Abstract
Chromatophoromas are neoplasms arising from pigment-bearing cells (chromatophores) of the dermis. While isolated cases have been reported in the literature, the prevalence and biological behavior of chromatophoromas in snakes are unknown. Forty-two chromatophoromas were identified among 4663 submissions (0.9%) to a private diagnostic laboratory in a 16-year period. The most commonly affected snakes were colubrids (23 cases, 55%) and vipers (8 cases, 19%). The San Francisco garter snake was the most commonly affected species (6 cases; 14% of all affected snake species and 3.7% of all garter snake submissions). No sex predilection was found. The age of 28 snakes ranged from 5 to 27 years. Single cutaneous chromatophoromas were most commonly observed and presented as pigmented cutaneous masses or plaques along any body segment. Euthanasia or death due to progressive neoplastic disease or metastasis was reported in 8 (19%) and 4 (10%) cases, respectively. The survival time of 4 animals ranged from 4 to 36 months. Microscopically, xanthophoromas, iridophoromas, melanocytic neoplasms, and mixed chromatophoromas were identified, with melanocytic neoplasms being most common. Microscopic examination alone was generally sufficient for the diagnosis of chromatophoroma, but immunohistochemistry for S-100 and PNL-2 may be helpful for diagnosing poorly pigmented cases. Moderate to marked nuclear atypia appears to be consistently present in cutaneous chromatophoromas with a high risk of metastasis, while mitotic count, lymphatic invasion, the level of infiltration, and the degree of pigmentation or ulceration were not reliable predictors of metastasis.
The skin of reptiles, amphibians, and fish is characterized by a wide pattern of colors. This phenotypical feature is mediated by chromatophores, a group of pigment-bearing and light-reflecting cells that reside in the dermis. In reptiles, chromatophores include xanthophores (containing carotenoids and pteridine [red or yellow]), iridophores (containing birefringent purine crystals [variable color]), and melanophores (containing melanosomes [black]). 2,13 The nomenclature of tumors arising from chromatophores has been historically based on their cell type origin (eg, melanophoroma), which is often characterized by either light microscopy or ultrastructural features. 7,8 In the current literature, there are multiple single-case reports of chromatophoromas in snakes, including benign melanophoromas in an eastern yellowbelly racer 21 and a pine snake; 9 malignant chromatophoromas in a canebrake rattlesnake 7 and a gopher snake; 17 malignant melanophoromas in death adder, 10 yellow rat, 11 Florida pine, 4 and western terrestrial garter snakes; 5 and malignant iridophoroma in a pine snake. 9 Also, a recent report of chromatophoromas in reptiles described 5 benign melanophoromas and 1 malignant melanophoroma. 8 To the authors’ knowledge, there are no large retrospective studies regarding the prevalence and clinicopathological features of cutaneous chromatophoromas in snakes. Likewise, specific prognostic markers for this type of neoplasm in this species have not been evaluated. Therefore, the aims of this retrospective study were 1) to provide a comprehensive clinicopathological description of 42 cases of cutaneous chromatophoromas in snakes, 2) to identify prognostic markers that accurately identify the more aggressive cases of these neoplasms, and 3) to determine the usefulness of immunohistochemical markers to distinguish melanocytic neoplasms in snake chromatophoromas.
Materials and Methods
Animals
Cases submitted to the Northwest ZooPath during the years 1994 to 2010 were considered for this study. Essential criteria for inclusion were snake cases with cutaneous masses or plaques with gross and/or histologic features consistent with chromatophoroma (ie, tumors composed of pigmented neoplastic cells). All the samples included in this study correspond to either surgical biopsy specimens or postmortem tissues collected by clinicians for diagnostic purposes.
