Abstract
The decidua is the superficial portion of endometrium that transforms, or decidualizes, under the influence of progesterone to nourish the early embryo during pregnancy. Deciduae outside the uterus are found in nearly 100% of human pregnancies. This condition, known as deciduosis, may mimic malignancy, resulting in additional diagnostic procedures that place the mother, baby, or both at risk. Deciduosis has been described in both Old World and New World nonhuman primates in conjunction with pregnancy and after treatment with exogenous progestins. Here the authors present 6 cases of deciduosis associated with endometriotic lesions in female rhesus and cynomolgus macaques (Macaca mulatta and Macaca fascicularis). Full diagnostic necropsies with histologic analyses were performed on all animals. Deciduae were stained with hematoxylin and eosin and by immunohistochemistry for vimentin, CD10, progesterone receptor, estrogen receptor, desmin, cytokeratin, kermix P8, chorionic gonadotropin, human placental lactogen, and calretinin. The most common clinical signs were abdominal pain (4 of 6) and anorexia (2 of 6). At necropsy, macaque uteri were often enlarged or disfigured (4 of 6) with abundant fibrous adhesions (5 of 6). Affected tissue consisted of epithelial-lined cysts and decidualized stroma with scattered gamma/delta T cells. Decidualized stromal cells were large and polyhedral with abundant cytoplasm and round vesicular nuclei. They stained positive for vimentin, CD10, progesterone, and estrogen. In summary, these cases illustrate deciduosis in 6 nonhuman primates with endometriosis. Understanding decidualization in nonhuman primates will aid in elucidating the pathophysiology of deciduosis during pregnancy or endometriosis and potentially lead to new interventions.
Keywords
Decidua consists of a layer of endometrial stromal cells that support early embryonic development and are required for maintenance of normal pregnancy. Endometrial stromal cells transform, or decidualize, under the influence of progesterone. 29 Decidualization begins near the base of the spiral arteries. There, cell nuclei become rounded and vesicular, and the cytoplasm is enriched with glycogen and lipids. Decidualized stromal cells may cause processes such as oxidative stress, immune modulation, and endometrial remodeling. 14 Ectopic decidualized stroma, or decidua outside the uterus, is termed deciduosis. Deciduosis is an asymptomatic condition commonly observed during pregnancy in humans and nonhuman primates, reported at a rate of up to 100%. 20,26,30 Ectopic deciduae in women are most often identified on the ovaries and uterine serosa, but there are reports of peritoneal, omental, intestinal, lymphatic, pulmonary, and cutaneous decidualization. 33 For humans, the medical literature contains many reports of ectopic deciduae mimicking metastatic lesions. 20,23,25,26 Endometrial decidualization, while not a neoplastic process, appears both grossly and microscopically similar to malignant neoplastic lesions. The lesion infiltrates the serosal surface of all abdominal viscera, as occurs in carcinomatosis from metastatic adenocarcinomas, squamous cell carcinomas, and malignant deciduoid mesothelioma. 16,23,25 Decidualized endometrial stromal cells observed on a Papanicolaou smear may confound diagnosis of a cervical intraepithelial neoplasia. 9 Rarely, decidualized aberrant stroma or endometriosis tissue may also directly complicate pregnancy through hemoperitoneum, intestinal perforation, or pneumothorax. 11,19,20
Endometrial stroma in nonhuman primates decidualizes in response to progesterone stimulation as in humans. Deciduosis has been described in various species of monkey, including Goeldi’s (Callimico goeldii) and common marmosets (Callithrix jacchus), 21 squirrel monkeys (Saimiri sciureus), 22 and rhesus macaques (Macaca mulatta). 5 It typically occurs within the endometrium as well as on the serosal surface of the uterus and other abdominal organs. Tissue changes with endometrial cell morphology similar to that of pregnancy-induced deciduae have been reported after progestin administration for menstrual cycle control or treatment of endometriosis. 5,25,34 In many of these cases, animals received melengestrol acetate or medroxyprogesterone acetate for contraception or endometriosis treatment. 5
In this study, we report on the clinical, macroscopic, histopathologic, and immunohistochemical features of 6 cases of exuberant deciduosis caused by endometriosis tissue decidualization in 4 rhesus macaques (M. mulatta) and 2 cynomolgus macaques (Macaca fascicularis). Three of these cases developed without stimulation from exogenous progesterone, in contrast to previous reports of deciduosis. The purpose of this article is to characterize endometriosis decidualization in nonhuman primates and further develop them as a model for studying decidualization, developing new methods for differentiating exuberant deciduae from malignancies, and identifying new treatments.
