Abstract
Lymphoproliferative disorders of natural killer (NK)-cell lineage are well documented in humans but have yet to be documented in non-human primates (NHPs). Here we describe a case of NK-cell lymphoproliferative disorder/leukemia in a 20-y-old captive female rhesus macaque (Macaca mulatta). The animal clinically had mild splenomegaly and marked lymphocytosis with small-to-medium lymphocytes in blood smears. By flow cytometry and cluster differentiation, the lymphocytes were CD3-negative, CD8-positive, CD4-negative, and CD20-negative for cell surface markers; immunohistochemistry revealed the presence of intracellular CD3 and granzyme B. This immunoprofile is consistent with a NK-cell phenotype. Histologically, these cells were predominantly intravascular within the splenic red pulp, liver sinusoids, and to a lesser degree bone marrow. Oncogenic viruses, such as Mason-Pfizer monkey viruses (MPMV; formerly, and commonly known as, simian retroviruses or SRV; Retroviridae, Betaretrovirus maspfimon); simian immunodeficiency virus (SIV; Retroviridae, Lentivirus simimdef), and primate T-lymphotropic virus 1 (PTLV1; commonly known as simian T-lymphotropic virus type 1, STLV1; Retroviridae, Deltaretrovirus priTlym1), were not detected in this animal by serology. Immunohistochemistry using EBNA2 antibody to detect rhesus and cynomolgus monkey lymphocryptovirus (McGHV4/RLV and McGHV10 respectively; Orthoherpesviridae, Lymphocryptovirus macacinegamma4 and Lymphocryptovirus macacinegamma13, respectively) was negative. Together these findings are consistent with a diagnosis of naturally occurring NK-cell lymphoproliferative disorder. NK-cell lymphoproliferative disorder has not been reported previously in rhesus macaques, to our knowledge.
Natural killer (NK) cells are cells of the innate immune response that have cytotoxic activity similar to CD8+ cytotoxic T lymphocytes. They have similar anti-viral and anti-tumoral functions, and share common markers with T cells. 6 Both in animals and humans, identifying NK cells is complicated by the lack of unique NK-cell markers; the existence of subpopulations of NK cells also complexifies their identification.6,16 CD56 is a widely used NK-cell marker in humans but is a monocyte rather than a lymphocyte marker in rhesus macaques. 2 Other NK-cell markers such as CD94/NKG2A and CD16 also have disputed expression patterns in NK cells of non-human primates (NHPs), which means that, in practice, these cells are typically isolated by using combinations of markers. 16
The World Health Organization (WHO) classification of human hematolymphoid tumors separates NK-cell neoplasia into large granular lymphocytic leukemia (NK-LGLL), aggressive NK-cell leukemia (ANKL), indolent NK-cell lymphoproliferative disorders of the gastrointestinal tract, and extranodal NK/T-cell lymphoma (ENKTL). 1 Both ANKL and ENKTL are clinically aggressive and often associated with infection with human gammaherpesvirus 4 (HuGHV4; formerly, and commonly known as Epstein-Barr virus, EBV; Orthoherpesviridae, Lymphocryptovirus humangamma4), an oncogenic herpesvirus.
Similar to humans, lymphoproliferative disorders also occur in NHPs. 12 Retroviruses, such as primate T-lymphotropic virus 1 (PTLV1; commonly known as simian T-lymphotropic virus type 1; Retroviridae, Deltaretrovirus priTlym1), and gammaherpesviruses, such as lymphocryptoviruses (LCVs) and rhadinoviruses, are often reported in association with various lymphomas.5,8,9,15 LCVs have genomic and biologic similarities with human EBV, and can immortalize B cells.3,13 Although most of the reports are of B-cell or T-cell lymphoma, NK-cell lymphoma is incredibly rare; only 3 cases of simian EBV-associated NK/T-cell lymphoma have been reported in Japanese macaques.9,15
In contrast, reports of lymphoproliferative disorders in NHPs seronegative for the common oncogenic viruses are sparse. Reports include a T-cell lymphoproliferative disorder in a rhesus macaque seronegative for oncogenic herpesviruses and PTLV1, and one case of chronic T-cell leukemia in a rhesus macaque seronegative for macacine alphaherpesvirus 1 (McAHV1; also known as herpes B virus; Orthoherpesviridae, Simplexvirus macacinealpha1), PTLV1, simian immunodeficiency virus (SIV; Retroviridae, Lentivirus simimdef), and Mason-Pfizer monkey viruses (MPMVs; formerly, and commonly known as, simian retroviruses; Retroviridae, Betaretrovirus maspfimon).4,7
Here we report a case of NK-cell leukemia in a captive rhesus macaque not associated with a viral etiology. We retrieved no cases of spontaneous NK-cell lymphoproliferative disorder in rhesus monkeys in a search of Google, PubMed, CABI Direct, Web of Science, and Scopus using the search terms “NK cell lymphoproliferative disorder” or “NK cell leukemia” and “rhesus monkey” or “rhesus macaque” or “Macaca mulatta,” suggesting that this condition has not been reported in rhesus monkeys.