Histopathology
Tissue samples were submitted and maintained in 10% neutral buffered formalin for up to 5 days prior to being processed routinely for histology. When required, decalcification of specimens with 12% HCl solution was performed for approximately 1 day. Chromatophoromas were histologically categorized based on the predominant type of intracellular pigment. Since melanophoromas closely resemble melanocytic neoplasms in mammalian species, we will refer to them as melanocytic neoplasms and their malignant forms as malignant melanomas. 17 Histologic categories include melanocytic neoplasm (ie, cells containing dark brown to black pigment), iridophoroma (ie, cells containing distinct birefringent crystals under polarized light), xanthophoroma (ie, cells containing red to brown pigment), and mixed chromatophoroma (ie, containing 2 different chromatophore types). Chromatophoromas were microscopically evaluated according to the histologic factors proposed for canine cutaneous melanocytic neoplasms, including lymphatic invasion, mitotic count, degree of pigmentation, nuclear atypia, level of infiltration, and ulceration. 19 A high-power (40×) objective with a field number of 22 mm was used to count mitotic figures in 10 representative fields (ie, densely populated areas of the tumor that excluded areas of necrosis, hemorrhage, histologic artifacts, ulceration, and edema). Thus, the diameter of the field in which mitotic figures were counted is 2.37 mm2. 12 The degree of pigmentation was categorized as low (<30% of neoplastic cells), moderate (30%–60% of neoplastic cells), and high (>60% of neoplastic cells). Nuclear atypia was classified as mild (<30% of neoplastic cells), moderate (30%–60% of neoplastic cells), and marked (>60% of neoplastic cells). Also, intranuclear pseudoinclusions, which were at some point anecdotally significant in human melanomas, 1,16 were counted in 10 high-power fields (HPFs). One metastatic malignant melanoma (case No. 33) was excluded from this assessment as only the tissue with metastasis but not the primary neoplasm was available for microscopic examination.
Immunohistochemistry
An immunohistochemistry (IHC) panel was performed on 5-μm serial sections from 10 randomly selected chromatophoromas that represented the different histologic categories (case Nos. 2, 3, 9, 10, 14, 16, 24, 25, 27, and 28). The panel included antibodies to detect protein S-100, melanocytic antigen PNL2, melan A, and HMB45. IHC was performed on a Bond autostainer automated system (Leica Microsystems, Bannockburn, IL), using a biotin-free, polymeric alkaline phosphatase–linked antibody detection system with fast red chromogen and hematoxylin counterstain (Bond Polymer AP Red Detection Kit; Leica Microsystems) as previously described. 18 For negative controls, antibodies were replaced with homologous immune sera. Further technical information regarding immunohistochemical staining can be found in Supplemental Table S1.
Results
Animals
A total of 4663 snake submissions were identified at the laboratory during the period of study. Submissions from 42 (0.9%) snakes met the criteria of inclusion of this study. Thirty-seven cases corresponded to snakes that belonged to zoological collections, and 5 cases were from snakes kept as pets that were seen at private veterinary clinics.
Signalment and Clinicopathological Findings
Supplemental Table S2 summarizes the signalment and relevant clinical and pathological findings from each of the cases included. Chromatophoromas were identified in 23 colubrids (55%), 8 vipers (19%), 7 boids (17%), 3 pythons (7%), and 1 elapid (2%). Chromatophoromas were most commonly detected in the San Francisco garter snake (Thamnophis sirtalis tetrataenia) with 6 cases (14% of all affected snakes and 3.7% of all garter snake submissions), followed by the bullsnake (Pituophis catenifer sayi) with 3 (7%) cases, and the bush viper (Atheris squamigera), rosy boa (Lichanura trivirgata), corn snake (Elaphe guttata), tricolor hognose snake (Lystrophis pulcher), and the Mojave desert sidewinder (Crotalus cerastes cerastes) with 2 (5%) cases each. The remaining 25 cases were found in 25 different species (Suppl. Table S2). Fifteen (36%) snakes were female, 8 (19.1%) were male, and the sex was not reported for 19 (45%) cases. The age was known for 28 snakes and ranged from 5 to 27 years (12.9 years on average).
Neoplasms varied in size from 3 to 70 mm in greatest dimension. Clinical signs and the anatomical location of the neoplasms were not described for all the cases. Single masses were reported in 38 cases (91%; Figs. 1, 2, 4), and multiple masses (Fig. 3) were present in 4 cases (10%). In 6 cases (14%), neoplasms were described as swollen scales or plaque-like lesions (Figs. 1, 3). The anatomical location of the chromatophoromas was known for 20 cases. The most common location for tumor development was the caudal third of the body with 7 (35%) cases, followed by the mid third of the body with 4 (20%) cases. The remaining neoplasms were located in the skin of the cranial third of the body (3 cases), pericloacal skin (3 cases, all of which involved the mucocutaneous junction), and head (3 cases). Concurrent diseases in snakes with cutaneous chromatophoroma included oophoritis (n = 2), renal adenocarcinoma (n = 2), and 1 case of each of hemangiosarcoma, cutaneous hemangioma, biliary cystadenoma, weight loss, anorexia, regurgitation, spondylosis, renal disease, systemic granulomatous inflammation, chronic cestodiasis, cryptosporidiosis, and hypercholesterolemia with atherosclerosis of great vessels. Euthanasia due to poor prognosis associated with progressive neoplastic disease (ie, local tumor recurrence, local invasion, and/or presence of multiple masses) was reported in 8 cases (case Nos. 5, 16, 23, 27, 29, 31, 32, and 33). Four snakes were found dead, which was attributed to invasive chromatophoroma and concurrent granulosa cell tumor (case No. 2) and metastatic chromatophoroma (case Nos. 14, 20, and 28). The survival time was known for 4 cases and was 4 months (case No. 20), 6 months (case Nos. 14 and 28), and 36 months (case No. 2).
