Abstract
Pigment cell tumors, also known as chromatophoromas, are cutaneous spindle cell neoplasms originating from pigment cells (chromatophores) in the dermis of teleosts, amphibians, and reptiles. Chromatophoromas share similar histologic morphology to other spindle cell tumors and are not always pigmented. Therefore, immunohistochemical analysis may be useful in distinguishing these neoplasms from tumors of other cellular origin when poorly pigmented. We performed 3 immunohistochemistry assays (PNL-2, melan A, and SOX10) on 8 cutaneous neoplasms from 8 teleosts diagnosed as chromatophoromas based on histologic morphology. Semiquantitative analysis of immunoreactivity was evaluated on each immunohistochemical assay using a 0–3 scale. PNL-2 exhibited mild-to-moderate (1 or 2) immunoreactivity in 7 of the cases, and resident chromatophores (internal control) were also immunoreactive in these cases. Melan A exhibited mild-to-moderate (1 or 2) immunoreactivity in 4 cases (and with resident chromatophores in these cases); SOX10 was not immunoreactive in any cases. Our results indicate that PNL-2 may be a useful marker in teleosts to distinguish tumors of chromatophore origin. Melan A could also be useful, but appears to be less sensitive, and SOX10 is likely not a useful marker for these neoplasms in teleosts.
Chromatophoromas are common cutaneous neoplasms in many species of fish. 2 These tumors are locally invasive and commonly become ulcerated. Secondary infections often occur, contributing to ulceration and leading to large necrotic wounds that are difficult to treat. Tumor invasion alone, or in combination with secondary infection, can lead to death or euthanasia.2,10
Chromatophores are the cells of origin in these tumors and are of neural crest origin.1,4 The 4 major types of chromatophores are identified by the pigment type they produce, and therefore their color. Melanophores produce melanin, and are dark brown to black; erythrophores produce both carotenoids and pteridines, and are red; xanthophores produce carotenoids, and are yellow; and iridophores produce purines, and are colorless and refractile.1,3 Melanomas are the most commonly diagnosed chromatophoromas in fish.2,10
Etiology of the neoplastic transformation of these cells is unknown; however, genetic, age-related, and environmental components are indicated, and these tumors have been induced in laboratory fish exposed to chemical agents.2,4,7 In neoplastic transformation, chromatophores can lose their pigmentation, making some chromatophoromas difficult to definitively diagnose based on routine (hematoxylin and eosin) histologic examination. Moreover, common responses to tumor invasion, such as fibroplasia and granulation tissue, can have a similar histologic appearance, making tumor margins difficult to distinguish. Immunohistochemistry (IHC) is a powerful tool when trying to identify poorly pigmented variants of these neoplasms in other species. PNL-2, melan A, and SOX10 are commonly used melanocytic immunohistochemical markers in humans and other species, and have been used to identify chromatophoromas in amphibians and reptiles. 5 Melan A is a cytoplasmic protein recognized by cytotoxic T cells6,9; it is a specific melanocytic differentiation antigen and is highly sensitive for melanomas in humans and other domestic species (75–92% sensitivity in human melanomas). 9 PNL-2 is a monoclonal antibody with high specificity to a fixative-resistant melanocytic antigen, and the sensitivity is considered to be comparable to melan A in detecting human melanomas.6,9 A 2016 study in snakes also showed that PNL-2 was a useful marker in diagnosing chromatophoromas of various chromatophore origins. 5 In other mammalian species, melan A and PNL-2 are comparable, but melan A is considered to be more sensitive.8,9,12 The role of PNL-2 and melan A in melanogenesis is not fully known. 9 SOX10 is a nuclear transcription factor in the differentiation of neural crest progenitor cells and melanocytes. 13 Because SOX10 reacts to other neural crest cells, it is considered less specific for melanomas, but could prove useful when melan A and PNL-2 are not reactive.
We compared the histologic morphology of a series of cutaneous neoplasms considered to be of chromatophore origin, and identified a useful immunohistochemical assay to diagnose chromatophoromas in teleosts, particularly when the chromatophoromas are poorly pigmented. Although prognosis of chromatophoromas versus prognosis of other spindle cell neoplasms is not fully described in fish, melanocytic neoplasms in some mammalian species have a much poorer prognosis than that of other neoplasms with similar histologic morphology. Therefore, determining neoplasms of chromatophore origin may prove useful in prognostic determination in the future.
