Abstract
The rhesus macaque (Macaca mulatta) is one of the most extensively used nonhuman primate models for human diseases. This article presents a literature review focusing on major organ systems and age-associated conditions in humans and primates, combined with information from the Wisconsin National Primate Research Center Electronic Health Record database to highlight and contrast age-associated lesions in geriatric rhesus macaques with younger cohorts. Rhesus macaques are excellent models for age-associated conditions, including diabetes, osteoarthritis, endometriosis, visual accommodation, hypertension, osteoporosis, and amyloidosis. Adenocarcinoma of the large intestine (ileocecocolic junction, cecum, and colon) is the most common spontaneous neoplasm in the rhesus macaque. A combination of cross-sectional and longitudinal studies is required to truly define mechanisms of maturation, aging, and the pathology of age-associated conditions in macaques and thus humans. The rhesus macaque is and will continue to be an appropriate and valuable model for investigation of the mechanisms and treatment of age-associated diseases.
Keywords
Increasing life spans and expanding aging populations worldwide have led to greater scientific focus on the mechanisms of aging and the treatment of age-associated diseases. The US population of older Americans (>65 years) is projected to almost double from 43.1 million in 2012 to 83.7 million in 2050, constituting >20% of the total population. Aged individuals in the United States (>85 years) gained a year in survival between 1972 and 2010. This trend is expected to continue, and the aged are projected to compose approximately 4.5% of the US population in 2050. 63
The rhesus macaque (Macaca mulatta) is one of the most extensively used nonhuman primate models for human diseases.* Rhesus macaques share ∼95% genetic homology with humans and have been designated as a high-priority organism for whole-genome sequencing by the National Human Genome Research Institute, since genetics and genomics are increasingly integral to biomedical and evolutionary research. 67 –69 Pathologists and investigators should always be mindful of the origin and history (management, medical, and experimental) of the rhesus macaque to be evaluated. Spontaneous background lesions and/or incidental findings common to imported rhesus macaques often vary greatly from those of animals born in “closed” indoor research colonies. Background lesions may be exacerbated or masked by the process of aging. Excellent resources describing incidental and background lesions in rhesus macaques are available. †
Rhesus macaques age at about 3 times the rate of humans, with puberty occurring between 2.5 and 4.5 years, menopause at ∼26 years, a median life span of 27 years, and a maximum life span of ∼40 years. 19 Rhesus macaques and humans have very similar maturation progression, anatomy (including visual, placental, and neural anatomy), metabolism, endocrine and immune systems, reproduction, accommodative function, and aging.
The National Institute on Aging supports aging research at 5 National Primate Research Centers: the California National Primate Research Center, University of California–Davis (http://www.cnprc.ucdavis.edu); the Oregon National Primate Research Center, Oregon Health and Science University (http://onprc.ohsu.edu); the Tulane National Primate Research Center, Tulane University (http://tulane.edu/tnprc/); the Washington National Primate Research Center, University of Washington (http://www.wanprc.org); and the Wisconsin National Primate Research Center (WNPRC), University of Wisconsin–Madison (http://www.primate.wisc.edu). Longitudinal studies of aging and caloric restriction in rhesus macaques have taken place at the National Institute on Aging and the WNPRC since 1987 and 1989, respectively. 45,71 The National Institutes of Health website for National Primate Research Center–Research and Capabilities (http://nprcresearch.org/) is available as an additional resource for investigators interested in pursuing or transitioning to research with nonhuman primate models.
The focus of this article is age-associated pathologic findings in rhesus macaques. A literature review focusing on major organ systems and age-associated conditions in humans and primates was combined with information from the WNPRC Electronic Health Record (EHR) database. Age-associated disease conditions were highlighted by contrasting findings in geriatric rhesus macaques with younger cohorts. The ages of onset of many conditions within the WNPRC colony were reported for juvenile (<5 years), adult (5 to <20 years), geriatric (20–25 years), and aged rhesus macaques (>25 years).
Evaluation of the WNPRC Database
The WNPRC is funded by the National Institutes of Health to support behavioral, biomedical, and translational research with nonhuman primates. The WNPRC is fully accredited under the US Department of Agriculture and the Association for Assessment and Accreditation of Laboratory Animal Care International. All animal care and research are approved by an Institutional Animal Care and Use Committee and are carried out in accordance with all applicable national, local, and institutional guidelines. The founding population of rhesus macaques for the WNPRC’s specific pathogen–free (SPF) breeding colony was imported from India in the 1960s and 1970s. Monkeys are housed indoors on a 12-hour light/dark cycle with controlled temperature and humidity, fed a nutritionally balanced nonhuman primate diet (Diet 2050; Harlan-Teklad), and allowed water ad libitum. All monkeys are closely observed daily. They receive enrichment opportunities (foraging, sensory, and structural) on a regular schedule in accordance with the WNPRC enrichment plan. Housing environments include small social groups (3–10 individuals), pairs, and single housing with clinical or scientific justification. The WNPRC has housed a yearly average of 1050 rhesus macaques for the past 50 years. Monkeys that exhibit signs of clinical illness receive comprehensive diagnostic testing—including but not limited to physical examination, complete blood counts, serum chemistry analysis, urinalysis, radiographs, ultrasound, ultrasound-guided biopsy, surgical biopsy, and exploratory laparotomy. A complete necropsy with histologic analysis is performed on all monkeys following humane euthanasia as prescribed by Institutional Animal Care and Use Committee–approved research and management protocols.
Data for the prevalence of age-associated and background lesions in purpose-bred macaques were gathered via a search of WNPRC medical, biopsy, and necropsy records in the EHR database using Systematized Nomenclature of Medicine–coded keywords and terms. Electronic records for the entire colony included ∼9575 individual animals and spanned a >35-year period. Records from animals enrolled in infectious disease, vaccination, pharmaceutical, transplant, and other studies associated with or known to promote conditions covered in this review were excluded from the data set. The SPF breeding colony served as the untreated control group, when appropriate. Animals had varied clinical and experimental histories; however, commonalities in care, aging, and pathology were observed, and these formed the basis of comparisons.