Materials and Methods
Six intact female macaques aged between 14 and 31 years (mean age, 19.8 years) were identified during clinical evaluations at the Armed Forces Radiobiology Research Institute (AFRRI) or at the Wake Forest Primate Center at the Wake Forest School of Medicine (WFSM). Macaque care and experimentation were approved by each institution’s Institutional Animal Care and Use Committee and maintained in accordance to federal and state laws, including the National Academy of Sciences’ Guide for the Care and Use of Laboratory Animals. 24 In addition, the WFSM and AFRRI are accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International. Two rhesus macaques were part of a chronic gastric carcinogenesis study being conducted at the AFRRI, were receiving nitrosating carcinogen ethyl-nitro-nitrosoguanidine, and were experimentally infected with Helicobacter pylori. A third rhesus was part of a study at WFSM about opioid and pain receptors. The remaining rhesus macaque and the cynomolgus macaques were either on holding protocols or part of a WFSM breeding colony. Of the 6 cases, 3 had been treated with progesterone analogs (case Nos. 1, 3, and 6; medroxyprogesterone acetate) and 1 with an aromatase inhibitor (case No. 4; letrozole) for endometriosis.
After the initial clinical workup, cases were presented to pathologists at the Division of Comparative Pathology at the AFRRI or the Department of Pathology at the WFSM for pathologic evaluation. Diagnostic necropsies were performed on each animal, and tissues were collected. Affected tissues to include grossly recognized decidua were stained with hematoxylin and eosin and various combinations of vimentin (clone SP20, rabbit monoclonal, 1:40; Vector Laboratories, Burlingame, CA), common acute lymphoblastic leukemia antigen (CALLA, CD10, clone 56C6, mouse monoclonal, 1:20; Abcam, Cambridge, UK), progesterone receptor (Novocastra, clone 1A6, mouse monoclonal, 1:100; Vision Biosystems, Norwell, MA), estrogen receptor alpha (clone 6F11, mouse monoclonal, 1:100; Leica Microsystems, Wetzlar, Germany), desmin (clone Y66, rabbit monoclonal, 1:250; Epitomics, Burlingame, CA), cytokeratin AE1/AE3 (clone LU-5, mouse monoclonal, 1:50; BioCare Medical, Concord, CA), kermix P8, human chorionic gonadotropin (rabbit monoclonal, 1:200; BioCare Medical), human placental lactogen (Novocastra, U22 HPL, rabbit monoclonal, 1:200; Vision Biosystems), and calretinin. Secondary antibodies for the appropriate species were used at a 1:20 dilution (Super Sensitive Kit; BioGenex). Vector Red (Vector Laboratories) or 3,3′-diaminobenzidine were used as chromogens, and slides were counterstained with Mayer’s hematoxylin. Negative controls included slides treated with only secondary antibody. All immunohistochemical stains were validated and performed by histology cores at each location. Board-certified veterinary pathologists or a board-certified medical pathologist evaluated all cases and slides.
Results
Clinical Presentation and Gross Pathology
A summary of clinical findings is shown in Table 1. The 6 cases included 4 rhesus macaques and 2 cynomolgus macaques. The age of affected animals was 19.8 ± 6.0 years (mean ± SD). Affected macaques commonly presented with anorexia with or without weight loss and abdominal pain. Only 3 of the 6 macaques had a history of progesterone treatment (medroxyprogesterone acetate, Depo-Provera). Serosanguineous or sanguineous abdominal effusion was present in 3 of 6 macaques. One rhesus (case No. 6) was in heart failure at the time of death and had serous abdominal effusion. Only 1 macaque (case No. 5) had a history of heavy menses.
Clinical Findings in Animals With Decidualized Endometriosis.a

aCase Nos. 1, 3, and 6 were treated with exogenous progesterone. Case No. 4 was treated with an aromatase inhibitor (letrozole).
At necropsy, the uterus was commonly enlarged and disfigured or replaced by dense connective tissue. Fibrous adhesions were commonly observed, especially between the omentum and the uterus, liver, spleen, and urinary bladder (Fig. 1). In 3 cases, one or both ovaries were not identified and were suspected to be incorporated or effaced by the fibrous adhesions. Diffuse tan, red, or black solid or cystic nodules were identified on the uterus, urinary bladder, intestines, and ovaries of 4 of the 6 macaques. The gross lesions were similar in treated and untreated macaques (Table 1).
