Abstract
Pigment-containing and light-reflecting cell neoplasms, generically termed chromatophoromas, affect fish, reptiles, and amphibians. Chromatophoromas of light-reflecting cells are named iridophoromas. In this study, we aimed to describe the gross, histologic, and ultrastructural findings of 71 cases of iridophoromas in farmed Siamese fighting fish (Betta splendens). Macroscopically, iridophoromas appeared as whitish, gray, or black friable masses or plaques in the fin, trunk/tail, or head of the fish. Forty-five tumors (63%) were malignant and invaded the adjacent skeletal muscle and/or metastasized to other organs, whereas 26 (37%) tumors were restricted only to the skin, but due to the cytologic similarity to the malignant counterpart, we were not able to classify them as malignant or benign. Sixty-five (91%) tumors were classified as iridophoromas, whereas 6 (8%) were diagnosed as mixed chromatophoromas. Despite immunolabeling for PNL-2, melan A, or S-100 failing to demonstrate antigen expression, ultrastructural analysis identified light-reflecting neoplastic cells, unequivocally confirming iridophoromas as the predominant tumor. The high incidence of iridophoromas in Siamese fighting fish from the same breeding facility, coupled with a higher occurrence in royal blue and fancy copper color patterns and in young males, suggests a potential genetic/hereditary factor in the tumorigenesis of these neoplasms.
The Siamese fighting fish (Betta splendens) is a highly sought-after ornamental fish globally, known for its diverse phenotypic color patterns, a crucial factor determining the quality and market value in the ornamental fish trade.47,49 Similar to reptiles and amphibians, fish skin color patterns result from the spatial arrangement of specialized pigment-containing and light-reflecting cells. 17 In teleost fish, the group of pigment-bearing or light-reflecting cells has been generically referred to as chromatophores and are categorized into distinct types, such as melanophores, xanthophores, erythrophores, cyanophores, leucophores, and iridophores, the latter being light reflectors and responsible for the production of iridescent colors.13,40 Thus, neoplasms originating from pigment or light-reflecting cells in fish, reptiles, and amphibians have been generically referred to as chromatophoromas, and subdivided depending on their chromatophore of origin. For instance, melanophoromas arise from melanophores, xanthophoromas arise from xanthophores, erythrophoromas arise from erythrophores, and iridophoromas arise from iridophores. Histologically, iridophoromas are the only chromatophoroma subtype that is birefringent under polarized light. 23
Chromatophoromas have been more frequently reported in reptiles and fish, with iridophoromas and melanophoromas the most common types.23,29 In reptiles, these pigmented or light-reflecting cell tumors have been reported in Serpentes,25,44 Lacertilia,7,12,16 and less frequently, in Testudines.2,8 There are no reports in Crocodylia and Rhynchocephalia. In amphibians, chromatophoromas have been reported in newts 19 and axolotls. 14
The field of oncology in ornamental fish remains relatively unexplored. 46 Neoplasms in fish are less commonly reported than in domestic animals, and they are usually benign and mainly affect the skin.10,11 Despite occurring in both marine and freshwater species,27,28,33 chromatophoromas are rarely reported in ornamental fish, and only a few studies have explored their pathological, immunohistochemical, and ultrastructural characteristics. 27 Melanophoromas, iridophoromas, and mixed chromatophoromas in fish may present similar gross features, typically appearing as white to gray or black friable masses on the skin.27,36 Histologic examination and electron microscopy have been indicated as important confirmatory techniques to differentiate these neoplasms.6,36
The present study aimed to describe the gross, histologic and ultrastructural features of 71 cases of iridophoromas in Siamese fighting fish in Northeastern Brazil.
Materials and Methods
Case Selection
The 71 cases of chromatophoromas in Siamese fighting fish were identified in the archive of the Veterinary Pathology Laboratory of the Federal University of Paraiba, Northeastern Brazil. From a total of 135 cutaneous fish tumors examined between 2018 and 2022, 71 were histologically confirmed as chromatophoromas and included in this study. All cases originated from a single commercial fish farm specialized in Siamese fighting fish and located in the State of Paraiba, Northeastern Brazil. Cases of skin tumors with characteristic macroscopic appearances compatible with chromatophoromas, but did not have histologic confirmation, were not included in this study. This study was authorized by the University Institutional Ethics on Animal Use Committee of the Federal University of Paraíba, permit CEUA Nº 2161060621.
Necropsy and Histology
Due to the small size of the fish (approximately 5 cm in length), the whole animal was fixed in 10% buffered formalin. To better preserve the neoplasm and internal organs for histologic examination, serial cross-sections of the affected carcasses, at 0.5-cm intervals, were performed. The whole body and cross-sections were photographically documented. Tissues were processed, sectioned at 4 µm and stained with hematoxylin and eosin. Glass slides were evaluated under an optical light microscope, with and without polarized light.
Immunohistochemistry
Among the cases diagnosed as chromatophoromas through histology, 6 cases (cases 117, 161, 162, 163, 164, and 165) were selected for immunohistochemistry. The cases were selected based on the presence of characteristic features of iridophoromas or mixed chromatophoromas. For each case, immunohistochemistry was performed using the following immunomarkers: PNL-2 (1:100 dilution; LSBio, Seattle, WA), melan A (1:50 dilution; Biocare Medical, Pacheco, CA), and S-100 (1:100 dilution; Novocastra Laboratories, Newcastle upon Tyne, UK). Antigen retrieval was performed by heat-induced epitope retrieval and application of the respective antibodies in the aforementioned dilutions. A biotin-free, polymeric alkaline phosphatase-linked antibody detection system with fast red chromogen was used, and sections were counterstained in Fisher’s hematoxylin (Thermo Fisher Scientific, Waltham, MA) followed by Fisher’s staining reagent (Thermo Fisher Scientific), air-dried, and coverslipped. Sections from a dog melanoma were used as positive controls and normal skin from a Siamese fighting fish was used as a negative control.