A 20-y-old, indoor-housed, female rhesus macaque at the California National Primate Research Center (CNPRC) was cared for in accordance with the Animal Welfare Act and Guide for the Care and Use of Laboratory Animals. 14 The animal care and use program at the University of California–Davis (Davis, CA, USA) is USDA-registered, maintains a Public Health Services Assurance, and is fully AAALAC-accredited. During routine geriatric examination, there was mild splenomegaly, and severe leukocytosis (92.3 × 109/L; RI: 5.3–11.3 × 109/L) composed of 95% lymphocytes (87.7 × 109/L; RI: 1.3–5.1 × 109/L). Repeat bloodwork 4 d later revealed increased leukocytosis (103 × 109/L; RI: 5.3–11.3 × 109/L) composed of 98% lymphocytes (101 × 109/L; RI: 1.3–5.1 × 109/L), with mild neutropenia (1.0 × 109/L; RI: 2.5–5.7 × 109/L), and slight normocytic normochromic anemia with low hemoglobin at 100 g/L (RI: 137–155 g/L) and hematocrit of 33 L/L (RI: 41–48 L/L). On blood smears, the lymphocytes were mature with morphology within normal limits. By flow cytometry for cell surface markers, this lymphocyte population was CD3–/CD8+/CD4–/CD20– (Fig. 1). The absence of surface CD3 T-cell receptor (TCR) is inconsistent with a T-cell phenotype. Instead, this profile of cell surface markers suggested a NK-cell phenotype. Based on the presumptive diagnosis of a lymphoproliferative disorder of NK-cell lineage, euthanasia was elected.
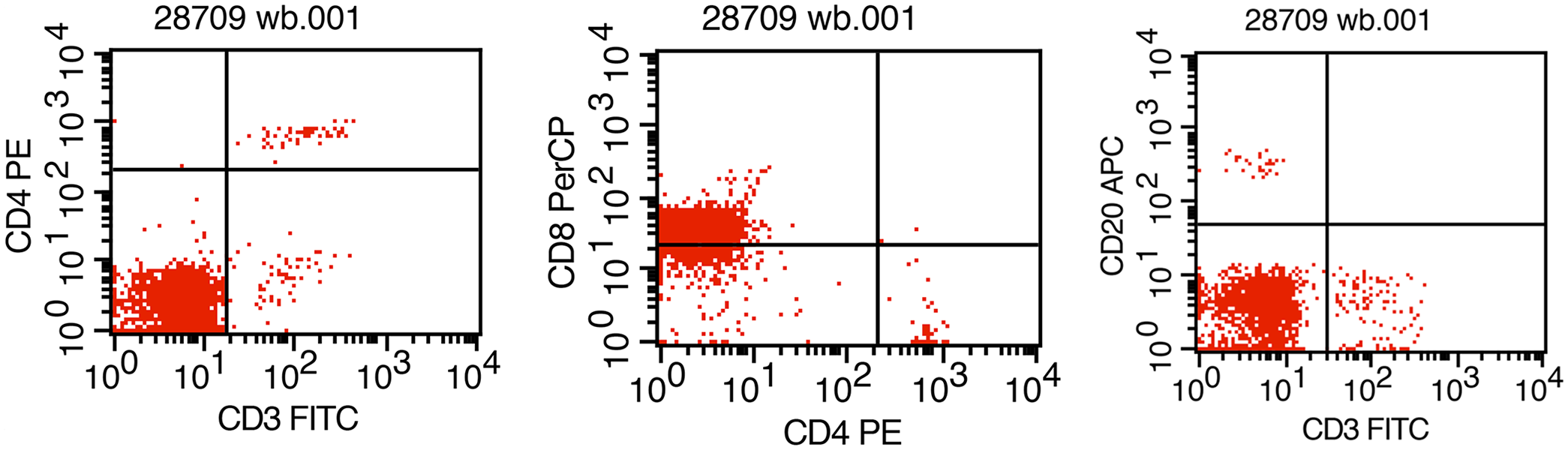
Flow cytometry of the blood from a rhesus macaque with a spontaneous natural killer–cell lymphoproliferative disorder. The cells of interest are CD3-negative, CD8-positive, CD4-negative, and CD20-negative for cell surface markers.
