Abstract
Five cases of Rhodococcus equi infection in dogs were identified from 2003 to 2014. Three of the dogs had severe, internal lesions attributable to R. equi that have not been previously described: endophthalmitis, endocarditis, and suppurative pleuropneumonia. Isolates from 4 of the dogs were analyzed by polymerase chain reaction for Rhodococcus virulence-associated plasmid (vap) genes. One isolate was vapA-positive, 2 lacked a virulence plasmid, and 1 carried the novel vapN-associated plasmid (pVAPN) recently characterized in bovine isolates. The pVAPN plasmid has not been described in isolates cultured from companion animals. Four of the dogs either were receiving immunosuppressive drugs or had endocrinopathies. R. equi has the potential to cause significant infections in dogs, and immunocompromised animals should be considered at risk for infection.
The genus Rhodococcus includes numerous species of nocardioform, aerobic, facultative, intracellular bacteria frequently found within the environment and feces of diseased and healthy animals. 1 Most rhodococcal infections in animals and humans are attributable to Rhodococcus equi. 14 R. equi coccobacilli stain positive in cytologic and histologic Gram’s and Fite’s acid-fast preparations. 4 The Rhodococcus spp virulence-associated proteins are presumed to aid bacterial survival in macrophages 15 and are encoded on circular and linear plasmids. A highly conserved conjugal transfer protein gene (traA) is common to all strains carrying plasmids. 9 Pathogenic R. equi strains are classified as virulent or intermediately virulent based on carriage of large plasmid encoding genes for either virulence-associated protein A (VapA) or B (VapB), respectively. 14 Strains that lack vapA or vapB are classified as avirulent. 2,14 A novel R. equi host-adapted linear virulence plasmid, pVAPN, was recently characterized in isolates from cattle 15 and encodes virulence-associated protein N (VapN). Rhodococcal infections are most common in foals, swine, 10 and cattle. 16 Infections in companion animals are comparatively rare, and contrary to the classic presentation in horses, most infections in dog and cats are extrapulmonary. 10,16 In dogs, R. equi rarely has been cultured from feces, 10 skin, nasal mucosa, and conjunctiva of healthy dogs, 14 and from dogs with skin lesions, 10,14 tracheitis, and vaginitis. 14 A single case of disseminated R. equi infection was reported in a 3-month-old Basenji puppy with myositis, hepatitis, and osteomyelitis. 2 This paper reports the clinical and pathologic features of canine R. equi culture-positive cases that presented with significant internal lesions attributable to R. equi.
The findings for each case are summarized in Supplemental Table 1, and complete narratives are provided as online Supplemental Material. All of the dogs (case Nos. 1–5) presented from 2003 to 2014. The cases included a 2.5-year-old male English Bulldog (dog No. 1), a 3-year-old female Pug (dog No. 2), a 3-year-old spayed-female Belgian Malinois (dog No. 3), a 10-year-old castrated-male Papillon (dog No. 4), and a 12-year-old spayed-female Boston Terrier (dog No. 5). All dogs had access to the outdoors, and 3 dogs (dog Nos. 1–3) lived in rural areas. Presenting clinical signs varied widely and included cardiopulmonary distress (dog Nos. 1–4), corneal ulceration (dog Nos. 1 and 5), endophthalmitis (dog No. 1), gingivitis (dog No. 1), and skin ulceration (dog Nos. 1, 3, and 5). Duration of clinical signs was 3 weeks, 1 week, 1 day, 1 week, and 2.5 weeks for dog Nos. 1–5, respectively. One dog (dog No. 1) had a history of dirofilariasis and ehrlichiosis within 1 year of presentation and was being treated for a chronic, unspecified skin condition. Four of the dogs had clinical evidence of severe, concurrent endocrine or autoimmune disorders: iatrogenic hyperadrenocorticism (dog No. 1), hyperadrenocorticism (dog No. 5), hypoadrenocorticism (dog No. 4), and immune-mediated polyarthritis (dog No. 3). Dog No. 4 had severe hypocholesterolemia (59 mg/dL) and panhypoproteinemia (2.7 g/dL). Three of the dogs had recently received corticosteroids: prednisone (dog No. 1, 10 mg per 24 hours; dog No. 3, 38 mg per 12 hours) and dexamethasone (dog No. 2, single injection of unknown dosage). Additional historical medications included clindamycin (dog No. 1), cyclosporine (dog No. 3, 150 mg per 12 hours), doxycycline (dog No. 1), and tobramycin (dog No. 5).