Transmission Electron Microscopy
For ultrastructure, neoplastic tissue from 4 individuals were fixed in 3% glutaraldehyde in 0.1 M sodium cacodylate buffer. Two neoplasms compatible with iridophoromas and 2 mixed pigment-containing/light-reflecting chromatophoromas were subjected to electron microscopy. Tissue sections were postfixed with 1% osmium tetroxide in 0.1 M sodium cacodylate buffer. Samples were processed as described elsewhere. 1 Briefly, samples were dehydrated in a 25% to 100% ethyl alcohol gradient series for 2 hours using an automated tissue processor (Leica Microsystems). Tissue was then infiltrated with EMbed 812 resin, embedded, and incubated at 58°C for 24 hours for polymerization (all reagents were from Electron Microscopy Sciences). To select areas of interest, samples were trimmed, sectioned at 1 µm, and stained with toluidine blue. From selected areas, thin sections (70–90 nm) were obtained and contrasted with 5% uranyl acetate and Satos’ lead citrate. To improve contrasting, a second grid was only contrasted with 5% uranyl acetate. All samples were visualized using a JEOL 1400Plus transmission electron microscope (JEOL LTD). Images were obtained and analyzed using an OneView Camera Model 1095 with the Gatan Microscopy Suite 3.0 (Gatan Inc).
Results
Animals
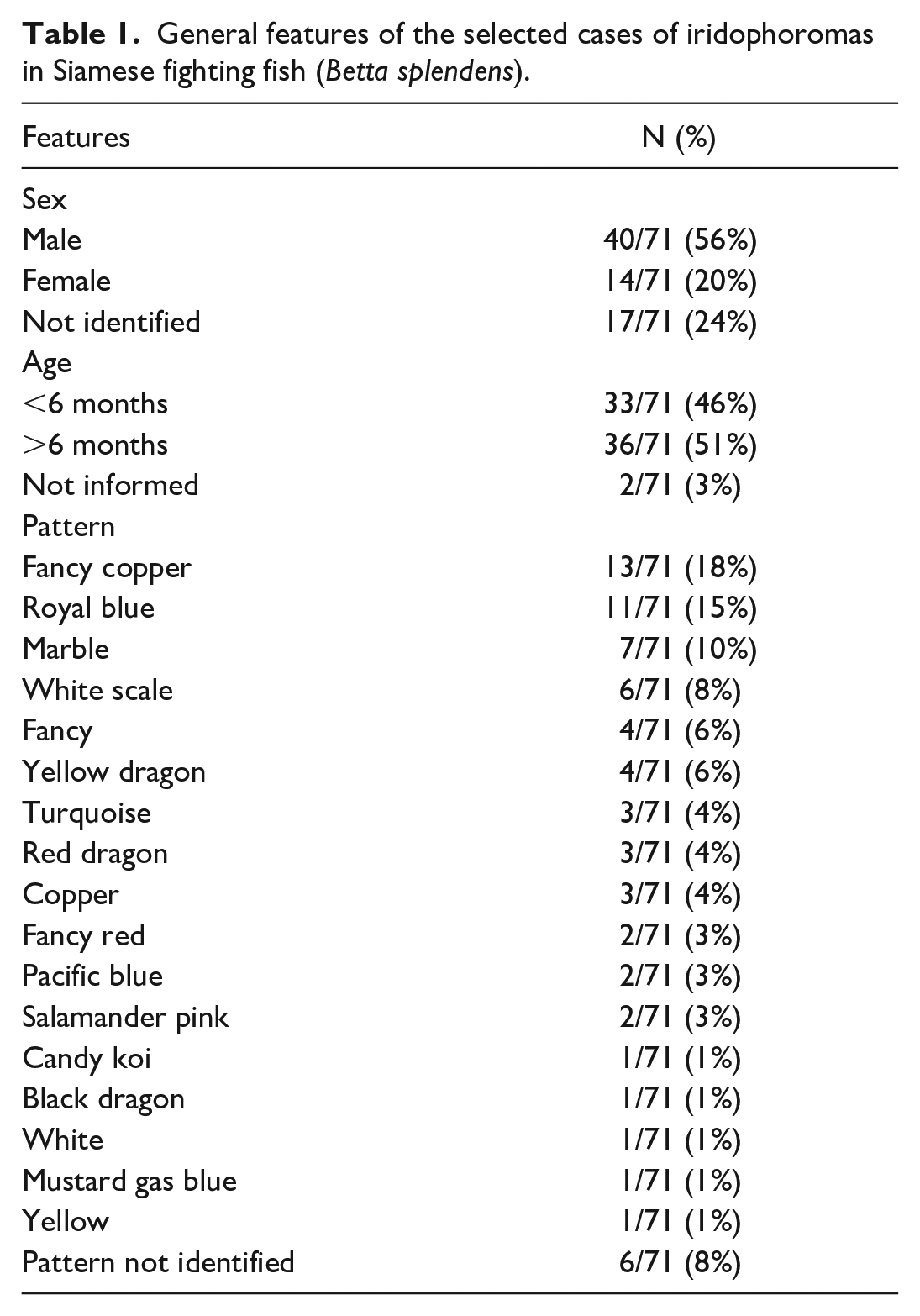
The general characteristics of the 71 cases are presented in Table 1. The fish ranged in weight from 0.8 to 4.6 g and in length from 3.67 to 8.92 cm. Of the 71 fish included in this study, 40 (56%) were male, 14 (20%) were female, and the gender could not be identified in 17 (24%) cases. Fish aged 6 months or older accounted for 51% of the cases (36/71), whereas those younger than 6 months represented 46% of the cases (33/71). The age was not informed in 2 cases. The most frequently observed color pattern in this study was fancy copper (18%), followed by royal blue (15%), marble (10%), and white scale (8%). Other patterns were present in 1 to 4 cases each (1%–6%).
General features of the selected cases of iridophoromas in Siamese fighting fish (Betta splendens).