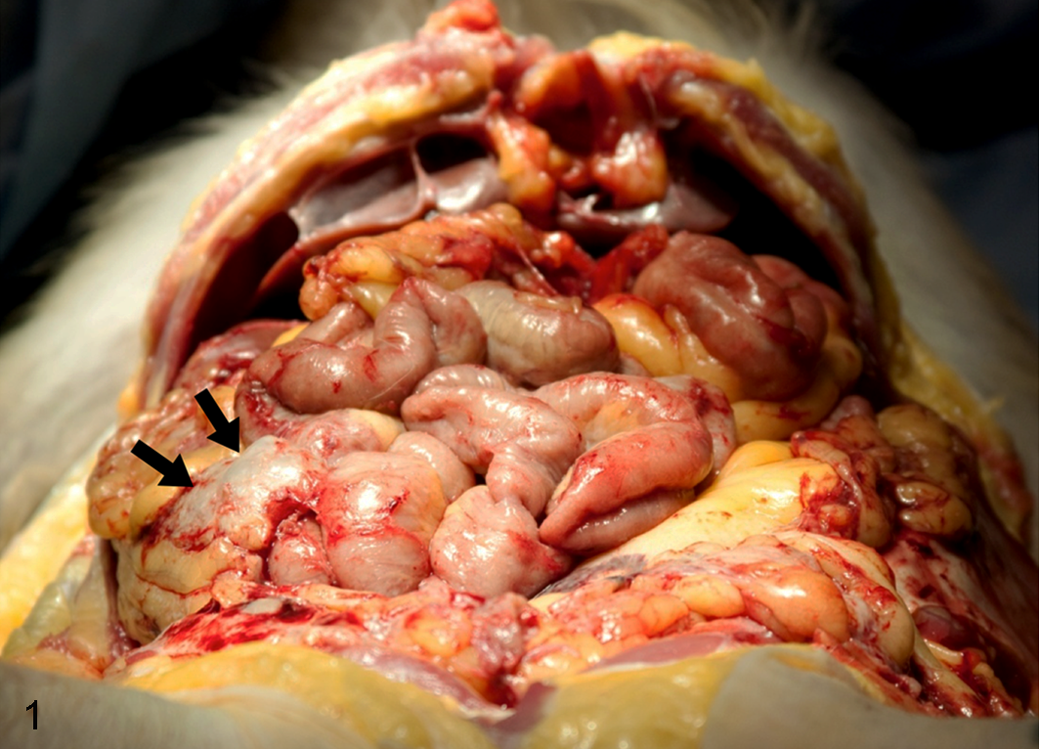
Decidualized endometriosis, abdominal cavity, rhesus macaque, case No. 1. Fibrous adhesions between the omentum and other abdominal organs causing a mass of roughened fused intestinal loops with irregular serosal surfaces (arrows).
Histopathology and Immunohistochemistry
Histopathologic features of the exuberant decidualized endometriosis tissue were very similar to lesions previously described in humans. Endometriotic cysts and islands of decidualized stroma were separated by variably dense bands of fibrous connective tissue (Fig. 2). Hemosiderin-laden macrophages were often identified near cysts (Fig. 3). Cysts were lined by pseudostratified ciliated columnar epithelium and filled with erythrocytes, cellular debris, and degenerate neutrophils and macrophages (Fig. 4). Decidualized endometriotic stromal cells were polygonal with distinct cell borders, abundant pale eosinophilic cytoplasm, and brightly eosinophilic intracytoplasmic globules, which occasionally marginalized the nucleus. Nuclei were round to oval with finely stippled chromatin and 1 or 2 nucleoli (Fig. 5). No mitoses were noted in any of the cases, but anisocytosis and anisokaryosis were present.