General Appearance
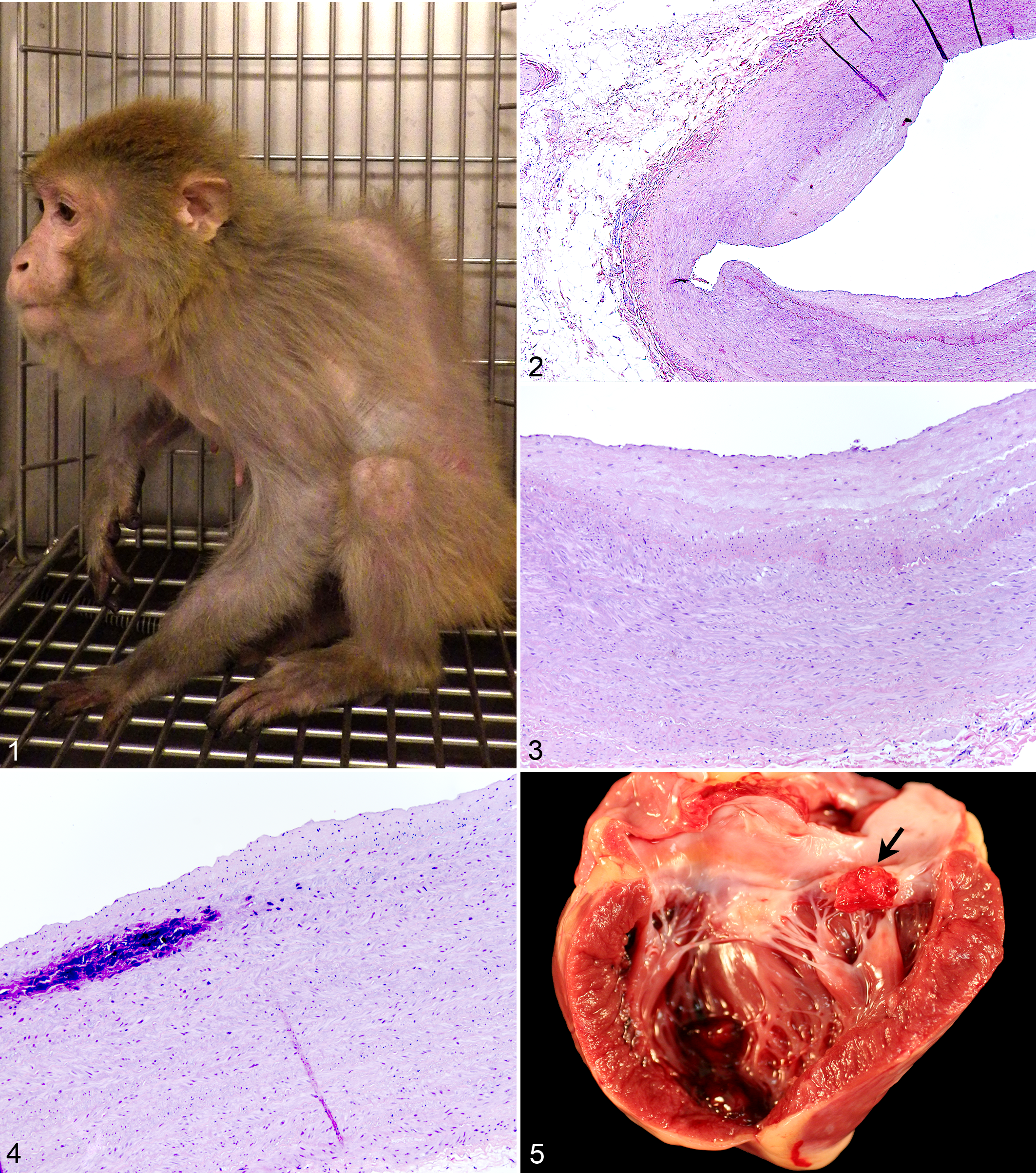
The external resemblance between geriatric rhesus macaques and geriatric humans is quite striking (Fig. 1). The dermis is thinned, wrinkled, and fragile, especially on the face and around the eyes. The spine, with mild to severe kyphosis and scoliosis, is covered by thin faded pelage. Muscle mass is moderately to markedly diminished and with obvious loss of joint mobility. Teeth are worn and missing. Many common age-related changes are reviewed below.
Cardiovascular
Cardiovascular disease develops in humans, great apes, and many nonhuman primates as they age, and it is identified as a primary cause of mortality in rhesus macaques and humans. ‡ Structural changes in blood vessels progress with increasing age in humans and include intimal and medial fibrosis in the aged to elderly. 33 Calcification of the vascular wall is associated with fragmentation of the intimal elastica and may involve as much as a quarter of the vascular circumference in humans. 33 Pale streaking of the intima of the aorta (fibrous intimal plaques) has been described during gross necropsy in wild-caught and purpose-bred rhesus macaques (Figs. 2–4). 13,16,82 A previous study of WNPRC rhesus macaques identified fibrous intimal plaques in 80% (23 of 29) of animals >30 years of age. 82
Arteriosclerosis and arteriolosclerosis, vascular thickening with or without luminal narrowing, have been noted to be a background finding in control rhesus macaques and have been shown to increase with age in rhesus macaques and humans. 16,82 There is a strong association between arteriosclerosis and hypertension and diabetes mellitus in rhesus macaques and humans. 14,15,54,73,74,86 Hypertension is associated with hyaline arteriolosclerosis (homogeneous hyaline thickening of vascular walls with loss of structural detail) and hyperplastic arteriolosclerosis (concentric lamellar fibrous and muscular thickening of vascular walls). 33,73 Although rhesus macaques are a model species for hypertension research, diagnosis of spontaneous hypertension in clinical cases is problematic. 29,34,72,93 Measuring blood pressure in unanesthetized monkeys typically requires restraint, thus increasing measured intravascular pressures, while chemical restraint usually lowers pressures.
Unlike humans, free-ranging and laboratory raised rhesus macaques rarely exhibit atherosclerosis, unless monkeys are fed an experimental high-fat diet similar to that of most people in Western industrialized countries. 13,16,76,80,81,90,93 Lesions develop first in the abdominal aorta, followed by the thoracic aorta and then the coronary arteries. 76 Myocardial infarction and stroke have been recorded in rhesus macaques fed high-fat diets but are not common findings in most research colonies fed commercial primate diets. 76,80,81
Common cardiac changes in geriatric and aged rhesus macaques include valvular endocarditis, valvular endocardiosis, valvular mineralization, focal to multifocal interstitial fibrosis, multifocal cardiomyocyte degeneration, cardiomyocyte hypertrophy, and generalized cardiac hypertrophy. 10,82,86 Vegetative valvular endocarditis occurs at any age and is associated with systemic bacterial, fungal, or rickettsial infection (Fig. 5). 15,72,74 Bacteria are the most common causative agents in human cases of infectious endocarditis. 74 Staphylococci and streptococci are the most common cause of bacterial endocarditis in nonhuman primates. 72 Valvular endocardiosis—characterized by chronic nodular thickening of the valve leaflets with fibrous proliferation and/or acid mucopolysaccharide deposition—is noted most often in the atrioventricular valves (Figs. 6, 7). Although considered an age-associated lesion, valvular endocardiosis occurs earlier than age-associated conditions such as neoplasia or cataracts in rhesus macaques. The majority (82%) of 117 recorded cases of valvular endocardiosis at the WNPRC were in rhesus macaques >10 years of age. The age distribution of WNPRC cases was as follows: 4% juvenile (<5 years), 45% adult (5–20 years), 22% geriatric (20–25 years), and 29% aged (>25 years). The prevalence of valvular endocardiosis in WNPRC SPF breeding colony animals was 5.5% with a mild upward shift in the age distribution in this smaller population to 3% juvenile (<5 years), 41% adult (5–20 years), 21% geriatric (20–25 years), and 35% aged (>25 years).
Degenerative and fibrosing cardiomyopathy is identified when interstitial fibrosis with degeneration of individual cardiomyocytes and variable hypertrophy of cardiomyocytes is observed in the myocardium; the incidence of this disorder increases with age (Figs. 8, 9). As with atrioventricular valve endocardiosis, only 6% of the recorded WNPRC cases occurred in young adult rhesus macaques (<10 years). The age distribution of recorded WNPRC cases was as follows: 0% juvenile (<5 years), 36% adult (5–20 years), 30% geriatric (20–25 years), and 33% aged (>25 years). The prevalence in SPF breeding colony animals was 7.3% with a similar age distribution.
Pulmonary
Although lung mites (Pneumonyssus simicola) are considered a background finding in almost 100% of wild-caught rhesus macaques, the introduction of ivermectin treatment prior to importation and during quarantine has made identification of active and/or severe infection with intact mites less common. 1,3,14,32,52,54,79 The importation of Indian-origin rhesus macaques into the United States was discontinued in 1978. 43,47 Thus, there is variable prevalence of lung mites in Indian-origin rhesus macaques bred and raised in US facilities. Rhesus macaques imported from other countries may have numerous small subpleural cysts (Fig. 10). Histologically, there are intact mites (in active infections) or fragments of chitinous exoskeleton (in treated animals) surrounded by eosinophilic to mixed granulomatous inflammation and variable amounts of brown-black pigment within bronchi and bronchioles with moderate to marked inflammation expanding the peribronchiolar interstitium (Figs. 11, 12). The prevalence of pulmonary acariasis in the entire WNPRC colony was 4%, while a 2% (39 of 2205) prevalence was recorded in the WNPRC SPF breeding colony. The age distribution in the SPF breeding colony was 0% juvenile (<5 years), 46% adult (5–20 years), 28% geriatric (20–25 years), and 26% aged (>25 years).