At postmortem examination, the spleen was diffusely enlarged, dark red, and measured 6.1 × 2.4 × 1.9 cm (Fig. 2). Regionally, the capsular surface was multifocally pale-gray to purple, and the parenchyma was slightly firm, diffusely dark-red, and had indistinct lymphoid follicles. The liver weighed 180 g (2.1% of body weight; RI: 2–2.5% based on unpublished data collected at the CNPRC) and was pale-brown throughout with sharp margins. All peripheral and intracavitary lymph nodes were homogeneously pale-tan and <1.5-cm diameter, considered to be within normal limits. Tissues and organ samples were collected, fixed by immersion in 10% neutral-buffered formalin, and routinely processed to produce 4-μm thick, H&E-stained sections.
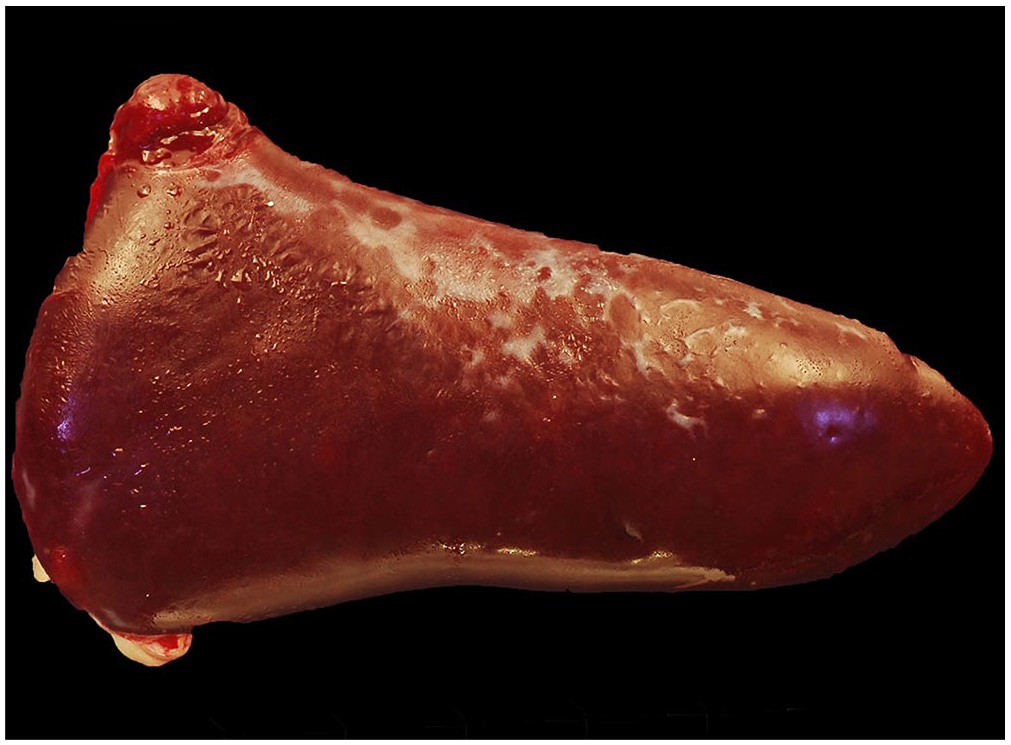
Spleen from a rhesus macaque with a spontaneous natural killer–cell lymphoproliferative disorder is diffusely enlarged, dark-red, and 6.1 × 2.4 × 1.9 cm with numerous pale-gray areas on the capsular surface.
Histologically, the red pulp sinusoidal architecture in the spleen was accentuated by a population of intravascular round cells that expanded the lumina and were ~1–1.5× the diameter of a RBC (Fig. 3A). Round cells had distinct cell borders and a high nuclear:cytoplasmic ratio. Nuclei were irregularly round with coarsely stippled chromatin and indistinct nucleoli. Anisocytosis and anisokaryosis were mild, and mitotic figures were not observed (Fig. 3B). Sinusoids were lined by reactive endothelial cells with a moderate amount of smudgy eosinophilic cytoplasm and plump nuclei. White pulp lymphoid follicles were within normal limits.