Necropsy and Histopathology
All animals (71/71) exhibited skin nodules or plaques resembling chromatophoromas grossly. Most tumors were located in the trunk/tail region (29/71, 41%). In 31% of the cases (22/71), the masses had a multifocal distribution, affecting multiple areas of the body simultaneously. Tumors located exclusively in the fins or head of the fish were observed in 14% of the cases each (Fig. 1). The masses ranged from 0.05 to 9.86 mm in diameter, while the size of the plaques ranged from 2.72 to 8.3 mm in length. The majority of the masses were completely white or gray, nodular, expansile, and sometimes pedunculated, with an irregular, soft, and friable surface (Fig. 2). In 12% of the cases, blackened areas were interspersed with the neoplasm. The plaques extended across multiple scales and occasionally elevated slightly above the skin surface. The neoplasms typically displayed a multifocal or multifocal to coalescing arrangements. The number of masses/plaques per fish varied greatly, with most fish having 2 masses or plaques (36/71, 51%), followed by 3 or more masses/plaques (22/71, 31%). Only 18% of the cases (13/71) presented the tumor as a single cutaneous mass/plaque.
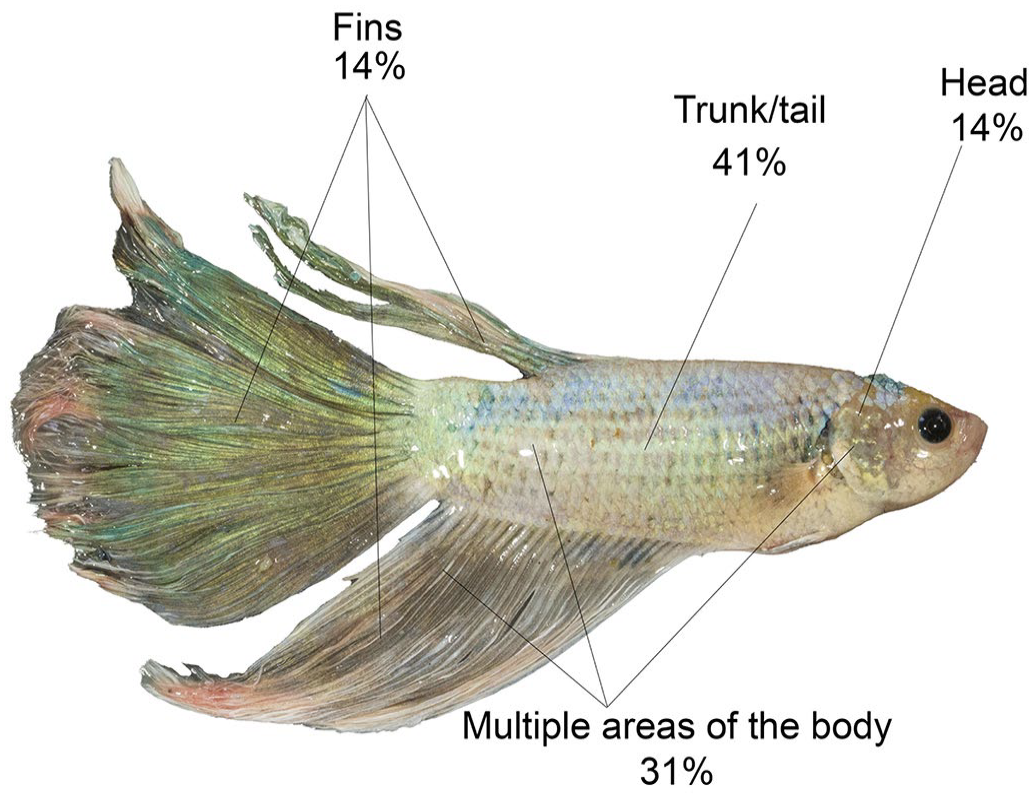
Distribution of iridophoromas in 71 Siamese fighting fish (Betta splendens).

Gross appearance of iridophoromas and mixed chromatophoromas in Siamese fighting fish (Betta splendens). (a) Iridophoroma. Whitish mass interspersed with small iridescent areas in the head. (b) Iridophoroma. Irregular silver mass in the trunk. (c) Iridophoroma. Multifocal to coalescing white masses in the caudal fin. (d) Mixed chromatophoroma. Round black mass interspersed by small whitish areas in the head. (e) Iridophoroma. A focal white plaque interspersed with bluish areas in the trunk. (f) Iridophoroma. A small single white mass in the dorsal fin. (g) Mixed chromatophoroma. A blackened mass interspersed by greenish and white areas invades and expands the left eye. (h) Iridophoroma. Multifocal white masses in the tail. (i) Iridophoroma. Multifocal to coalescing white masses in the tail and caudal fin. (j) Iridophoroma. Multilolubar white mass in the head. (k) Iridophoroma. Irregular friable white mass in the dorsal surface of the trunk. (l) Iridophoroma. Multifocal white masses in the dorsal and caudal fins.
Microscopically, iridophoromas appeared as poorly delimited, nonencapsulated, expansive and high cellular neoplasms (Fig. 3). Ninety-one percent of the tumors (65/71) were composed only of light-reflecting cells, hence classified as iridophoromas, whereas 8% (6/71) of the neoplasms were composed of light-reflecting and melanin-containing cells therefore classified as mixed chromatophoromas. Tumors were composed of poorly defined polygonal cells, separated by a discrete collagenous stroma and arranged in nests, strings, or in a solid pattern. In most cases, the neoplastic iridocytes had indistinct cell contours and inconspicuous nuclei, containing a moderate to marked amount of intracytoplasmic olive-green crystals (Fig. 4a) that were birefringent under polarized light. In cases of mixed chromatophoromas, in addition to neoplastic iridocytes, there were randomly distributed, multifocal areas of well-differentiated cells containing nonbirefringent brown to black intracytoplasmic pigment (melanin, Fig. 4b). Of the 71 neoplasms (Supplemental Table S1), 45 (63%) were classified as malignant and besides the integument, they infiltrated at least the adjacent skeletal musculature. Of these, 42 cases (93%) were iridophoromas and 3 cases (7%) were mixed chromatophoromas. Notably, metastases (Fig. 5) were observed in 14 cases (20%). In 5 cases (7%), iridophoromas affected the spinal cord and the spinal canal. Metastases were also found in the kidneys (4 cases), reproductive tract (2 cases), eyes (2 cases), and gastrointestinal tract (1 case). In 26 cases (37%), neoplasms were restricted to the skin.