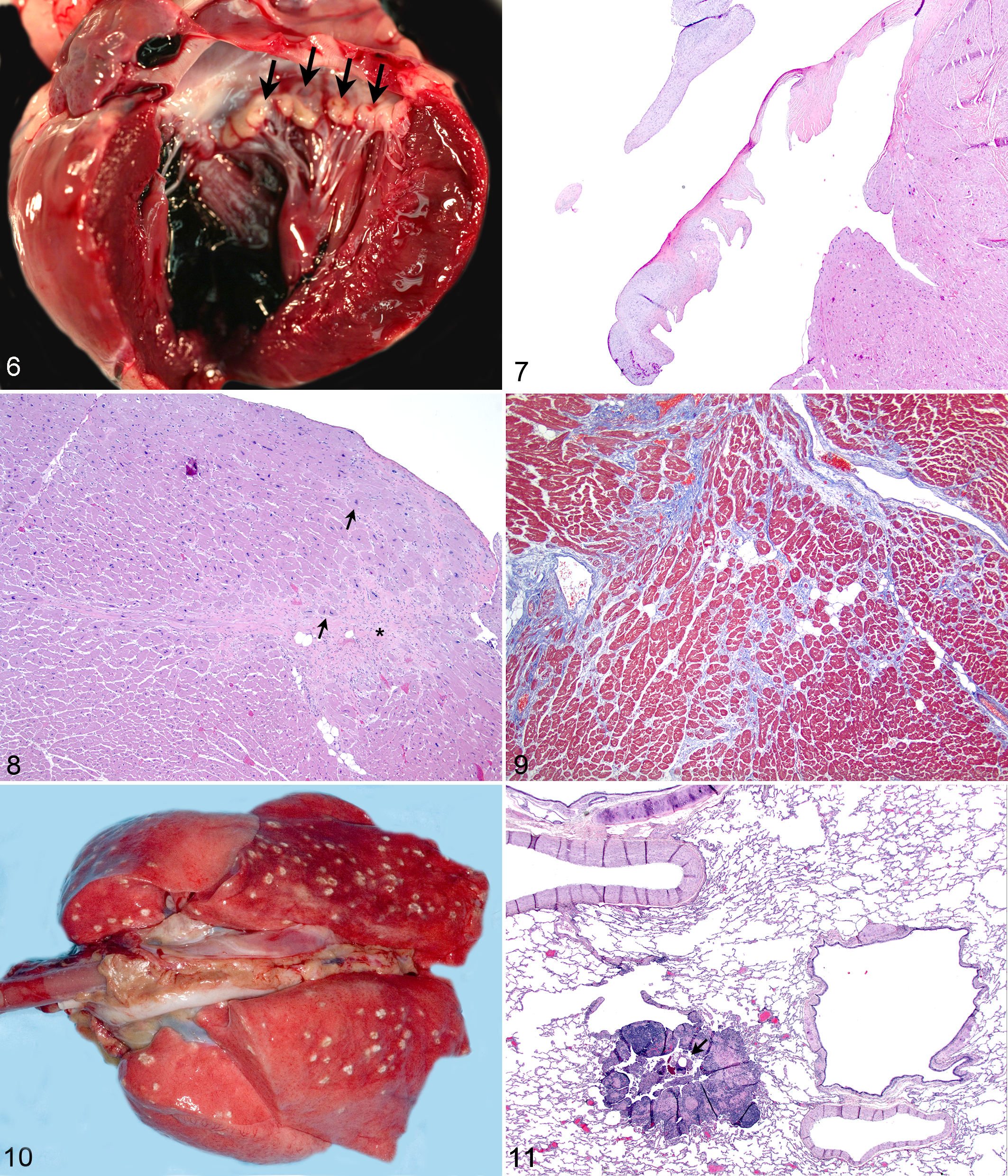
There is increased prevalence of pulmonary/pleural emphysema and subpleural bullae in the periphery of lung lobes in geriatric and aged rhesus macaques (Fig. 13). 82 These changes are not clinically significant but should be noted during gross and histologic evaluation. The presence of pulmonary edema, increased numbers of alveolar macrophages, and hemosiderin within alveolar macrophages (siderophages) is significant in aged individuals and suggests left-sided heart failure. 54
Musculoskeletal
During necropsy of older animals, bones may be found to be subjectively brittle during collection of samples such as brain or bone marrow. Like humans, rhesus macaques show increasing bone mineral content between birth and skeletal maturity (peak bone mass) with subsequent bone loss (decreased bone mineral content) with advancing age in both sexes. 19 –22,39,66 The female rhesus macaque is an excellent model for pre-, peri-, and postmenopausal osteoporosis due to marked similarities in menopausal hormonal transition and skeletal response. 36,39
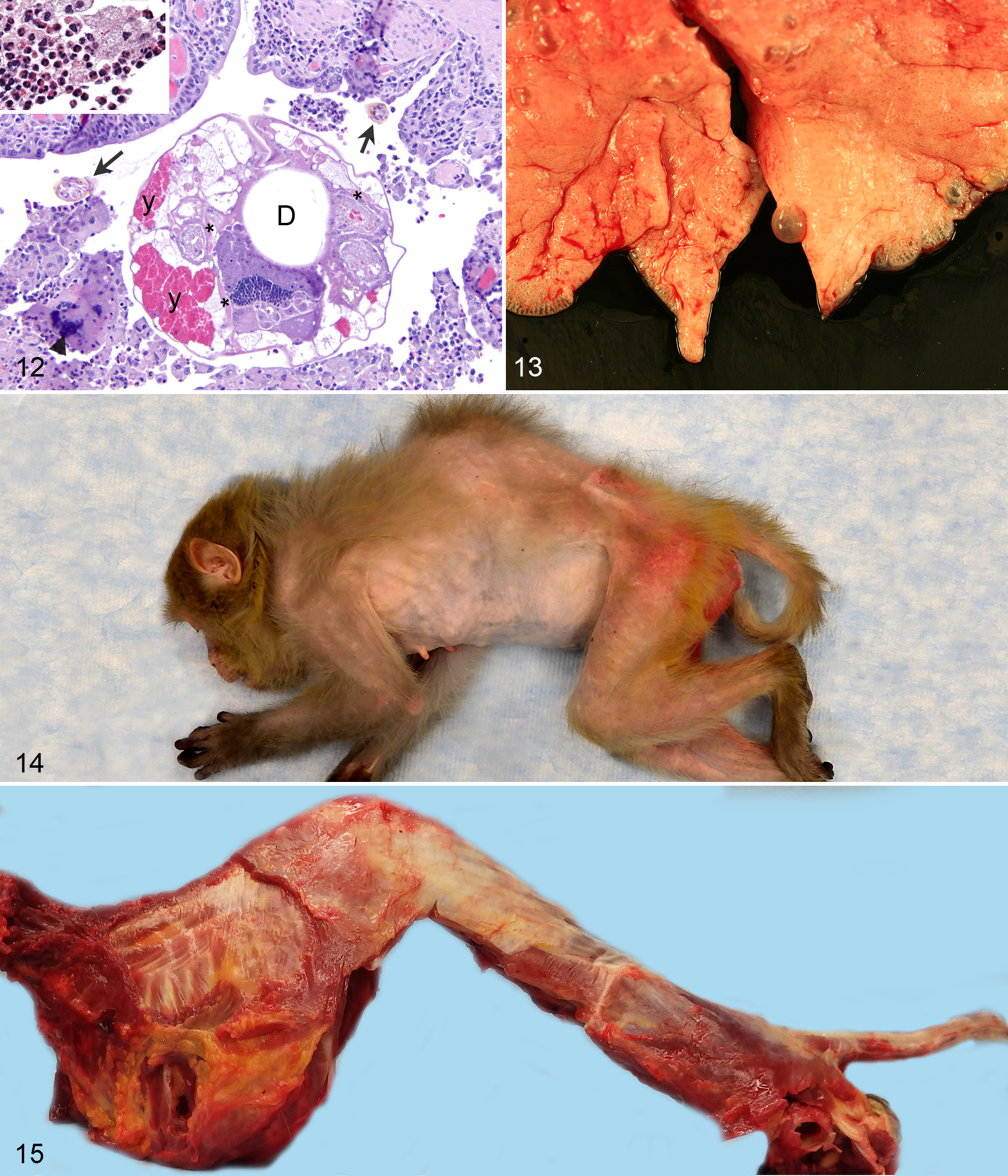
Primary osteoarthritis or degenerative joint disease is associated with aging and mechanical wear in weight-bearing joints, while secondary osteoarthritis is associated with traumatic joint injuries. 70 Primary osteoarthritis typically occurs in humans in their fifth decade and consists of degeneration of articular cartilage, eburnation of bone, and osteophyte formation. 70 Osteoarthritis in rhesus macaques has also been shown to increase with age. 7,20,21,66,70,82 Gross and histologic changes in rhesus macaques with vertebral osteoarthritis are quite similar to those seen in humans and include degeneration of intervertebral discs with concentric and radial tears, cartilage degradation, and osteophytosis. 7 Vertebral thoracic osteoarthritic changes in older rhesus macaques tend to be more impressive than in humans with conspicuous thoracic kyphosis, osteophyte formation, and bridging spondylosis on the anterior aspect of vertebrae (Figs. 14–16). Human vertebral osteophytosis occurs anterior and posterior with a lower prevalence of spondylosis. 7 The prevalence of kyphosis in the SPF breeding colony was 0.3%. The distribution by age of kyphosis, diagnosed at necropsy in 31 WNPRPC animals, was as follows: <1% juvenile (<5 years), 43% adult (5–20 years), 19% geriatric (20–25 years), and 37% aged rhesus macaques (>25 years of age).