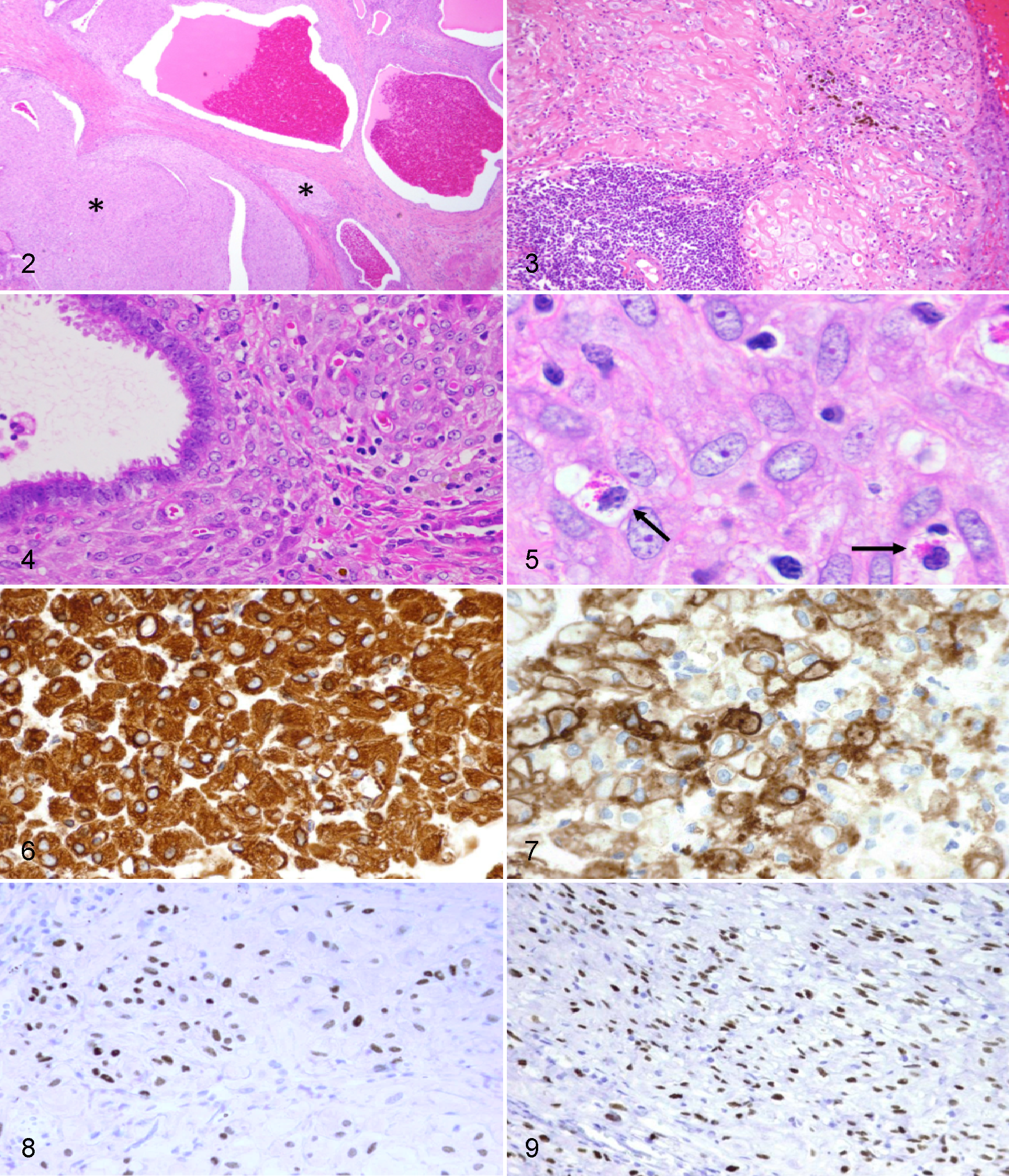
Endometrial granular leukocytes, a specialized type of gamma/delta T cell, were scattered in the decidualized endometriosis stroma. These were small cells with a high nuclear-to-cytoplasmic ratio (Fig. 5, small arrows). They contained eosinophilic granules eccentrically clustered within scanty cytoplasm. The nuclei were dense and deeply basophilic without recognizable nucleoli.
Decidualized stroma in these cases consistently stained positive for nuclear progesterone and estrogen receptors, C10, and cytoplasmic vimentin (Table 2, Figs. 6–9). Desmin, calretinin, cytokeratin, chorionic gonadotropin, cytokeratin AE1/AE3, U22 HPL, and kermix P8 were inconsistent or negative.
