Abstract
Murine noroviruses (MNVs) are highly prevalent in laboratory mice, can cause persistent infections, and have been shown to infect macrophages, dendritic cells, and B cells. To address the potential impact of MNV infection on research outcomes, numerous studies have been conducted with various mouse models of human disease and have generated mixed results, ranging from no impact to significant disease. Many of these studies included histologic evaluations after MNV infection, and these results have similarly been variable in terms of whether MNV induces lesions, despite the fact that localization of MNV by viral culture and molecular techniques have demonstrated systemic distribution regardless of mouse immune status. The aim of this review is to summarize the histologic findings that have been reported with MNV infection in several mouse models. The studies demonstrate that experimental infection of MNV in wild-type mice results in minimal to no histologic changes. In contrast, immunodeficient mice consistently have detectable MNV-induced lesions that are typically inflammatory and, in the most severe cases, accompanied by necrosis. In these, the liver is commonly affected, with more variable lesions reported in the lung, gastrointestinal tract, mesenteric lymph nodes, brain, and spleen. In specific disease models including atherosclerosis, MNV infection had a variable impact that was dependent on the mouse model, viral strain, timing of infection, or other experimental variables. It is important to recognize the reported MNV lesions to help discern the possible influence of MNV infection on data generated in mouse models.
Keywords
Noroviruses are nonenveloped, single-stranded, positive-sense RNA viruses in the
Prevalence
MNV was the first norovirus to be grown in cell culture. 50 The ability to grow the virus in vitro allowed for the development of diagnostic assays (including immunohistochemistry) to detect viral antigens, serologic assays for detection of anti-MNV antibodies, and molecular assays to detect viral RNA. 9,15,16,22,49 Initial large-scale prevalence data on MNV infection revealed that 22.1% of 12 639 serum samples from laboratory mice in the United States and Canada were positive for antibodies to MNV. 16 Another large-scale study likewise confirmed the high prevalence of MNV infection, with a rate of 32.4% for laboratory mice in North America and Europe, and other studies have reported rates as high as 64%. 26,27,32,40 These data indicate that MNV infection in laboratory mice is widespread and worldwide and therefore has the potential to interfere with research results.
Effects on Research
As a result of this novel discovery of a norovirus in laboratory mice, the variable clinical disease seen depending on the type of mouse infected, and the high prevalence of infection in research mice, further study is warranted to reveal and characterize the potential impact of this virus on research using MNV-infected mice. Importantly, MNV has a tropism to infect macrophages and dendritic cells and, more recently, has been reported to infect B cells.
18,50
Infection of immune cells therefore raises the possibility that MNV may be a confounding factor in mouse models of inflammatory disease. Surprisingly, results have been variable in that some mouse models are altered by MNV infection, while others do not show any changes. For example, MNV has been shown to cause Paneth cell abnormalities in a mouse model of Crohn’s disease and to exacerbate inflammatory bowel disease (IBD) in a
Histologic Lesions Caused by MNV Infection in Mice
As described above, MNV infection may alter some mouse models of disease but may have little or no impact on other models. This variable response may be due to a number of factors, including the particular disease model being studied or differences in pathogenicity of the infecting MNV strain. A large number of MNV strains have been described and are likely the result of genetic recombination and/or the lack of a proofreading activity in the RNA-dependent RNA polymerase of RNA viruses. 2,6,14,32 Therefore, determining whether MNV infection is a confounding factor in a particular research study using infected mice should be evaluated on a case-by-case basis.
A comprehensive summary has been compiled of whether histologic changes have been reported in particular tissues as a result of MNV infection in laboratory mice (Tables 1 –5). Additional information, including histologic changes, and detailed methods, including infectious challenge, the mouse genetic background, sex, health status, housing type (if reported), and any other infections or treatments, are provided in the Supplemental Tables 1–5. The intent of these summary tables is to assist the pathologist, veterinarian, and scientist with identifying the currently available and published information on the histologic findings in MNV-infected mice. The intent is not a comprehensive review of MNV itself, and so the reader is referred to a number of excellent reviews for more information on MNV and noroviruses in general. 20,21,23,51
Histologic Changes in Mice Naturally Infected With MNV.