A noninvasive imaging study of the lumbar spine in 114 rhesus macaques of both sexes revealed a 31% prevalence of vertebral osteoarthritis in animals <18 years of age with a marked increase to 95% in animals >18 years of age. 20,21 Scoliosis of the spine was also noted in rhesus macaques and may occur independently or concurrently with kyphosis. When the age distribution of all rhesus macaques in the WNPRC database with scoliosis was determined and compared with the radiographic study by Colman et al, 85% of monkeys diagnosed with scoliosis and 85% of monkeys with kyphosis were >18 years of age.
Osteoarthritic changes may affect any, often multiple, joints in the abaxial skeleton, from the digits to the shoulders and hips. The most commonly affected joint in the rhesus macaque was the stifle (82% of arthritis cases), followed by the hip (18% of cases), in WNPRC records (Figs. 17, 18).
As humans and rhesus macaques age, abdominal fat increases, and lean body mass declines. 20 –22,66,71 Sarcopenia, an age-associated decrease in muscle mass, begins in middle age (fifth decade in humans and between 14 and 16 years of age in rhesus macaques) and reaches significance in the eighth decade in humans (50% loss of leg muscle area) and between 23 and 24.5 years of age in rhesus macaque (>20% loss of lean muscle mass in the legs). 22,66
Eyes
The rhesus macaque has been proven to be the best model for visual accommodation and presbyopia. 24 Rhesus macaques’ accommodation diminishes at a similar relative rate with age to that of humans. 60 Cataracts in humans are age associated in approximately 90% of cases. 59 Lens opacity typically begins to develop in 20% of rhesus macaques in the WNPRC colony at 20 to 22 years of age with a significant increase in prevalence after 26 years of age (Fig. 19). 82 Current WNPRC medical and necropsy records identified cataracts in rhesus macaques with an age range from 9.6 to 40.4 years and the following distribution: 0% juvenile (<5 years), 20% adult (5–20 years), 6% geriatric (20–25 years), and 74% aged (>25 years). Pathologists should use caution when interpreting lenticular changes histologically, since the type of fixative and duration of fixation may cause artifacts mimicking cataracts (personal observation).
Renal
Mild multifocal nonsuppurative interstitial nephritis is considered an incidental change in the rhesus macaques. 14,17 As individuals age, there is increasing interstitial fibrosis causing capsular contraction or undulation. Membranous thickening of Bowman capsule, multifocal segmental to global membranous glomerulopathy, synechiation, and glomerulosclerosis all increase in incidence and severity with increasing age (Figs. 20, 21). Glomerulosclerosis, severe global fibrosis, and scarring of the glomerulus with synechiation were noted in WNPRC rhesus macaques with an age distribution of 6% juvenile (<5 years), 29% adult (5–20 years), 19% geriatric (20–25 years), and 45% aged (>25 years). Glomerular basement membrane thickness increases with age and plateaus at the age of 18 years in rhesus macaques. 27 Infectious and inflammatory conditions may also contribute to thickening of basement membranes through the deposition of fibrin, immunoglobulins, and/or amyloid. 39,46,79
Diabetic nephropathy follows myocardial infarction as the second-most common cause of diabetic mortality in humans and affects 25% to 40% of type 2 diabetics with a comparable prevalence in type 1 diabetics. 27 Diabetic nephropathy is characterized by diffuse thickening of the glomerular capillary membranes (membranous glomerulopathy), diffuse mesangial sclerosis, and hyaline arteriolosclerosis involving the afferent and efferent arterioles. 2,27 The rhesus macaque is one of the many species used in diabetes research, and there are drug-induced and spontaneous models of type 2 diabetes and nephropathy. 27,89 Obese rhesus macaques that develop type 2 diabetes have increased glomerular volume and glomerular basement thickening that do not correlate with increasing age. 27 Further review of medical and necropsy records with retrospective sample analysis is necessary to define the differing causes of glomerular changes noted in the WNPRC database. Membranous glomerulopathy, defined as membranous thickening of glomerular capillary basement membranes, across age groups in the WNPRC colony was as follows: 7% juvenile (<5 years), 57% adult (5–20 years), 11% geriatric (20–25 years), and 24% aged (>25 years).