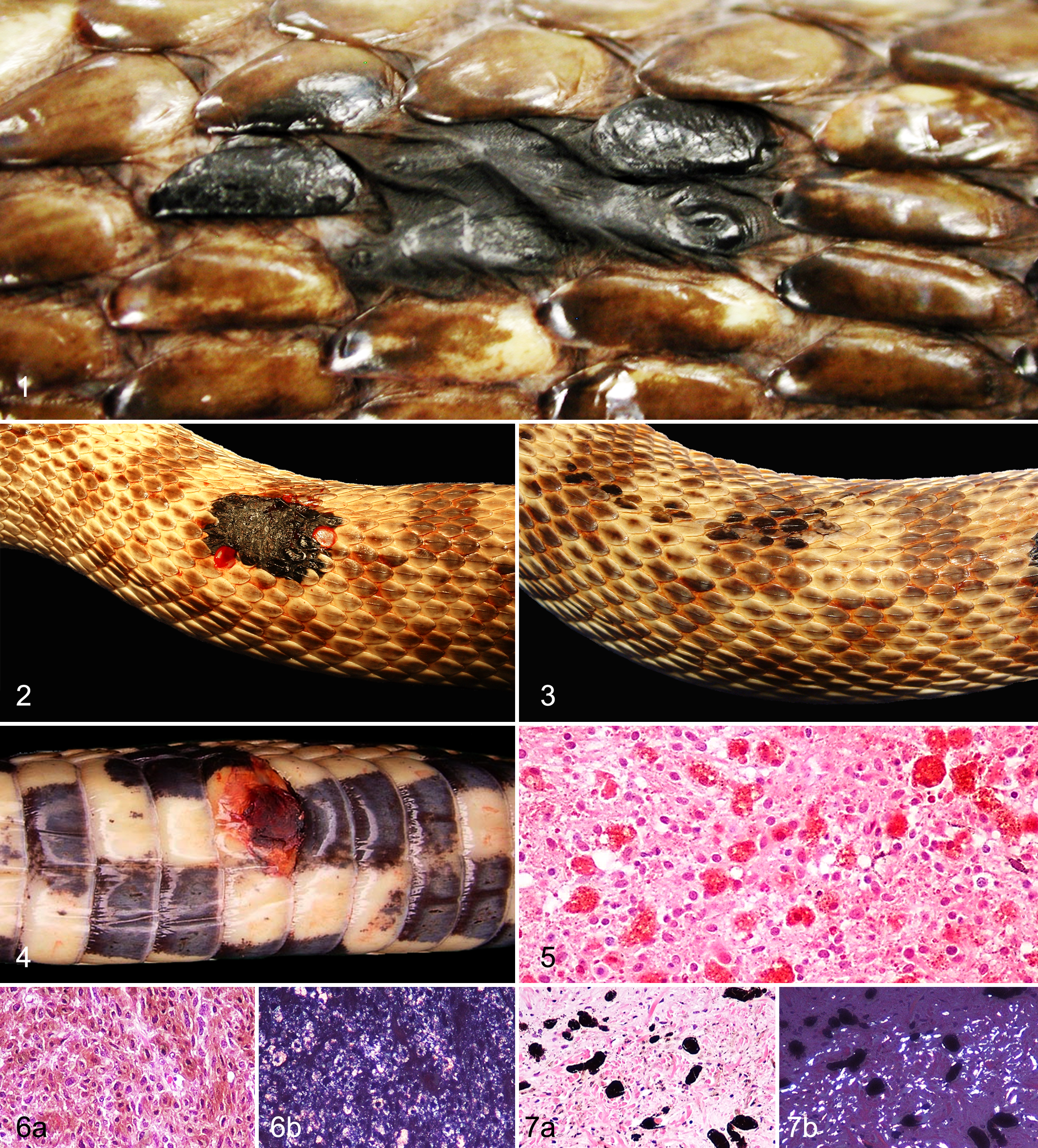
Histopathology
Chromatophoromas were poorly demarcated, unencapsulated, expansile, and moderately to highly cellular. Neoplastic cells were mainly spindle-shaped (31 cases). Tumors with polygonal (8 cases) and epithelioid (3 cases) neoplastic cells were also observed. Chromatophoroma cells were separated by variable amounts of collagenous stroma and were arranged in streams, bundles, and solid or loose sheets. Neoplastic cells had variably distinct margins with moderate to abundant cytoplasm and variable amounts of intracytoplasmic pigment. Pigment was either red to brown (xanthophores), brown to amber and birefringent (iridophores), or brown to black (melanocytic neoplasms). Thus, chromatophoromas were categorized as xanthophoromas (1 case; Fig. 5), iridophoromas (6 cases; Fig. 6a,b), or melanocytic neoplasms (26 cases; Fig. 8). In some cases, chromatophoromas were composed of mixed populations of neoplastic melanophores and iridophores, and these were categorized as mixed chromatophoromas (9 cases; Fig. 7a,b). The degree of pigmentation was low for 22 cases (Figs. 9, 10), moderate for 5 cases, and high for 15 cases (Fig. 8).
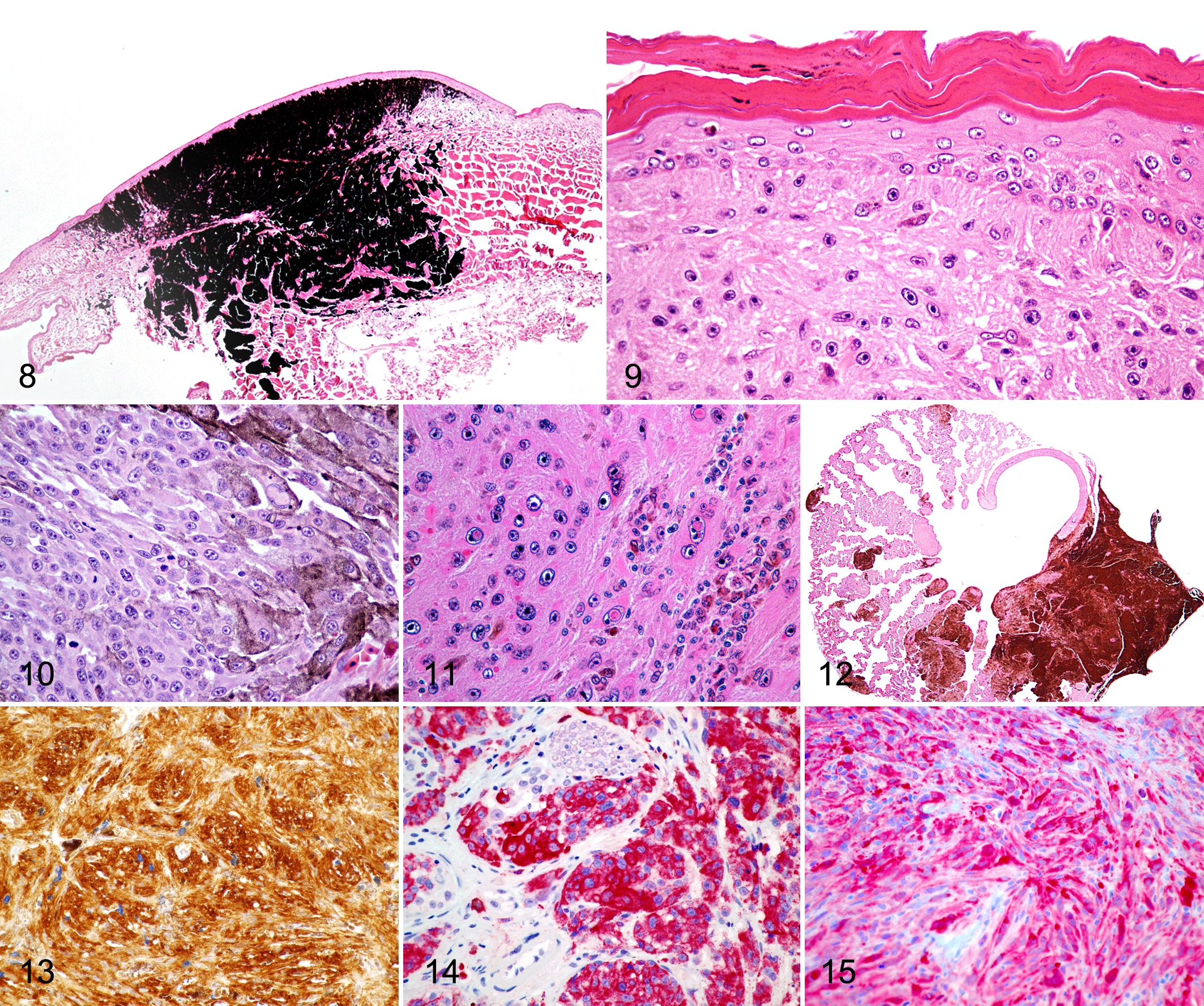
Nuclei were round to oval with finely stippled (6 cases) to vesicular (36 cases) chromatin and contained 1 to 5 small nucleoli. Nuclear atypia was mild in 15 neoplasms, moderate in 17, and marked in 9. The mitotic count ranged from 0 to 33 in 10 representative HPFs but was frequently low. In 11 cases, neoplastic cells