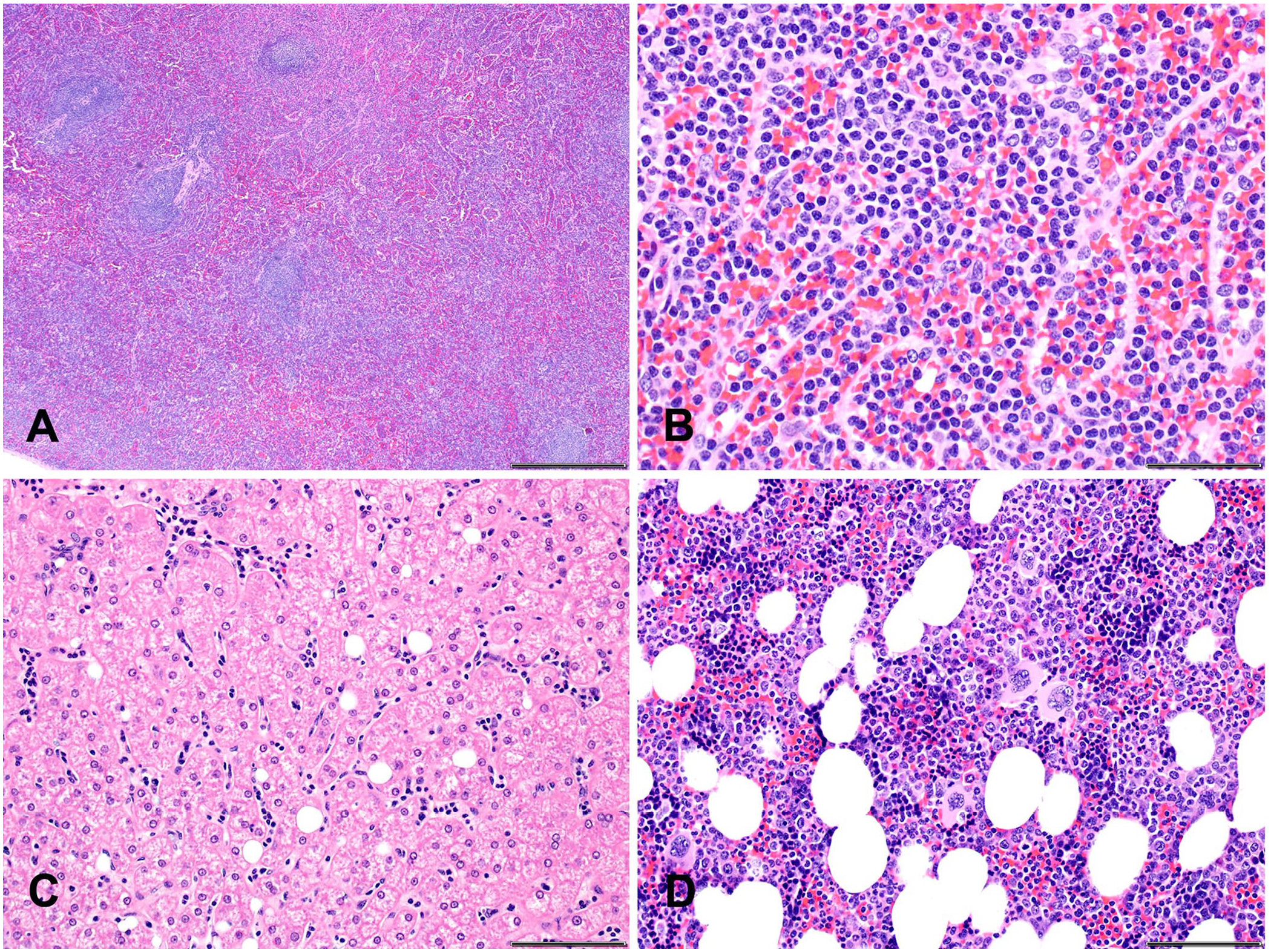
Histologic findings in a rhesus macaque with a spontaneous natural killer–cell lymphoproliferative disorder.