Reproductive
Female
Endometriosis is a common disease in women and rhesus macaques and was first reported in an adult female rhesus macaque in 1929. 17,18,23,30,31,94 Prevalence of endometriosis increases with age and may reach ∼ 30% in some rhesus macaque colonies. 94 Predisposing factors include but are not limited to long-term estrogen treatment, follicle aspiration, embryo transfer, cesarean section, hysterectomy, and radiation (personal communication, L. Colgin). 18,30,31 Evidence from retrospective kinship investigations of humans and rhesus macaques with endometriosis suggests that there may be a genetic basis for cases of spontaneous endometriosis in both species. 94
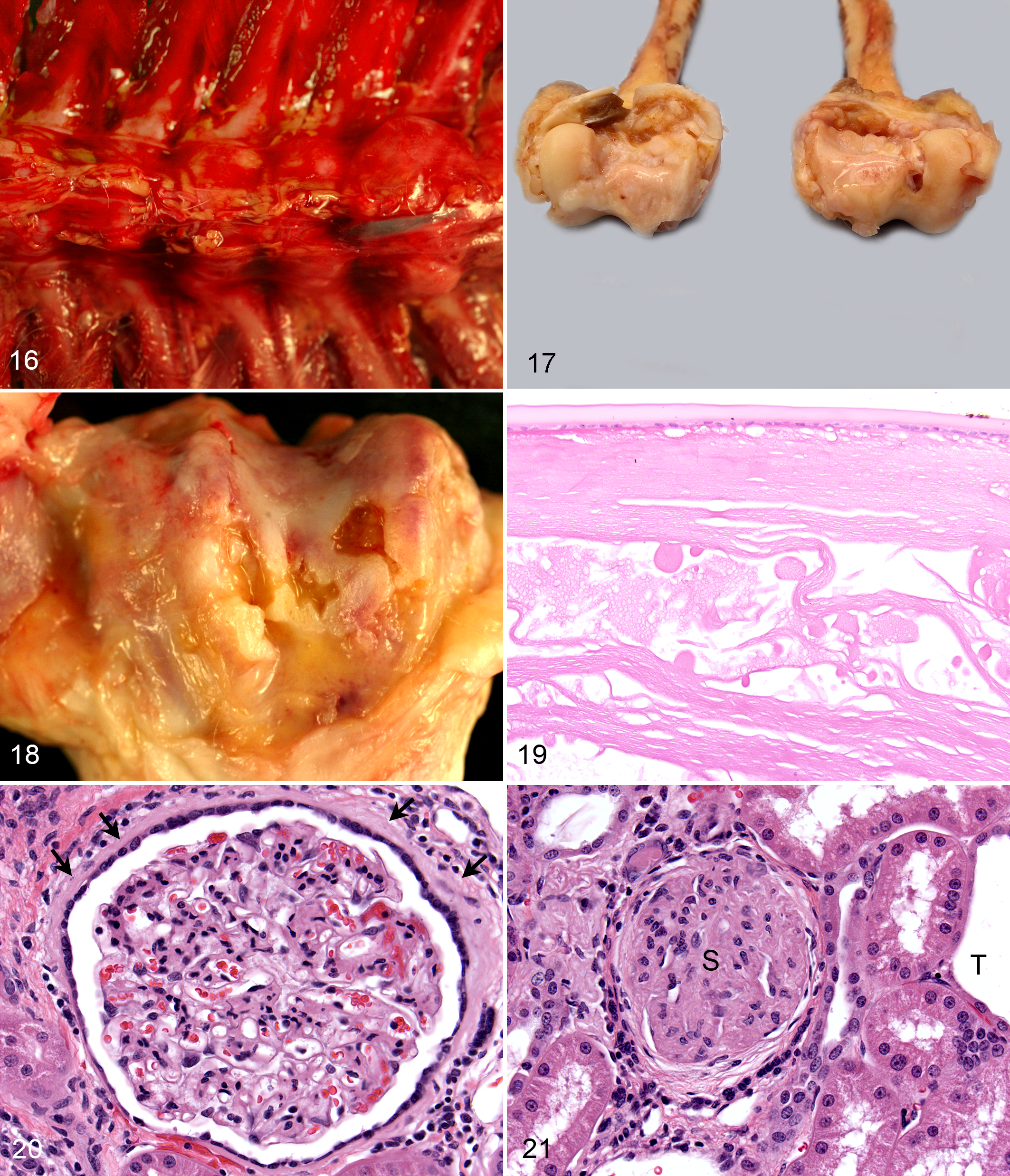
A diagnosis of endometriosis renders rhesus macaques unsuitable for many studies, and necropsy is often performed shortly after diagnosis (Fig. 22). The uterus (serosa and external aspect of the myometrium) is the most commonly affected site in the WNPRC database (63% of endometriosis cases). The ovary (one or both) was the most common extrauterine location (47% of cases) in the WNPRC colony (Fig. 23). These data agree with other retrospective studies of endometriosis in rhesus macaques. 18,31 The prevalence of ovarian infiltration in other colonies of rhesus macaques may be as high as 80%. 31 Animals and humans with endometriosis often have multicentric lesions affecting the colon, urinary bladder, and abdominal organs, such as the liver, spleen, and diaphragm, with occasional invasion of the retroperitoneum and migration into the thorax and lungs. 5,6 The prevalence of endometriosis within the SPF breeding colony was 9%, while it was only 1% in the full WNPRC colony. The age distribution in the SPF breeding colony was as follows: 1% in juvenile (<5 years), 59% in breeding age adults (5–20 years), 25% in geriatric (20–25 years), and 15% in aged (>25 years). There was a mild shift to greater numbers of aged monkeys (24% of 256 animals) with endometriosis in the general colony, reflecting the large number of geriatric and aged animals enrolled in long-term longitudinal studies at the WNPRC.
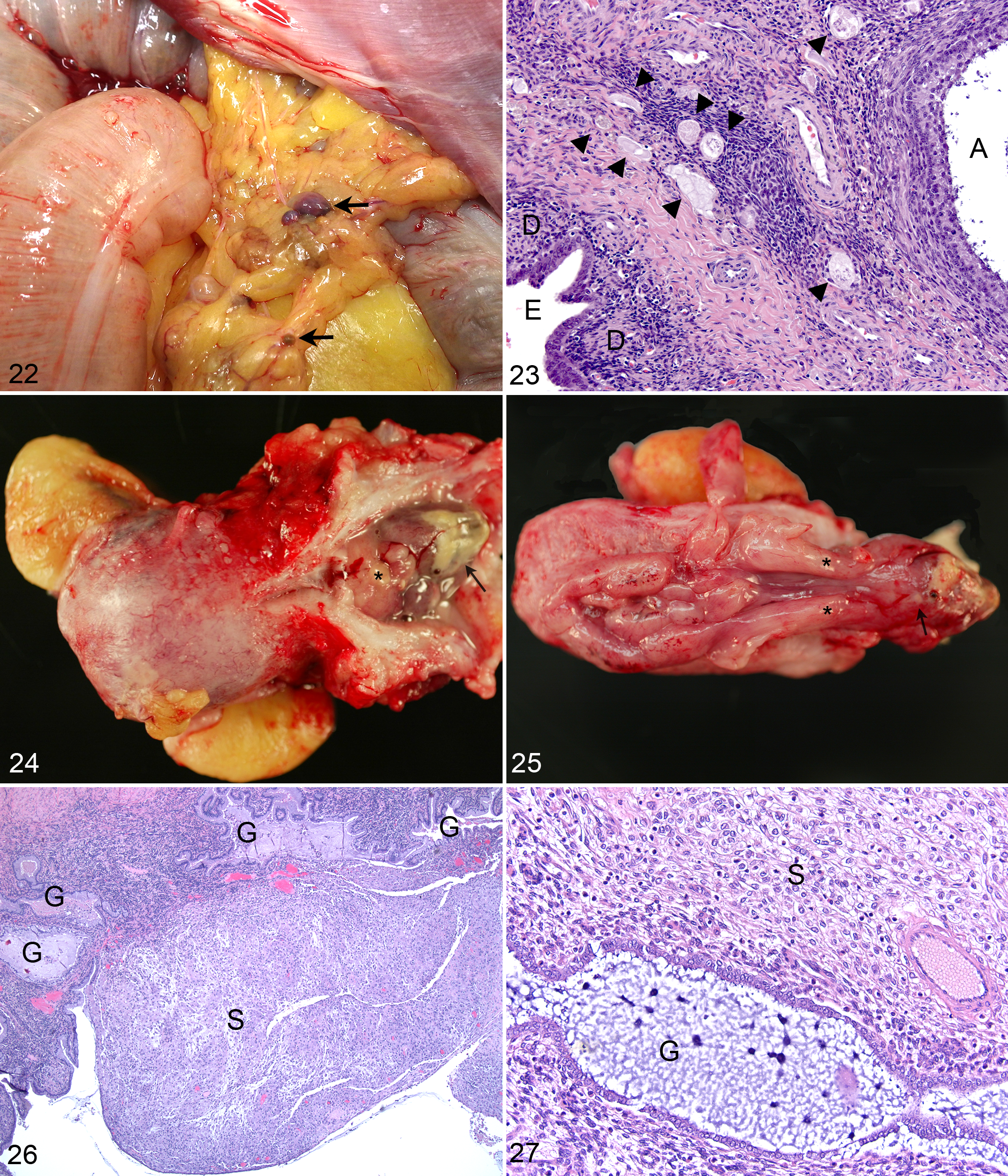
Numerous females in longitudinal studies and a focus on characterizing the rhesus macaque model of endometriosis have led to routine management of endometriosis at the WNPRC. Animals are treated with a combination of monthly intramuscular injections of synthetic progesterone, 150 mg of medroxyprogesterone acetate (MPA), to suppress the estrous cycle and analgesics to ameliorate the clinical signs of discomfort (anorexia, hunched posture, and lethargy). 26 Administration of MPA may reduce insulin sensitivity and glucoregulatory function in rhesus macaques within months of initiation of therapy, as compared with age- and weight-matched controls. 26 The package insert for MPA (Depo-Provera, 150-mg/mL suspension for injection; Pfizer Pharmaceuticals) lists this as an unreferenced side effect in women with additional specific cautions to monitor diabetic women carefully. 65 Thus, clinical management of animals with endometriosis requires periodic evaluation for the development of diabetic signs. Multiple WNPRC animals on MPA therapy have had breakthrough cycling. Abdominal ultrasound has shown progressive uterine enlargement in a number of cases. The uteri of MPA-treated animals are often mildly to moderately enlarged with thickened pale soft friable endometrium that may have polypoid proliferations that can protrude through the cervix into the vagina (Figs. 24, 25). Histologic evaluation of biopsy and necropsy specimens of these lesions reveals proliferative thickened endometrium with irregularly shaped to elongated glands and decidualized secretory stroma between and often overlying glands (Figs. 26, 27). These changes are comparable to changes noted in women treated with MPA. 49 Of animals recently diagnosed with progestin-induced endometrial proliferation, 3 of 4 (75%) had evidence of active endometriosis. Thus, MPA therapy may ameliorate external signs of menstrual cycling, but it does not provide complete control of endometriosis, necessitating consistent monitoring of animals maintained on long-term therapy.