contained eosinophilic, nuclear membrane invaginations (pseudoinclusions; Fig. 11). Ulceration in association with heterophilic and mononuclear inflammation was observed in 9 cases. No junctional activity or lentiginous spread was present in any of the tumors (Fig. 9). Lymphatic invasion was observed in a malignant melanoma (case No. 2) and an iridophoroma (case No. 24). Tumor infiltration into the subcutaneous tissue was observed in 6 cases (case Nos. 3, 8, 28, 29, 36, and 41), including 3 melanocytic tumors, 1 mixed chromatophoroma, 1 malignant melanoma, and 1 malignant iridophoroma. In 2 cases, 1 malignant melanoma and 1 malignant mixed chromatophoroma (case Nos. 5 and 14), the neoplastic cells infiltrated skeletal muscle. Moreover, 2 melanocytic tumors (case Nos. 2 and 38) infiltrated vertebral bone. In 5 biopsy cases (12%), complete excision was histologically achieved, and in 28 cases (67%), the tumor infiltrated the excisional margins. Recurrence of incompletely excised melanocytic neoplasms was reported in 7 cases for which follow-up clinical history was available. Metastases (Fig. 12) were found in 10 cases, and these primarily involved kidneys (case Nos. 14, 19, 20, 23, 28, and 33), liver (case Nos. 14, 16, 18, 20, and 28), and lungs (case Nos. 5, 18, and 20). Other less common sites of metastatic spread included spleen, aorta, heart, mesentery testis, intestine, bone marrow, and trachea. Metastatic chromatophoromas were primarily malignant melanomas (7 cases), while only 2 malignant iridophoromas and 1 malignant mixed chromatophoroma had evidence of metastasis in the study population.
