Abstract
Alzheimer disease is a dementing disorder characterized pathologically by Aβ deposition, neurofibrillary tangles, and neuronal loss. Although aged animals of many species spontaneously develop Aβ deposits, only 2 species (chimpanzee and wolverine) have been reported to develop Aβ deposits and neurofibrillary tangles in the same individual. Here, the authors demonstrate the spontaneous occurrence of Aβ deposits and neurofibrillary tangles in captive cheetahs (Acinonyx jubatus). Among 22 cheetahs examined in this study, Aβ deposits were observed in 13. Immunostaining (AT8) revealed abnormal intracellular tau immunoreactivity in 10 of the cheetahs with Aβ deposits, and they were mainly distributed in the parahippocampal cortex and CA1 in a fashion similar to that in human patients with Alzheimer disease. Ultrastructurally, bundles of straight filaments filled the neuronal somata and axons, consistent with tangles. Interestingly, 2 of the cheetahs with the most severe abnormal tau immunoreactivity showed clinical cognitive dysfunction. The authors conclude that cheetahs spontaneously develop age-related neurodegenerative disease with pathologic changes similar to Alzheimer disease.
Alzheimer disease (AD) is a neurodegenerative disorder that causes progressive deterioration of cognitive function and is characterized pathologically by 3 histologic features: (1) cerebral atrophy with neuronal loss; (2) senile plaques, consisting of extracellular Aβ deposits; and (3) neurofibrillary tangles (NFTs), which are twisted aggregates of argyrophilic cytoskeletal proteins within neuronal cell bodies and processes, consisting mainly of abnormally phosphorylated tau. 44 AD is also classified as one of the tauopathies, a group of heterogeneous dementias and cognitive disorders characterized neuropathologically by prominent intracellular accumulations of abnormal tau filaments. 27
It is generally believed that AD is a human-specific disease, since there has not been any species that spontaneously develops the disease. Hence, strains of transgenic mice have mainly been used as experimental models of AD, although these animals replicate only some aspects of the disease and do not represent its systemic pathologic features. Aβ deposits and accumulation of abnormally phosphorylated tau have been described in the brains of a variety of species. 6,17 –19 However, argyrophilic NFTs have been reported in few species, including the chimpanzee, 43 wolverine, 42 Asiatic brown bear, 7 sheep, and goat. 5 To our knowledge, the presence of Aβ deposits and NFTs in the same individual has been detected in only 1 wolverine and 1 chimpanzee. 42,43 Since both of these were only case reports and intracranial infarction or hemorrhage was present in each and may have been the primary lesion responsible for NFTs, it remains unclear whether both findings of AD (NFTs and Aβ deposits) spontaneously developed in these cases. Some researchers assume that AD is not present in animals because they do not live long enough to develop this slow-progressing disease. 33
The cheetah (Acinonyx jubatus) is a felid and well known as the fastest hunter of all land animals, with longevity of 17 years. 12 Cheetahs are unique in the extent to which they have lost polymorphism, and this genetic homogeneity has been suspected to be one of the causes of their poor health and reproductive failure in captivity. 35,36,38 In addition, these animals are susceptible to environmental stresses under captive conditions, which accounts for the occurrence of diseases in this species. 31
In Japan, systemic AA amyloidosis is an important cause of morbidity and mortality in cheetahs, and life expectancy is around 9 years. 51 However, the number of aged cheetahs has increased due to recent improvement in reproductive management and veterinary treatment. We found Aβ deposition, NFT formation, and cerebral atrophy (neuronal loss) in the brains of aged cheetahs, which correspond to the lesions observed in human AD. We report here the pathologic observation of a novel disease in cheetahs, which we think is a counterpart of human AD.
Materials and Methods
Animals
Twenty-two brains obtained from postmortem cheetahs examined from 2002 to 2007 were used. They were collected from 7 facilities (WA, HC, FS, YA, TT, AT, ST) in Japan and ranging in age from 10 months to 19 years. Six cheetahs were imported from Namibia and 5 from South Africa, while the others were bred in Japan. Individual sex, age, facility, place of birth, and existence of systemic AA amyloidosis are listed in Table 1. All procedures in this study were in accordance with the guidelines approved by the Animal Research Committee of Azabu University.
