Abstract
Ovine pulmonary adenocarcinoma (OPA) is a naturally occurring and experimentally inducible lung cancer of sheep caused by Jaagsiekte sheep retrovirus (JSRV). The first aim of this study was to monitor the development of OPA with minimally invasive, real-time observations of animals experimentally infected with JSRV as well as ovine lentivirus (maedi-visna virus). Worldwide, simultaneous infection of sheep with these 2 retroviruses is a common occurrence, naturally and experimentally; consequently, the lung tumor homogenates used as inocula contained both viruses. Following inoculation, computed tomography was used to detect tumor nodules early, before the onset of clinical signs, and to monitor tumor advancement. However, not only was OPA disease progression observed, but the apparent spontaneous regression of OPA was witnessed. In fact, regression was more common than progression following JSRV inoculation of neonatal lambs. Immune responses were detected, particularly involving CD3+ T cells and the production of antibodies against JSRV that may mediate the spontaneous regression of JSRV-induced OPA. The second aim of this study was to determine whether OPA tumors harbor genetic alterations similar to those found in human lung adenocarcinoma. No mutations were found in the tyrosine kinase domain of the epidermal growth factor receptor, KRAS codons 12 and 13, or the DNA-binding domain of p53 in tumor DNA from naturally occurring and experimentally-induced OPA cases. Overall, the genetic profile combined with the disease development data provides further important characterization of OPA and describes, for the first time, spontaneous regression of OPA tumors in experimentally infected sheep.
Keywords
Ovine pulmonary adenocarcinoma (OPA), also known as sheep pulmonary adenomatosis (SPA), is a naturally occurring and experimentally inducible lung cancer of sheep that shares symptomatic and histopathologic characteristics with human bronchioloalveolar carcinoma (BAC). 10,14,20,37,43,57 Symptomatically, OPA presents with the same clinical signs as BAC in humans: progressive dyspnea, abundant bronchorrhea, cough, anorexia, and cachexia. 37 Histopathologically, in BAC and OPA, the tumor originates from differentiated alveolar type II cells and nonciliated bronchiolar epithelial (Clara) cells. 39,45,46,55 Also, in both cancers, the neoplastic tissues are preferentially localized at the periphery of the lungs, 43 and the lesions tend to be multifocal, 43 presumably owing to either intrapulmonary metastasis 35 or a multiclonal origin. 5 Additionally, there may be molecular signaling parallels between these two lung cancers, including aberrant activation of the phosphatidylinositol-3 kinase/Akt and Ras/Raf/extracellular signal–regulated kinase 1/2 pathways. 31
Although the exact mechanism of OPA oncogenesis is unknown, the Jaagsiekte sheep retrovirus (JSRV) envelope (Env) protein has been found to transform cells in culture 1,9,13,32-34,44,48,63 and to induce lung adenocarcinomas in sheep 8 and immunodeficient mice. 62 Thus, in vitro and in vivo, JSRV Env has been shown to function as an oncoprotein and to be the viral component that is necessary and sufficient to induce transformation. Specifically, deletion and mutation experiments show that the transmembrane subunit of JSRV Env is the primary determinant for cell transformation. 9,23,25,31
OPA induction in sheep is predictable and reproducible following JSRV inoculation of neonatal lambs. 17,52,56,57 Currently, diagnosis of OPA is possible only when clinical signs or lesions at necropsy are observed 15 and the presence of JSRV is confirmed in lung fluid or tumors by immunoblotting, 58 enzyme-linked immunosorbent assay, 42 or polymerase chain reaction (PCR). 3,41 It is difficult to identify infected animals during the preclinical period because of the lack of detectable JSRV proteins outside the tumor 42 and the absence of circulating JSRV-specific antibodies. 40,58 A blood test was recently developed that used PCR to detect JSRV; however, this test was unreliable for detection of disease in affected sheep that did not exhibit clinical signs of OPA. 16 Consequently, a dependable method is lacking for minimally invasive detection of OPA before the onset of clinical signs.
The first aim of this study was to test the utility of computed tomography (CT) as a minimally invasive imaging modality to detect OPA earlier than possible with clinical observations and to follow the development of disease over time. The second aim of this study was to compare OPA and human lung adenocarcinoma on a molecular level. Specifically, we investigated whether OPA tumors harbor genetic alterations similar to those found in human lung adenocarcinoma by analyzing OPA-derived tumor DNA for mutations in the tyrosine kinase (TK) domain (exons 18–21) of the epidermal growth factor receptor (EGFR), exon 1 of KRAS (v-Ki-ras2 Kirsten rat sarcoma viral oncogene homolog), with particular interest in codons 12 and 13, and the DNA-binding domain (exons 5–8) of p53.
Materials and Methods
Experimental Infection and Archival Samples
Twelve colostrum-fed neonatal female lambs were obtained from a local flock reportedly free of ovine lentivirus (OvLV) infection (Josh Livestock, Greeley, CO). At approximately 1 week of age, lambs were given inocula prepared from the lung tissue (stored at –70°C for 8 to 16 years) of 3 sheep with confirmed OPA as previously described. 17 Inocula A and C were prepared from natural cases of OPA, and inoculum B was prepared from an experimentally induced OPA case. Inocula were tested for lentiviral reverse transcriptase (RT) activity using a Lenti RT Activity Kit (Cavidi Tech, Uppsala, Sweden) in accordance with the manufacturer’s instructions. For inoculation, the lamb was restrained, an 18-gauge needle was inserted into the distal trachea between 2 tracheal rings, and 5 mL of the respective inoculum was injected. With inhalation, the inoculum was aspirated into the airways. Animals were monitored daily for clinical signs of respiratory disease suggestive of OPA. Lambs were euthanized humanely. At necropsy, gross examination of the lungs was performed to identify any lesions suggestive of OPA. Tumorlike areas and/or tissue from all other lobes were obtained for microscopic examination and further analysis. For microscopy, tissue samples were fixed in 10% neutral-buffered formalin, processed routinely, sectioned at 5 µm, and stained with hematoxylin and eosin (HE). Other tissue samples obtained at the time of sacrifice were rapidly frozen in liquid nitrogen and stored at –80°C. All experimental procedures were approved by Colorado State University’s Animal Care and Use Committee. For comparison, ovine lung tumor samples were obtained from frozen tissue collected from sheep naturally or experimentally affected by OPA. 17,50,51 Tissue was collected at the time of sacrifice, rapidly frozen in liquid nitrogen, and stored at –80°C. If recognized grossly, tumor tissue was dissected to enrich the tumor cell population.
CT Imaging
Disease progression was monitored by acquiring serial sheep thorax images via spiral CT using a modified Picker PQ CT scanner. Scan factors were as follows: 130 kV, 150 mA, 1.5 pitch, 512 × 512, and 25-cm field of view using a transverse imaging plane and reconstructed as 5.0-mm contiguous slices. Scanning was performed before and after intravascular contrast material (Hypaque 76 [370 mg/mL iodine], GE/Amersham, Pittsburgh, PA) administration (2.2 mL per kg), with the exception of the preinoculation scans (no contrast material). Postinoculation (PI) CT images were scans acquired after contract material administration. For all scans, sheep were anesthetized with morphine and isofluorane and subsequently situated in the supine position. Images were acquired before inoculation, and PI on a monthly basis through 1 month (1 animal), 4 months (2 animals), 5 months (3 animals), and 6 months (6 animals).
Using a lung window (width: 1500, level: –450), a board certified veterinary radiologist (S. L. Kraft) evaluated the CT scans. Disease characteristics were recorded, including location, presentation, appearance, extent, internal features, and nodule margin characteristics.
Disease location
The location of disease was arbitrarily considered to be central if the disease was within the inner two thirds of the lung and peripheral if the disease was within the outer one third of the lung. Dorsal and ventral disease location was also noted. Additionally, disease location was described by lobe (right cranial lobe, right middle lobe, right caudal lobe, right accessory lobe, left cranial lobe, or left caudal lobe).
Disease presentation
The presentation of disease was described as a solitary nodule, multiple nodules, or diffuse lung pattern (DLP). A pulmonary nodule was defined as a discrete focal opacity less than 3 cm in maximum diameter. 2 An area of opacity larger than 3 cm was described as a DLP.