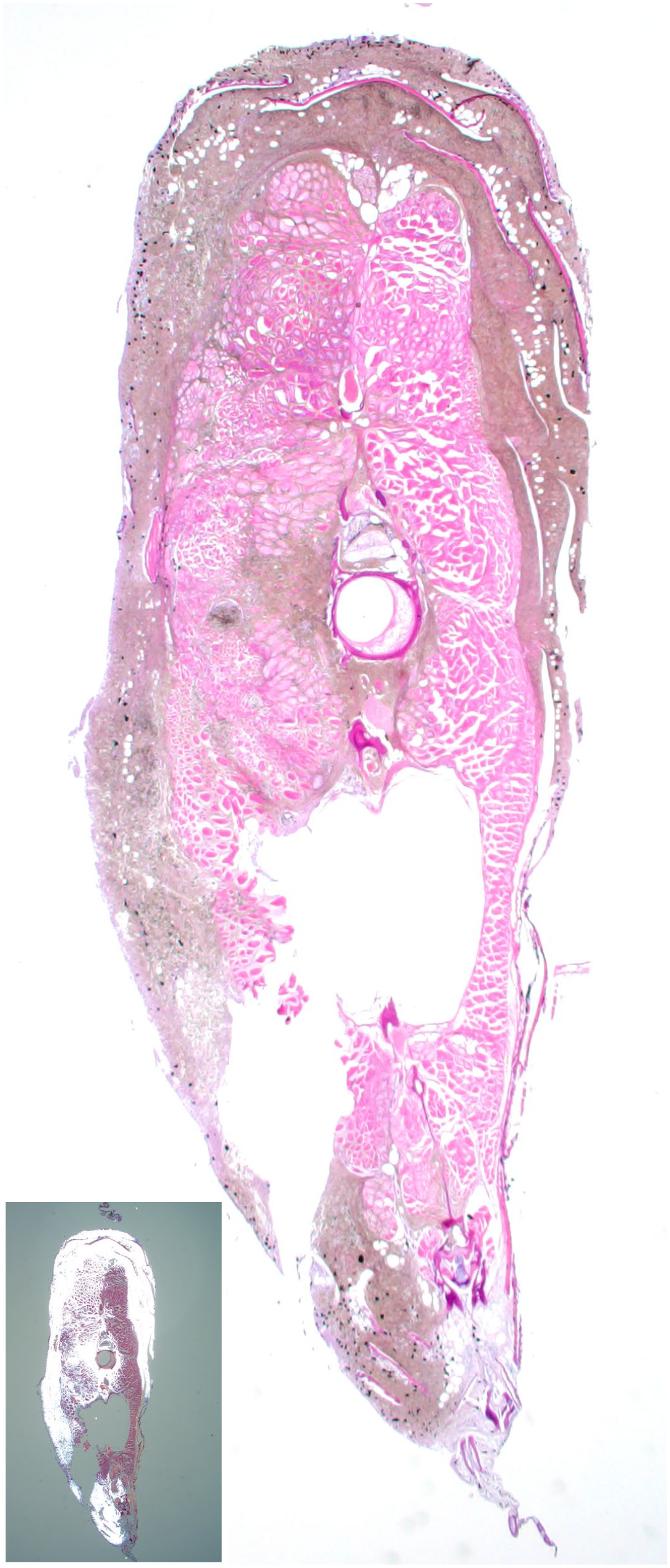
Malignant iridophoroma, skin and skeletal muscle, transverse section through the trunk of a Siamese fighting fish (Betta splendens). Olive-green neoplastic cells form a poorly demarcated mass that extends into the underlying skeletal muscle and spinal canal. The neoplasm is birefringent under polarized light (inset). Hematoxylin and eosin.
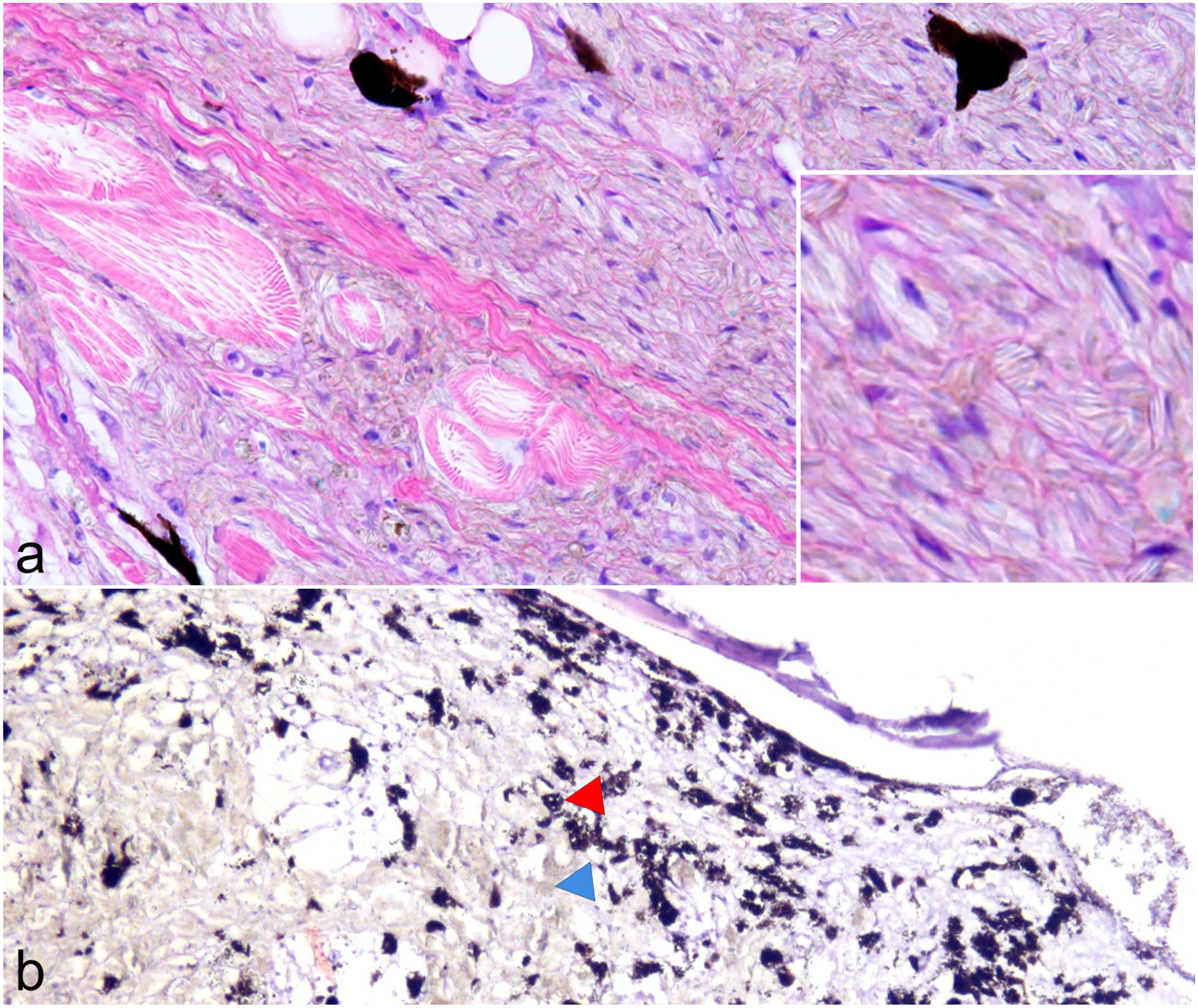
Cytologic features of neoplastic iridophores and melanophores. Hematoxylin and eosin. (a) Iridophoroma, skin and skeletal muscle. Neoplastic iridocytes have indistinct cell contours and inconspicuous nuclei, containing a moderate to marked amount of intracytoplasmic olive-green crystals (inset). (b) Mixed chromatophoroma, skin. The neoplasm is composed of olive-green neoplastic cells (iridophores, blue arrowhead) interspersed by well-differentiated cells containing nonbirefringent brown to black intracytoplasmic pigment (melanin, red arrowhead).
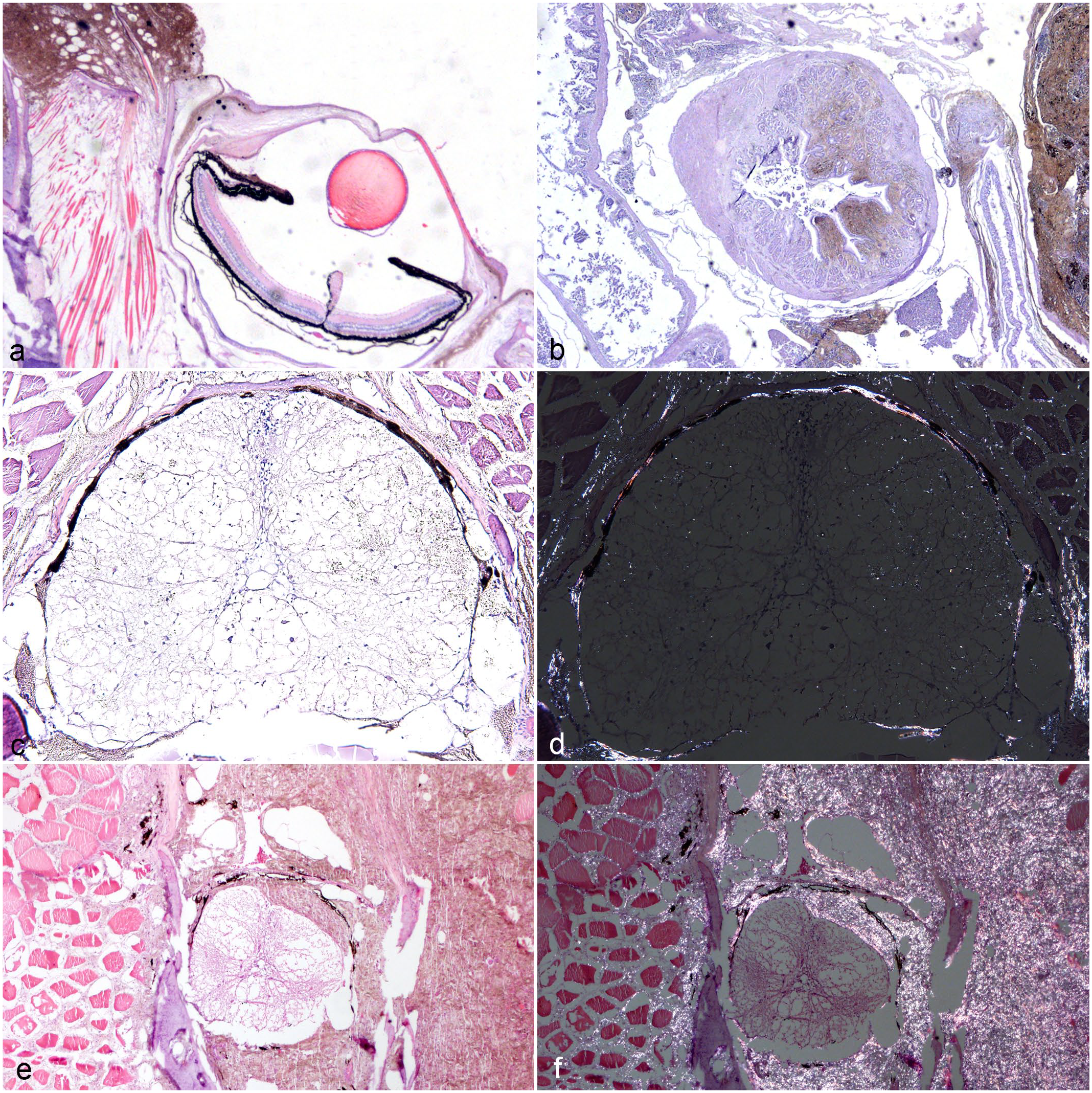
Histopathologic features and sites of invasion of malignant iridophoromas and mixed chromatophoromas in Siamese fighting fishes (Betta splendens). Hematoxylin and eosin. (a) Malignant iridophoroma. A poorly demarcated tumor extends into the adjacent skeletal muscle, ocular conjunctiva, and sclera. (b) Malignant iridophoroma. Gastrointestinal tract infiltrated by olive-green neoplastic cells. (c) Malignant iridophoroma. Neoplastic cells surround and infiltrate the spinal cord. (d) Malignant iridophoroma. Spinal cord. Neoplastic cells are birefringent under polarized light. (e) Malignant mixed chromatophoroma. The tumor consists of olive-green neoplastic cells with discrete multifocal areas of brown to black cells and extends into the adjacent skeletal muscle, infiltrating and compressing the spinal cord. (f) Malignant mixed chromatophoroma. Spinal cord and adjacent skeletal muscle under polarized light. Note that only the olive-green cells exhibit birefringence when viewed under polarized light.
