Abstract
Ovine pulmonary adenocarcinoma (OPA) is a contagious lung cancer of sheep caused by jaagsiekte sheep retrovirus (JSRV). It is generally accepted that transmission by the respiratory route occurs under natural conditions. However recent studies strongly indicate that JSRV can also be transmitted to lambs perinatally via colostrum and milk (C/M). The aim of this work was to confirm that C/M can transmit JSRV infection to lambs under natural conditions and investigate the initial events associated with this transmission route. We have analyzed the presence of JSRV in C/M samples from 22 naturally infected, asymptomatic ewes throughout a lactation period, and in various tissues collected from a group of 36 of their lambs that were fed naturally. The lambs were euthanized at 12, 24, 48, and 72 hours and at 5 and 10 days after birth. We detected JSRV-provirus by PCR in the somatic C/M cells from 10/22 ewes (45.45%). The virus was also detected in 9/36 lambs (25%). JSRV-infected cells, with lymphoreticular-like morphology, were observed by immunohistochemistry (IHC) and in situ hybridization (ISH) in Peyer’s patches (PP) from the small intestine of the youngest lambs and in mesenteric lymph nodes (MLN) from lambs older than 72 hours. The virus was also detected by PCR in white blood cells (WBC) in 2/36 lambs (5.5%). These results confirm colostral transmission of JSRV to lambs under natural conditions. Infected lymphoreticular cells contained in C/M appear to be involved. These cells can cross the intestinal barrier of newborn lambs, reach the MLN and enter into circulation.
Keywords
Ovine pulmonary adenocarcinoma (OPA, Jaagsiekte, sheep pulmonary adenomatosis), is an important infectious disease of sheep caused by jaagsiekte sheep retrovirus (JSRV). 7,18 JSRV induces the transformation of secretory epithelial cells of the distal respiratory tract. The disease is clinically characterized by a chronic respiratory process as a consequence of the development of lung adenocarcinoma. 27 In addition, JSRV infects cells of the lymphoreticular system and JSRV-provirus can be detected by PCR in white blood cells (WBC), lymph nodes, and other lymphoid organs prior to the onset of OPA lesions. 3,5,9,12,24 Epidemiological studies using PCR techniques have shown that in clinically affected flocks JSRV-infected animals may be present in a high proportion (up to 80%). However, only a minority (5–17%) develop tumors, following a long incubation period. 3
OPA can be experimentally transmitted by intratracheal inoculation with lung fluid produced by clinically affected animals, 4,24,26,30 and it is generally accepted that transmission by the respiratory route occurs under natural conditions. 27 However, other transmission routes may contribute to the spread of infection in commercial sheep farms. Thus, in endemically OPA affected flocks it is possible to detect, by PCR, a high proportion (10–43%) of JSRV-positivity in the blood from lambs during the first months of life. 3 This finding indicates that the perinatal period may be relevant for the transmission of JSRV in commercial flocks. This hypothesis is also supported by a study performed with a German flock, containing high numbers of sheep with OPA. This survey showed the dramatic reduction of the disease by creating a new flock with lambs separated at birth from their mothers and artificially fed with colostrum and milk (C/M). 31 This suggests that C/M may be a relevant transmission route for JSRV infection under natural conditions. A further study added more support to these findings, providing evidence that JSRV proviral-DNA could be detected in somatic cells from colostrum in sheep belonging to a flock with a long history of OPA, and that lambs born to mothers testing JSRV-positive in blood cells but with no detectable OPA tumors became blood-infected at a very early age. 10 In addition, these authors have also shown that JSRV provirus could be detected in the blood of lambs artificially fed with C/M obtained from JSRV-infected ewes. 10 This combination of data strongly indicates that JSRV can also be transmitted to lambs perinatally via C/M. Nevertheless, the method by which C/M from infected ewes could transmit the infection to lambs has not been elucidated yet.
The objective of the work we present here was to verify that JSRV can be transmitted to the lambs during natural suckling and investigate the initial events associated with this transmission route. Thus, we have tried to identify JSRV by several techniques, in different tissues collected from a group of very young lambs naturally fed with C/M from ewes, which are JSRV-positive in the blood but have no clinical signs of OPA.