Profile of Cheetahs a
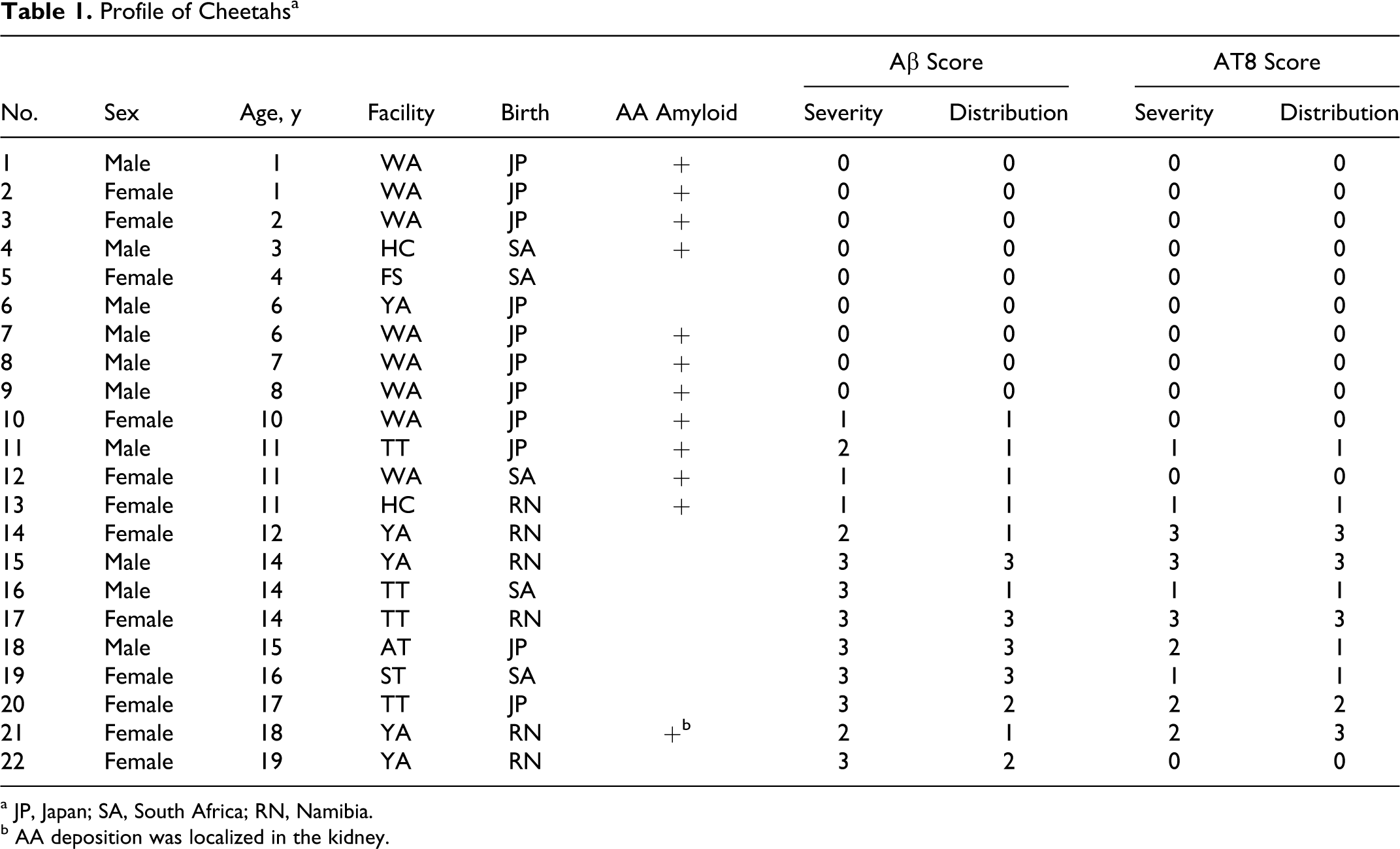
a JP, Japan; SA, South Africa; RN, Namibia.
b AA deposition was localized in the kidney.
Histopathology
The midportion of the cerebral hemispheres (including the cortex, hippocampus, parahippocampus, and thalamus) was fixed with 10% neutral buffered formalin for 24 to 48 hours and cut transversely. The slices were routinely embedded in paraffin wax and cut into 3- or 7-μm-thick sections (used for Congo red staining). These sections were stained with hematoxylin and eosin, Klüver-Barrera, Congo red, methenamine silver stain, and the Gallyas-Braak method with pretreatment with 0.3% potassium permanganate for 10 minutes, followed by 0.1% oxalic acid for 1 minute. 20
For immunohistochemistry, antibodies to Aβ, hyperphosphorylated tau, and ubiquitin were used to detect AD-related lesions. Additional antibodies used for other purposes are listed in Table 2.