Disease appearance
Disease attenuation was described as ground glass opacity (GGO), solid, or mixed (GGO/solid). GGO was defined as hazy increased attenuation of the lung without obscuration of underlying vessels. 2 Solid attenuation was defined as a homogeneous increase in pulmonary parenchymal attenuation with obscuration of underlying vessels. Mixed (GGO/solid) consisted of heterogeneous attenuation with both GGO and solid components.
Extent of disease
Extent of disease, whether diffuse or nodular, was expressed according to lobar or sublobar locations or general occurrence. Nodules were described in terms of size (mm) and number (1, 1–10, 10–20, 20–30, > 30).
Internal features of disease
An air bronchogram was described as the appearance of an air-filled bronchus surrounded by airless lung parenchyma. 60 A cystic air space was described as a well-defined, circumscribed, air-containing lesion surrounded by a wall of variable thickness. 2 CT angiogram sign was described as enhanced branching pulmonary vessels in a low-attenuating consolidation of the lung parenchyma. 26 In addition, for nodules, a halo sign was described as GGO surrounding the circumference of a nodule or mass, 2 and pseudocavitation was described as lucency within a nodule that did not represent a cavity. 60
Nodule margin characteristics
Nodule margins were considered ill defined if the perimeter was hazy or indistinct. If the nodule was sharply and distinctly separated from surrounding lung parenchyma, the margins were considered well defined. Well-defined margins were described as smooth or irregular. Nodule margins were considered smooth if the margin was round and distinctly separated from surrounding lung parenchyma. Nodule margins were considered irregular if the margin was erratic and/or indistinct. Nodules with irregular margins were further examined for spiculations and pleural tags. Spiculations were defined as fine linear strands extending from the nodule margin into lung parenchyma without contacting the pleural surface. 30 A pleural tag was defined as a linear area of high attenuation that was surrounded by aerated lung, originated from the margin of the nodule, and extended peripherally to contact the pleural surface. 30
Nodular Disease Development
Progression of nodular disease was defined as an increase in the size and/or number of nodules over time. Partial regression of nodular disease was defined as a decrease in the size and/or number of nodules over time. Complete regression of nodular disease was defined as a total disappearance of nodules over time.
Polymerase Chain Reaction
DNA was extracted from samples using a DNeasy Tissue Kit (Qiagen, Chatsworth, CA). The exogeneous JSRV (exJSRV) transmembrane domain, the first 4 exons (exons 18-21) of the TK domain of the EGFR gene, exon 1 of the KRAS gene, and exons 5–8 of the highly conserved core domain of the p53 gene were amplified in a one-step PCR using the MultiBlock PCR System (Thermo Electron Corporation, Waltham, MA). Each 50 µL PCR reaction contained 45 µL Platinum PCR SuperMix (Invitrogen, Carlsbad, CA), 200 nM each of the forward and reverse primers (Table 1), and 500 ng template DNA. PCR cycling parameters were as follows: one template denaturation/enzyme activation cycle at 94°C for 2 minutes, 35 cycles of denaturation at 94°C for 30 seconds, annealing at indicated temperature (Table 1) for 30 seconds and extension at 72°C for 60 seconds, followed by one final extension cycle at 72°C for 10 minutes. PCR amplification products were then separated and identified by electrophoresis in 3% agarose gels containing ethidium bromide.
Primer Sets and Annealing Temperatures Used for Amplification in Polymerase Chain Reactions a
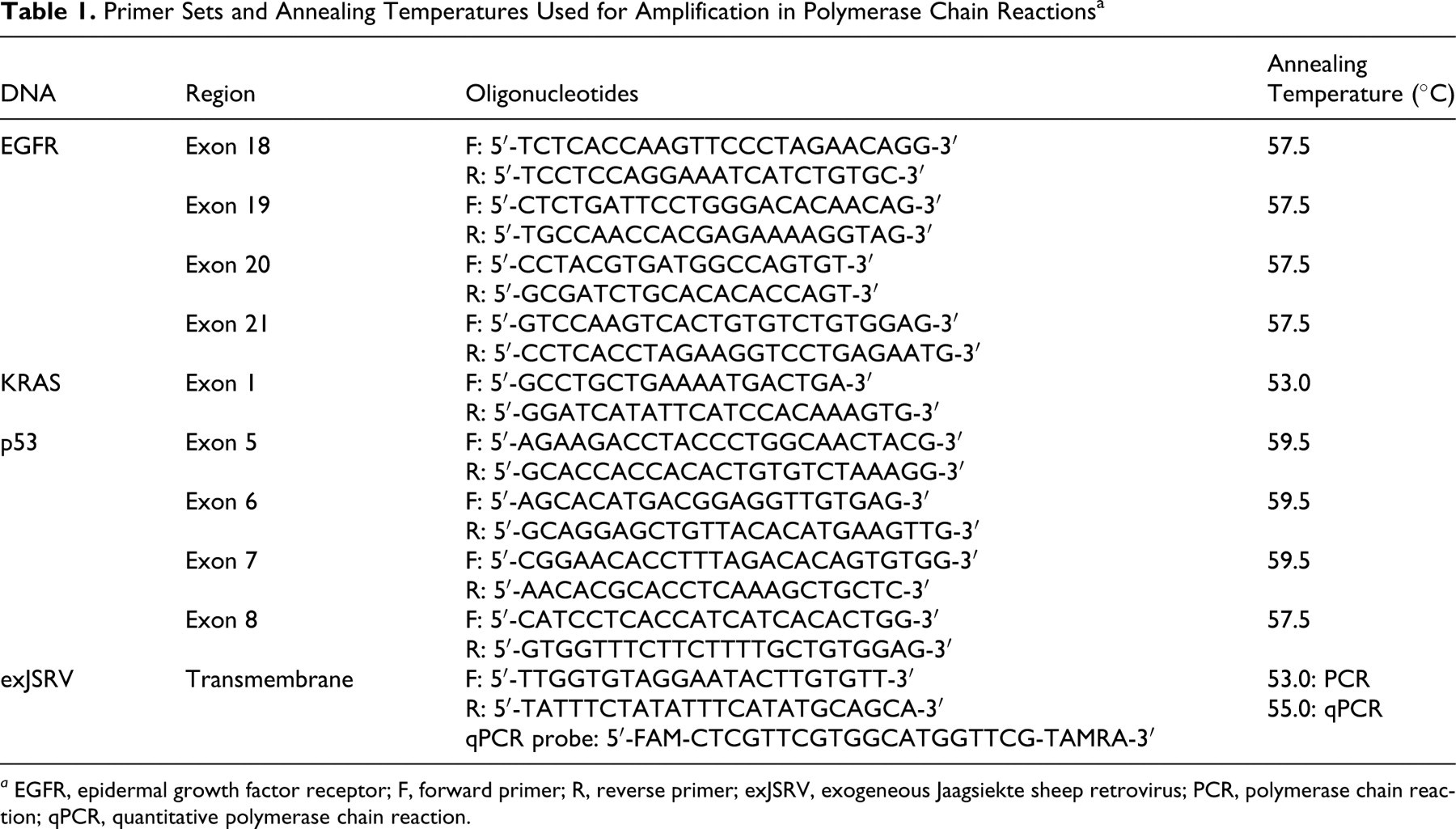
a EGFR, epidermal growth factor receptor; F, forward primer; R, reverse primer; exJSRV, exogeneous Jaagsiekte sheep retrovirus; PCR, polymerase chain reaction; qPCR, quantitative polymerase chain reaction.
DNA Sequencing of EGFR, KRAS, and p53
The resulting PCR products were purified using a QIAquick 96 PCR Purification Kit (Qiagen). High-throughput (96-well plate) bidirectional dideoxynucleotide sequencing of PCR-amplified gene products was performed at Polymorphic DNA Technologies, Inc. (Alameda, CA) using a BigDye Terminator v3.1 Cycle Sequencing Kit (Applied Biosystems, Foster City, CA) and an ABI 3730xl DNA Analyzer (Applied Biosystems). Sequence variants were confirmed by an additional, independent PCR amplification and sequencing reaction in sense and antisense directions.