Immunohistochemistry Results for Endometriosis Decidualized Stroma by Case.a
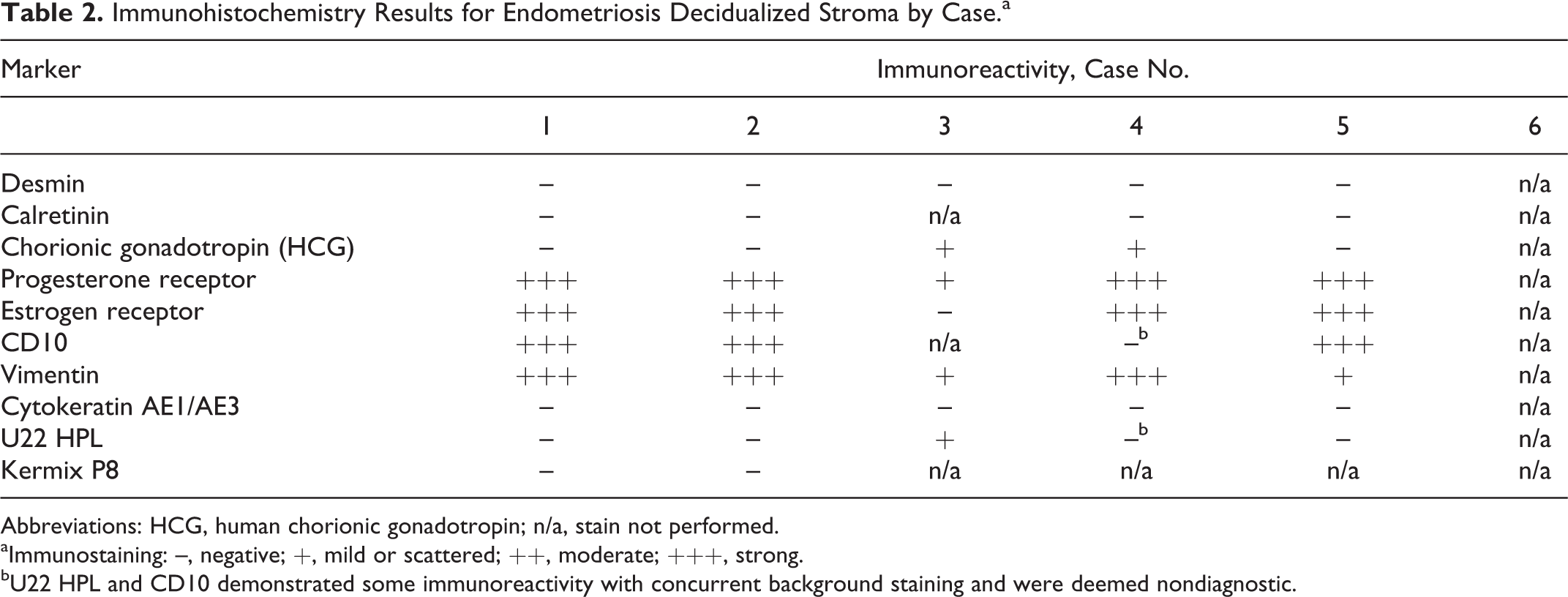
Abbreviations: HCG, human chorionic gonadotropin; n/a, stain not performed.
aImmunostaining: –, negative; +, mild or scattered; ++, moderate; +++, strong.
bU22 HPL and CD10 demonstrated some immunoreactivity with concurrent background staining and were deemed nondiagnostic.
Discussion
Decidualization of the endometrium or ovarian tissue is a normal physiologic process and occurs in up to 100% of human pregnancies. However, exuberant decidualized tissue—especially when it occurs in combination with endometriosis—may endanger the mother and/or the fetus by causing pregnancy complications or being mistaken for neoplasia. 1,18,20 Stroma of endometriosis lesions and endometrium with hormonal stimulation from high progesterone may transform into decidualized cells. The typical microscopic appearance of the transformed endometrial stroma within the myometrium (internal), peritoneum, thoracic cavity, or skin (external) is seen as islands of large polygonal cells with abundant eosinophilic cytoplasm frequently containing intracytoplasmic droplets. These droplets have been described as sulfated mucin and glycogen. 12,17 Decidualized stroma cells have oval, centrally located nuclei with distinct magenta nucleoli and finely stippled chromatin. While no mitoses are typically observed, there is often pronounced anisocytosis and anisokaryosis. The population of transformed cells is often embedded in swaths of histopathologically typical endometriosis: endometrial stroma, endometrial glands lined by ciliated epithelium, pools of hemorrhage, and hemosiderosis. Furthermore, there is often abundant lymphoplasmacytic inflammation, hemorrhage, and varying degrees of necrosis. 26
The cellular and nuclear atypia, as well as the implantation of these cells into the serosa of a myriad of abdominal organs, may resemble a malignant neoplastic process, such as deciduoid mesothelioma, squamous cell carcinoma, mucin-producing adenocarcinoma, or carcinomatosis. 8,17,27,35 Gross differential diagnoses for a decidualized stroma within the abdomen include decidualized mesothelioma, endometriosis with granulomatous inflammation, fibroplasia, granulation tissue, and carcinomatosis, especially if tissue invades and destroys the structural integrity of abdominal organs. 27 Atypical extraplacental trophoblastic implantation of the peritoneal cavity is another differential in animals with a history of pregnancy. 6,10,31 The rhesus macaques in case Nos. 1 and 2 (Table 1) were both exposed to a nitrosamine carcinogen. Genital papillary mesotheliomas have been described in male rats given nitrosamines. 15 Finally, differential diagnoses for abdominal neoplasms following aromatase inhibitor treatment, as in case No. 3, should include atypical mesenchymal proliferation induced by the aromatase inhibitor treatment or, potentially, a neoplastic mesenchymal transformation, as noted in cases of retroviral-induced retroperitoneal fibromatosis. 13 Retroperitoneal fibromatosis is histologically distinct from deciduosis since it is composed of dense connective tissue without the large polygonal decidualized stromal cells. 28
In nonhuman primates, decidualization of endometriosis tissue occurs most often under the influence of exogenous progestins or during pregnancy. 5,25,34 Here only 3 of the 6 cases had received exogenous progestins (medroxyprogesterone acetate). None of these animals were pregnant, so we speculate that the endometriotic tissue decidualized under the influence of elevated endogenous progesterone.