Menopause, the absence of ovulation and menstrual cycling, typically occurs in female rhesus macaques at approximately 26 years of age. 17,18,46,61,88,92 Hormonal changes in the aging rhesus macaque are quite similar to aging women with lower circulating estradiol levels and increased GnRH, LH, and FSH levels. 36 Interestingly, GnRH pulse frequency within the brain does not differ between young and aged rhesus macaques, suggesting aging changes within the hypothalamus itself. 36
The ovary in the rhesus macaque has diminishing numbers of follicles and stroma with increasing age, similar to humans. 61 There is loss of stroma, and the proportions of primordial and primary follicles shift from ∼75% and ∼20% to ∼ 55% and ∼45%, respectively. 61 Although numbers of antral follicles decrease with increasing age, the proportion respective to primordial and primary follicles remains consistent throughout life. 61
Endometrial polyps are noted to occur in postmenopausal rhesus macaques. The ovaries of these animals should be evaluated for the presence of granulosa cell tumors or luteinized follicular cysts. 18 Of the 25 recorded endometrial polyps in the WNPRC database, the prevalence was as follows: 0% juvenile (<5 years), 32% adult (5–20 years), 24% geriatric (20–25 years), and 40% aged (>25 years), and 4% in adults with unrecorded birth dates.
Mammary gland changes in aged rhesus macaques include cystic dilatation of mammary ducts and lobules and focal lobular hyperplasia. 18 One retrospective study of mammary neoplasia identified lobular carcinoma in situ, ductal carcinoma in situ, and invasive ductular carcinoma with diagnoses predominantly in rhesus macaques >19 years of age. 91
Male
Fertility in males persists longer than in females in most species. Cryopreservation studies have shown that older rhesus macaques (∼19 years) have fresh sperm concentrations and grade (quality) comparable to that of young males but significantly reduced motility in thawed cryopreserved samples. 35
The aged rhesus macaque prostate has been extensively evaluated due to the need for models of benign prostatic hyperplasia and prostatic carcinoma. Studies and case reports of rhesus macaque prostatic pathology have identified stromal hyperplasia, cystic hyperplasia, squamous metaplasia, focal glandular pleomorphism, nonsuppurative prostatitis, rare single adenocarcinomas, and more commonly, benign basal cell hyperplasia. 8,40,42,53,56,58 In the above-cited references, these lesions were noted most often in older adult and aged rhesus macaques. The age distribution of prostatic hyperplasia in the WNPRC colony was as follows: 0% juvenile (<5 years), 17% adult (5–20 years), 50% geriatric (20–25 years), and 33% aged (>25 years).
Metabolic
Humans and rhesus macaques exhibit decreased glucoregulatory function with increasing age, especially in obese individuals. 4,19,27,46,64,71,87,93 Significant disorders associated with diabetes in humans include hypertension, platelet dysfunction, macrovascular disease, myocardial infarction, renal vascular insufficiency, cerebrovascular accidents, diabetic nephropathy, diabetic neuropathy, dyslipidemia, microalbuminuria, diabetic retinopathy, glaucoma, cataracts, and enhanced susceptibility to infections. 48 Hyaline arteriolosclerosis in humans is more prevalent and severe in diabetics than nondiabetics but may be diagnosed in elderly individuals without diabetes or hypertension. 4 Amyloid accumulation occurs within the pancreatic islets of ∼90% of human type 2 diabetics, is well described in the diabetic rhesus macaques, and may be independent of amyloid deposition in other tissues (Fig. 28). 4,28,64,87 Rhesus macaques are one of many animal models of diabetic neuropathy and retinopathy. 41
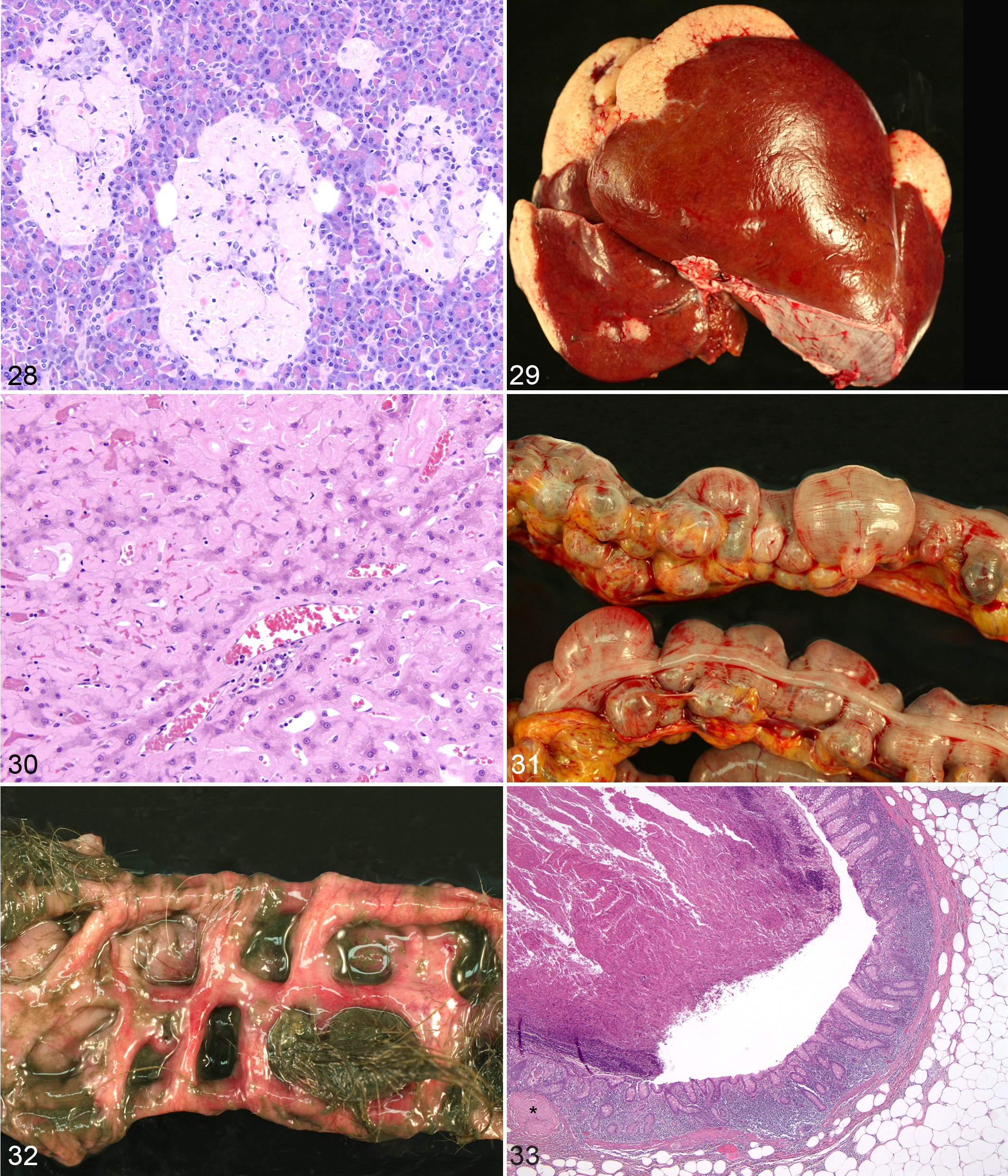
There are excellent papers describing systemic (secondary) amyloidosis in nonhuman primate. Systemic amyloidosis can develop at any age, although there is an overall incidental increase with advancing age. 9 Secondary (AA) amyloidosis is associated with generalized inflammation, production of serum amyloid A by the liver, and conversion and deposition of AA amyloid in 1 or multiple tissues, including the spleen, pancreas, large and small intestinal lamina propria, lymph nodes, kidneys, and liver (Figs. 29, 30). Amyloid accumulation associated with plasma cell dyscrasias, multiple myeloma, and other conditions that cause immunoglobulinemia is considered primary amyloidosis. 9 When animals infected with simian immunodeficiency virus, other infectious diseases, and individuals in vaccine studies were removed from the WNPRC data set, increasing age correlated with increases in amyloid prevalence. The study by Blanchard et al reported prevalences by age of systemic amyloid in the Tulane National Primate Research Center colony as follows: ≤5 years, 30%; 6–10 years, 10%; and >10 years, 60%. 9 The age distribution for the WNPRC colony was as follows: ≤5 years, 2%; 6–10 years, 7%; and >10 years, 91%. The striking difference between the juvenile and adult populations at the 2 National Primate Research Centers is likely due to the differences in colony management and housing. The Tulane National Primate Research Center has outdoor enclosures and large groups of rhesus macaques living together, while WNPRC rhesus macaques are housed indoors in pairs, mother-infant quartets, and small groups not exceeding 10 individuals. Animals housed outdoors have a greater likelihood of dietary indiscretions through the ingestion of pests (birds, squirrels, and rodents) and/or soil- or water-borne pathogens (eg, Listeria) that may cause illness. Dominance and alliance behaviors within large groups of primates often lead to injuries as well. In addition, the focus on longitudinal aging investigations at the WNPRC and behavioral research studies at the Harlow Center for Biological Psychology has ensured a large population of geriatric animals in the WNPRC data set. When amyloid cases were evaluated according to age distribution, the accumulation of amyloid was strongly associated with advanced age: 3% juvenile (<5 years), 30% adult (5–20 years), 24% geriatric (20–25 years), and 43% aged (>25 years of age).