Eight teleosts of 2 cyprinid species, common carp (Cyprinus carpio; n = 5) and goldfish (Carassius auratus; n = 3), from different facilities were presented to the University of California–Davis Veterinary Medical Teaching Hospital in 2012–2018 (Table 1). Fish were 9–20 y old. Three of the cases were euthanized fish, and neoplasms were diagnosed on postmortem examination. The other 5 cases were submitted as biopsies. All of the neoplastic lesions were located along the dorsum or dorsal skull and/or periocular region of the fish (Fig. 1), and the lesions were moderately to severely ulcerated, invasive, and variably pigmented to non-pigmented. All fish had similar masses diagnosed as pigment cell tumors or chromatophoromas, based on characteristic histologic features.
Clinical details of the 8 cases of chromatophoroma in cyprinids.

NA = not available.
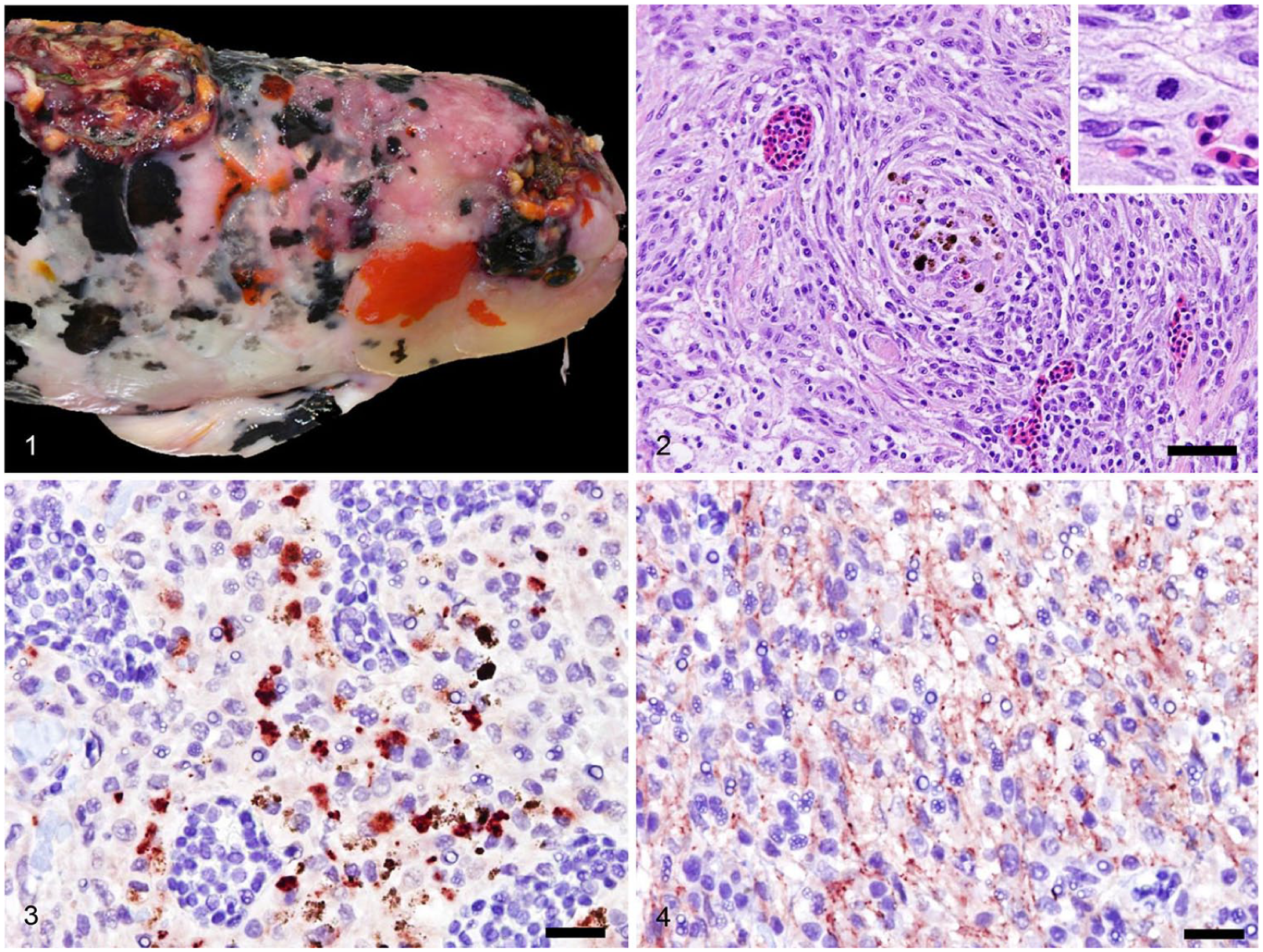
Chromatophoromas in a common carp (Cyprinus carpio, case 3).