Assessment of Prognostic Markers in Metastatic Chromatophoromas
We sought to determine prognostic markers of chromatophoroma based on the current knowledge of melanocytic tumors in dogs. 19 Parameters examined in the primary tumor of 9 metastasizing cutaneous chromatophoromas (6 malignant melanomas, 2 malignant iridophoromas, 1 malignant mixed chromatophoroma) included lymphatic invasion, mitotic count, degree of pigmentation, nuclear atypia, level of infiltration, presence of intranulcear pseudoinclusions, and ulceration (Table 1). One metastatic case (case No. 33) was not included in this assessment as the primary cutaneous neoplasm was not available for microscopic examination. Lymphatic vascular invasion was not observed in any metastasizing neoplasm. Across all types of chromatophoromas, the mitotic count in 5 tumors ranged from 2 to 31; however, no mitoses were seen in 4 malignant melanomas that metastasized. The degree of pigmentation was variable, and highly pigmented chromatophoromas were more common (4 cases) than poorly pigmented (3 cases) and moderately pigmented (2 cases) neoplasms. Nuclear atypia was moderate in 7 metastasizing neoplasms and marked in 2 cases. Two metastasizing chromatophoromas infiltrated the subcutaneous adipose tissue, and 2 other neoplasms infiltrated the skeletal muscle. Only 3 metastasizing chromatophoromas contained intranuclear pseudoinclusions, and these ranged from 3 to 7 in 10 HPFs. Ulceration was seen in only 2 cases.