Recognizing the histopathologic appearance of the decidualized cells and correlating their presence with endometriosis are important criteria toward appropriate diagnosis. Furthermore, immunohistochemical markers aid in confirming the transformed endometrial stroma. We found that the decidualized stroma stained positive for nuclear progesterone and estrogen receptors and cytoplasmic vimentin as well as CD10 (Table 2). These histopathologic characteristics were consistent with reported results in studies of affected humans and nonhuman primates. Typical endometrial stroma stains positively for common acute lymphoblastic leukemia antigen, or CD10, which is expressed by hematopoietic neoplasms as well as the endometrium. 2,4,5,32 Additionally, the nuclei of the stroma have positive staining for progesterone receptor and estrogen receptor. Human chorionic gonadotropin and U22 HPL staining are often assessed to rule out a trophoblastic implantation, while cytokeratin markers are evaluated to reject carcinomas as possible diagnoses. 6,31 Scattered immunopositivity to both human chorionic gonadotropin and U22 HPL observed in 2 of the 5 cases stained may represent a variation in laboratory-specific sensitivity. Both positive cases were processed within the same immunohistochemistry laboratory. Desmin has been reported to have variable staining in decidualized endometriosis in humans, interpreted as metaplastic transformation of the stroma into myofibroblasts or leiomyocytes, reinforcing the argument of endometrial stroma plasticity. 7 In all our cases, the stromal cells were negative to desmin. A recent report of pleural endometriosis in a rhesus macaque found that the endometrial stroma was positive to calretinin, which is an immunohistochemical marker noted to demonstrate strong nuclear and cytoplasmic staining of the normal endometrial stroma while showing decreased or negative reactivity in pathologic uterine tissues to include deciduosis—a finding supported in the immunohistochemical evaluation of all 4 cases evaluated with calretinin. 3,4
In summary, endometrial stromal decidualization was confirmed on the basis of the histopathologic features and immunohistochemical analyses of each case. Macroscopically, the extensive invasive spread of the endometriosis, multifocal islands of decidualized stromal cells, abundant hemorrhage, and variable regional necrosis mimicked malignancy. Histopathologic atypia noted within the decidualized cells represents a further confounding feature. These observations aid in identifying and diagnosing such lesions in primates to serve as models for decidualized endometriosis lesions in humans. Understanding endometrial decidualization in nonhuman primates with endometriosis will aid to elucidate the etiology of decidualized endometriotic stroma in women and further the investigation of deciduosis diagnosis and treatment.
Footnotes
Authors’ Note
Dr Dubois passed away on June 30, 2012, after making significant contributions to the conception, design, and initial drafts of the manuscript.
Acknowledgements
We acknowledge Hermina Borgerink, Jean Gardin, and Joseph Finley from the Wake Forest School of Medicine for their histology technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded in part by National Institutes of Health training grant T32OD010957-37.