Antibodies Used in This Study
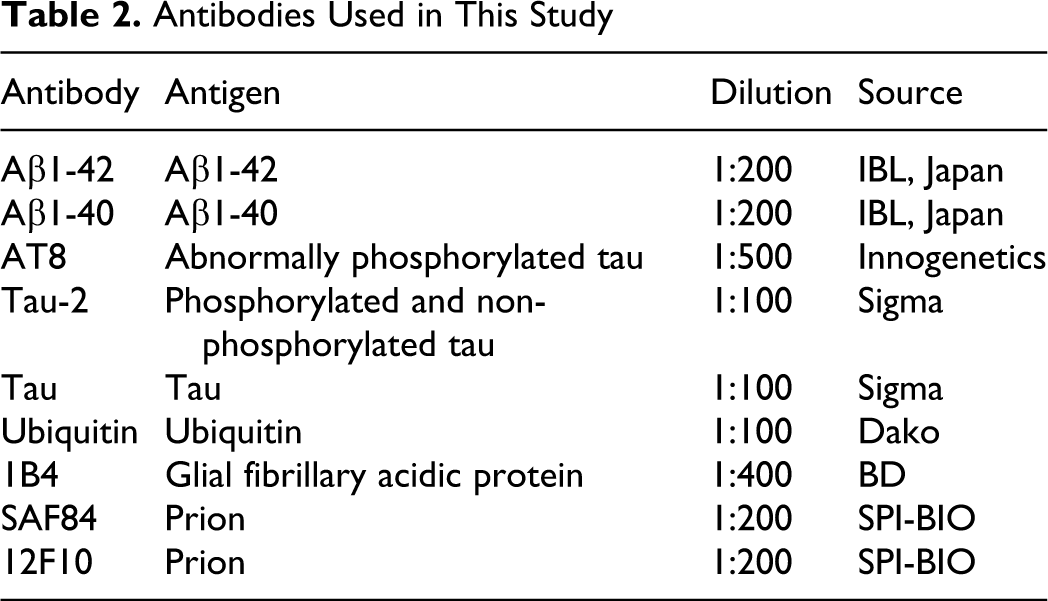
After deparaffinization and rehydration, 3-μm-thick sections were incubated in 1% hydrogen peroxide–methanol for 20 minutes to inactivate endogenous peroxidase. Samples were then pretreated with 100% formic acid for 10 minutes (for Aβ1-42, Aβ1-40), microwave irradiated at 90°C for 9 minutes in pH 6.0 citrate buffer (for AT8, GFAP), or autoclaved at 121°C for 5 minutes (for tau, tau-2, ubiquitin) to enhance antigenicity. Some rehydrated sections examined for PrPSC were pretreated with 98% formic acid and proteinase K, followed by 3% hydrogen peroxide and 100mM sodium hydrate (kindly performed by Dr Hiroyuki Okada, National Institute of Animal Health).
To block nonspecific immunoreactivity, the sections were then treated with 5 or 10% skim milk for 20 minutes. They were subsequently incubated overnight at 4°C with primary antibodies. After being washed in phosphate buffered saline, the sections were sequentially incubated with the peroxidase-conjugated secondary antibody Histofine-Simple Stain MAX-PO (MULTI; Nichirei, Tokyo, Japan) for 1 hour at room temperature. Immunolabeled antigen was visualized using diaminobenzidine. Negative controls were obtained by omitting the primary antibodies.
Electron Microscopic Analysis
To clarify the ultrastructural characteristics of NFTs observed in the cheetah brains, electron microscopic analysis was performed. A brain sample of case No. 17 was examined since it showed large number of NFTs with minimal postmortem change under light microscopic examination. Formalin-fixed cortex was cut into 1-mm cubes and postfixed in 1% osmium tetroxide in 0.1M cacodylate buffer (pH 7.2). The tissues were dehydrated in graded ethanols, cleared with propylene oxide, and embedded in Epon 812. One-micrometer sections were stained with toluidine blue. Ultrathin sections from selected areas were contrasted with uranyl acetate and lead citrate and examined with a Hitachi transmission electron microscope (H7500).