Brain
Cerebral plaques composed of amyloid-β have been described in the brains of rhesus macaques and humans with increasing plaque density paralleling advancing age. 75,78,83 Plaque accumulation has not been noted in rhesus macaques <19 years of age, is quite variable among animals 20 to 25 years of age, and is significantly increased in the cerebrum after 30 years. 78,83 Plaque formation has been noted in the basal ganglia, prefrontal gyri, amygdala, inferotemporal regions, and parietal cortices with the parietal and hippocampal gyri less commonly affected. 75,78,82 Neurofibrillary tangles, a feature of Alzheimer disease, are not noted in the aged rhesus macaque brain. 75,78
Spontaneous cerebral infarcts have been detected by magnetic resonance imaging (MRI) in the parietal and temporal lobes in aged rhesus macaques. 82 A cross-sectional study provides an indication of the presence of infarction in different age categories, but a review of longitudinal aging studies with serial MRI data is needed. Gross evidence of cerebral infarction with focal regions of atrophy and/or loss of cerebral parenchyma is an uncommon finding in the WNPRC necropsy database. Infarcts were identified in 5 untreated animals between 15.8 and 33.8 years of age (mean age, 25.7 years).
Age-related iron accumulation in the brains of rhesus macaques and humans has been identified by MRI and confirmed histologically in the globus pallidus and substantia nigra. 38,82 The red nucleus, basal nuclei, and parietal, temporal, and perirhinal cortices have also been identified as sites of iron accumulation. 44,55
Alimentary
Diverticulosis is age associated in humans and rhesus macaques and may occur at any level of the gastrointestinal tract. The colon is the most commonly affected region in both species. 10,12,48,55 Outpouchings of the mucosa and submucosa through the muscularis are similar in both species and may be focal or multifocal along the entire length of the colon (Figs. 31–33). Human populations <30 years of age have almost no incidence of diverticulosis, while populations >60 years of age in many industrialized nations have prevalences of 50%. 48 The age distribution of diverticulosis in the WNPRC colony was as follows: 0% juvenile (<5 years), 40% adult (5–20 years), 18% geriatric (20–25 years), and 42% aged (>25 years of age). Ninety-five percent of WNPRC cases involved only the colon, with the cecum and colon affected in the remaining 5%.
Neoplasia
The nonhuman primate literature—specifically, investigations involving rhesus macaques—provide a very good overview of the numerous types of cancer that occur in the species. § Most retrospective surveys of neoplasia focus on “spontaneous” tumors and exclude cases involving treatment with known carcinogens or infections with cancer-associated viruses such as lymphocryptovirus, Epstein-Barr virus, simian T-cell leukemia virus, simian immunodeficiency virus, and papilloma viruses. 77 When these cases are not excluded, hematopoietic neoplasia is the most common cancer of nonhuman primates. 51 When viral and chemically induced neoplasms are excluded from studies, intestinal adenocarcinoma of the ileocecocolic junction and colon is the most commonly diagnosed neoplasm in rhesus macaques. ||
Large intestinal neoplasia in the rhesus macaque is believed to be significantly different from that in humans due to the absence of polyp formation, although there are similarities in histologic appearance and immunohistochemical characteristics. 37 Polyps were noted in only 10 WNPRC animals. Locations included the stomach, duodenum, ileum, ileocecocolic junction, colon, and rectum. Concurrent adenocarcinoma was present in only 2 of 10 (20%) polyp cases, affecting the ileocecocolic junction and colon (Fig. 34). Mucosal ulceration and/or enterocolitis was present in 6 of 10 (60%) polyp cases. Polyps occurred in monkeys >21 years of age in 9 of 10 (90%) cases, with a single 4.6-year-old rhesus macaque with a benign polyp at the ileocecal junction.
A previous survey of spontaneous neoplasms diagnosed in the WNPRC and National Institute on Aging colonies of rhesus macaques was updated for this review (Table 1). 77 No animal had >3 distinct types of neoplasia. Adenocarcinoma of large intestine (ileocecocolic junction, cecum, and colon) remained the most common neoplasm in the rhesus macaque (Figs. 35, 36).
Prevalence of Neoplasms in Rhesus Macaques.a
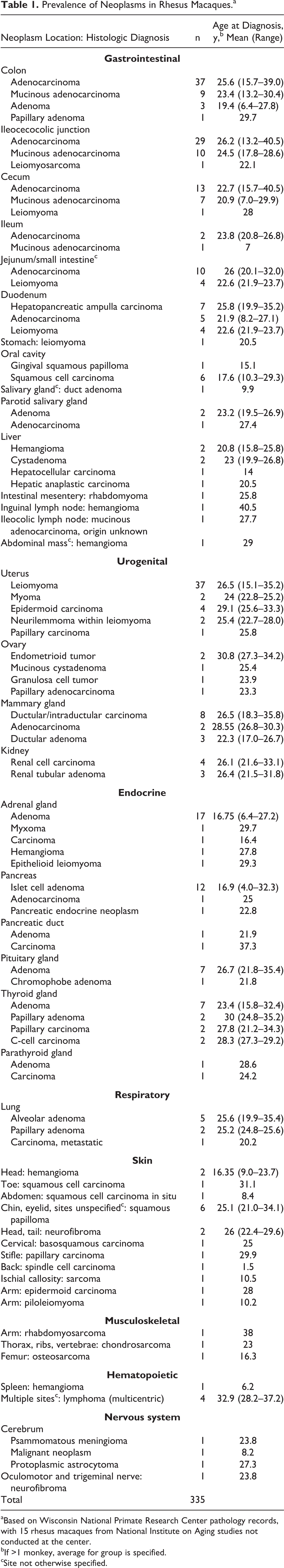
aBased on Wisconsin National Primate Research Center pathology records, with 15 rhesus macaques from National Institute on Aging studies not conducted at the center.
bIf >1 monkey, average for group is specified.
cSite not otherwise specified.
