Abstract
The European brown hare (Lepus europaeus) plays an important role in the ecology of tularemia, and it may serve as a significant source of human infection. The aim of the present study was to examine the lesions induced by Francisella tularensis in 50 cases of naturally infected seropositive European brown hares. Gross pathological examination revealed scant to numerous grayish-white foci with diameters of 0.1 to 1.0 cm in single organs (24 cases) or multiple organs (20 cases) in 44 of 50 cases (88%). These lesions proved to be areas of granulomatous inflammation, frequently encompassing necrosis. F tularensis antigen was detected with immunohistochemistry in 46 of 50 cases (92%), whereas F tularensis ssp holarctica was isolated by culture and identified by polymerase chain reaction from 35 of 50 cases (70%). Infection by the respiratory route is suggested by the presence of the tissue lesions in thoracic organs of 44 of 50 cases (88%). These results emphasize the importance of the European brown hare as a reservoir of tularemia.
Francisella tularensis, the etiological agent of the life-threatening zoonotic disease tularemia, has been known since 1912, when McCoy and Chapin identified it during an outbreak of a “plague-like” disease of ground squirrels in Tulare County, California. 13 Three subspecies of F tularensis are recognized: the highly virulent F tularensis ssp tularensis and the moderately virulent F tularensis ssp holarctica and F tularensis ssp mediasiatica. 3 F tularensis ssp tularensis has been almost exclusively found in North America; however, several isolates of this subspecies were obtained in the 1980s from ticks in Slovakia. 7 F tularensis ssp holarctica is found throughout the northern hemisphere, whereas the distribution of F tularensis ssp mediasiatica is restricted to Central Asia. 10
The broad host range of F tularensis comprises primarily mammalian species and, to a lesser extent, birds, amphibians, and fish and even invertebrates. 15 Lagomorphs, along with rodents, are generally known as the main reservoirs of F tularensis, whereas hematophagous arthropods have a substantial role in disease transmission. 4,9,10 In the New World, the cottontail rabbit (Sylvilagus spp), the black-tailed jackrabbit (Lepus californicus), and the snowshoe hare (Lepus americanus) are important in the ecology of tularemia. The European brown hare (Lepus europaeus), the mountain hare (Lepus timidus), and the Japanese hare (Lepus brachyurus) are the lagomorphs associated with tularemia in the Old World. 4 The European wild rabbit (Oryctolagus cuniculus) and, thus, the domestic rabbit are both relatively resistant to tularemia. 2 Human infections often result from injuries during the skinning of infected lagomorphs (eg, European brown hare) and bites from infected ticks. 16 To a lesser extent, individuals may become infected after inhaling the agent, eating inadequately cooked contaminated meat, or drinking water from public water supplies contaminated by F tularensis originating from infected carcasses. 1,5,6,10 F tularensis is a highly infectious agent; as few as 10 bacteria can infect animals and humans; and it has been placed on the list of class A biothreat agents. 23
The European brown hare is reported to be a common reservoir of tularemia, and its public health importance is accentuated by being one of the most significant European game species. 10,16,20,22 Despite the significant role of this lagomorph in the ecology of tularemia and its importance as a human infection source, no report has been published so far about the pathological and microbiological examination of naturally infected European brown hares. The aim of the present study was to identify the gross and histological lesions characteristic for F tularensis infection of European brown hares. This essential information can facilitate both the detection of tularemia in brown hares by hunters and the recognition and identification of this dangerous zoonotic disease by diagnosticians.
Materials and Methods
Samples: Postmortem Examination
European brown hares, shot at different locations in Hungary during several hunting events over two winter hunting seasons (2007–2008 and 2008–2009), were screened by the slide agglutination test using stained bacteria (Bioveta Inc, Ivanovice na Hané, Czech Republic) and whole blood. Forty-seven animals (16 males, 31 females; 32 adults, 15 juveniles) were found to be seropositive and were thus used for laboratory examinations. Carcasses of 3 dead seropositive adult male hares submitted for diagnostic examination were included in the study, so 50 seropositive animals were examined in total. Tissue samples of 20 seronegative hares were collected and thus served as negative controls. All carcasses were necropsied under appropriate biosafety conditions at the Central Agriculture Office Veterinary Diagnostic Directorate, Budapest, Hungary. Animals were categorized as same-year juveniles and older, based on the so-called Stroh mark. 21 The body condition was estimated with a simplified categorical (good, moderate, weak) version of the kidney fat index. 21 Tissue samples were collected for histology and fixed in 10% buffered formalin: from 50 animals, heart, pericardium, lung, liver, spleen, kidney, small and large intestine, and bone marrow; from 19 animals, testicle and epididymis; from 31 animals, ovarium and mammary gland; and from 35 animals, mediastinal lymph node. The same tissue samples of 20 seronegative hares served as negative controls. Four-micron-thick sections of formalin-fixed, paraffin-embedded tissue samples were stained with hematoxylin and eosin and examined by light microscopy.