Material and Methods
Experimental Design
A sample of 40 lambs, born to a group of 22 JSRV-infected but asymptomatic ewes (JSRV (+) group) and another sample of 7 lambs from a control group of 6 ewes (JSRV (-) group) were used in this experiment. The infected ewes (aged 4–6 years) were selected from 2 flocks of Rasa Aragonesa local breed and Rasa Aragonesa-Romanov crosses, with a long history of OPA. The sheep with JSRV-positive blood were identified by heminested PCR analysis (U3hn PCR) 17 and animals with no clinical signs of OPA were selected. The JSRV (-) group of ewes (aged 4–6 years) belonged to a flock of Rasa Aragonesa-Romanov crosses, from the same geographical area, with no clinical record or pathological findings suggestive of OPA in the last 10 years. Blood samples were taken at 2 different times and tested by U3hn PCR and all the replicates were found to be negative. Both groups were selected when the ewes were pregnant. They were then transported to the animal facilities at the University of Zaragoza and housed in 2 separate locations. JSRV (+) ewes were retained for 2 lambing periods to obtain enough lambs for the study. To induce a second gestation, ewes were oestrous synchronized by routine methods and presented to the ram. The resulting lambs were born and housed with their mothers under the same conditions. From the JSRV (+) group 36 lambs and 6 from the JSRV (-) group were naturally nursed. They were randomly euthanized, following approved procedures by the Animal Research Committee of the University of Zaragoza, at 12, 24, 48, and 72 hours and at 5 and 10 days after birth, generating groups with the same number of lambs. Furthermore, 4 lambs from the JSRV (+) group and 1 lamb from the JSRV (-) group were deprived of colostrum by covering the udders and nipples of their mothers with a cloth bag for 10 days prior to the expected parturition and were humanely killed shortly after birth. Blood samples were collected from each of the 47 lambs just prior to euthanizing and the WBC were isolated as described by De las Heras et al. 5 After necropsy, duplicate samples from the small intestine (duodenum, jejunum and ileum with Peyer’s patches), mesenteric and mediastinal lymph nodes, lung and tonsil were collected, using individual, sterile instruments for each tissue. One set of samples was fixed in formalin and embedded in paraffin wax for evaluation by immunohistochemistry (IHC) and in situ hybridization (ISH). Another set of samples were snap frozen and stored at −80°C for PCR analysis.
Throughout the first lactation period, samples (50–200 ml) of precolostrum (at the end of the pregnancy), colostrum at 12, 24, and 48 hours, and milk at 5 and 10 days and at 2 and 4 weeks after parturition, were collected under aseptic conditions from ewes belonging to both groups. When the lambs were removed for slaughter, milk production was maintained by daily milking.
Somatic cells (SC) from these milk samples were obtained by several steps of centrifugation and washing with PBS, as described by Extramiana et al. 8 The total number of cells was determined by counting microscopically and aliquots of 5×106 cells were stored at –80°C until PCR processing.
The ewes were euthanized at the end of the experimental period, followed by a careful postmortem examination of the lungs. Samples from various areas and from any gross lesion suggestive of OPA were taken for histopathological and IHC evaluation. Unfortunately 3 infected ewes died during the second lambing (2 ewes at the end of the gestation period and the other one during the delivery), and we could not perform postmortem examination or collect samples from these animals.
PCR Detection of JSRV
PCR analysis for the detection of JSRV proviral DNA was performed with different types of milk secretions collected from the mothers and blood and tissue samples taken from the lambs. Total DNA from SC of precolostrum and colostrum samples was extracted by a modified phenol-chloroform method, 25 and from milk, lamb’s WBC and frozen tissues samples using the QIAamp DNA Mini Kit (QIAGEN GmbH, Hilden, Germany). DNA purity and concentration were estimated by optical density at 260 and 280 nm, and DNA was tested by U3hn PCR. 17 Three replicates, each containing 500 ng of test DNA, were prepared for each sample. Appropriate controls, positive and negative, were used at each step. Amplification products were analyzed by electrophoresis in 2% (w/v) agarose gels with 0.5 mg/ml ethidium bromide and visualized on a bioimaging system of automated gel documentation (Gene Genious, Syngene, Cambridge, UK). Samples were considered positive when amplification was found in at least 1 of the replicates. To confirm DNA quality and evaluate the possible inhibitors of PCR, negative samples were tested by the Glyceraldehyde-3-phosphatase dehydrogenase (GAP-DH) PCR described by Palmarini et al. 17
Immunohistochemistry (IHC)
Sections of tissues from the lambs were evaluated by immunohistochemical procedures previously described by Palmarini et al, 16 using an automated stainer (Autostainer Link 48, Dako, Glostrup, Denmark). Rabbit polyclonal antibodies against 2 different JSRV proteins: matrix (MA) and p10 protein, both encoded by gag gene, and a mouse monoclonal antibody against surface protein (SU), 32 encoded by env gene, were utilized as primary antisera. These antibodies were kindly supplied by Dr Palmarini (anti MA and anti p10) and by Dr Miller (anti SU).