Abbreviations: +, histologic changes reported; –, no histologic changes; MLN, mesenteric lymph node; MNV, murine norovirus; n/r, not reported; perit, peritoneum; ref, reference.
Histologic Changes in Wild-Type Mice Experimentally Infected With MNV.

Abbreviations: +, histologic changes reported; –, no histologic changes; MLN, mesenteric lymph node; MNV, murine norovirus; n/r, not reported; perit, peritoneum; ref, reference.
Histologic Changes in Immunodeficient Mice Experimentally Infected With MNV.
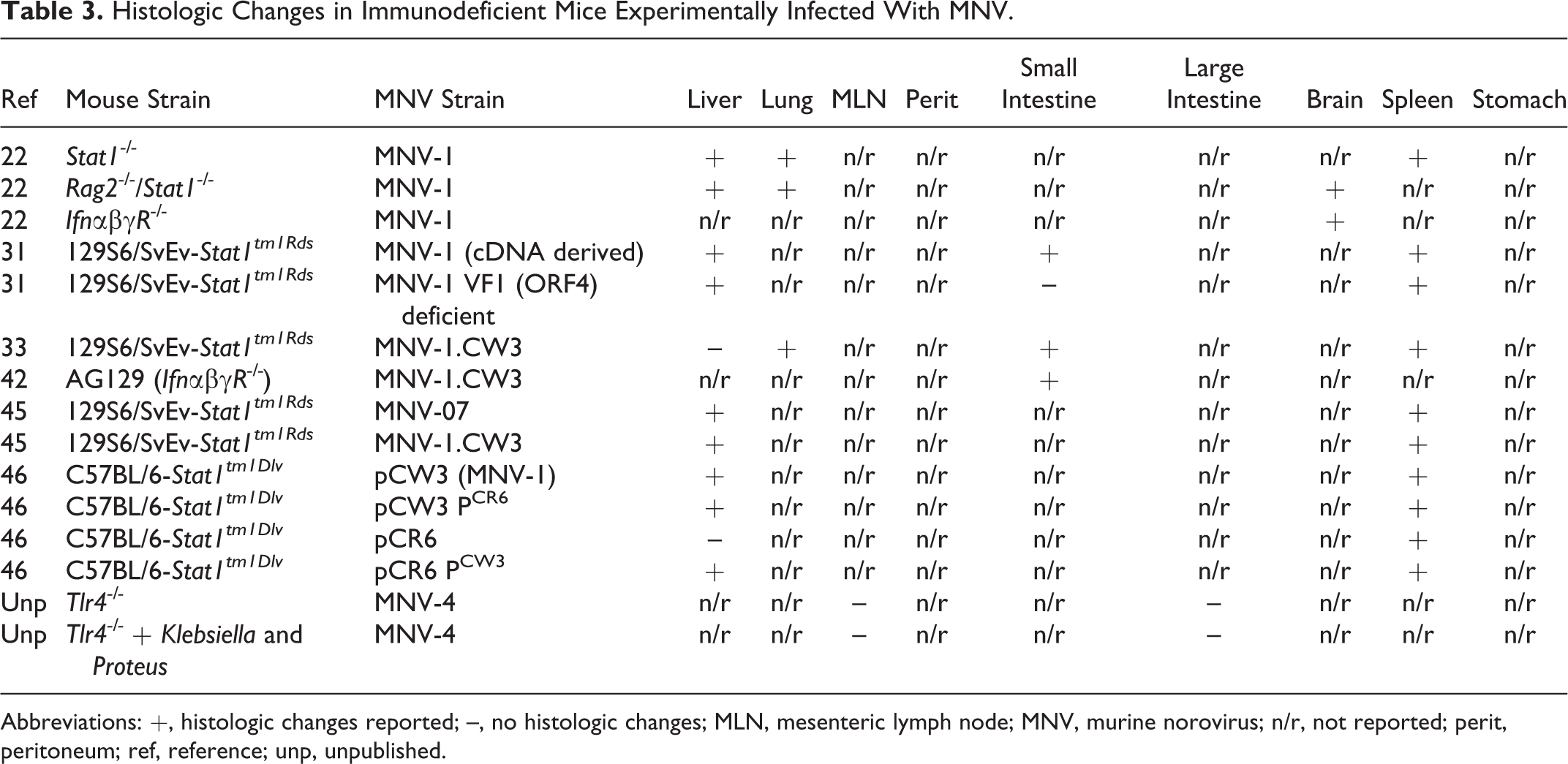
Abbreviations: +, histologic changes reported; –, no histologic changes; MLN, mesenteric lymph node; MNV, murine norovirus; n/r, not reported; perit, peritoneum; ref, reference; unp, unpublished.