Scoring
The assessment of Aβ deposition and abnormally phophorylated tau were based on the severity and distribution of immunoreactive lesions visualized by Aβ1-42 and AT8. The severity of Aβ1-42 deposition was scored as 0 (none), 1 (few deposits are observed on close examination at a low magnification), 2 (multiple deposits are observed easily at a low magnification), and 3 (aggregation of Aβ immunoreactivity can be observed grossly); for distribution, 0 (none), 1 (present in parietal cortex or temporal cortex), 2 (present throughout the entire cerebral cortex), and 3 (present in the entire cerebral cortex and hippocampus).
A method of staging of neurofibrillary pathology based on immunohistochemical distribution and severity of AT8-positive tangles has recently been proposed. 1 In our study, the severities of abnormally phosphorylated tau were scored as 0–3 by using AT8: 0 (none), 1 (few AT8-positive cells were observed on close examination at a low magnification), 2 (multiple AT8-positive cells were observed easily at a low magnification), and 3 (AT8-positive aggregates are observed grossly); for distribution, 0 (none), 1 (present in the hippocampus or parahippocampal cortex), 2 (present throughout temporal cortex), and 3 (present throughout entire cerebral cortex). The scores of both Aβ1-42 and AT8 are listed in Table 1.
Results
On gross examination and basic histopathologic analyses, the brains of 2 cheetahs (case Nos. 14 and 15) showed severe atrophy of the cerebral cortex due to neuronal loss accompanied by astrocytosis (Figs. 1, 2). Further details of these cheetahs are mentioned later.
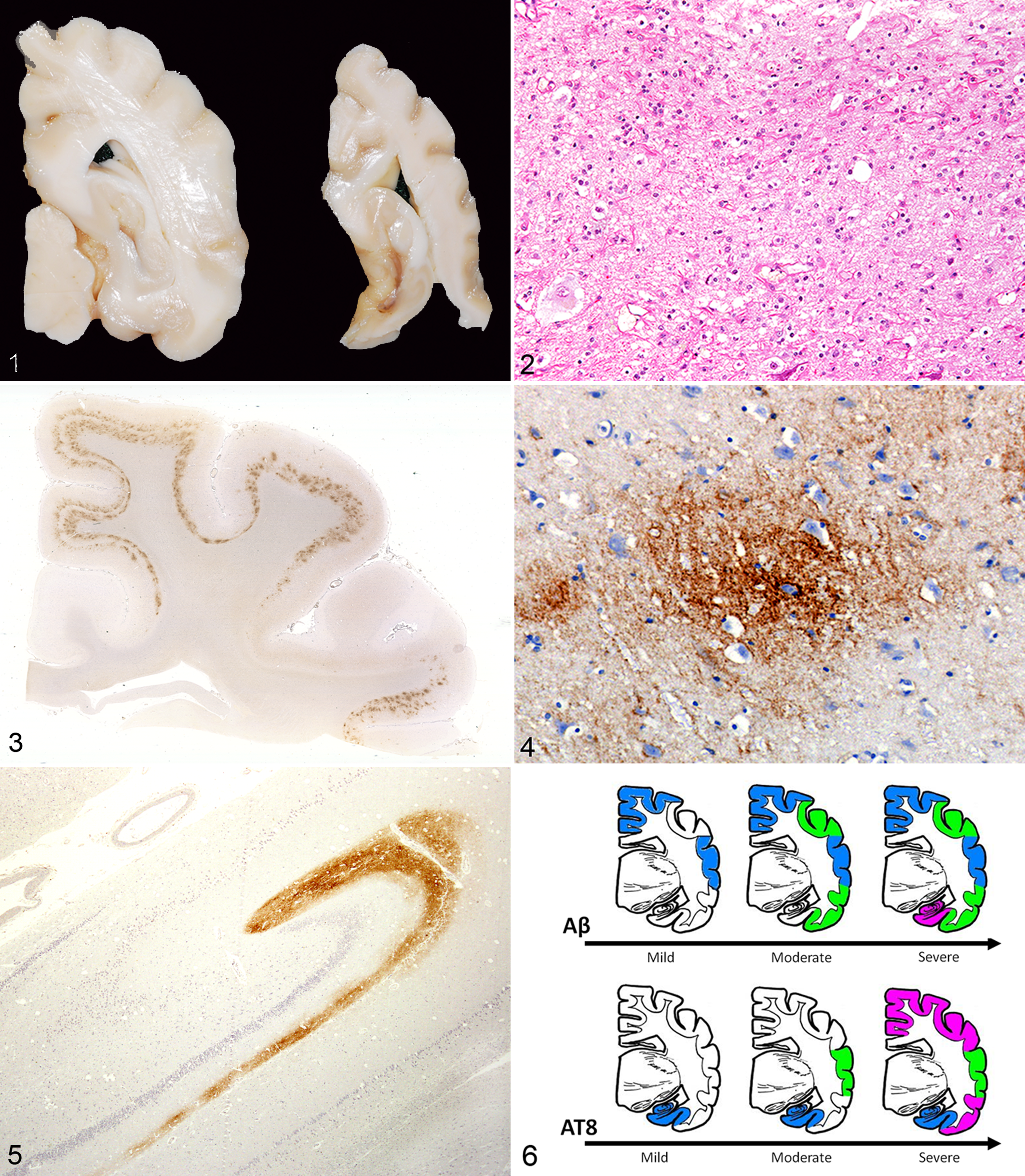
Cerebrum; case No. 14. Gross view of normal (left) and severely atrophied brain (case No. 14). Note the severely atrophied cerebral cortex (brainstem is missing in the right).
Cerebral cortex; No. 17. Intracellular argyrophilic neurofibrillary tangles within the perikaryon of neural cells. Modified Gallyas-Braak method.