Lung sections collected from the mothers were histopathologically examined and lesions compatible with OPA were evaluated immunohistochemically using the mouse monoclonal antibody against JSRV SU.
In Situ Hybridization (ISH)
Lamb sections of small intestine and mesenteric lymph nodes (MLN), from pieces of tissues previously identified as JSRV-positive by IHC procedures, were used for ISH evaluation.
A single strand DNA 5′biotinylated probe comprising of 176 nucleotides (nts) was chemically synthesized (IDT, Integrated DNA Technologies, Coralville, Iowa, USA) according to nts 7210–7385 in the cDNA JSRV sequence. 33 This fragment is included in the U3 region of the long terminal repeat (LTR) of the virus and is located at the same position that the exogenous specific JSRV sequence that is amplified in the first round of the U3hn PCR. 17
Sections of 4 µm thickness were placed on silanized slides (Dako, Glostrup, Denmark) for ISH. After dewaxing and rehydration, the sections were treated with a target retrieval solution (Dako, Glostrup, Denmark) for 20 min at 98°C and allowed to cool at room temperature (RT) for 20 min. After washing, the sections were digested with pepsin (Dako, Glostrup, Denmark) under acid conditions, according to the manufacturer’s recommendations. Concentration of pepsin and duration of treatment were optimized for each sample of tissue examined. It was determined that for MLN sections optimal pepsin treatment was 0.5 mg/ml at RT with a digestion time ranging between 1 and 3 min. For sections of small intestine the optimal duration of treatment was shorter, ranging from 30 seconds to 1 min. Pepsin treatment for OPA tumor sections, used as positive controls, was 2 mg/ml at RT for 10 min. The digestion was stopped by washing. To avoid nonspecific staining by endogenous peroxidase and/or biotin, tissue derived peroxidase and biotin were blocked prior to hybridization. Slides were immersed in H2O2 0.3% in methanol for 20 min at RT, and then washed before using a streptavidin-biotin blocking system kit (Dako, Glostrup, Denmark), according to the manufacturer’s protocol. The sections were then submitted to hybridization using 2 µg/slide of biotin-labeled probe in hybridization solution (deionized formamide 50%, dextran sulphate 10%, SSC 2X, salmon sperm DNA 400 µg/ml). Hybridization was performed in a Dako hybridizer (Dako, Glostrup, Denmark), where samples were subjected to a heat denaturation (95°C for 5 min) followed by an overnight incubation at 37°C. After washing, a tyramide signal amplification (TSA) system (Dako GenPoint TSA System for Biotinylated Probes, Dako, Glostrup, Denmark) was used, according to the manufacturer’s instructions, to detect and enhance the biotin signal. This system provides all the reagents required to develop the reaction (primary and secondary streptavidin-horse radish peroxidase (HRP) conjugates, biotinyltyramide and diaminobenzidine (DAB) (chromogen). The sections were counterstained with Carazzi’s hematoxylin. Positive controls consisted of sections of OPA tumor. MLN and small intestine tissue sections from colostrum-deprived lambs were used as negative controls. For each sample stained, a parallel procedure was performed using a section in which the biotin-labeled probe was omitted.
Results
Detection of JSRV in Milk Secretions and in Lung Samples From the Ewes
Results of JSRV detection by U3hn PCR in the SC obtained from the different types of milk secretions collected from the group of infected ewes are shown in Supplemental Table 1. Ten out of the 22 ewes in the JSRV (+) group (45%) were JSRV-positive in at least 1 of the milk secretion samples analyzed. When samples from each individual animal were studied, it was noted that the detection of the virus was not consistent throughout the lactation period. None of the animals were positive in more than 2 analyzed samples. Nevertheless, when taking into account the total group of sheep, the virus was detected in all types of milk secretions (precolostrum, colostrum, milk), though in the perinatal period the detection of JSRV appeared to increase and the virus was found in precolostrum or colostrum samples from 7/10 C/M-positive animals (70%). None of the 22 ewes in this study showed clinical signs of OPA throughout the experimental period but, as shown in Supplemental Table 1, necropsy revealed small (1–2 cm diameter) single OPA lesions in 5 of the remaining 19 animals (26%). This finding was confirmed by IHC. Only 2 out of the 5 animals with OPA lesions (40%) were JSRV-positive in some of the milk secretions previously analyzed by PCR.
None of the samples of milk secretions from any of the ewes in the JSRV (-) group, analyzed by PCR, were JSRV-positive and no OPA lesions were identified.