Immunohistochemistry
Immunohistochemistry (IHC) was applied for the demonstration of F tularensis lipopolysaccharide antigen in tissue sections. Briefly, after deparaffinization and antigen retrieval (in a microwave oven at 750 W for 20 minutes in citrate buffer, pH 6.0), the sections were incubated in 3% H2O2 solution for 10 minutes and then in a 2% solution of skimmed milk powder for 20 minutes. The samples were incubated overnight at 37°C with a 1:6,000 dilution of a F tularensis lipopolysaccharide-specific mouse monoclonal antibody (clones FB11 and T14, MAB8267, Chemicon International Inc, Southhampton, UK). Antibody binding was detected by a horseradish peroxidase–labeled polymer (EnVision+ Kit, Dako, Glostrup, Denmark). A serial section incubated with phosphate buffer solution was used as a negative control.
Isolation and Identification of Bacteria
F tularensis strains were isolated from tissue samples on a modified Francis agar (chocolate agar plate containing 1% glucose and 0.1% cysteine) after a mouse passage (mouse, approximately 20 g, Naval Medical Research Institute, Bethesda, MD). Isolates were identified with the MicroLog MicroStation System, GN2 Microplate (Biolog Inc, Hayward, CA) and a 16S rRNA gene based polymerase chain reaction method as described previously. 8 All procedures were performed under biosafety level 3 conditions.
Statistical Methods
Formerly described formulas were used to calculate the specificity, sensitivity, and predictive value of diagnostic tests. 14
Results
Gross Pathological Lesions: General Remarks
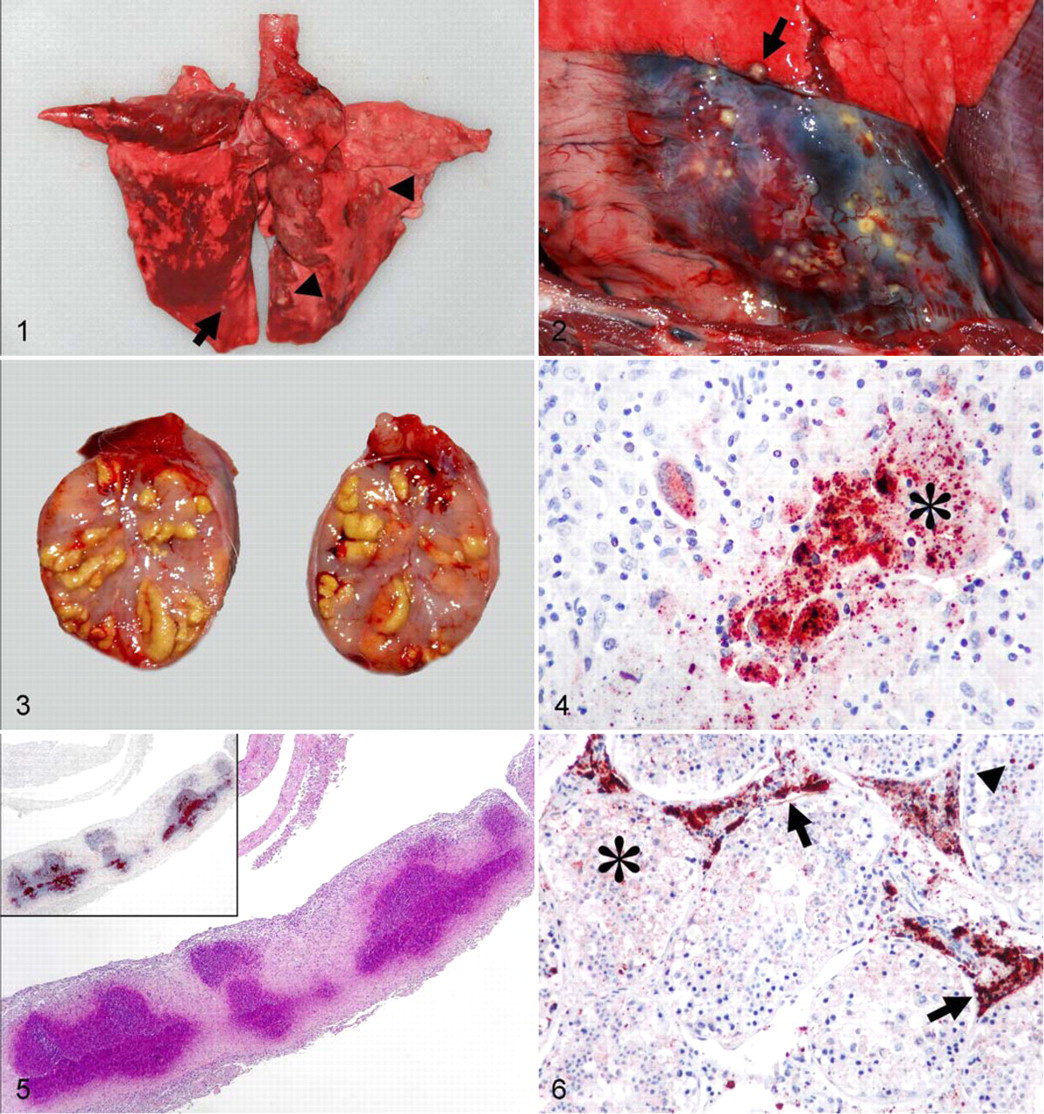
The body condition of the seropositive animals was good (12 of 50 cases, 24%), moderate (11 cases, 22%), or weak (27 cases, 54%). The main gross pathological feature of the disease was the presence of few to numerous necrotic foci in different organs in 44 cases (88%) (Table 1 ). Necrotic foci were evident in single organs (24 cases), 2 organs (13 cases), and multiple organs (7 cases). Foci were randomly distributed, well demarcated, round, white, grayish-white, or yellowish-white, with diameters of 0.1 to 0.5 cm, and they were often surrounded by a dark hyperemic