Immunohistochemistry
None of the cases showed immunoreactivity for melan A, S-100, and PNL-2.
Electron Microscopy
Ultrastructurally, iridocytes identified in unaffected regions of the skin contained cytoplasmic stacks of light-reflecting platelets with up to 8 platelets. These platelets measured from 1.14 to 2.37 µm in length. In tumors, cells that were birefringent under polarized light were identified as neoplastic iridocytes. Neoplastic iridocytes were polygonal, elongate to stellated with cytoplasm:nucleus ratio up to 10:1. The nucleus was indented to lobulated and contained granular and marginated heterochromatin. The nucleolus was compact with nucleus: nucleolar ratio of up to 10:1. Neoplastic iridocytes had small intercellular attachments and cytoplasm with a low number of small organelles, including rounded mitochondria, endoplasmic reticulum, and vesicles. Stacks of light-reflecting platelets were present in the cytoplasm of neoplastic iridocytes (Figs. 6, 7). In some tumors, the cytoplasm was abundant and contained stacks of small light-reflecting platelets, whereas in others, the cytoplasm was occupied by large numbers of randomly oriented stacks composed of up to 20 parallel-arranged light-reflecting platelets (Fig. 6a, b). Individual platelets within the stack were approximately 48.4 to 56.3 nm thick, 5.66 to 8.12 µm long (Fig. 6b) and were lined by a double membrane with a discrete intermembrane space (Fig. 6c). Guanine-base crystals contained in the reflecting platelet were not contrasted on lead citrate-uranyl acetate preparation, whereas purine-crystals were electron-dense on uranyl acetate contrasted preparation (Fig. 6c, d). In mixed chromatophoromas, there were melanocytes interspersed with the neoplastic iridocytes (Fig. 7a, b). These melanocytes had a diameter of 0.65 to 0.75 µm and membrane-bound vesicles containing an electron-dense fine fibrillary substance characteristic of mature melanosomes (Fig. 7c). Melanocytes were well-differentiated and had well-demarcated stellated to spindle-shaped cytoplasm.

Malignant iridophoroma, skin, Siamese fighting fish (Betta splendens). Transmission electron microscopy. (a) Neoplastic iridocytes contain intracytoplasmic haphazardly oriented stacks of light-reflecting platelets; N nucleus. Bar = 5 µm. (b) Magnification of the white box in (a). Stacks of light-reflecting platelets are composed of parallel-arranged platelets oriented in a random fashion (double-headed arrows). The cytosol is scare (black arrows) and contains few mitochondria (M). Bar = 1 µm. Uranyl acetate contrast. (c) Individual platelets within the stack lined by a bilipid membrane (arrows). The platelet crystals are not stained. Bar = 0.2 µm. Lead citrate-uranyl acetate contrast. (d) Platelets are electron-dense after guanine-base crystals are contrasted with uranyl acetate. Bar = 0.2 µm.
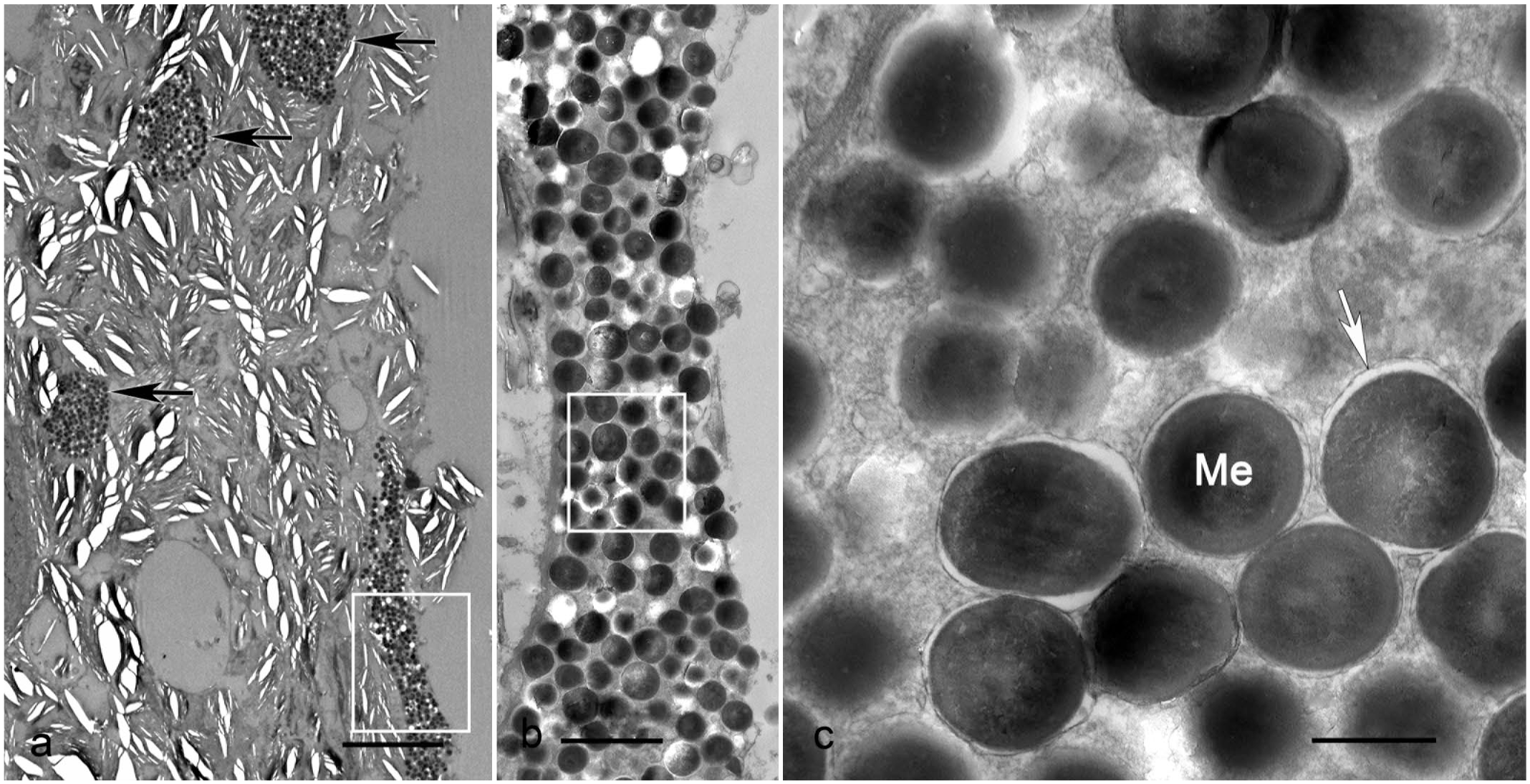
Mixed chromatophoroma, skin, Siamese fighting fish (Betta splendens). Transmission electron microscopy. (a) Neoplastic iridocytes are interspersed by aggregates of spindle-shaped melanosome-containing cells (melanocytes, black arrow). Bar = 10 µm. Clear elliptical spaces are an artifact due to splitting of the neoplastic iridocytes light-reflecting platelets. (b) Magnification of the white box in (a). Cytoplasm of a melanocyte carrying numerous melanosomes. Bar = 2 µm. (c) Magnification of the white box in (b). Numerous membrane-bound organelles containing a fine fibrillary electron-dense matrix (arrow), characteristic of melanosomes (Me). Bar = 2 µm. Lead citrate-uranyl acetate contrast.