Aβ deposits were observed in all 13 cheetahs that lived to over 10 years of age, with the use of anti-Aβ1-42 antibody (Table 1). Diffuse staining for extracellular Aβ deposits was found within layers III to VI of the cerebral cortex. These deposits were also slightly labeled by anti-Aβ1-40 antibody and typically seen in the parietal and temporal lobes as diffuse granular foci within the neuropil (Figs. 3, 4). All these deposits were negative for Congo red staining and methenamine silver staining.
In 5 of the 13 brains examined, diffuse clouds of Aβ-positive staining were observed in the outer molecular layer of the dentate gyrus (bandlike deposition), all from cheetahs over 14 years of age (case Nos. 15–19) (Fig. 5).
As shown in Figure 6, substantial differences in the distribution of Aβ deposits were found among aged cheetah brains. Mildly affected brains had few Aβ deposits distributed in the parietal and temporal cortex. In moderate cases, many deposits were observed throughout the cerebral cortex. Additional bandlike deposits in the dentate gyrus were observed in severely affected brains.
There were no Aβ immunoreactive meningeal vessels, and only a few capillary blood vessels immunopositive for both Aβ1-42 and Aβ1-40 were found in 2 aged cheetahs (case Nos. 14, 15).
Immunohistochemical analysis using antibodies to abnormally phosphorylated tau (AT8) revealed intracellular immunoreactivity in 10 of 13 cheetahs with Aβ deposits, all of which were over 11 years of age.
Abnormally phosphorylated tau was detected within neuronal cell bodies, dendrites, and the neuropil and was most abundant in the parahippocampal cortex and CA1 of the hippocampus. In some strongly affected cases, they were detected throughout the cerebral cortex. The initial locations of abnormally phosphorylated tau and Aβ deposits in mildly affected cases differed. Abnormally phosphorylated tau was observed in the hippocampal area, whereas Aβ deposits were observed in the parietal and temporal areas (Fig. 6).