Hepatic sinusoids contained moderate numbers of intravascular round cells similar to those described within the spleen (Fig. 3C). The bone marrow was moderately hypercellular. Both the myeloid and erythroid lineages were present in appropriate amounts with a myeloid-to-erythroid ratio of ~3:1 (Fig. 3D). Scattered throughout were occasional low numbers of lymphocytes with morphologic features similar to those described in the spleen. Together, the presence of bland small round cells within the bone marrow without effacing the bone marrow architecture, similar cells in the splenic sinusoids and hepatic sinusoids, and the absence of lymphadenopathy, suggests leukemia.
Immunohistochemistry (IHC) for granzyme B, CD3, and CD20 was performed on formalin-fixed, paraffin-embedded sections of spleen and bone marrow. The infiltrating population of cells had moderate-to-strong intracytoplasmic immunoreactivity to CD3 (Fig. 4A). Granzyme B IHC demonstrated diffuse, punctate, intracytoplasmic immunoreactivity (Fig. 4B). The cells were immunonegative for the B-cell lymphocyte marker CD20. CD20 IHC highlighted germinal centers in the spleen. Flow cytometry was repeated on total cells isolated from blood and spleen collected at postmortem examination. Similar to antemortem flow cytometry data, the cells of interest were CD3–/CD8+/CD4–/CD20–. As mentioned previously, the absence of TCR-CD3 at the cell surface is inconsistent with a T-cell phenotype; however, the absence of surface CD3 with the presence of surface CD8 suggested a NK-cell phenotype. 11 Furthermore, although NK cells do not express the TCR-CD3 complex on their surface, they are known to contain intracytoplasmic CD3.7,11 Interpreted together, the flow cytometry and IHC results indicate a cell surface immunophenotype CD3–/CD8+/CD4–/CD20– with intracytoplasmic CD3 and granzyme B, which is consistent with a NK-cell phenotype.6,11 The marked leukocytosis with the distribution of the cells in the spleen, bone marrow, hepatic sinusoids, and lack of lymph node involvement are consistent with a diagnosis of NK-cell lymphoproliferative disorder or leukemia with similarities to NK-LGLL in humans. 10
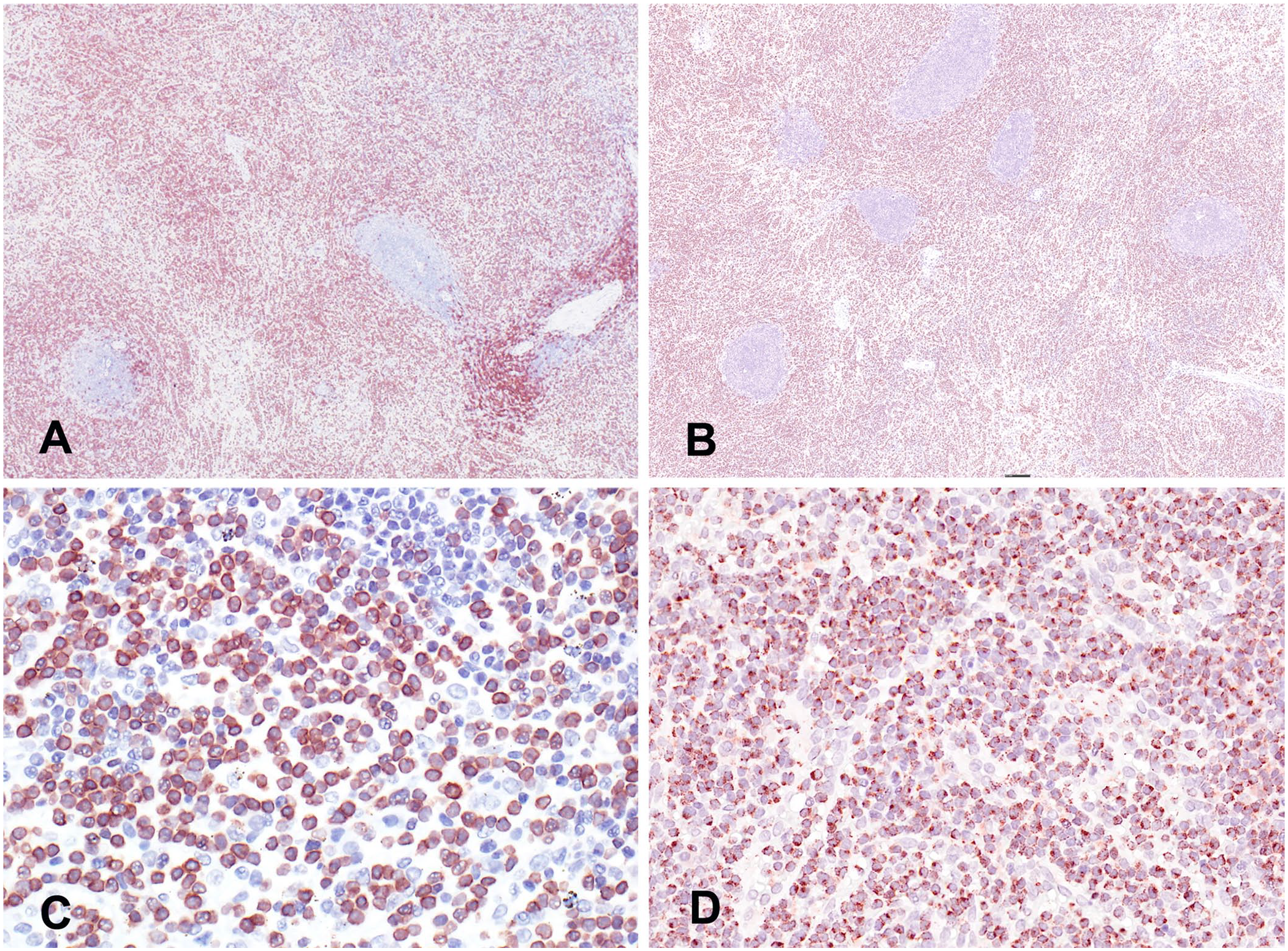
Immunohistochemistry (IHC) of samples of spleen from a rhesus macaque with a spontaneous natural killer (NK)-cell lymphoproliferative disorder.