ring. The foci in the lungs and kidneys coalesced to nodules of up to 1 cm in diameter in several cases. Foci were frequently observed on the serosal surface of the lungs (Fig. 1) and kidneys. These foci and the foci in the pericardium (Fig. 2) were raised, dome shaped, or flattened; furthermore, their serosal surface was dry and granular, so the lesion corresponded to focal serositis. Affected testicles were enlarged, and several yellowish-white foci or nodules were evident on their cut surface (Fig. 3). Low numbers of foci were seen in the bone marrow and the mammary gland. Two of the 3 natural mortality cases were adult male hares in weak body condition. They had swollen lungs, kidneys, livers, and spleens. Petechial hemorrhages were observed in the lungs, and large numbers of grayish-white or yellowish-white foci were seen in both lungs and pericardia, as well as in the testicles of a single case. No lesions resembling tularemia or other infection were observed in seronegative hares.
Cases With Gross Pathological Lesions in the Organs of Seropositive European Brown Hares (n = 50)

Histological Examination
The foci identified by gross pathological examination corresponded to focal or coalescing granulomatous inflammation, which completely replaced the normal tissue structure of the affected organs. These foci were randomly distributed in the organs, and serosal membranes were frequently involved. Macrophages were the dominant constituent cell type, but other cells were found occasionally, including lymphocytes, heterophil granulocytes, multinucleated giant cells, and fibrocytes (Fig. 4). Focal or multifocal necrosis was often observed in the center of these lesions (Fig. 5).
Granulomatous inflammation was found with microscopic examination but not with gross pathological examination in the mediastinal lymph nodes of 12 of 35 cases, in the liver and spleen of 2 of 50 cases, and in the bone marrow of 1 of 50 cases. Two of 50 carcasses (4%) showed no visible gross lesions, but they did exhibit microscopic changes indicative of the infection. Lesions were not found in 4 of 50 (8%) seropositive animals and 20 seronegative control animals.