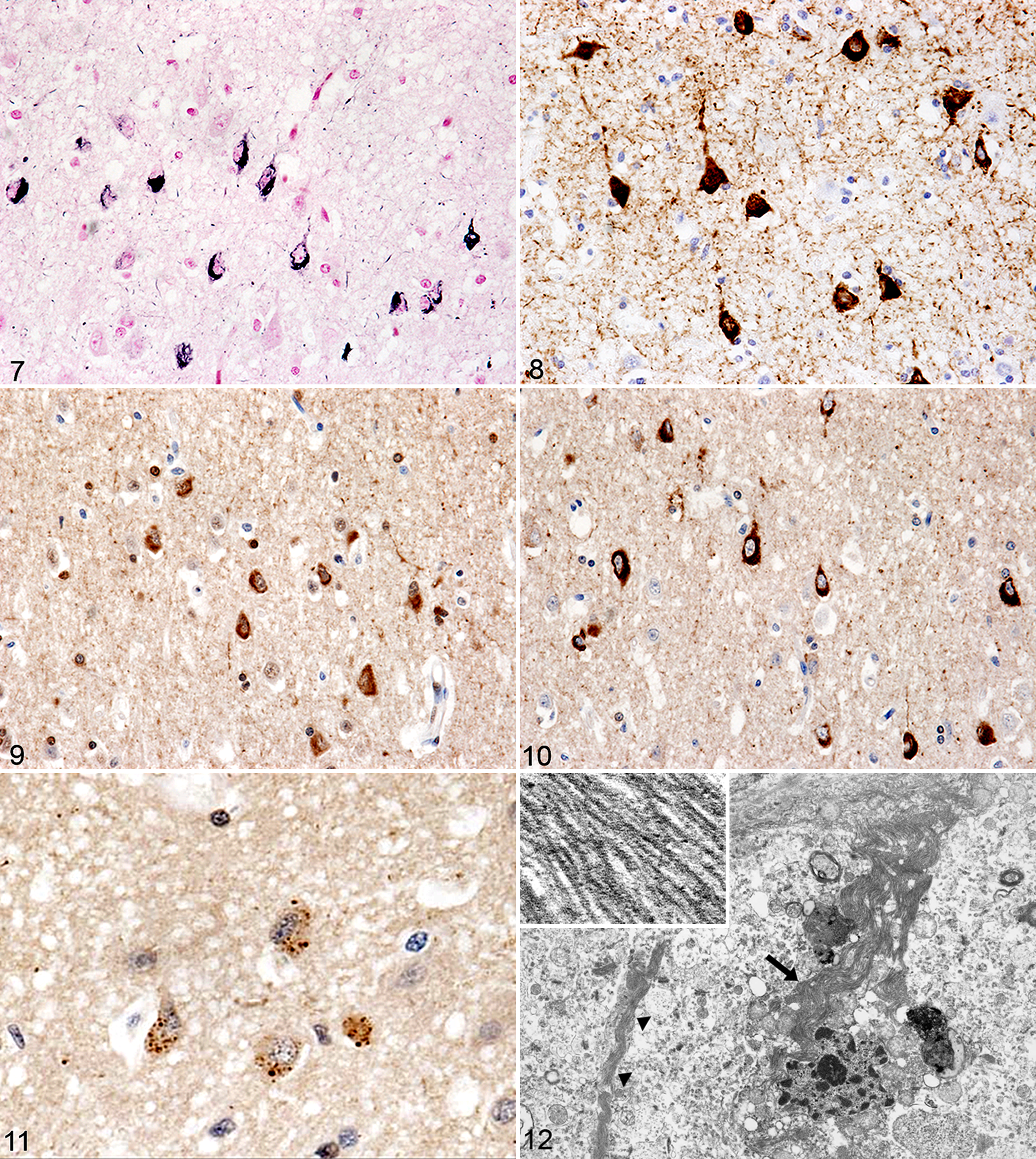
In association with AT8 immunoreactivity, argyrophilic fibrillar structures were found in neuronal cell bodies and the surrounding neuropil (suggestive of NFTs and neuropil threads, respectively) in 6 of 10 cheetah brains with the use of modified Gallyas-Braak silver stain (Fig. 7).
These argyrophilic changes were also positive for tau and tau-2 (a marker of normal and abnormal tau conformation) as well as AT8, suggesting that they consisted of excessive accumulation of both normal and abnormal tau (Figs. 8–10). In addition, granular immunolabeled neurons were observed, consistent with AT8-positive neurons, with the use of antibody to ubiquitin (Fig. 11).
Ultrastructurally, bundles of straight filaments filled the neuronal soma and axons, consistent with the tangles observed on light microscopy. The filaments had a uniform diameter of 15 nm and exhibited no helical structure (Fig. 12).