The animal was seronegative for oncogenic viruses often associated with lymphoproliferative disorders in NHPs: MPMV, no specific serotype; SIV; and PTLV1. Although not considered oncogenic and therefore unrelated, this animal was seropositive for McAHV1, macacine betaherpesvirus 3 (McBHV3; formerly, rhesus cytomegalovirus; Orthoherpesviridae, Cytomegalovirus macacinebeta3), and simian foamy virus (SFV; Retroviridae, Simiispumavirus). IHC using an EBV-encoded nuclear antigen 2 antibody (EBNA2) that cross-reacts with LCVs was performed on the spleen and bone marrow. No immunoreactivity was observed, supporting the absence of LCV and simian EBV homologues in the neoplastic population.
Leukemia is defined as a clonal proliferation of malignant progenitor cells in the blood and/or blood-forming organs. 1 Detecting clonality is based on molecular techniques detecting T cell or immunoglobulin receptor gene rearrangements. Unlike T or B lymphocytes, NK cells do not rearrange such receptors, making the proof of clonality difficult. 6 This is further complicated by the overall lack of available PCR primers needed to detect clonality in the rhesus macaque. The distinction between clonal and reactive NK-cell expansion can be observed in several situations, such as autoimmune diseases and viral infections, thus the distinction between the two can be challenging without receptor rearrangement. Hence, we chose to use “lymphoproliferative disorder or leukemia” for our case.
Our case had similarities with NK-LGLL in humans, which occurs mainly in adult patients with mild-to-moderate lymphocytosis and splenomegaly, and in which lymphadenopathy and hepatomegaly are usually absent. Neoplastic cell infiltrates are usually also present in the spleen and liver, and despite the relative paucity of marrow infiltration, neutropenia and anemia are often present. 1 In humans, the identification of recurrent mutations provides new tools to prove clonality, to further recognize these important diseases. 10 Although these tools are not available for NHPs, lymphoproliferative disorders consisting of NK-cell lineage should be considered on the differential list, particularly if there is concurrent anemia and neutropenia. Testing for an underlying viral etiology should also be performed in all cases of lymphoproliferative disease.
Footnotes
Acknowledgements
We thank Sarah Lockwood for excellent technical assistance; Dr. Zhong-Min Ma for invaluable help with the figures; Lindsay Parmelee at the Integrated Pathology Core of the Oregon National Primate Research Center for the EBNA2 IHC; and Abigail Spinner and Dr. Peter Moore for helpful discussions.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research did not receive any specific grant from the public, commercial, or not-for-profit funding agencies. We received CNPRC base grant P51 OD011107.
