well established in lymphoreticular organs. In the inflamed CNS, both subarachnoid and perivascular spaces can be infiltrated by macrophages that express MHC class II and present antigens to lymphocytes. 1,6,28 B cells are virtually confined to the perivascular spaces until they mature into plasma cells. Instead differentiated CD8+ and CD4+ T cells can migrate into the parenchyma. It is likely that other compartments could be populated by different homing and tracking mechanisms of neoplastic lymphocytes. An example from this study is the highly aggressive intravascular BCL subtype in which neoplastic lymphocytes aggregate exclusively within blood vessels in the brain and some extraneural organs. 5,19 This vascular targeting is believed to be the result of defective lymphocyte homing receptors, which prevent normal extravasation of lymphocytes. 7,25
Moreover, studies have recently implicated interleukin (IL)-15 in the increased risk of CNS involvement in human B-cell acute lymphoblastic leukemia. Interleukin 15 upregulates P-selectin glycoprotein ligand-1 (PSGL-1) and chemokine receptor 3 (CXCR3), which are associated with normal CNS lymphocyte trafficking, thus providing some mechanistic evidence for increased trafficking in lymphoma. 39 Further evidence for the aberrant targeting hypothesis is the selective CNS tropism of a primary human CNS BCL, which when subcutaneously implantated into an athymic mouse developed an exclusively intravascular pattern of lymphoma restricted to the brain. 11 It is suspected that chemokine regulation of lymphocyte homing to the CNS may also play a role in the pathogenesis of metastatic CNS lymphoma. 14 Both C-X-C motif chemokine ligands 12 and 13 (CXCL12 and CXCL13) are chemokines that elicit homing of B helper CD8+ cells and a subset of T-helper cells within the lymphoid follicle and have been demonstrated on malignant lymphocytes and vascular endothelium in primary human CNS lymphomas. 4,29 More pragmatically, elevated CSF levels of CXCL13 and IL-10 had a 99.3% diagnostic specificity for both primary and secondary lymphomas in a large case series (n = 220) of human patients with lymphoma. 26 None of these markers have been identified yet in canine DLBCL. 9,10,16,22,23 Molecular studies in domestic animals may thus be of diagnostic value in addition to identifying the mechanisms of invasion and homing of neoplastic cells.
In conclusion, the main goal of this study was to characterize the histological and immunophenotypic subtypes of canine nervous system lymphomas and their relation to CSF analysis and neuroanatomic localization. Because the diagnosis of primary CNS lymphoma ultimately depends on a rigorous postmortem examination to exclude underlying involvement of other organs, 2 the few case reports of primary CNS lymphoma in the veterinary literature are controversial. 3,17,32,37 It is hoped that this rigorous approach will lead to better designed prospective studies to confirm the hypothesis developed from this study that these histological patterns, together with their MRI profiles (Sturges et al, in preparation), accurately reflect targeting of specific anatomical compartments, which is in turn dependent on the expression of different homing molecules among the lymphoma subtypes. Such studies would lead to improved clinical diagnosis and prognosis, better understanding of the molecular pathogenesis, and innovative therapeutic targeting nervous system lymphoma.
Footnotes
Acknowledgments
The authors are grateful to Dr Martí Pumarola, DVM, PhD, for his help with the AGAUR grant. The authors thank Mike Manzer, Barry Puget, Kurt Takahashi, and Cristopher Kwong in the VMTH histology laboratory for their superb histological and immunohistochemical technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially funded by the Fellowship in Neuropathology awarded to Dr Sílvia Sisó i Llonch, University of California, Davis. Dr Marco-Salazar was supported by an AGAUR PhD grant [2012 BE100584] from the Generalitat de Catalunya, Barcelona, Catalunya.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