Histomorphologic Features of Metastatic Chromatophoromas in Snakes.
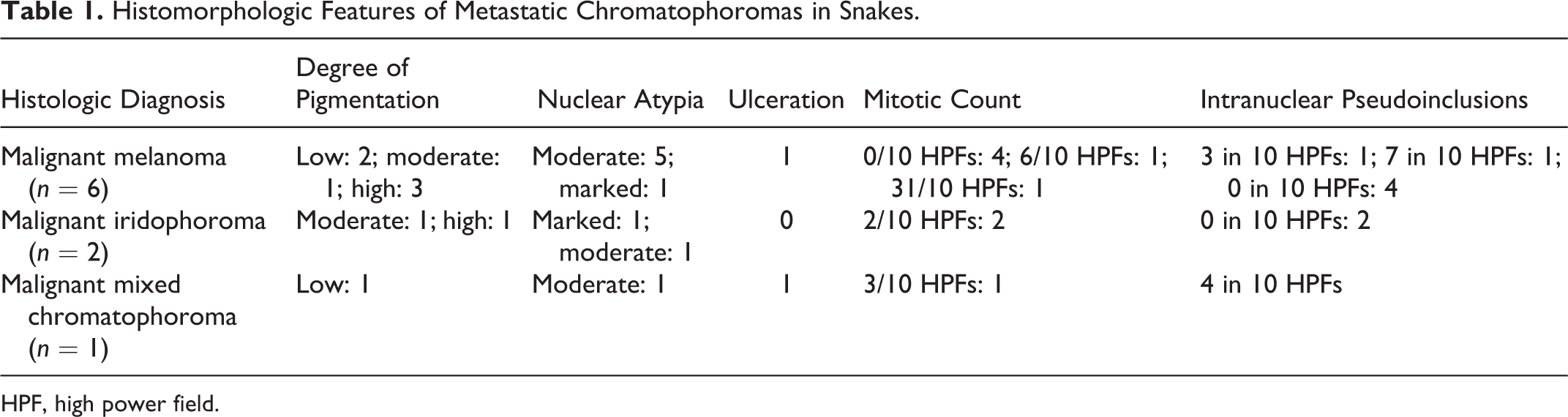
HPF, high power field.
Immunohistochemistry
Five melanocytic neoplasms (2 malignant melanomas), 2 iridophoromas, and 3 mixed chromatophoromas (1 malignant mixed chromatophoroma) were labeled with S-100, PNL-2, melan A, and HMB45 (Table 2). Neoplastic cells in all cases except 1 melanocytic neoplasm were positive for S-100 (Fig. 13). Three melanocytic neoplasms, 2 mixed chromatophoromas, and 1 malignant iridophoroma were positive for PNL-2 (Fig. 14). Only the malignant mixed chromatophoroma was positive for melan A (Fig. 15), and neoplastic cells in 1 melanocytic neoplasm, which was negative for S100 but positive for PNL-2, had trace reactivity for melan A. Only 1 mixed chromatophoroma was positive for HMB45.
Immunohistochemical Staining Profiles of Cutaneous Chromatophoromas in Snakes.a

+, positive; –, negative; +/–, trace.
aCase numbers correspond to those given in Supplemental Table S1.
Discussion
This retrospective study characterizes the clinicopathological features of cutaneous chromatophoromas in 42 captive snakes. According to our results, the relative prevalence of chromatophoromas in snakes is low (about 1% of the histopathology submissions to a private laboratory), which is consistent with a previous report of chromatophoromas in reptiles. 8 Likewise, the relative prevalence of pigmented neoplasms in snakes is lower than that reported for mammalian species, including dogs (7%) and horses (15%). 8,20 The reason for the lower prevalence is unclear but may depend on differences in cell type origin and environmental conditions in which reptiles are maintained in captivity. Chromatophoromas were more commonly seen in colubrids, and the San Francisco garter snake was overrepresented. The sex for most of the cases was unknown, and a sex predisposition cannot be determined. Similar to melanocytic neoplasms in mammalian species, cutaneous chromatophoromas occurred in middle- to old-age animals, and the average age of affected snakes was 12.9 years. The macroscopic presentation of these neoplasms was highly variable, and practitioners should be aware that cutaneous chromatophoromas may occur not only as expansile or nodular masses but also as flat, pigmented plaques affecting single or multiple scales. In addition, chromatophoromas appear to have the potential to occur at any body site, and invasion of subcutis, skeletal muscle, or bone is not uncommon. However, euthanasia or death due to progressive or metastatic disease was reported in only 29% of cases. Unfortunately, due to the relatively small sample size (n = 4), overall survival times and biological behavior of pigmented tumors in snakes cannot be reliably interpreted based on our data. Metastatic disease was only reported for 10 cases, and metastases primarily involved the kidneys, liver, and lungs. Nevertheless, the metastatic rate may have been higher, since not all tissues were examined microscopically for each case, and microscopic metastatic foci may have been missed at necropsy.