Quantitation of exJSRV Copy Number
DNA was extracted from samples as above. Integrated exJSRV copy number was detected using a fluorogenic probe in combination with real-time PCR using probes and primers based on the nucleotide sequence of the envelope region of JSRV. Each 25 µL PCR reaction contained 12.5 µL Platinum Quantitative PCR Supermix-UGD With ROX (Invitrogen), 200 nM (final concentration) forward primer, 200 nM (final concentration) reverse primer, 100 nM (final concentration) fluorogenic probe, and 250 ng template DNA (Table 1). Quantitative PCR cycling parameters were as follows: one UDG incubation cycle at 50°C for 2 minutes, one template denaturation/enzyme activation cycle at 95°C for 2 minutes, 45 cycles of denaturation at 95°C for 15 seconds and annealing at 55°C for 45 seconds (this step was enabled for data collection). Reactions were done in triplicate. A full-length JSRV provirus clone (pBlue-JSRV) was serially diluted and used as a standard. The sensitivity of this assay (developed by Nuno F. Carreiro and Thomas E. Allen in the DeMartini laboratory) was 1 copy of pBlue-JSRV. JSRV copy numbers are presented as the mean of 3 replicates ± standard error.
Immunohistochemistry
Immunohistochemistry (IHC) labeling of lung tissue was performed using standard techniques on an automated stainer (Discovery, Ventana Medical Systems, Tucson, AZ). The antiserum against the JSRV Env was collected from C57BL/6 mice at 8 weeks after infection with an AAV vector expressing JSRV Env (ARJenv). 62 The antiserum was diluted 1:500. The pan–T cell antibody used was a polyclonal rabbit antihuman CD3 antibody (catalog No. CP 215, Biocare, Concord, CA), diluted 1:50. Slides were analyzed for CD3 immunolabeling using an image analysis workstation (AxioVision Release 4.6, Carl Zeiss, Maple Grove, MN). Polychromatic high-resolution images (1300 × 1030 pixels) were obtained at 20× magnification. The percentage of tissue surface area with CD3 immunolabeling was recorded from 5 independent image analyses, and the mean was used for statistical analysis.
Virus Neutralization Assay
The ability of serum to neutralize the infectivity of virus bearing the JSRV Env protein was measured as described previously. 62 The virus used was a retroviral vector encoding placental alkaline phosphatase that was packaged using gag-pol proteins from Moloney murine leukemia virus and Env protein from JSRV. 47 Briefly, approximately 150 infectious units of virus were mixed with various dilutions of sheep serum in a total volume of 40 µl, and the mixtures were incubated at 20°C for 30 minutes. The serum–virus mixtures were then added to HT-1080 human fibrosarcoma cells in 6-well plates seeded the day before at 5 × 104 cells per well. Infections were done in the presence of 4 µg/ml Polybrene. Two days after virus exposure, the cells were stained for alkaline phosphatase expression, and positive foci were counted. Antibody titers were expressed as the reciprocal of the highest dilution of serum that reduced the number of viral foci by ≥ 50%. Assays were performed on both heat-inactivated and non-heat-inactivated serum with identical results, showing that heat-labile serum components such as complement were not responsible for virus inactivation. Last, serum from sheep No. B2 at 6 months PI was unable to neutralize (titer < 10) an otherwise identical retroviral vector bearing a mouse amphotropic retrovirus Env protein instead of the JSRV Env, whether the serum was heat inactivated or not, showing that the neutralizing activity observed for this serum against the JSRV vector was JSRV Env specific.
OvLV Detection
OvLV DNA was measured by PCR and serum antibodies to OvLV were detected by agar-gel immunodiffusion serology, both at Colorado State University’s Veterinary Diagnostic Laboratories.
Statistical Analysis
Two-tailed unpaired Student’s t-test (normal distributions and equal variances), two-tailed unpaired Student’s t-test with Welch’s correction (normal distributions and unequal variances), and one-way analysis of variance with Bonferroni’s multiple comparison posttest were used for statistical analysis. Differences were considered significant when P < .05. Statistical analysis was performed using Prism v4.03 (GraphPad Software, San Diego, CA).
Results
RT Activity and Viral DNA in Inocula
Three inocula prepared from 3 sheep with confirmed OPA were used in this study to maximize the likelihood of OPA induction. All 3 inocula exhibited RT activity indicating the presence of retroviruses. JSRV proviral DNA was detected by PCR in 2 of the preparations (A and B), as described in the methods, and in the other preparation (C) using a different PCR assay (J. Bishop, unpublished). Further analysis was performed using qPCR: JSRV copy numbers per 250 ng of DNA isolated from inoculum A and inoculum B were 3,800 and 860 copies, respectively. JSRV DNA was not detected in inoculum C by qPCR. We also looked for an alternative source of RT activity in the inocula and found that DNA from all 3 tested positive by PCR for OvLV. Consequently, the RT activity in the preparations cannot be attributed solely to JSRV. Thus, in the following study, 3 cohorts of sheep (n = 4 each) received inocula containing both JSRV and OvLV proviral DNA, although the cohort exposed to inoculum C likely received a preparation that contained JSRV at a lower titer than the others. Sheep exposed to inocula A, B, and C will subsequently be referred to as A1, A2, A3, A4; B1, B2, B3, and B4; and C1, C2, C3, C4.
Clinical Signs
Sheep were monitored closely for clinical signs of OPA, including audible breathing, moist rales, progressive dyspnea, abundant bronchorrhea, nasal discharge, and cachexia. Mild audible breathing was observed in all sheep during the study and was not specific to nodular disease development group but did progress, though not substantially, over time. No other clinical signs were evident, even at 6 months PI. At euthanasia, the “wheelbarrow test” was performed on all sheep, in which the hindquarters of the animal are lifted above the head to check for lung exudate flow out the nose and mouth; no fluid was observed. Overall, only mild clinical signs of respiratory distress were apparent in the sheep involved in this study.
Measurements of Disease by CT
Before inoculation, ovine lung on CT appeared as a pattern of hypoattenuating air-filled lung parenchyma interspersed by a honeycomb pattern of soft tissue–attenuating connective tissue of the lungs. The bronchial tree was visible as tubular gas-filled structures with soft tissue–attenuating walls that became smaller as they branch peripherally, accompanied by soft tissue–attenuating tubular pulmonary vascular branches throughout the lungs. Following virus administration, pulmonary abnormalities were visible on CT scans in all 12 sheep (100%) and were predominated by two distinct disease presentations: nodules (considered to represent OPA) and a DLP (considered to represent OvLV-induced ovine progressive pneumonia [OPP]) (Table 2). In total, nodules were seen in 8 of 12 sheep (67%) and a DLP was evident in all 12 sheep (100%). These two disease presentations occurred individually and concurrently, and, when found together, nodules were seen within the DLP and/or independent of the DLP.