In several cases, the coalescing granulomatous inflammation in the lungs contained no or only minor necrotic areas. Remnants of necrotic blood vessels and bronchioli were occasionally observed in affected areas. Inflammatory exudate and debris were found in several bronchioli not adjacent to the areas of granulomatous inflammation; alveolar edema was prominent in these areas. The foci in the pericardium showed excessive central necrosis, and fibrinous inflammation was obvious on adjacent serosal surfaces (Fig. 5). Multiple foci of granulomatous inflammation were found in the cortex and medulla of affected kidneys. The entrapped tubuli found in these areas were partly or completely necrotic and filled with necrotic debris. Multifocal granulomatous inflammation was found in the suburothelial area of the renal pelvis, and adjacent inflammatory necrotic exudate was present in the lumen. In the testis and epididymis, the granulomatous inflammation expanded the intertubular connective tissue lining over large areas. Several tubuli were partly or completely necrotic, and their lumens were filled with necrotic inflammatory exudate (Fig. 6). Foci of granulomatous inflammation with central necrosis were found in the liver, bone marrow, mammary gland, spleen, and mediastinal lymph nodes (Table 2 ).
Occurrence of Histological Lesions and Francisella tularensis Lipopolysaccharide Antigen in the Organs of Seropositive European Brown Hares (n = 50)

a One of them natural mortality case.
b Natural mortality case.
Histological lesions were essentially the same in the 3 natural mortality cases and in the shot animals. However, in 2 of the former cases, numerous necrotic foci were evenly distributed in 4 or 5 organs (Table 2). No lesions were found in heart muscle, ovaries, and the intestines.
Immunohistochemical Examination
F tularensis antigen was detected in all cases presenting tissue lesions (46 of 50, 92%) (Table 2). The bacterial antigen revealed granular or amorphous staining and was clearly associated with histological lesions. In several cases, F tularensis antigen occurred in small quantities or was absent in some areas of granulomatous inflammation. In all cases, the bacterial antigen was observed in large amounts, mostly extracellularly, in foci of tissue necrosis. Intracellular F tularensis was found in macrophages and giant cells in the majority of cases and less frequently in other cell types, such as tubular epithelial cells of the kidney, testis, and epididymis; epithelial cells of the renal pelvis; hepatocytes; and bronchiolar epithelial cells. Extracellular and intracellular labeling was present in the inflammatory exudate situated in the lumen of airways, renal tubuli and pelvis, and tubuli of the testis and epididymis. Additionally, in the 2 severely affected natural mortality cases, F tularensis antigen was found in histologically unaffected tissue areas of lungs, livers, and spleens. Bacterial antigen was visualized as fine intracellular granular structure within intact alveolar epithelial cells, hepatocytes, and intravascular macrophages whereas extracellular labeling was present in blood vessels. Seronegative animals and the negative control tissue sections remained negative. Four seropositive animals were negative with IHC. Compared to the IHC assay, slide agglutination test exhibited 100% sensitivity but only 83% specificity. The predictive value of a negative test was 100%, whereas the predictive value of a positive test was 92%.
Isolation and Identification of Bacteria
Bacteria showing F tularensis–like colony morphology were isolated in pure culture from 35 of 50 cases (70%). All these cases were positive with IHC. Grayish-white 1-mm colonies could be seen after 4 days of incubation. These bacteria were small (< 1 μm) coccoid, nonmotile, Gram-negative rods. They were oxidase negative, with weak positive catalase activity, and they were not able to utilize glycerol. The strains were identified as F tularensis with the MicroLog MicroStation System, and because they were unable to use glycerol as a sole carbon source, they were further characterized as F tularensis ssp holarctica. This diagnosis was confirmed by polymerase chain reaction and sequencing of the amplified partial 16S rRNA gene. The resulting sequences showed 100% identity with the F tularensis ssp holarctica (accession no. CP000803) and only 99% identity with the F tularensis ssp tularensis (accession no. CP000608) 16S rRNA sequences deposited in the GenBank. When compared with the IHC assay, bacterial culture showed 100% specificity but only 76.1% sensitivity. The predictive value of a negative test was 68.6%, and the predictive value of a positive test was 100%.