Radiographs were performed in dog Nos. 1, 3, 4, and 5 and revealed lysis of the mandible (dog No. 1), calcinosis cutis (dog No. 1), an enlarged heart (dog No. 5), and pulmonary pathology ranging from large, discrete lung masses with pleural effusion (dog No. 3) to a diffuse interstitial pattern (dog Nos. 4 and 5). Hypoadrenocorticism was confirmed in dog No. 4 via corticotropin stimulation test (pretest <1.0 μg/dL; posttest <1.0 μg/dL) and abdominal ultrasound, which revealed bilateral adrenal cortical atrophy. Pituitary dependent hyperadrenocorticism was suspected in dog No. 5 due to bilaterally enlarged adrenal glands observed on ultrasound. Cytological examination was performed by a veterinary clinical pathologist on a fine-needle aspirate of a skin mass (dog No. 1) and by the attending clinician on purulent fluid obtained via thoracentesis (dog No. 3). In dog No. 1, numerous neutrophils and foamy macrophages were observed in a proteinaceous background that frequently contained variable numbers of cocci (Fig. 1), diplococci, and phagocytosed cellular debris; fewer bacteria were seen in dog No. 3.
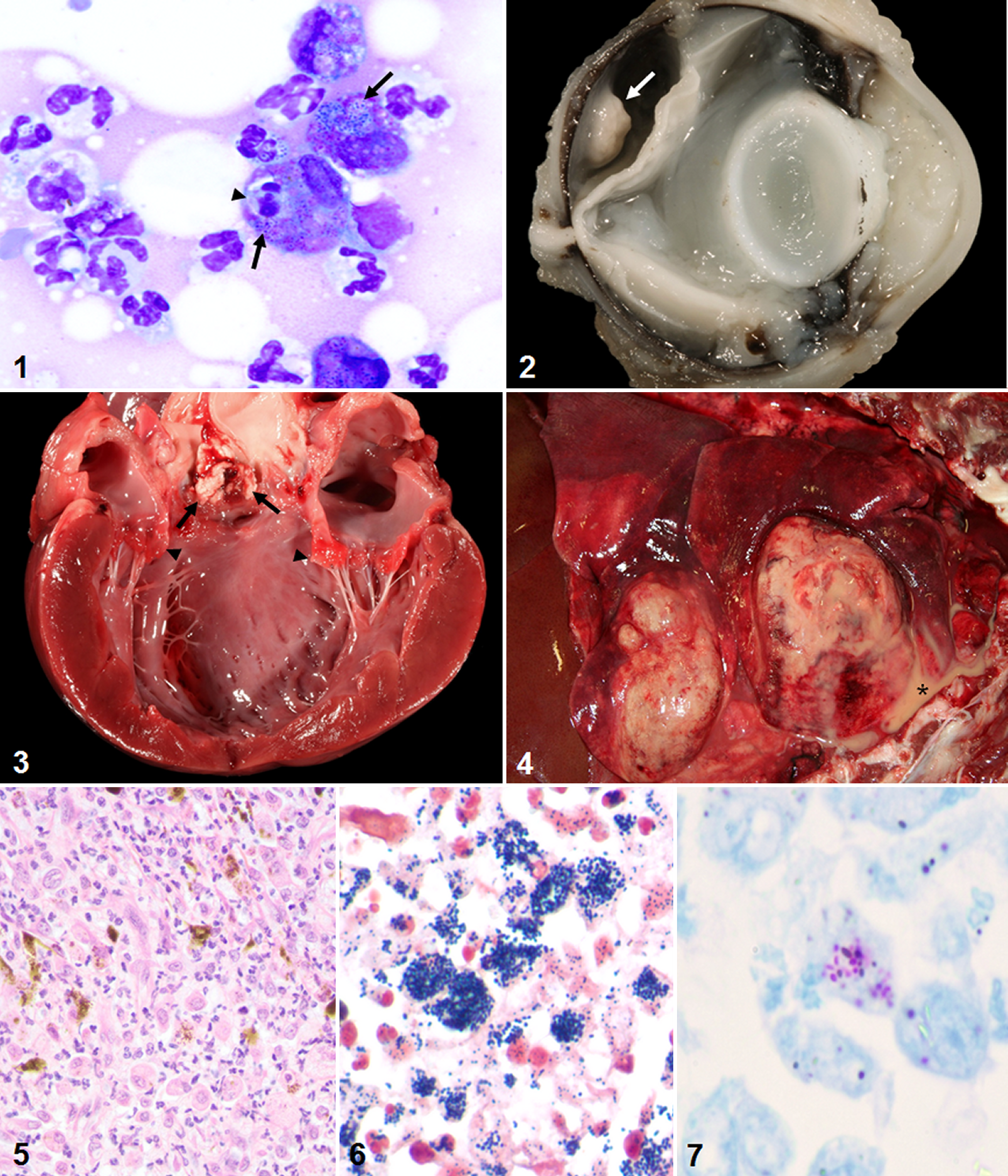
Dog Nos. 1–4 were euthanatized and submitted for complete necropsies. One dog (dog No. 5) died of cardiopulmonary arrest, but necropsy was not performed and only the eye was examined. Sections of major organs were fixed in 10% neutral buffered formalin and processed routinely for histopathologic examination by hematoxylin and eosin, Hucker-Twort Gram’s, and Fite’s acid-fast stains. The affected eyes from Dogs 1 and 5 were fixed (intraocularly and extraocularly) in Davidson’s solution and processed similarly. Gross lesions attributable to R. equi infection found at necropsy included pyogranulomatous dermatitis (dog Nos. 1 and 3), endophthalmitis (dog No. 1; Fig. 2), aortic vegetative valvular endocarditis (dog No. 2; Fig. 3), gingivitis (dog No. 1), hepatitis (dog No. 3), pulmonary abscessation with pyothorax (dog No. 3; Fig. 4), pulmonary edema (dog Nos. 1 and 4), lymphadenitis (dog Nos. 1 and 3), and keratitis and partial anterior staphyloma (dog No. 5). The inflammatory component in all dogs was primarily histiocytic to pyogranulomatous (Fig. 5) with variable amounts of necrosis, hemorrhage, mineralization, and edema in all affected tissues. Numerous intracytoplasmic Gram-positive (Fig. 6) and Fite’s acid-fast-positive (Fig. 7) coccobacilli were observed in the subretinal mass of dog No. 1 and the lung and liver of dog No. 3. Similar bacteria were rarely observed on the aortic valve (dog No. 2), within alveolar macrophages (dog Nos. 1 and 4), and within a perforated corneal ulcer (dog No. 5). Additional lesions unrelated to R. equi infection included vertebral disk herniation (dog No.1), appendicular polyarthritis (dog No. 3), lymphocytic adrenalitis (dog No. 4), and cutaneous phaeohyphomycosis (dog No. 3).