Summary of Computed Tomography Disease Presentation and Nodular Disease Progression/Regression in Inoculated Sheep a
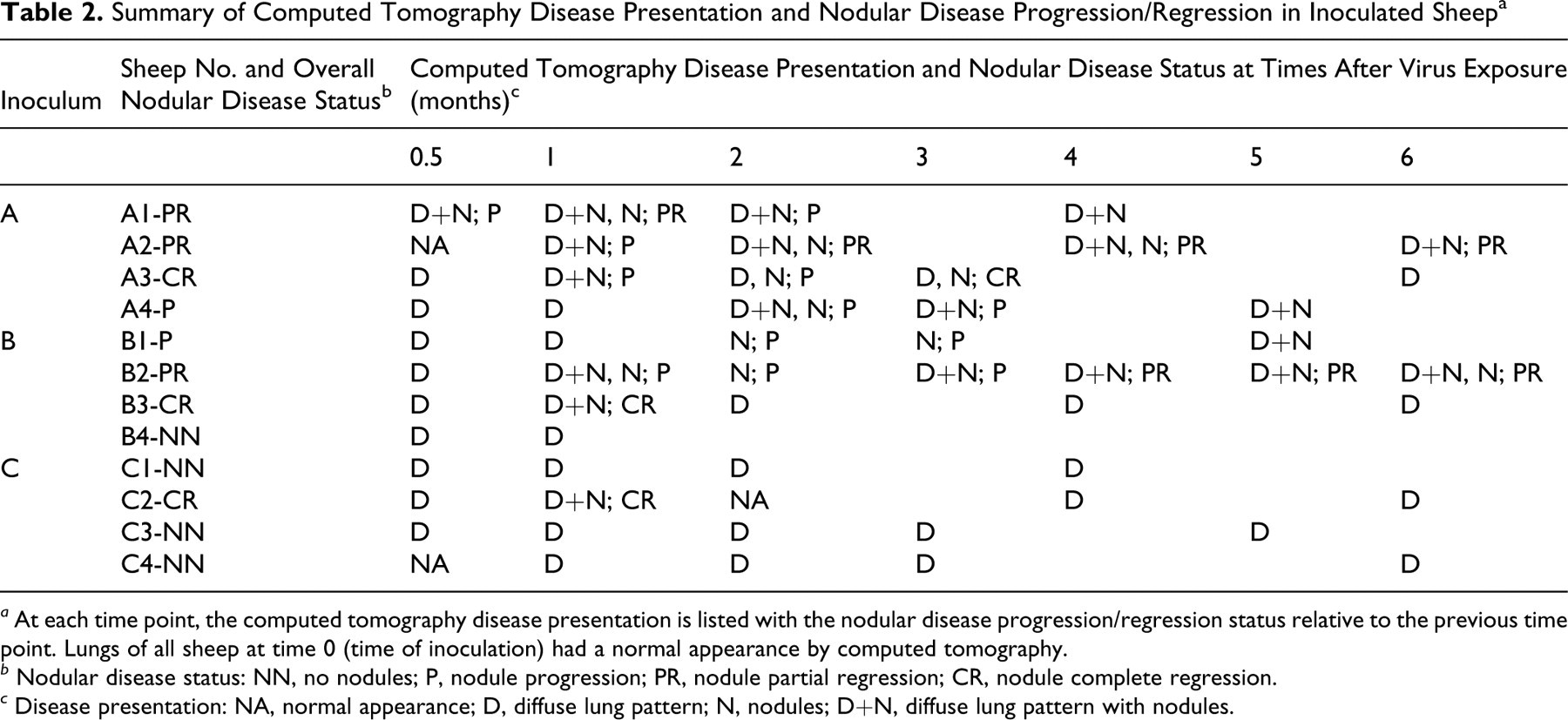
a At each time point, the computed tomography disease presentation is listed with the nodular disease progression/regression status relative to the previous time point. Lungs of all sheep at time 0 (time of inoculation) had a normal appearance by computed tomography.
b Nodular disease status: NN, no nodules; P, nodule progression; PR, nodule partial regression; CR, nodule complete regression.
c Disease presentation: NA, normal appearance; D, diffuse lung pattern; N, nodules; D+N, diffuse lung pattern with nodules.
Nodule presentation varied from a solitary nodule (n = 2) to multiple nodules (n = 6). The attenuation of these nodules ranged from GGO to mixed (GGO/solid) to solid, with mixed opacity nodules being the most prevalent. Halo signs and psuedocavitation were occasionally observed within the nodules. Both ill-defined and well-defined nodule margins were seen. The well-defined margins were sometimes smooth but more commonly irregular, with occasional spiculations and pleural tags. Nodules were found unilaterally and bilaterally, with numbers often exceeding 30. The sizes of the nodules were typically less than 6 mm, although nodules as large as 10 mm were observed. Size did not vary significantly between nodules with different attenuations. All animals receiving inoculum A developed nodules, 3 of 4 animals receiving inoculum B developed nodules (B1, B2, B3), and only 1 of 4 sheep receiving inoculum C developed nodules (C2).
Tumor Histopathology
At euthanasia, 3 of the 8 sheep that received inocula A and B (with easily detectable JSRV DNA) had grossly apparent lesions (A4, B1, B2) (Fig. 1a, b), 5 had evident nodules on CT (A1, A2, A4, B1, B2), and these same 5 had histopathologically visible tumor. Of the remaining 3 animals (A3, B3, B4) with no grossly or histopathologically discernable tumor at euthanasia, 2 (A3, B3) had nodules visible by CT at some point during the study that completely regressed by 6 months PI, and 1 (B4) was sacrificed early, at 1 month PI. The 4 sheep receiving inoculum C did not have nodules on their final CT scan or grossly or histopathologically discernable tumor at euthanasia, although 1 animal (C2) did have a solitary nodule visible on CT at 1 month PI.
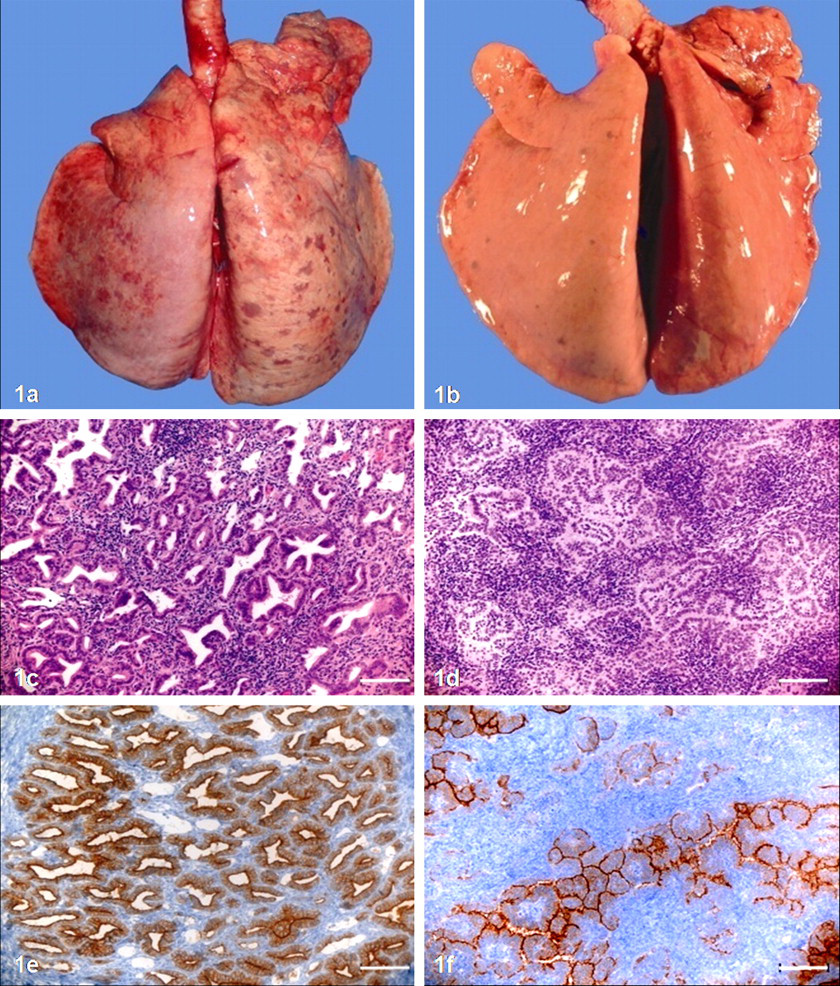
Lung. P, progressor; PR, partial regressor.
The lesion appearances in the 5 sheep with histopathologically confirmed cancer varied from a few tiny well-circumscribed neoplasms to innumerable discrete-to-coalescing tumor masses. Two animals (A1, A2) showed the former pattern whereas the other 3 (A4, B1, B2) showed the latter (Fig. 1c, d). Histologically, the cells that composed these neoplasms were cuboidal to low columnar, with basally located round to oval nuclei having an open chromatin pattern and a central nucleolus. The majority of neoplasms appeared with a regular alveolar pattern and general preservation of the pulmonary architecture; only rarely were there papillary formations in small and terminal bronchioles or a tubulopapillary pattern superimposed on the alveolar appearance. Mitoses were rare but perceptible in 4 of 5 animals with tumor (A1, A4, B1, B2). Morphological features of apoptosis were almost never seen, except in 1 sheep (B2), in which 1 apoptotic cell per 10 high-power fields was observed.
For comparison, we looked at these features in archival HE-stained sections from 22 JSRV-infected sheep with confirmed OPA; observations were similar to those found in our tumor-bearing sheep: Lung neoplasms varied greatly, from one or a few tiny tumors to large coalescing masses; mitotic figures and apoptotic cells were rare.