Detection of JSRV in the Tissues From the Lambs
The results of the detection of JSRV, by different techniques, in the tissue samples from the lambs born to the JSRV (+) group of ewes are summarized in Table 1. From all the tissue samples analyzed only 2 lambs, euthanized at 72 hours and 10 days after birth, respectively, gave PCR-positive results, with JSRV detected in the WBC. However, immunohistochemical (IHC) methods yielded a higher number of positive results, with labeled cells observed in the tissues from 8 of the 36 lambs (22%) that suckled infected ewes. The virus was only detected in 2 types of tissue: small intestine in the youngest animals, where staining was observed in Peyer’s patches (PP) but not in epithelial cells of the villi; and in the mesenteric lymph nodes (MLN) from lambs older than 72 hours. PP samples were positive in two 12-hour-old and two 24-hour-old lambs. MLN samples were positive for 4 animals aged between 72 hours and 10 days. One MLN-positive 10-day-old lamb was also found to be blood-positive. JSRV was detected using primary antisera against JSRV matrix (MA) and p10 proteins (one 24-hour-old lamb was PP-positive with both antisera). However no virus was detected in any tissue samples from any of the lambs using antisera against JSRV surface protein (SU). ISH analysis, used to confirm the presence of exogenous virus in the IHC positive samples, detected JSRV-infected cells in all the MLN samples analyzed, but only in half of the PP samples tested.
Jaagsiekte Sheep Retrovirus (JSRV) Detection in the Tissues of the Lambs From JSRV-Blood Positive Ewes.
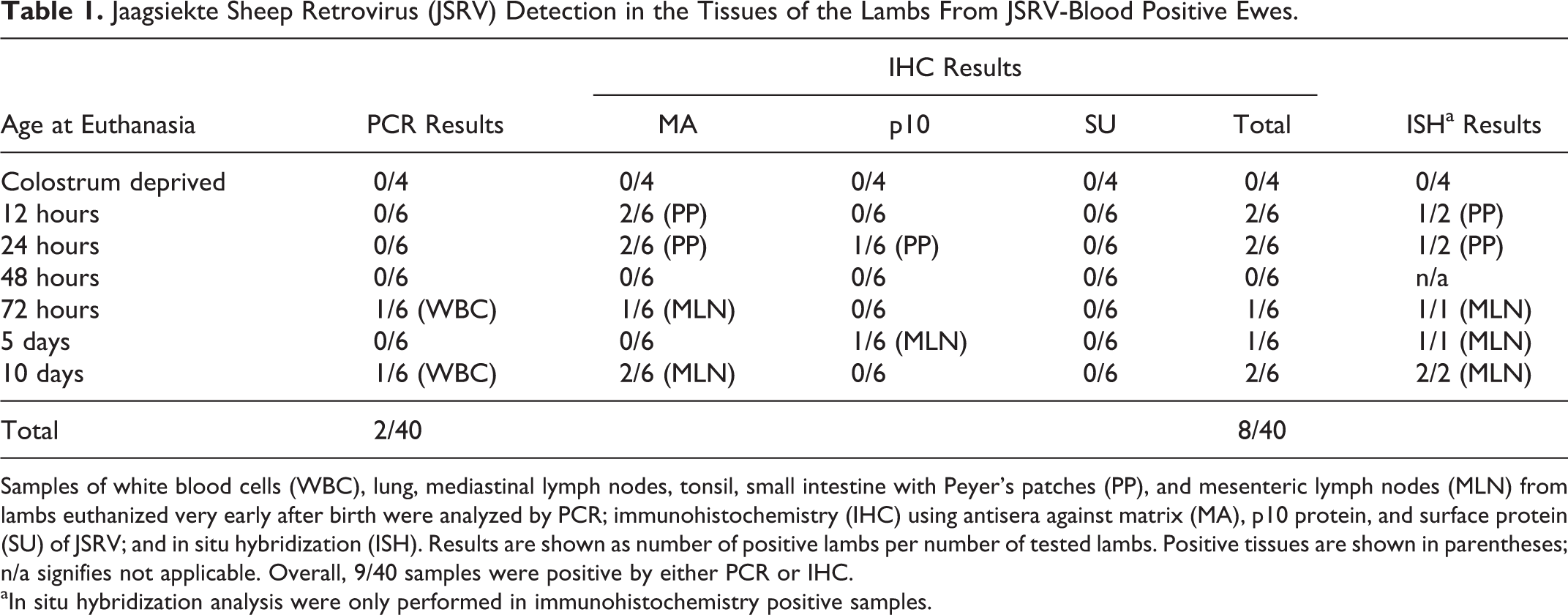
Samples of white blood cells (WBC), lung, mediastinal lymph nodes, tonsil, small intestine with Peyer’s patches (PP), and mesenteric lymph nodes (MLN) from lambs euthanized very early after birth were analyzed by PCR; immunohistochemistry (IHC) using antisera against matrix (MA), p10 protein, and surface protein (SU) of JSRV; and in situ hybridization (ISH). Results are shown as number of positive lambs per number of tested lambs. Positive tissues are shown in parentheses; n/a signifies not applicable. Overall, 9/40 samples were positive by either PCR or IHC.