Surgical excision with intestinal resection and anastomosis remains the preferred treatment for intestinal adenocarcinoma in rhesus macaques in the WNPRC breeding colony and for individuals on long-term projects with Institutional Animal Care and Use Committee protocols that include approval for surgical interventions. Postsurgical survival (> 5 days) was determined for 25 rhesus macaques with intestinal adenocarcinoma (Table 2). At the time that this article was prepared, 12% (3 of 25) of animals with surgical resection of intestinal adenocarcinomas were alive, with a mean survival time of 1.5 years. The other 22 of 25 animals were euthanized due to deterioration of clinical health. Surgical excision was determined to be curative in 55% (12 of 22) of cases with no gross or histologic evidence of recurrence at the time of necropsy. The presence of serosal and mesenteric invasion with metastasis was noted in 55% (12 of 25) of cases at the time of biopsy (surgical excision) but was not predictive of recurrence or metastasis at the time of necropsy. Sites of metastasis included mesenteric and regional lymph nodes, omentum, liver, pancreas, uterus, spleen, diaphragm, and lungs.
Postsurgical Survival of Rhesus Macaques With Intestinal Adenocarcinoma.a

aAnimals that survived <5 days postsurgery are excluded. Data are from Wisconsin National Primate Research Center biopsy and necropsy records between October 1, 1996, and August 1, 2015. All animals were euthanized due to deterioration of clinical health. Values presented as number of rhesus macaques unless noted otherwise.
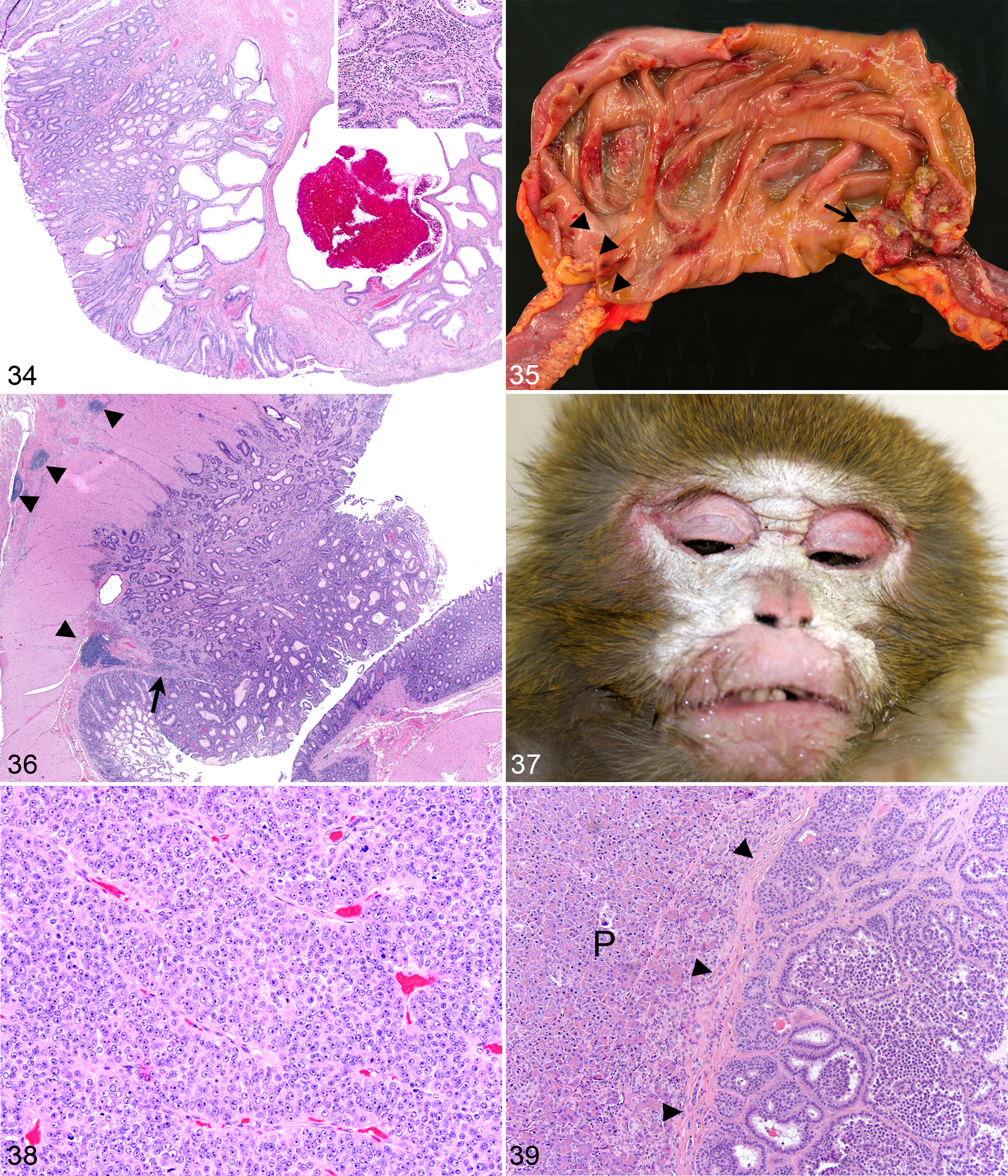
Paraneoplastic syndromes are best described in humans and dogs but also affect the cat and horse. 48,50 These syndromes occur in approximately 10% of people with malignant neoplasms and may be the earliest manifestation of disease. 48 Endocrinopathies are often encountered, and hypercalcemia is the most common paraneoplastic syndrome. 48 There has been 1 case of a paraneoplastic syndrome in a rhesus macaque at the WNPRC, characterized by severe cutaneous hyperkeratosis of the face, flanks, and legs (Fig. 37). Histologically, there was severe orthokeratotic hyperkeratosis, epidermal hyperplasia, superficial lymphoplasmacytic dermatitis, and rare intracorneal pustules. The initial biopsy diagnosis was a metastatic carcinoma in the axillary lymph node (Fig. 38). Thoracic and abdominal radiographs, thoracic and abdominal ultrasound, positron emission tomography scan, and abdominal exploratory surgery failed to identify the primary neoplasm, but the primary tumor, a 1-cm nodular pancreatic mass adjacent to the primary pancreatic duct, was identified during necropsy (Fig. 39). Because definitive diagnosis of a paraneoplastic syndrome is made through the treatment and/or removal of the primary tumor with subsequent resolution of clinical signs, this is a presumptive case of a paraneoplastic syndrome due to a pancreatic endocrine neoplasm.
Summary and Conclusions
The rhesus macaque is one of the most extensively used nonhuman primate models for human diseases. It is hoped that this combined literature review and records from the WNPRC colony will serve as a resource for pathologists and investigators working with rhesus macaques of all ages. There is great value in cross-sectional studies of populations for identification of morbidities and mortalities at different ages. Banking samples and data for retrospective studies are absolutely necessary to maximize the scientific value of rhesus macaques. Longitudinal studies in rhesus macaques will continue to be necessary to properly define mechanisms of maturation, aging, and age-associated diseases to better serve ageing populations.
Footnotes
Acknowledgements
No retrospective study can be performed without the work of one’s predecessors and the support of current colleagues, especially veterinary staff, animal care staff, behavioral management, colony managers, and the many investigators who work with nonhuman primates. I specifically thank Robert A. Becker for invaluable assistance with figures; Peter Pierre, Ruth Sullivan, and Susan M. Williams for editorial assistance; and the pathologists and residents of the WNPRC: James R. Allen, Sang Kee Paik, Etsuro Uemura, Hideo Uno, James A. Thomson, Sheree Beem, Prachi Sharma, Amy L. Usborne, Ruth Hurley, Daniel I. Shenkman, Raman Muthuswamy, and Andres F. Mejia.