Disease Development
Surprisingly, not all sheep with nodules visible by CT during the course of the study had progressive OPA (Table 2). In fact, only 2 of 8 animals exhibited progressive nodular disease, as evidenced by increasingly more and/or larger nodules over time (Fig. 2a). The CT scans from the other 6 sheep showed nodules that apparently regressed over time, either partially (Fig. 2b) or completely. In the latter case, there was neither CT nor gross indication of nodules at the end point of the study (euthanasia); additionally, no evidence of OPA was found histopathologically. Based on nodular disease development, sheep were divided into 4 groups for subsequent analyses: no nodules (NN; n = 4), comprising sheep Nos. B4-NN, C1-NN, C3-NN, C4-NN; progressors (P; n = 2), comprising sheep Nos. A4-P, B1-P; partial regressors (PR; n = 3), comprising sheep Nos. A1-PR, A2-PR, B2-PR; and complete regressors (CR; n = 3), comprising sheep Nos. A3-CR, B3-CR, C2-CR.
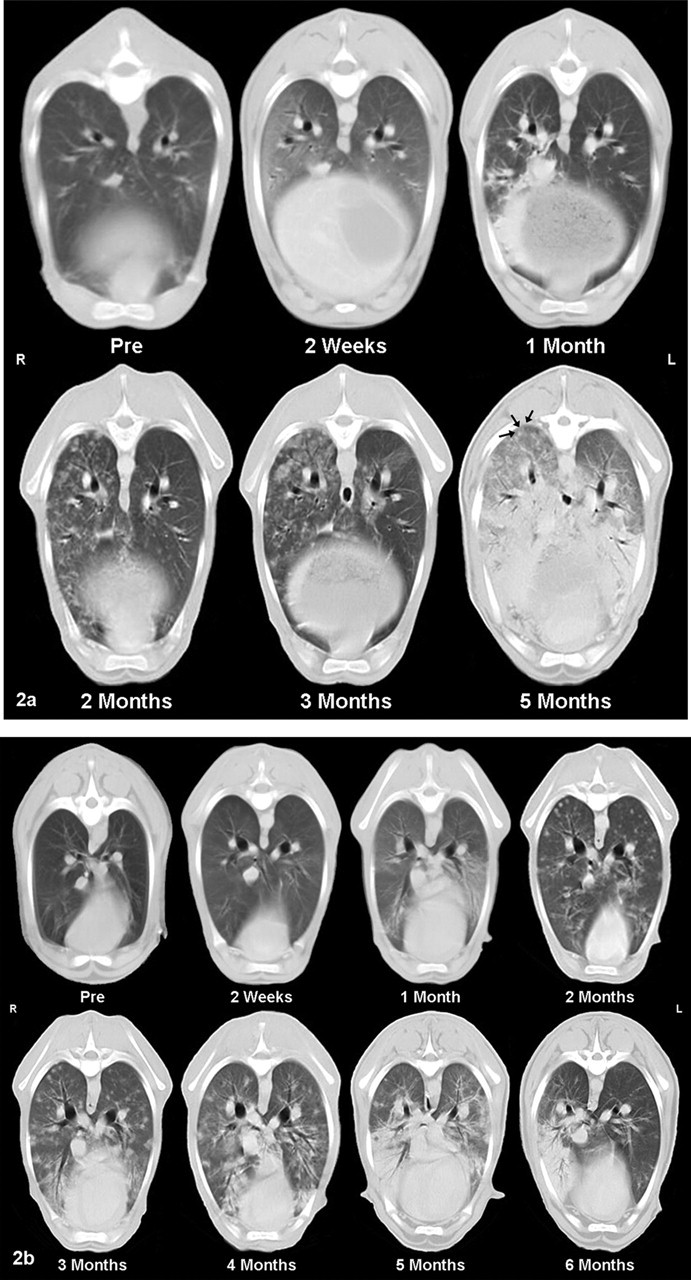
Lung. P, progressor; PR, partial regressor.
CT Characteristics of Nodules
To determine if the nodule characteristics present on CT scans varied between nodular disease development groups, we compared presentation, lung involvement, location, appearance, and extent of nodular disease among progressors, partial regressors, and complete regressors over time. A solitary nodule was detected in only 2 sheep and this occurred early in the study, at 2 weeks PI (A1) and 1 month PI (C2). One of these sheep was a complete regressor (C2-CR), whereas the other developed multiple nodules that partially regressed (A1-PR). The other 6 sheep (A2-PR, A3-CR, A4-P, B1-P, B2-PR, B3-CR) presented with multiple nodules at some point during the study. Nodular lesions always involved both lungs in the progressors, whereas unilateral and bilateral nodules were both seen in the partial regressors. Interestingly, the nodular disease was unilateral in 2 of the 3 complete regressors. Taken together, these results suggest that multiple bilateral nodules are more likely to occur in progressive disease, whereas regression was associated with an appearance of singular or unilateral nodules. There was no clear relationship between anatomic location of nodules in the lung (central, peripheral, dorsal, or ventral) and disease development over time. Attenuation, size, and number of nodules were assessed serially; none of these characteristics were found to vary significantly between nodular disease development groups or over time.
CT Characteristics of DLP
A DLP was observed in all 12 sheep in 90% of PI CT scans. The DLPs consisted of diffuse sublobar to lobar nonstructured hyperattenuation of the pulmonary parenchyma to that of soft tissue density. The diffuse attenuation varied from patchy and nonuniform to uniform (also described as consolidative). In as few as 2 weeks PI, 83% of sheep presented with a DLP, and at 1 month PI, 100% of sheep had a DLP. This resolved in 25% of the sheep by 2 months PI but recurred and was then consistently found in all sheep at all time points, including and after 4 months PI. DLPs were present primarily in the central, peripheral, and ventral parts of the lungs and, less often, dorsally.
Histologically, we observed follicular hyperplasia (FH) of the bronchus-associated lymphoid tissue (BALT), hypertrophy of the BALT, alveolar septal thickening owing to collagen deposition, and increased numbers of mature plasma cells, macrophages, lymphocytes, and occasional neutrophils. These lymphocytes were seen in the lung parenchyma as well as within tumors. In 11 of 12 sheep, both FH and lymphocytic infiltration of lung parenchyma were observed at euthanasia.
To determine if the DLP characteristics present on CT scans varied among nodular disease development groups, we compared lung involvement, location, and appearance of DLP disease among progressors, partial regressors, and complete regressors over time. As mentioned previously, all sheep developed a DLP, so this disease presentation was not unique to a particular group. Also, the colocalization of a DLP and nodules was seen in all groups; it was not time specific; and, accordingly, it was not determined to be indicative of the eventual progression or regression of nodular disease. DLP lung involvement (unilateral versus bilateral disease), lobe location, and appearance were not distinct within any group or over time. Overall, we found no obvious association between a DLP and nodule presence, progression, or regression.
JSRV DNA Quantitation and Env Immunohistochemistry
We were able to detect and quantitate JSRV DNA in lung tissue from 11 of the 12 sheep at euthanasia. In the 4 sheep with no nodules, the mean JSRV copy number was 22 ± 6. In comparison to the sheep with no nodules, the progressors had about 350 times more JSRV DNA (7,700 ± 700 copies); the partial regressors had about 30 times more JSRV DNA (670 ± 240 copies); and the complete regressors had about 5 times more JSRV DNA (112 ± 36 copies). These copy numbers were significantly different (as determined using unpaired t-tests with Welch’s correction) between the no-nodules group and the progressors, partial regressors, and complete regressors groups, with P values of .0001, .0295, and .0375, respectively. In addition, the differences were significant between the progressors and the partial regressors (P = .0001) and between the progressors and the complete regressors (P = .0001). However, there was not a significant difference between the partial regressors and the complete regressors. Overall, JSRV copy number at euthanasia was significantly related to nodule disease development.
In the cohort receiving inoculum C, JSRV DNA was detected in 3 of 4 sheep at the time of euthanasia, although at significantly lower levels than those of the other 2 inocula groups. This may be the result of infection by lower levels of JSRV virus in inoculum C.
To investigate the presence of JSRV and verify that the tumors were indeed OPA induced by JSRV, we immunostained the lung tissue samples with an antibody specific for the Env protein of this virus. We found tumor-specific staining in samples from the 2 progressors (A4-P, B1-P) and 1 partial regressor (B2-PR); the other 2 partial regressors had insufficient tumor tissue available for JSRV IHC. Thus, of the 3 animals with evident tumor on the IHC sections, all expressed JSRV Env. However, the staining intensity differed between nodular disease development groups: On a staining-intensity scale ranging from weak to intermediate, moderate, and strong, the 2 progressors showed strong immunolabeling whereas the partial regressor only showed moderate immunolabeling (Fig. 1e, f). In all animals, this staining was most intense at the apical membrane of tumor cells; it was homogeneous in the cytoplasm of the tumor cells; and it was not observed in the nuclei.