Discussion
Chromatophoromas in Siamese fighting fish have been documented as case reports,29,31,32,36 and to the best of the authors’ knowledge, this is the largest study (71 cases) to describe the gross, histologic, and ultrastructural features of iridophoromas in Siamese fighting fish (Betta splendens). Approximately 52% of suspected neoplastic cases in Siamese fighting fish evaluated in this study were diagnosed as chromatophoromas. These results corroborate with one study that evaluated the occurrence of chromatophoromas in free-ranging Hawaiian butterflyfish (Chaetodon spp.) and found an occurrence of 50%. 27 In snakes, the occurrence of chromatophoromas corresponded to only 1% of all histopathology submissions in a private laboratory. 25 In mammalian species, the reports of melanocytic tumors range from 3% to 9% (dogs and cats)41,45 to 15% (horses). 41
In the present study, it is conceivable that the actual number of chromatophoromas exceeds 71, as several other neoplasms observed in Siamese fighting fish specimens received by our laboratory exhibited gross characteristics resembling the confirmed cases of chromatophoromas. However, histopathological confirmation could not be obtained in 64 cases, primarily due to challenges encountered during tissue handling, fixation, and processing. These tumors, being frequently small and highly friable, tended to detach and disintegrate easily when manipulated.
The reason for this high occurrence of chromatophoromas in Siamese fighting fish is unclear; however, since all cases evaluated in this study originated from a single fish farm and were more frequently observed in certain phenotypic patterns (fancy copper and royal blue), a genetic component might play a crucial role in the development of these neoplasms. This is supported by genetic studies with the occurrence of melanomas in Xiphophorus spp., where genetic hybrids between pigmented platyfish (Xiphophorus maculatus) and nonpigmented swordtails (Xiphophorus helleri) spontaneously developed melanomas,18,30 suggesting the involvement of genes related to pigmentation patterns, such as the spotted dorsal (Sp) and spotted side (Sd) genes, in tumorigenesis.
Interestingly, the oncogenes involved in tumorigenesis in Xiphophorus spp. are sex-linked18,30 and the occurrence of pigmented tumors in Xiphophorus spp. hybrids seems to be higher in males.22,37 These results are in agreement with our study, where 56% of the fish that developed chromatophoromas were male, whereas only 20% were female. It is worth noting that sex identification was not possible in 24% of the cases, which might further contribute to the observed difference in tumor incidence between male and female fish.
Other proposed etiologies for tumors of pigment-containing or light-reflecting cells in fish include carcinogenic factors3,9,15,27,48 and oncogenic viruses.27,46 Papillomaviruses have been linked to the occurrence of cutaneous nodules in fish. 42 Recently, the complete genome sequence of a novel papillomavirus recovered from a Siamese fighting fish has been published, 4 although its significance for the development of cutaneous tumors in this species remains unclear. In addition, UV light exposure has been implicated as a significant predisposing factor for the development of certain tumors in both wild and laboratory fish, including cromatophoromas.26,43 However, we were not able to investigate or rule out the influence of these factors in our study.