Histologic Changes in Mice of Gastrointestinal Disease Models Experimentally Infected With MNV.
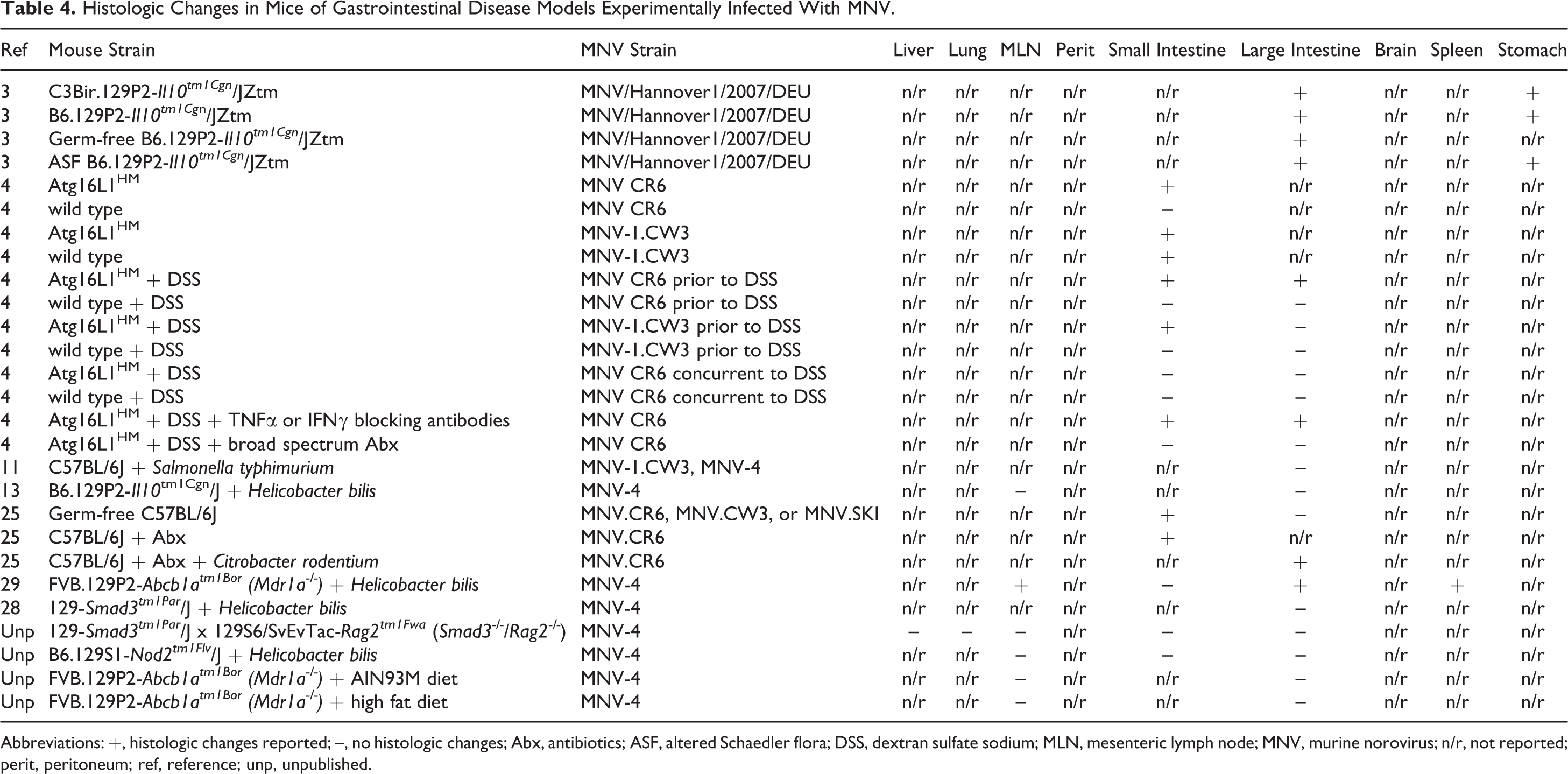
Abbreviations: +, histologic changes reported; –, no histologic changes; Abx, antibiotics; ASF, altered Schaedler flora; DSS, dextran sulfate sodium; MLN, mesenteric lymph node; MNV, murine