OvLV
At 2 weeks PI, OvLV antibodies were detected in the serum of 1 animal (B4-NN). This lamb was subsequently euthanized to prevent OvLV transmission to the other 11 animals. However, by 2 months PI, 10 of the remaining 11 sheep had seroconverted, and at euthanasia all animals were seropositive for antibodies against OvLV. Seroconversion likely was due to the presence of OvLV in the virus preparations used to infect the animals and not to preexisting OvLV infection.
Lymphocyte Infiltration and CD3 Immunohistochemistry
We observed lymphocytic infiltration within the lung parenchyma as well as within the tumors. For the intratumoral lymphocyte infiltration (scored on a scale from 0 to 5), 1 partial regressor (A1-PR) with neoplasms < 500 µm in diameter showed only minimal lymphocyte infiltration (score of 1); however, the 2 progressors exhibited marked lymphocyte infiltration (score of 4) and the other 2 partial regressors exhibited severe lymphocyte infiltration (score of 5). Thus, infiltration of lymphocytes within the neoplasms was not specific to a nodular disease development group. This feature was also noted in the archival sections from 22 JSRV-infected sheep with confirmed OPA and no documented regression.
Next, we sought to further characterize the lung parenchyma and intratumoral lymphocytes by IHC staining of the ovine lung tissue samples with an antibody specific for CD3, a pan–T cell marker. There were no obvious differences in lung parenchyma CD3+ T-cell infiltration among nodule disease development groups, although the sheep with the greatest CD3+ infiltrate at euthanasia did not develop nodules during the study. Of the 3 sheep with tumor in the IHC sections, the partial regressor had more intratumoral CD3+ lymphocytes than did both progressors (Fig. 3a, b). In fact, when intratumoral CD3+ cells were quantified with image analysis software, it revealed that more than 40% of the tissue surface area examined comprised CD3+ T cells in the partial regressor, whereas in both progressors, CD3+ T cells constituted about 7% of the tissue surface area (Figs. 3c, d, 4).
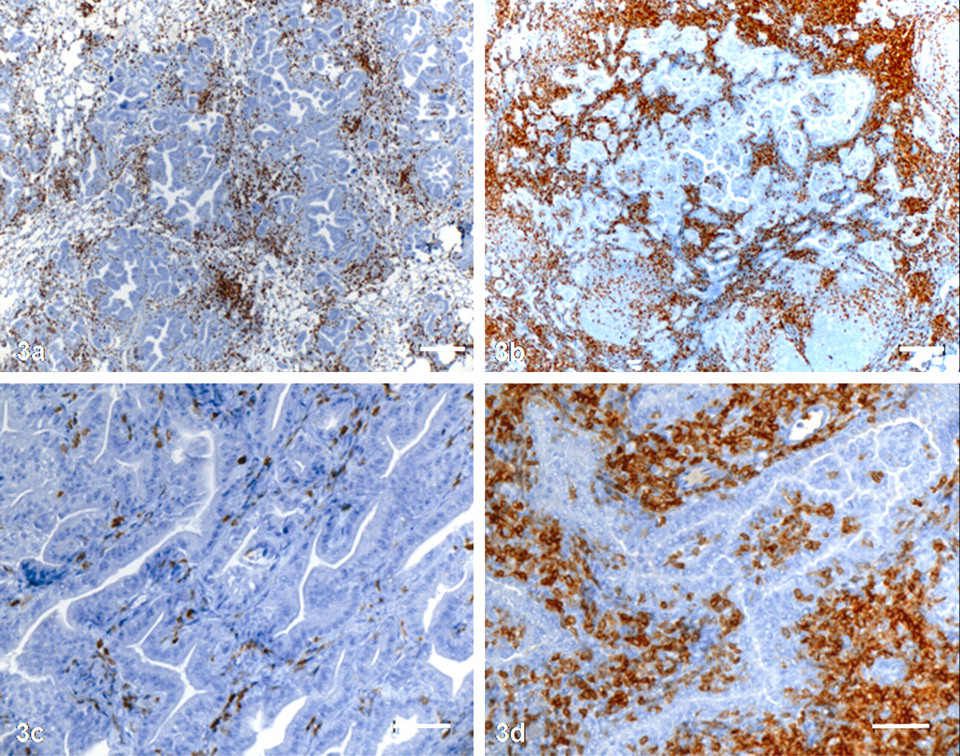
Lung. P, progressor; PR, partial regressor.
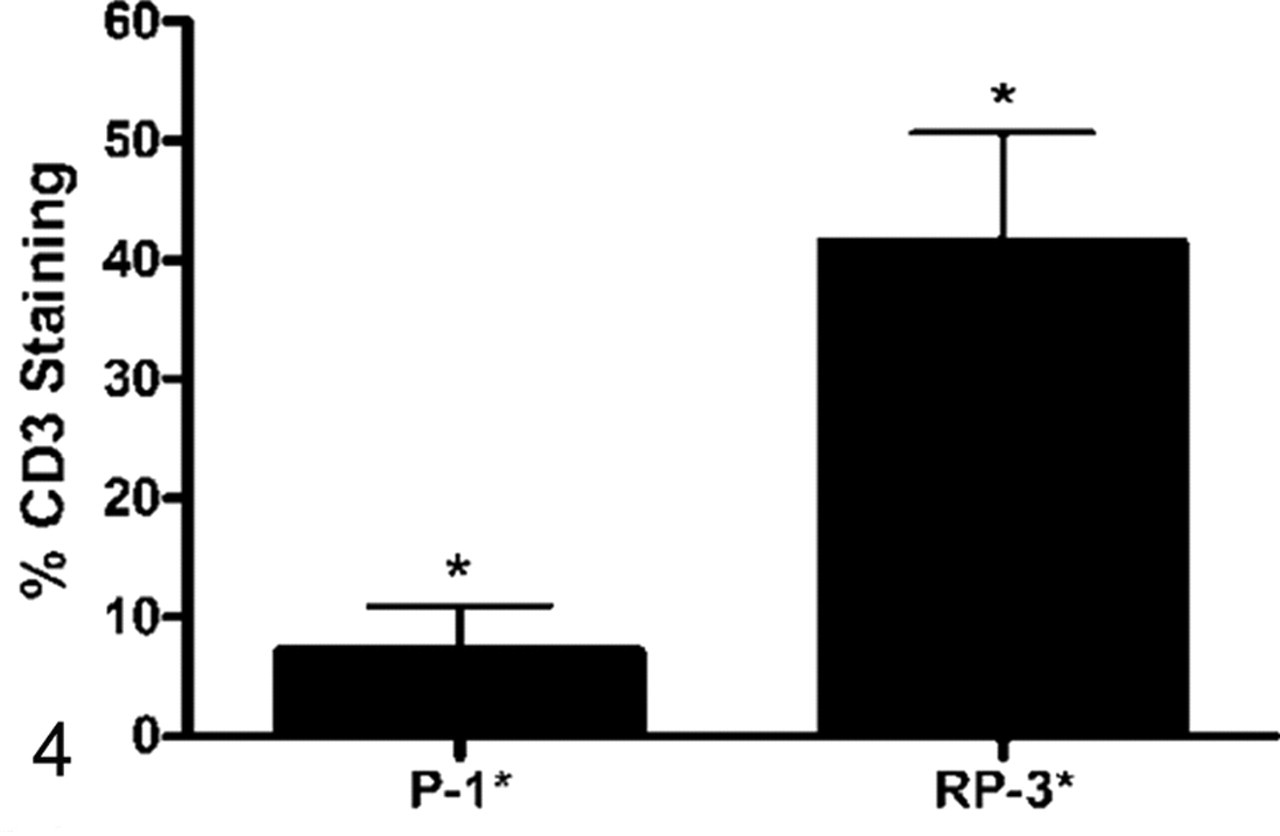
The percentage of tissue surface area expressing CD3 in sheep Nos. A4-P and B2-PR. P, progressor; PR, partial regressor. Columns show means ± SE of 5 independent images analyses. Asterisks (*) indicate a significant difference (P < .001).