aIn situ hybridization analysis were only performed in immunohistochemistry positive samples.
IHC detection of JSRV-positive cells in sections of PP from small intestine and MLN is shown in Figures 1 and 2, respectively. In both tissues, labeled cells with lymphoreticular-like morphology were identified, which presented an intracytoplasmic immunolabeling. Labeled cells were found at a low frequency, particularly in PP (1–2 positive cells after complete examination of the section). JSRV proviral DNA detected by ISH, performed on sections of PP and MLN, is shown in Figures 3 (PP) and 4 (MLN). A low number of infected cells were also revealed with this technique.
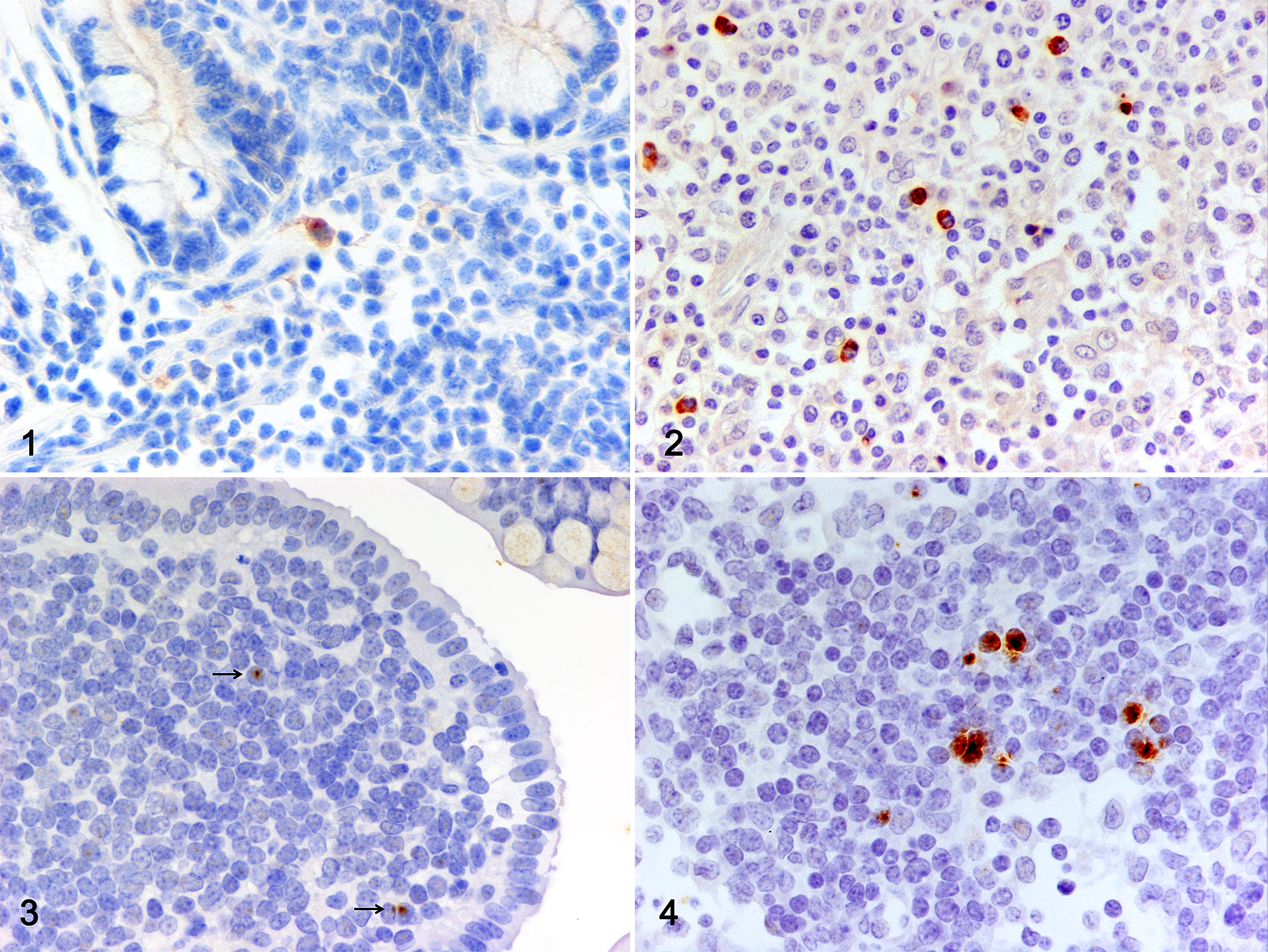
Jaagsiekte sheep retrovirus (JSRV) infection, lymphoid tissue, lamb. Detection of JSRV-positive cells in the tissues of lambs nursed by JSRV-infected ewes.