Discussion
This is the first report describing the gross and histopathological lesions of a large number of European brown hares naturally infected with F tularensis ssp holarctica and the subsequent results of the IHC examination and bacterial culture. Only a single case report of a European brown hare acutely infected with F tularensis is available in the literature, but no histological or IHC examinations were applied in that study. 18 Gross pathological lesions suggestive of the infection were observed in 44 of the 50 (88%) seropositive hares in the present study. These results indicate the excellent applicability of gross pathological examination as an initial screening method for the diagnosis of F tularensis in European brown hares. Numerous macroscopically visible grayish-white foci and nodules with a diameter of 0.1 to 1.0 cm in single or multiple organs should warn hunters and diagnosticians to handle carcasses with care and thus help prevent subsequent human infections. The most often affected organs were lungs, pericardia, and kidneys, which is in contrast to reports in rodents and other lagomorphs wherein liver and spleen were found to be the primary targets. 4,16 Overall, histological lesions resembled the subacute tularemia reported in the experimentally infected domestic rabbit. 11
Given the several infections that can produce similar lesions (eg, Brucella suis or Yersinia pseudotuberculosis infections), additional laboratory tests are necessary for final diagnosis of tularemia. 16 According to the present study, IHC was the most sensitive and specific method, in comparison with bacterial culture and serology. The advantage of IHC over serology and bacterial culture is that the infectivity of tissue samples is eliminated by the time of laboratory examination owing to formalin fixation. 12 Several authors have shown the IHC assay to be a useful and sensitive method for the detection of F tularensis in domestic and wild animals. 23 –25 The F tularensis lipopolysaccharide-specific reagent used in the present study was a blend of two clones of mouse monoclonal antibodies, which ensure the detection of all subspecies of F tularensis. The reagent cross-reacted with neither B suis nor Y pseudotuberculosis (data not shown), which can both cause infection in hares. In the European brown hare, F tularensis antigen was always associated with tissue lesions, especially with necrotic foci.
In areas of granulomatous inflammation, bacterial antigen was present in low quantities, or it was totally absent, which may be explained by the successful elimination of bacteria by the host’s immune system. Two of the 3 natural mortality cases presenting weak body condition showed severe disseminated lesions. Bacterial antigen was identified in affected tissues within lesions and within blood vessels in unaffected tissues. Similar findings were reported in experimentally infected African green monkeys, but no explanation of this phenomenon was presented. 23 We hypothesize that these 2 hares died with F tularensis septicemia, resulting in occurrence of bacteria within blood vessel of unaffected tissues.
Serological methods are used for the diagnosis of tularemia in humans, and they can be applied in animals. Serology has limited use in highly susceptible species of animals, which usually die before specific antibodies develop. 16 The slide agglutination test was found to be useful in the present study. It is simple and fast with 100% sensitivity and high specificity. F tularensis shares common antigens with Brucella spp, Yersinia spp, and Legionella spp, and subclinical, clinical, or previous infection with any of these organisms may be responsible for the false-positive results, 19 which may explain the occurrence of 4 seropositive cases with IHC-negative test results. However, F tularensis–induced tissue lesions may occur in small numbers and in only a few organs, so this may have prevented the recognition of these cases by IHC. In the present study, bacterial culture had low sensitivity compared with that of the IHC test; however, this method is necessary for the determination of species and type of F tularensis strain.
Previous experimental studies suggested that the European brown hare is moderately susceptible to F tularensis ssp holarctica–caused tularemia, compared with the mountain hare, and that it serves as a potential reservoir host of F tularensis ssp holarctica in Europe. 17 The present study supported this hypothesis in finding moderate to good body condition in 48% of IHC-positive animals (data not shown) and revealing the presence of subacute histological lesions in all cases, thereby indicating a protracted disease. The frequent lesions in thoracic organs (44 of 50 cases, 88%), lung, mediastinal lymph nodes, and pericardia suggest that the airborne route is the most common way of acquiring the infection in European brown hares.
In conclusion, the European brown hare is considered an important reservoir of F tularensis ssp holarctica. Macroscopically visible lesions can be found in almost all infected hare cases. The IHC assay for the diagnosis of F tularensis infection is sensitive and has a similar specificity as bacterial culture. Because the combination of plate agglutination and detection of gross pathological lesions has proven to be a sensitive tool for preliminary diagnosis of tularemia, we recommend the use of this method for the screening of F tularensis infection in brown hares.
Footnotes
The authors declared no potential conflicts of interest with respect to the authorship and of or publication of this article.
The authors received no financial support for the research and of or authorship of this article.