Humoral Antibody Response Against the JSRV Envelope Protein
We tested for circulating antibodies against JSRV Env by measuring the ability of sheep serum to neutralize the infectivity of a retroviral vector bearing JSRV Env (LAPSN[PJ4]) 47 (Table 3). Mice bearing tumors induced by expression of JSRV Env can develop high neutralizing antibody titers (3,000) against this vector, 62 showing that the assay can readily detect such antibodies.
Titers of Neutralizing Antibodies in Serum Directed Against the Jaagsiekte Sheep Retrovirus Envelope Protein
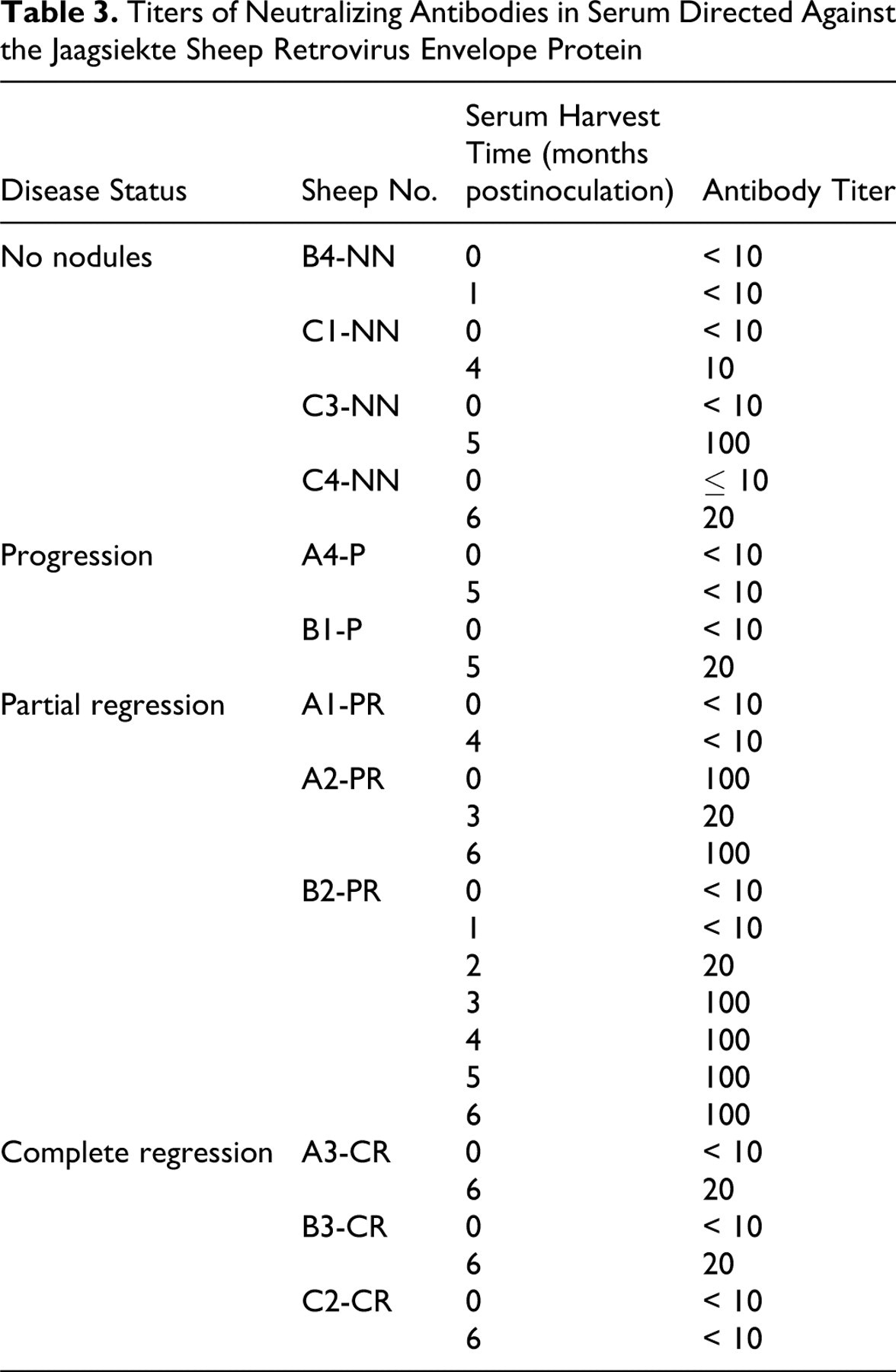
For 11 of 12 preinoculation serum samples, the antibody titer was low (≤ 10), but for sheep No. A2-PR, the titer was 100 for the preinoculation serum sample. Perplexed by this relatively high antibody titer against JSRV Env in the serum of a neonate, we tested the dam of this lamb and found that her serum had a neutralizing antibody titer of 100 as well (data not shown). These results suggest maternal transfer of the anti-JSRV Env antibodies from the dam to the lamb, most likely via colostrum. We tested the serum of sheep No. A2-PR again at 3 months PI and found that the titer was reduced to 20, consistent with time-dependent degradation of maternal immunoglobin G antibodies and not antibody production by the lamb. However, at 6 months PI, the antibody titer had increased to 100, indicating that this animal was independently making antibodies against the JSRV Env.
Interestingly, at 4 and 6 months PI, partial regression of nodules visible by CT was observed in sheep No. A2-PR. Additionally, another partial regressor (B2-PR) produced relatively high titers of anti-JSRV Env antibodies (titer of 100) during months 3 to 6, which coincided with nodule clearance. During this time, dramatic nodule regression was observed, specifically at 5 and 6 months PI. As for the other partial regressor (A1-PR), nodules regressed much earlier, at 2 months PI, and then progressed until euthanasia, at 4 months PI. At this time, the antibody titer was < 10, indicating little or no humoral immune response during nodule progression toward the end of the study. Similarly, serum antibody titers were low (≤ 20) at euthanasia for the two animals with nodular disease progression (A4-P, B1-P), again suggesting an inadequate immune response unable to combat JSRV-induced tumors.
Regarding the 3 complete regressors, nodule clearance occurred early (2 months PI) in 2 (B3-CR, C2-CR); at euthanasia, 4 months later, these sheep did not have appreciable antibody titers (≤ 20). As for the other complete regressor (A3-CR), nodules were apparent at 3 months PI and were cleared by the next CT scan, at 6 months PI, when the antibody titer was 20. However, because of the 3-month gap in CT scanning, it is unclear when nodule regression occurred; therefore, we cannot make an obvious association with anti-JSRV Env antibody titer in this animal.
Of the 4 sheep with no nodules visible by CT during the study, 3 (B4-NN, C1-NN, C4-NN) had low to undetectable antibody titers at euthanasia. In contrast, sheep No. C3-NN had a relatively high titer of 100 at this time, suggesting an active humoral immune response against JSRV that may have prevented tumor development.
Mutational Profiling of EGFR, KRAS, and p53
For mutational profiling, we used tissue samples from the 12 sheep involved with the CT imaging study, as well as additional archival samples from previous studies. 17,50,51
We sequenced the TK domain of the EGFR gene isolated from lung tumor DNA of JSRV-positive sheep and from lung tissue DNA of JSRV-negative sheep and found a total of 7 heterozygous single-base substitutions (Table 4). However, these substitutions were not exclusive to JSRV-positive sheep; that is, 4 of the 7 were also seen in JSRV-negative sheep. All 7 substitutions found in exons 18–21 of the EGFR gene were silent substitutions and did not alter the amino acid sequence of the EGFR protein; also, most substitutions were present in JSRV-infected sheep and uninfected sheep.