Hematoxylin and eosin (H&E) as well as 3 immunohistochemistry assays, PNL-2 (1:100 dilution; LSBio, Seattle, WA), melan A (1:50 dilution; Biocare Medical, Pacheco, CA), and SOX10 (1:100 dilution; Abcam, Burlingame, CA), respectively, were performed on formalin-fixed, paraffin-embedded samples of each mass. Briefly, all immunohistochemical assays followed the same protocol, which included antigen retrieval using heat-induced epitope retrieval (HIER) and application of the respective antibodies at the aforementioned dilutions. The sections were counterstained in Fisher hematoxylin 1 (Thermo Fisher Scientific, Waltham, MA) followed by Fisher bluing reagent (Thermo Fisher Scientific), air dried, and coverslipped. Positive and negative controls for each antibody were processed in parallel. The histologic appearance of each neoplasm on H&E was described, and the level of immunoreactivity was analyzed semiquantitatively for each immunohistochemical assay using a grading scale of 0–3, with 0 = non-immunoreactive, 1 = mildly immunoreactive, 2 = moderately immunoreactive, and 3 = strongly immunoreactive (Table 2). Immunoreactivity was expected to be cytoplasmic for PNL-2 and melan A, and nuclear for SOX10, based on previous studies in various species.
Semiquantitative evaluation of immunoreactivity for each immunohistochemical assay in each case of chromatophoromas using a scale of 0–3 (0 = non-immunoreactive, 1 = mildly immunoreactive, 2 = moderately immunoreactive, 3 = strongly immunoreactive).

Histologic examination revealed neoplastic cells to be arranged in short interlacing streams and fascicles as well as whorls with a neural appearance (Fig. 2). Neoplastic cells were predominantly round-to-fusiform with indistinct cell borders and a moderate amount of eosinophilic-to-amphophilic cytoplasm. Dark-brown cytoplasmic pigment granules were within groups of neoplastic cells, varying from moderate amounts to absent. No cytoplasmic refractive crystals were identified (as seen in iridophoromas). Nuclei were round-to-ovoid with finely stippled chromatin, and anisocytosis and anisokaryosis were mild-to-moderate. Mitotic activity was variable and ranged from 0 mitotic figures per 400× field to 6 mitotic figures per ten 400× fields.
Seven of the 8 cases had mild-to-moderate cytoplasmic immunoreactivity with PNL-2 (Fig. 3), including most of the non-pigmented cells; normal chromatophores within the skin also exhibited immunoreactivity. Four of 8 cases had mild-to-moderate cytoplasmic immunoreactivity with melan A (Fig. 4), including most non-pigmented cells; normal chromatophores within the skin also had immunoreactivity in these cases. No neoplasms were immunoreactive with SOX10, and neither were the resident chromatophores in the tissue.
A majority of the chromatophoromas contained cells with few or no cytoplasmic pigment granules, making these regions difficult to distinguish from other dermal spindle cell tumors. Chromatophoromas sometimes produce no pigment, and histologic distinction from other spindle cell tumors, such as the perivascular wall tumors formed in Hikui disease, 11 can be challenging. IHC could be used in these poorly pigmented and non-pigmented tumors; however, IHC analysis of neoplasms in fish, and specifically neoplasms of chromatophore origin, are not well-documented. Overall, the information gathered from our series of cases indicates that PNL-2 and melan A are likely useful IHC markers for chromatophoromas in teleost species. Additional testing with larger sample numbers is needed to further support this theory.
All of these dermal chromatophoromas were located on the dorsum and usually close to or on the head, which could indicate that these tumors are solar induced. Some histologic features found in cases of ultraviolet (UV)-induced skin damage in fish were present in the epidermis overlying the tumors as well as in the epidermis and dermis at the margin of the tumors. However, these features (separation of the epidermis from the underlying dermis; extracellular clefting and edema in the superficial stratum compactum; epidermal hyperplasia, epidermal degeneration) are nonspecific for UV damage and can be induced by other forms of insult as well (Bullock AM. The skin response of fish to ultraviolet radiation: a histological study [PhD thesis]. Scotland: University of Stirling, 1984. Available from: http://dspace.stir.ac.uk/bitstream/1893/2557/1/Bullock%20(1984)%20-%20The%20Skin%20Response%20of%20Fish%20to%20Ultraviolet%20Radiation.pdf).
Footnotes
Acknowledgements
We thank Drs. Verena Affolter, Kevin Keel, Chuck Mohr, and Patricia Pesavento of the University of California–Davis, Veterinary Medical Teaching Hospital Anatomic Pathology Department; Dr. Alvin Camus of the College of Veterinary Medicine, University of Georgia; and the University of California–Davis, Veterinary Medical Teaching Hospital histopathology laboratory and for their contribution to our study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