Aerobic culture (see Supplemental Material) of the lung (dog Nos. 3 and 4), aortic valve (dog No. 2), renal lymph node (dog No. 1), and spleen (dog No. 1) yielded 2+ growth, 3 colonies, 3+, 3+, and 1+ growth of R. equi, respectively. Bacterial growth from the corneal ulcer (dog No. 5) was not quantified. Isolates from dog Nos. 1–4 were screened via polymerase chain reaction (PCR) for the R. equi virulence plasmid genes: traA, vapA, vapB, and vapN (see Supplemental Material). The isolate from dog No. 5 was unavailable for analysis. The renal lymph node and splenic isolates from dog No. 1 were positive for vapN and negative for vapA and vapB by PCR (ie, traA+/vapAB–/vapN+). Two dogs (dog Nos. 2 and 3) had the avirulent plasmid phenotype (traA–/vapAB–/vapN–), and 1 dog (dog No. 4) had the virulent plasmid phenotype (traA+/vapA+B–/vapN–). The vapN sequence for dog No. 1 had 100% identity with the pVAPN reference sequence (KP851975.1) and was deposited in GenBank under accession number KT211244.
The R. equi isolates from dog Nos. 2 and 3 were traA-negative and were most likely avirulent strains, whereas dog No. 4 had the virulent phenotype. The isolates from the spleen and renal lymph node of dog No. 1 had the traA- and vapN-positive phenotype of isolates recently classified from cattle. To the authors’ knowledge, the isolates from dog No. 1 are the first confirmed report of pVAPN in a companion animal species. The majority of R. equi isolates collected from cattle are vapA and vapB negative 9 and carry pVAPN, 15 which is more related to the linear plasmid (pNSL1) carried by the recently discovered Rhodococcus sp NS1 strain than to the vapA and vapB plasmids. 15 The virulent and intermediately virulent plasmid phenotypes are associated with disseminated infections in foals 16 and pigs, 14 respectively. From previous limited studies in companion animals, vapA-positive isolates have been more frequently found in samples from cats than dogs, 14 while vapB carriage has not been described in dogs. 9,14 Most of the canine R. equi isolates studied previously have been classified as avirulent based on standard typing for vapA and vapB 14 and were not assessed for carriage of traA to determine whether another type of virulence plasmid was present. In the previous case of disseminated rhodococcal infection in a dog, the isolate was classified as avirulent, 2 but it is unknown whether the isolate actually lacked a plasmid or carried another type of virulence plasmid similar to pVAPN.