No positive results were detected by PCR or IHC in any samples analyzed from the colostrum-deprived lambs from the JSRV (+) group (Table 1) and from the lambs from the JSRV (-) group.
Table 2 describes the distribution of JSRV detected in the lambs from the JSRV (+) group of ewes, according to the C/M status of the mother. A total of 9 out of the 36 lambs (25%) that suckled C/M from the JSRV (+) group of ewes became positive. The percentage of lambs delivered from C/M-positive ewes in which JSRV was detected increased to 31.5%, with 6/19 positive lambs. The virus was also present in the tissues of 3 of 17 lambs (18%) that suckled from blood-positive ewes in which JSRV could not be detected in C/M.
Detection of JSRV in the Lambs From Blood-Infected Ewes According to Colostrum/Milk Status of the Mothers.
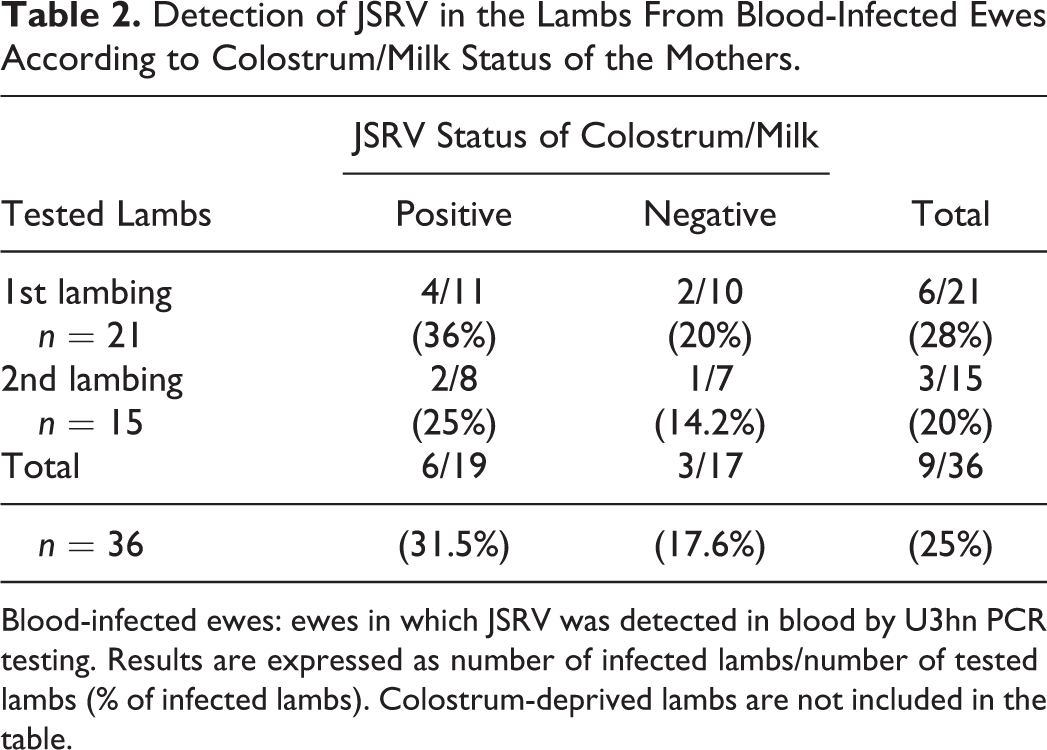
Blood-infected ewes: ewes in which JSRV was detected in blood by U3hn PCR testing. Results are expressed as number of infected lambs/number of tested lambs (% of infected lambs). Colostrum-deprived lambs are not included in the table.