Although widely used for confirmation of poorly or nonpigmented melanocytic tumors in mammals,34,35 immunohistochemistry was not useful in our study since all antibodies (PNL-2, S-100, and melan A) used had no observable reactivity with the fish tissues. These findings are in accordance with another study on iridophoromas in Siamese fighting fish, 29 which suggests that these negative results are possibly due to species-specific reasons. Interestingly, in one study on chromatophoromas in cyprinid species (Cyprinus carpio and Carassius auratus) 39 and in a second study that described a chromatophoroma in a crevice kelpfish (Gibbonsia montereyensis), 5 immunohistochemistry yielded positive results, particularly with the anti-melan A antibody. It is important to note that, although the investigators could not determine which specific type of chromatophoroma occurred in the cited cases, the tumors did not contain the typical birefrigent crystals observed in iridophoromas. In our cases, these crystals were present and may have interfered with our immunohistochemical results. In reptiles, immunohistochemical evaluation of chromatophoromas has yielded variable results, and antibodies widely used in mammalian melanomas do not have the same sensitivity and specificity for reptilian chromatophoromas. However, immunohistochemistry may be especially useful for diagnosing nonpigmented chromatophoromas in some reptile species. 23
In the present study, electron microscopy was essential to confirm that the fish presented 2 types of chromatophoromas: iridophoromas and mixed chromatophoromas. While iridophoromas were composed of only cells that contained light-reflecting intracytoplasmic platelets, mixed chromatophoromas were comprised by the neoplastic iridocytes, in addition to spindled to stellated melanosome-containing cells, consistent with melanocytes. These findings agree with the few published studies involving ultrastructural analysis of chromatophoromas in fish.27,36 Ultrastructural analysis of the iridophoromas in our study also revealed differences between normal and neoplastic iridocytes. While no difference regarding the orientation of intracytoplasmic light-reflecting platelets was observed (platelets were randomly oriented in both normal and neoplastic cells), differences in the size of platelet stacks and the length of platelets were noted. In normal cells, platelet stacks measured up to 8, whereas in neoplastic cells, they measured up to 20. In addition, the length of the platelets ranged from 1.14 to 2.37 µm in normal cells, compared with 5.66 to 8.12 µm in neoplastic cells. It is worth noting a discrepancy in the length of crystal platelets compared with the only prior study in fish that quantified this parameter using electron microscopy. In that study, platelets measured between 0.5 and 1.5 µm in iridophoromas found in Chaetodon multicinctus and Chaetodon miliaris. 27
One interesting feature of the neoplastic behavior of the iridophoromas in our study is that more than 60% of the tumors were not restricted to the skin and invaded adjacent tissues, especially the skeletal musculature. These tumors were classified as malignant iridophoromas/mixed chromatophoromas, and these findings are in accordance with what has been described in a case report of a malignant iridophoroma in a Siamese fighting fish. 32 It is important to note that, although malignant mixed chromatophoromas corresponded to only 3 cases, out of the 71 neoplasms that met the inclusion criteria, we only identified 6 cases of mixed chromatophoromas. Therefore, it was not possible to make assumptions about the correlation between malignancy and pigmentation. In addition, metastasis to the spinal cord was identified in 5 cases, similar to what has been described in another study where a Siamese fighting fish presented an iridophoroma invading the spinal canal and entrapping the spinal cord. 29 In one study on reptiles, the spinal facets of a rat snake (Elaphe sp.), but not the spinal cord, were invaded by a melanophoroma. 23
In 37% of the iridophoromas in this study, the cellular structure and overall arrangement of these neoplasms were similar to what was observed in the malignant counterpart; however, these tumors were restricted to the skin and did not invade adjacent tissues or metastasize to other organs. We were not able to classify these iridophoromas as malignant or benign, and it is essential to acknowledge the possibility that these tumors could have a malignant behavior, but we may have sampled them at a stage where they had not progressed enough to reach other tissues. Therefore, further research is needed to elucidate the nature of these tumors. A comprehensive ultrastructural analysis might be particularly helpful in highlighting distinct features in malignant and benign tumors that were not observable in light microscopy, providing a more detailed understanding of their characteristics and behavior. Mitotic figures were not observed in the current study, similar to what has been described in other cases of iridophoromas.20,29 In one study of cutaneous chromatophoromas in captive snakes, mitotic count was not considered a reliable predictor of metastases for these neoplasms. 25
Despite being widely used to describe pigment-containing or light-reflecting cells in fish, reptiles and amphibians, the terms chromatophores, melanophores, and iridophores have inconsistent interpretations depending on the species that these structures were identified. In some cases, the same terminology is used for different structures. For instance, the term chromatophore has been used on a subcellular level to designate photosynthetic organelles of amoebic organisms, 38 on a cellular level to characterize individual pigment-bearing or light-reflecting cells of vertebrates, 24 as well as on a tissue level to name complex color-producing cutaneous organs of cephalopods. 21 In the latter, chromatophores are defined as complex structures composed not only of a pigment-containing cell referred to as a chromatocyte, but also nerves, glial cells, radial muscles, and sheath cells. 21 Comprehensive descriptive studies of these cells in vertebrates are needed to determine whether the current terminology is adequate or should be revised.
In summary, presumptive diagnosis of iridophoromas should be performed when whitish, gray or black friable cutaneous masses or plaques are present in the fin, trunk/tail, or head of Siamese fighting fish. Histology should be sufficient for the diagnosis of these neoplasms, but electron microscopy could be useful to differentiate between iridophoromas and mixed chromatophoromas. Immunohistochemistry was not useful for the diagnosis of chromatophoromas in Siamese fighting fish in the present study. Comprehensive genetic and molecular studies are in need to elucidate the etiology of these neoplasms in Siamese fighting fish, but their occurrence in several individuals from the same breeding facility and its higher occurrence in fancy copper and royal blue color patterns and in young male suggests that a genetic/hereditary factor may play a crucial role.
Supplemental Material
sj-xlsx-1-vet-10.1177_03009858241281887 – Supplemental material for Gross, histologic, and ultrastructural features of iridophoromas in Siamese fighting fish (Betta splendens)
Supplemental material, sj-xlsx-1-vet-10.1177_03009858241281887 for Gross, histologic, and ultrastructural features of iridophoromas in Siamese fighting fish (Betta splendens) by Samuel L. P. Diniz, Hodias S. Oliveira Filho, Kin M. O. L. Santos, José L. C. Duarte, Rafael L. Oliveira, Felipe Pierezan, Aníbal G. Armién and Jeann Leal de Araújo in Veterinary Pathology
Footnotes
Acknowledgements
We thank the histology technicians at the Federal University of Paraiba and UC Davis for extensively working on H&E slides and Christina Heart at UC Davis for the outstanding TEM and special contrast preparation. We also thank the National Council for Scientific and Technological Development (CNPq), the Coordination for the improvement of Higher Education (CAPES), and the Paraiba Research Foundation (FAPESQ), for the financial support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