In our study, melanocytic neoplasms were the most common type of chromatophoroma, and 6 of the 9 cases with metastases were malignant melanomas. Moderate to marked nuclear atypia was the most consistent histologic finding among metastasizing cutaneous chromatophoromas and may be considered a potential prognostic marker. Further investigation, however, is required to definitively establish this microscopic feature as a prognostic marker of chromatophoromas in snakes. Neither junctional activity nor lentiginous spread was present in any of the neoplasms, likely because the cell of origin in reptiles is primarily dermal as opposed to epidermal. Ulceration was present in 2 metastatic chromatophoromas and 7 cases with no reported metastasis, and it was likely due to secondary trauma, vascular compromise, or direct extension through the epidermis by the tumor, rather than the more common junctional involvement seen in mammalian and avian counterparts. Although the mitotic count was generally low in this case series, it varied greatly across neoplasms with no reported metastases and appeared to be consistently low in metastasizing tumors. The absence of metastasis in neoplasms with a high mitotic count and moderate to marked nuclear atypia may be due to the lack of follow-up information (eg, no necropsy was performed or microscopic metastases that were inapparent to the prosector). Deep infiltration into the subcutaneous tissue, skeletal muscle, or bone was observed in 4 metastasizing neoplasms and 2 cases with no history or evidence of metastatic disease. Intranuclear pseudoinclusions were consistently low in metastasizing tumors.
Immunohistochemistry is the primary tool for the definitive diagnosis of poorly pigmented melanocytic neoplasms in different mammals. 14,15,18 All cases included in this study had at least a low degree of pigmentation (ie, 30% of neoplastic cells), and IHC was not necessary for the definitive diagnosis of chromatophoroma. Nevertheless, we studied the diagnostic value of some melanocytic immunohistochemical markers in snakes. Expression of protein S-100 was found in 9 of 10 cases and was the most reliable marker for a pigmented neoplasm. However, while S-100 immunolabeling has been shown to be highly sensitive for the diagnosis of melanocytic neoplasms in mammals, it has a low specificity. 10,14,15,18 For instance, we would expect expression of S-100 to occur in nerve sheath tumors, which have been reported in snakes. 6 PNL-2 has been demonstrated as a highly specific and sensitive marker for the diagnosis of melanocytic neoplasms in multiple animal species. 3,15,18 Nevertheless, PNL-2 was less sensitive than S-100 as only 6 chromatophoromas were immunopositive. More important, only 3 of the 6 melanocytic neoplasms and 2 of the 3 mixed chromatophoromas were positive for PNL-2, while 1 of the 2 iridophoromas was also positive. The sensitivity of melan A and HMB45 appears to be extremely low in reptile melanocytic tumors. While S-100 and PNL-2 may be used to support a diagnosis of chromatophoroma in snakes and PNL-2 is highly specific for a diagnosis of pigmented neoplasms in mammals, neither antibody was useful in distinguishing between the different types of chromatophoromas or to determine biological behavior and did not improve diagnostic accuracy over conventional histologic examination. Finally, these immunohistochemical findings highlight the urgent need for validating the reagents created for mammalian species in nonmammalian species to enhance diagnostics of reptile and other exotic species.
In summary, a presumptive diagnosis of chromatophoroma should be made when pigmented cutaneous masses are present along any body segment. Microscopic examination is generally sufficient for the diagnosis of chromatophoroma, but IHC for S-100 and PNL-2 may be helpful for supporting a diagnosis of chromatophoroma for poorly pigmented cases. Moderate to marked nuclear atypia appears to be consistently present in those cutaneous chromatophoromas that metastasize.
Footnotes
Acknowledgements
We thank the practitioners that provided the gross photographs and the histology laboratory at the Diagnostic Center for Population and Animal Health at Michigan State University for their support with the immunohistochemistry.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