Discussion
Exogenous retroviruses can be transmitted by different routes, with C/M being a very important mode of infection for several. Human immunodeficiency virus (HIV), mouse mammary tumor virus (MMTV) and maedi-visna virus (MVV) are good examples of this: HIV, MMTV, and MVV are found as free particles in the C/M and enter the newborn during suckling. Infected epithelial and/or lymphoreticular cells in the mammary gland produce these viral particles, and once they are inside the digestive tract they can cross the intestinal epithelial lining and/or infect epithelial cells and lymphoreticular cells in the gut and disseminate the infection to other organs. 1,13,21,23,29 In addition, cell-associated virus secreted in C/M, as in the case of HIV and MVV, 13,20 can also be involved. Colostrum is very rich in lymphocytes and monocytes and it has been demonstrated that these cells can breach the intestinal barrier of newborn, colonize mesenteric lymphoid tissues, and reach the circulation. 14,28 C/M also appear to be involved in the transmission of JSRV infection to lambs. 10 However the way in which C/M can transmit the virus has not, as yet, been elucidated.
It is generally accepted that JSRV can be transmitted from sheep to sheep by the respiratory route through lung secretions containing JSRV generated from OPA—affected animals. 27 With this mode of transmission, inhaled free JSVR-particles reach the alveolar type II cells, which are the target cells of the virus. In the C/M of infected ewes, JSRV proviral DNA has been detected in the SC using PCR techniques and it has been further demonstrated that the infection can be transmitted to recipient lambs artificially fed with C/M from infected ewes. 10
Results obtained in this study reinforce previous findings that JSRV-infected cells from C/M may play a role in the transmission of JSRV infection to the lambs. First, we detected JSRV proviral DNA by PCR in somatic C/M cells in 10/22 infected sheep (45%), at different sampling times throughout a lactation period. Our data confirm the findings of Grego et al, 10 that detected JSRV proviral DNA at the beginning and end of the lactation period, but also show that cell-associated virus can be excreted throughout lactation in JSRV-infected but asymptomatic ewes. In spite of the high number of samples analyzed for each animal throughout the period of the study, only very few of them were JSRV-positive. As inhibition of GAP-DH PCR was not found in the samples that tested JSRV-negative, the DNA from these samples was understood to be in good condition and inhibitors of PCR were ruled out. Consequently, our results suggest that the proviral load in C/M samples was not high enough to allow detection by this highly sensitive heminested PCR method that detects a single copy of JSRV template in a background of 500 ng of sheep genomic DNA. 17 These results in C/M samples resemble those obtained using PCR techniques to detect JSRV in WBC of infected but asymptomatic animals, that provided inconsistent detection, 2,3,5 probably as a consequence of the low number of WBC containing the proviral DNA. 12 Similarly in somatic C/M cells, a low level of infection may explain our results. Second, despite that the number of C/M cells containing proviral DNA seems to be very low, we have found, by IHC and ISH, JSRV-infected cells with lymphoreticular-like morphology in the PP or MLN in 8 of the 36 lambs (22%) nursed by infected ewes, in 2 of them as early as 12 hours after birth. These data indicate that JSRV from C/M, free or cell-associated virus or both, can cross the intestinal barrier. The possibility of free virus appears remote for JSRV as high levels of viral expression have only been evidenced in the transformed secretory epithelial cells of the distal respiratory tract of OPA-affected animals. 16 Our results are in agreement with this, and immunolabeling of positive cells in the PP and MLN was only evident when antisera against viral MA and p10 proteins were used, pointing to some viral proteins of gag gene being expressed in these cells. But there is no env gene expression or it is below the limit of detection, making virus replication possibility ineffective or at the least, very low. In contrast to MVV infection, 21 in which viral absorption or infection of intestinal epithelial cells occurs, labeling of these cells was not observed in any of the lambs of this study. Furthermore, efforts to obtain evidence of JSRV infection of mammary epithelial cells or free virus in C/M of infected sheep have been unsuccessful (Ortín, unpublished results). All these data strongly suggest that JSRV most likely crosses the intestinal barrier of newborn lambs as cell-associated virus, although passage of free viral particles cannot be completely ruled out. The intestinal barrier in normal lambs is permeable to the mother’s lymphoreticular cells for a few hours after birth, 28 being a relevant physiological event and important for the transmission of immunity from the mother to the offspring for species with epitheliochorial placentation, such as sheep. 14 Thus, infected and noninfected cells from colostrum would cross the intestinal barrier during this short period. The low frequency of infected cells in C/M would explain the low number of positive cells found in the PP and MLN of the lambs. Negative results obtained by PCR in these lamb tissues may also be explained by the very low level of infected cells present in the samples that seems to be below the detectable level for PCR.