Summary of Heterozygous Single-Base Substitutions Found in Ovine Epidermal Growth Factor Receptor Tyrosine Kinase Domain Nucleotide Sequences a
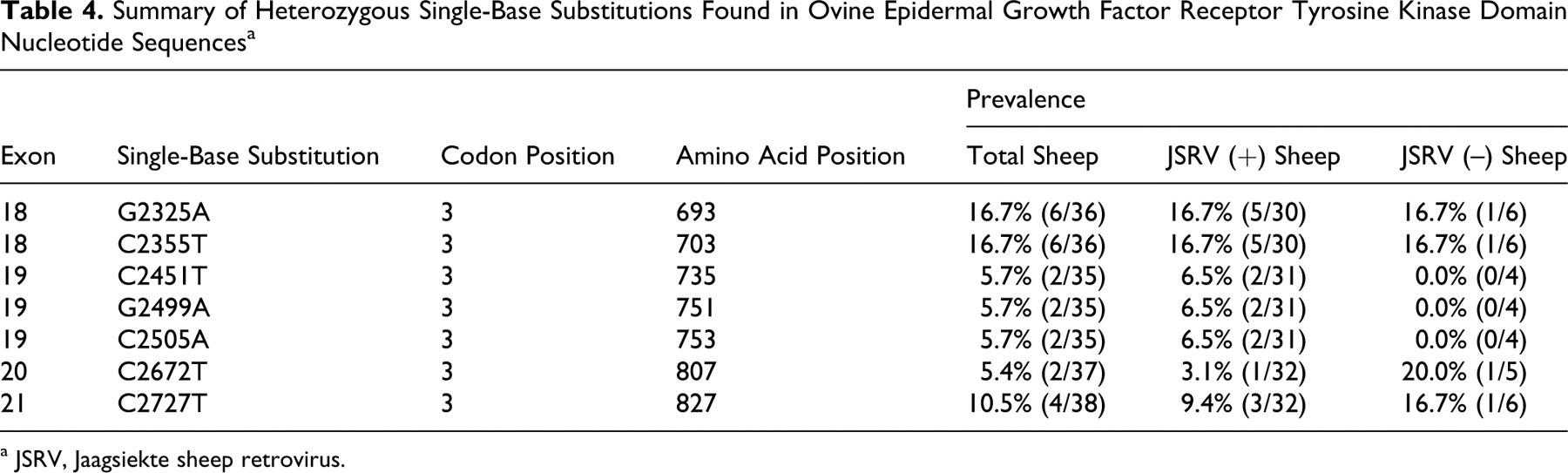
a JSRV, Jaagsiekte sheep retrovirus.
Additionally, we sequenced the interior of KRAS exon 1 isolated from lung tumor DNA. The first 27 and last 18 nucleotides of KRAS exon 1 were unreadable. For nucleotides 28 through 93, we found no variants in any of the samples.
Last, we sequenced the DNA-binding domain (exons 5–8) of p53 isolated from lung tumor DNA. In all 4 exons sequenced, we found no nucleotide alterations. Thus, p53 DNA-binding domain mutations do not appear to be important for JSRV-induced OPA.
Discussion
This is the first report describing the CT appearance of OPA and OPP disease development after inoculation of neonatal lambs with JSRV and OvLV. Our results demonstrate that CT is a suitable imaging modality for visualizing OPA disease development and can detect pulmonary abnormalities before the onset of clinical signs. However, a definitive diagnosis of OPA cannot be made using CT alone.
The CT appearance in conjunction with histopathological evidence at euthanasia indicates that the nodules visible by CT were indicative of OPA and not OPP. Additionally, the CT presentation of OPP has been detailed using high-resolution CT; in 5 experimentally infected lambs, no nodules were seen at 3 months PI. 7 Thus, nodules are not associated with early stages of OvLV infection, but the DLP revealed by CT scan is characteristic of infection by OvLV. 7 Thus, we conclude that the nodules observed in this study likely represent tumors induced by JSRV. In a human study assessing the high-resolution CT appearance of different types of pneumonia, nodules were seen in 78% of patients with viral pneumonias; all nodules were centrilobar and no random nodules were detected. 49 Also, in humans, interstitial pneumonias caused by viruses are associated with a reticulonodular pattern. 21,49 In our study, nodules were random, as opposed to centrilobar, and a reticulonodular pattern was not evident, again suggesting that the nodules were OPA and not pneumonic. Histopathological evaluation revealed that the 5 sheep with nodules evident on CT at euthanasia did indeed have tumors characteristic of OPA. Moreover, these 5 animals were from the cohorts receiving inoculum A or B; no sheep receiving inoculum C (with lower titers of JSRV) had nodules visible by CT or histopathologically discernable tumor at euthanasia. Of the 3 animals with tumor in the IHC sections, all expressed JSRV Env, indicating that the tumor nodules were indeed OPA induced by JSRV.
Unexpectedly, 6 of 8 animals with nodules visible by CT demonstrated either partial or complete regression of nodular disease during the course of the study. In retrospect, a detailed histopathologic correlation of the CT nodules throughout the study would have been ideal given the apparent tumor regression; however, for reasons relative to the overall mission of the study, serial biopsies or sacrifices were not practical. At euthanasia, nodular disease development was found to be associated with JSRV DNA copy number in the lung tissue (tumor tissue, if available): Sheep with progressive disease had the highest levels of JSRV DNA, whereas sheep showing complete regression of visible nodules had the lowest levels of JSRV DNA. Additionally, expression of the JSRV Env antigen was more intense in the progressors than in the partial regressor at the time of necropsy. Thus, these findings suggest a correlation between the level of JSRV expression and severity of lesions.
Nodular disease development was also found to be associated with CD3+ T-cell infiltration. The sheep with the greatest CD3+ T-cell infiltration of the lung parenchyma did not develop nodules during the study, and the partial regressor that had tumor on the IHC section had significantly more intratumoral CD3+ cells than did either of the progressors. These results suggest a role for a T cell–mediated immune response against JSRV and/or OPA in the spontaneous regression of lung cancer seen in our study, although these data should be interpreted with caution because of the potential impact of OPP on T-cell responses in the lungs of these cases.
The sheep with the greatest CD3+ lung parenchyma T-cell infiltration also had a relatively high anti-JSRV Env antibody titer; additionally, 2 partial regressors produced relatively high antibody titers that coincided with nodule clearance. These results suggest an association between an immune response against JSRV and partial tumor regression, although the serum-neutralizing activity was low (maximum titer of 100) compared to that observed in mice expressing JSRV Env in their lungs (titer of 3,000). 62
Stimulation of the host immune response is often implicated in the spontaneous regression of cancer. In human cancer cases, tumor-infiltrating lymphocytes have been shown to inhibit tumor growth and are associated with improved prognoses in melanoma, 11 breast cancer, 36 colorectal cancer, 4,18,27,38 esophageal cancer, 54 and ovarian cancer. 53,64 In a recent study, the density of T cells infiltrating colorectal tumors was a better predictor of survival than that of traditional staging based on tumor size and spread. 22 Additionally, in primary lung cancers, intratumoral infiltration of CD3+ T cells has been associated with a better patient outcome. 28
Numerous insults could act as stimulants to the immune system and thus cause regression of cancer, including trauma, infectious agents, and tumor cells themselves. 12 Thus, the spontaneous regression of JSRV-induced OPA observed in our sheep could be the result of the tumoricidal activity of cytotoxic T lymphocytes in response to cell surface–expressed JSRV or tumor antigens. Conversely, it has been postulated that the lymphocyte influxes observed in the lungs of OPA-affected sheep are due to concurrent infections, such as OvLV, and not JSRV or tumor antigens. 59 Accordingly, it is also possible that the host does not elicit an immune response against JSRV or OPA and that the lymphocyte influx, including CD3+ T cells, is the result of immune stimulation by OvLV, which might nevertheless have a role in OPA tumor rejection. In human malignancies, there are cases of cancer in which spontaneous regression was closely related to infections, such as measles, viral hepatitis, herpes zoster, and chickenpox. 6,19,24,29,61
In conclusion, we determined that JSRV-induced OPA can be detected early, before the onset of clinical signs, and that the cancer development could be monitored with minimal invasion using CT. Interestingly, the most common outcome seen in our study was that of spontaneous regression of OPA, possibly mediated by the immune system. We have not determined if the T-cell tumoricidal activity, if present, is directed toward JSRV, OPA tumor antigens, or concurrent OvLV infection, and further studies are warranted to investigate the basis for spontaneous regression of OPA. In addition, we found that OPA tumors do not harbor genetic mutations in the TK domain of EGFR, KRAS codons 12 and 13, or the DNA-binding domain of p53 and therefore are not genetically similar to human lung adenocarcinomas that contain these mutations. The mechanism of JSRV Env–induced oncogenesis is still uncertain and further elucidation of the means by which this virus acts as a carcinogen will improve our understanding of OPA and human lung cancer.
Footnotes
Notes
Acknowledgements
We are grateful to Alexandra DeLille, Thomas E. Allen, Karamjeet Pandher, Jeanette V. Bishop, Dennis J. Madden, and Laura S. Chubb for all of their help and guidance with this project.