R. equi–associated ophthalmitis has rarely been reported in horses 12 and humans. 3,5 Rhodococcus spp have been isolated as part of the normal flora of the canine conjunctival sac. 17 Although the perforated corneal ulcer in dog No. 5 did culture positive for R. equi and there are additional reports of superficial Rhodococcus spp corneal and conjunctival infections in dogs, 8,14 no other instances of Rhodococcus spp endophthalmitis have been characterized histologically in dogs. The wide dissemination of the rhodococcal granulomas in dog No. 1 is suggestive of hematogenous spread of the bacteria, which is similar to the presentation in foals with R. equi panophthalmitis and other extrapulmonary infections. 11 Infection in dog No. 1 may have disseminated from the ulcerative gingivitis or dermatitis, and tropism for lymph nodes, as is commonly observed in other pVAPN isolates in livestock, 15 may have facilitated spread of the infection. The R. equi infection in dog No. 5 most likely descended through the corneal defect, as is common in human and adult equine cases of rhodococcal keratitis. 3,12 Chronic, immunosuppressive corticosteroid treatment (dog No. 1) and endocrine dysfunction (dog No. 5) likely played a role in maintenance and spread of opportunistic infection of R. equi in the dogs. As a necropsy was not performed on dog No. 5, the cause of death and the possibility of bacterial spread to other tissues was not confirmed.
Infectious endocarditis most commonly involves the mitral and aortic valves in dogs. 6,13 Rhodococcal endocarditis is a rare occurrence in humans 7 and foals and is often associated with immunodeficiency, septicemia, or a concurrent heart defect. 11 Bacterial colonization of the aortic valve, as observed in dog No. 2, results in severe left cardiac insufficiency and shortened survival time compared with endocarditis of the mitral valve in dogs. 6 The most common cultured bacterial agents are Staphylococcus spp, Streptococcus spp, and Escherichia coli, with Bartonella spp often implicated in culture-negative cases. 6 When R. equi was classified as Corynebacterium equi, endocarditis related to Corynebacterium spp was often reported in dogs. 13 Because these historical Corynebacterium spp isolates from canine endocarditis cases were not further identified, it is unknown whether any of the cases were associated with the agent now known as R. equi. In more recent surveys of endocarditis in dogs, R. equi has not been identified in heart lesions. 6 To the authors’ knowledge, dog No. 2 is the first confirmed report of endocarditis associated with R. equi in a dog. The single dose of dexamethasone given to dog No. 2 shortly before euthanasia likely did not play a role in initial infection, and the predisposing cause of the endocarditis may have been the mitral valve endocardiosis or narrowing of the aortic outflow tract. Initial colonization of the aortic valve may have occurred secondary to turbulence-induced damage and bacteremia with slow progression of infection over time.
Suppurative bronchopneumonia with abscessation is a classic presentation of R. equi in foals, but pulmonary Rhodococcus spp infections in companion animals are rare. 10,16 In the previous canine case of disseminated R. equi infection, pulmonary abscessation was not a feature. 2 Although bacteria were only rarely observed in the alveolar macrophages of dog Nos. 1 and 4, numerous bacteria were in the lung of dog No. 3 within a lesion resembling the presentation in foals. Rhodococcal pneumonia is commonly observed in people with immunodeficiency and in individuals with hematologic cancers and alcoholism. 1 Dog No. 3 had been receiving immunosuppressive doses of corticosteroids that likely contributed to development of infection, which also locally spread to the liver and mediastinal lymph nodes in a manner similar to that observed in dog No. 1. The histologic changes in dog No. 4 were mild by comparison, and the initial clinical signs were likely favored by severe autoimmune hypoadrenocorticism, which may have been compounded by mild pulmonary infection with a virulent R. equi strain.
Disseminated R. equi infection can occur in dogs and especially in immunocompromised individuals. The virulence profiles of the R. equi strains cultured from 4 of the dogs are diverse: 2 avirulent, 1 virulent, and 1 carrying the new VapN-associated plasmid. This report is the first description of pVAPN in a companion animal species. Additionally, R. equi–associated endophthalmitis, endocarditis, and suppurative pleuropneumonia have not been reported before in dogs. R. equi is an important differential diagnosis in dogs with pyogranulomatous inflammation attributable to bacterial infection. Dog Nos. 1, 3, 4, and 5 either were receiving multiple doses of corticosteroids or had clinical or gross evidence of an endocrine disorder. The wide distribution of organ systems involved illustrates that R. equi has the potential to cause serious multisystemic infections in dogs, and patients with endocrine dysfunction or receiving immunosuppressive therapy likely have a heightened risk for infection.
Footnotes
Acknowledgements
The authors thank the technical staff of the Texas A&M Veterinary Medical Teaching Hospital Clinical Microbiology Laboratory for their efforts to culture and preserve the bacterial isolates used in this study and Dr Noah Cohen for critical review of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