Interestingly, numerous NFTs were observed throughout the cerebrum in 3 cases (case Nos. 14, 15, and 17), 2 of which were clinically diagnosed as cognitive dysfunction (case Nos. 14 and 15). Case No. 14 exhibited a decrease in threat posture, self-injurious behavior (namely, biting its own forelimbs), and lack of awareness of the gateway to the cage. Case No. 15 exhibited wandering, fecal/urinary incontinence, decreased awareness of other individuals, and running into the fence. Abnormal behavior was not observed in other aged cheetahs over their lifetime. In this study, systemic AA amyloidosis, a major cause of death in cheetahs, was not found in cheetahs that lived to over 11 years of age, except for 1 case that AA deposition was localized in the kidney (Table 1).
Two cheetahs diagnosed as cognitive dysfunction had been imported from Namibia and raised at the same facility (YA) until they died at the ages of 12 years (case No. 14) and 14 years (case No. 15). Their brains exhibited significant atrophy on gross observation, as shown in Figure 1. Histologically, they exhibited severe neuronal loss in the entire area of cerebral cortex with extensive reactive astrocytosis (immunolabeled by GFAP) and cerebral focal vacuolation suggestive of feline spongiform encephalopathy (FSE) (Fig. 2), the disease that has been reported in captive cheetahs in Europe. However, FSE was negative for staining by antibody to PrPSC (Table 2) in our case.
Discussion
In the present study, all Aβ deposits in the cheetah brains were large and diffuse, and none were silver stained. These characteristics appeared to be similar to the first step of formation of senile plaques reported in humans, 45 also consistent with a previous report on aged cats. 19 Interestingly, immunoreactivity for both Aβ1-42 and Aβ1-40 was observed in the cheetah Aβ deposits, despite the fact that Aβ1-40 is typically found in mature argyrophilic plaques after the accumulation of Aβ1-42. 10,21
Although senile plaques have been found in the brains of various species, including nonhuman primates, 14,15,24,49 dogs, 6 cats, 19,32 and bears, 47 it is still unclear whether senile plaques are related to cognitive dysfunction in animals. 8,9,16,39,41 In dogs, accumulation of Aβ in the brain is related to neuronal loss of certain area. 4 In this study, some of the very aged cheetahs with high Aβ burden developed severe NFTs accompanied by cerebral cortical atrophy (Table 1; case Nos. 14, 15, 17).
Cerebral amyloid angiopathy is frequently observed in the brains of patients with AD as well as in non-AD humans, and many animals with senile plaques also develop it. 6,48 In contrast, there was no Aβ deposition in the vessels of the cheetahs in this study, except in a few capillary vessels in 2 aged cheetahs. This is also consistent with the findings for cats, which are considered a species that does not develop severe cerebral amyloid angiopathy. 19,32 The differences that exist in the major clearance pathway of Aβ through the vessel walls into blood 11 suggest that the Aβ clearance system in felids may be somewhat different from that in other species.
The NFTs filling the neuronal soma and axons resembled the NFTs previously demonstrated in chimpanzees, sheep, wolverines, and Asiatic brown bears, as well as in humans. 34 The ultrastructural findings were consistent with so-called straight filaments, which comprise the NFTs in some types of tauopathies in humans. 2 However, it was different from the typical ultrastrucure of NFTs seen in AD (paired helical filaments).
Furthermore, the NFTs observed in cheetahs were immunoreactive to antibodies to abnormally phosphorylated tau and ubiquitin, suggesting that these accumulations were major components of the argyrophilic tangles described in humans. 29 These cytoskeletal abnormalities were considered very similar to the NFTs observed in aged humans in morphology on silver staining and immunoreactivity.
Interestingly, NFTs were initially located in parahippocampal and CA1 regions, complementary to the distribution of Aβ (Fig. 6), which is deposited mainly in the parietal and temporal cortex. This difference in distribution of the 2 lesions is similar to that noted in a previous report in nondemented and preclinical AD geriatrics. 40 Chronologic study with more individuals is necessary to clarify the relationship between Aβ and NFTs in cheetahs.
The group of heterogeneous disorders exhibiting dementia and behavior defects that are characterized neuropathologically by prominent intracellular accumulations of abnormal tau filaments is collectively termed the tauopathies and includes AD. 27 It has been reported that some animals develop abnormally phosphorylated tau accumulation in neuronal cell bodies, and a recently reported chimpanzee was the first animal diagnosed with tauopathy. 43 As described above, only 2 individual animals (a chimpanzee and a wolverine) have been reported to exhibit both argyrophilic NFTs and Aβ deposits. 42,43 Nevertheless, we detected abnormally phosphorylated tau in the brains of 10 aged cheetahs (which lived over 10 years) and NFTs in 6 aged cheetahs with diffuse Aβ deposits in the cerebrum.