The virus was detected in tissues of the lambs from mothers with infected C/M but it was also found (although in a lower proportion) in lambs from sheep that were blood-infected, but JSRV proviral DNA could not be detected in C/M. This result indicates that blood-infected ewes can transmit JSRV to lambs by suckling even if the virus is not detected in their C/M. Again as a consequence of the low frequency of infected cells, it is impossible to confirm with absolute certainty that C/M is not infected.
Positive cells were found first in PP (a few hours after birth), and later in MLN and WBC, and never in samples from lung and mediastinal lymph node. This indicates that infection of the lambs via the respiratory route was unlikely. As we mentioned we have detected positive cells in the PP at 12 and 24 hours after birth and later they have been found in the MLN (at 72 hours, 5 and 10 days) but also in the WBC (at 72 hours and 10 days). This means that JSRV-infected cells from colostrum can reach the circulation. Previous publications indicated that JSRV-positive WBC can be detected for several months after birth in lambs artificially fed with infected C/M. 10 In addition, JSRV-positive cells can be detected in lambs from commercial OPA-affected flocks for a period of years. 3 Thus, it could be inferred that JSRV infection of lambs, transmitted via C/M could persist for a longer time than the expected lifespan of the mother’s infected leukocytes, 34 establishing a permanent JSRV infection in these lambs. HYAL-2 is the cell surface receptor for JSRV and it is present in numerous cell types. 22 Consequently JSRV can infect many cell types including lymphocytes and monocytes, but the LTR of the virus is preferentially active in type II pneumocytes and club cells (formerly known as Clara cells) of the lung. 19 This means that JSRV has very high activity only in the cells where the tumor develops. In other cells such as lymphocytes and monocytes the level of replication appears to be low and the generation of viral particles is also presumably low. Hence, infected cells from the mother may have a poor level of replication in the lymphoid organs of the lamb, infecting only a few cells that are in close proximity. This would result in maintaining a very low level of infection in a few cells which can circulate and be detected occasionally in the WBC. Thus, JSRV infection would be maintained at a very low level in the lambs. These infected cells could be recruited to the lung in case of injury and participate in the sequence of events leading to infection of active progenitor cells for lung repair of bronchioloalveolar areas, and tumor development. Recent studies have demonstrated that naturally occurring initial OPA lesions are mixed tumors that might be generated after specific lung injuries with the active participation of lymphoreticular cells and the local progenitor/stem cells of lung repair. 6 However, this line of argument is only speculation as it is not supported by experimental evidence. More work is required to unravel the process of sustained JSRV infection and determine if C/M transmission route can lead to OPA development.
Investigation into routes of viral transmission is essential for the implementation of control strategies of an infectious disease such as OPA, where a limited number of control measures are available. According to the data presented here, heat treatment of colostrum and artificial rearing of lambs could be implemented as initial JSRV infection control measures, the same as for MVV infection, since after heat treatment the cells are not absorbed into the intestines of newborn lambs. 28 Nevertheless, heating can affect IgG concentration of colostrum 15 and new technologies for colostrum sanitation, not based on heating, are being developed, such as SDS treatment. 11 In any case, control strategies for JSRV and OPA should be implemented at the flock level, due to the low sensitivity of the tests for the detection of infected individuals.
The results obtained from this study have revealed that in JSRV-infected but asymptomatic ewes, even though the number of infected cells in C/M seems to be very low, these cells can cross the intestinal barrier of newborn lambs, reach the mesenteric lymph nodes and enter into circulation. Therefore it is confirmed that colostrum intake is a mode of transmission of JSRV to lambs suckling under natural conditions. More studies must be undertaken to determine the impact of this route in the transmission of JSRV to lambs, ascertain how resilient the resulting infection is and explore the possibility that C/M transmission of JSRV can result in OPA development.
Footnotes
Acknowledgements
We are grateful to Dr Massimo Palmarini and Dr Dusty Miller for generously providing the antisera against JSRV proteins, and to Dr Christina Summers for reading and revision of the manuscript. We do not agree with the use of terms with a connection to Nazi Germany, such as “Clara cell,” and in our opinion “Club cell” would be preferable. However, due to the widespread international use of “Clara cell,” and in order to not confuse readers, both terms have been used in this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from the Spanish Ministerio de Ciencia e Innovación, AGL2008-00803 and Marta Borobia was a predoctoral research fellow under the Gobierno de Aragón programme (B103/09).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