As noted above, the cheetah is unusual among felids in its genetic uniformity 30 and features high rates of morbidity and mortality associated with systemic AA amyloidosis. 38 Our findings revealed high prevalence of abnormally phosphorylated tau accumulation and NFTs among cheetahs over 10 years of age (Table 1). The high prevalence of NFT formation may suggest that a mutated tau gene may have spread in the wild cheetahs. Unfortunately, the degree of consanguinity of these animals is unclear, since they were captured in the wild. Genetic analysis is required to clarify whether they had a tau gene mutation.
As animals age, a variety of behavioral changes may occur, some of which are assumed to be due to cognitive dysfunction. 9,16 However, disorders including metabolic diseases, viral diseases, cardiac diseases, and primary neurologic diseases could be responsible for these changes. Though some criteria are available for dementia in animals, the definition and measurement of it are quite vague. In this study, we attempted to apply the diagnostic criteria for dementia in dogs based on behavior, which are employed in some clinics in Japan. 23 However, they could not be used for cheetahs, which are particularly fierce animals. Therefore, cognitive dysfunction of the 2 cheetahs were diagnosed subjectively by the clinician, who had been treating these cheetahs for a long period.
In this study, 2 cheetahs showed cognitive dysfunction whose brains were severely atrophied. The major pathologic findings in these brains were severe neuronal loss in the cerebral cortex, diffuse Aβ deposition, and NFT formation throughout the cerebrum (Figs. 1, 2). Thus, all of the major AD lesions were observed in these animals. It is important to exclude the possibility that NFTs developed secondary to other neuronal diseases, though no other causative lesions were observed in these brains, including FSE.
There is a report of an outbreak of FSE in cheetahs in Europe. 3,25,28 The cheetahs involved exhibited behavioral disorder, accompanied by vacuolization and astrocytosis in the brain. Palmer et al reported an outbreak of progressive encephalomyelopathy in captive-bred cheetahs, the cause of which remains unknown. 37 We initially suspected that the astrocytes observed in the present study were reactive to background neurologic disorders, including FSE. However, staining for PrPSC was negative on immunohistochemical examination. Another known neurologic disease of cheetahs is leukoencephalopathy, which mainly exhibits white matter degeneration. 13 In this study, degenerative lesions in the cheetah brains occupied the cortical gray matter. White matter myelin degeneration was not appreciable with Klüver-Barrera-stained sections. Moreover, NFTs and astrocytosis coexisted throughout the cerebral cortex, ruling out other possibilities, such as previous cerebral trauma (Fig. 6).
Of the 3 cheetahs with high AT8 scores, 1 (case No. 17) did not exhibit cognitive dysfunction and also developed huge numbers of Aβ deposits and NFTs, as observed in the 2 cheetahs with cognitive dysfunction. The difference between case No. 17 and the 2 animals (case Nos. 14 and 15) was the extent of neuronal loss and astrocytosis.
A recent study indicated that environmental enrichment reduces Aβ deposits and mitigates cognitive dysfunction in mice. 22,26 In addition, captive cheetahs are sensitive to chronic stress and are vulnerable to certain diseases. 46,50 These considerations suggest that the lesions identified in this study may have occurred not only because of genetic factors but also because of chronic stress in captivity.
In summary, we found that the cheetah spontaneously develops Aβ deposition with concomitant tauopathy in an age-related manner, which may or may not be clinically significant. These lesions have not been reported in animals except for single case reports. Although it is unclear why cheetahs develop these pathologic findings in such high proportion, further study of this species, especially the genetic background and environmental matter, could open new doors to elucidating the etiology of the lesions.
Footnotes
Acknowledgements
We thank Dr Hiroyuki Okada of the National Institute of Animal Health for performing immunohistochemical examination of FSE and Dr Hiroyuki Nakayama and Dr Kazuyuki Uchida of the Department of Veterinary Pathology, The University of Tokyo, for generous advice and donation of antibodies to tau protein. We thank the members of Cheetah working group of Japanese association of zoological gardens and aquariums, Mr Shu Ito of Adventure World, Mr Shigehisa Kawakami of Gunma Safari Park, and Mr Yusuke Ikebe of Akiyoshidai Safari Land, who provided us with very useful material and information.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