norovirus; n/r, not reported; perit, peritoneum; ref, reference; unp, unpublished.
Histologic Changes in Mice of Other Disease Models Experimentally Infected with MNV.
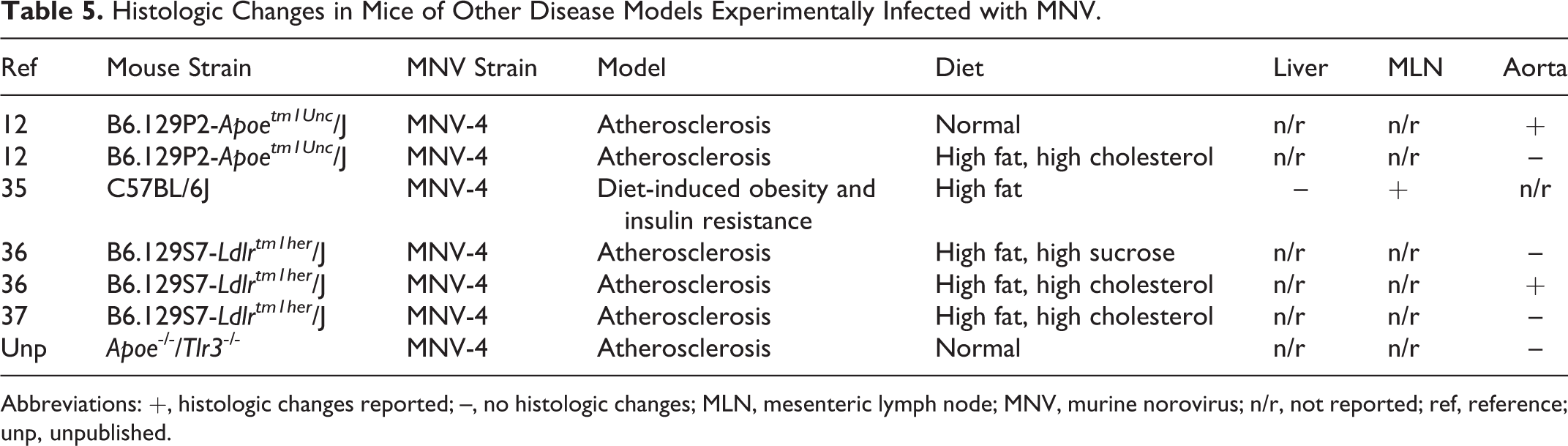
Abbreviations: +, histologic changes reported; –, no histologic changes; MLN, mesenteric lymph node; MNV, murine norovirus; n/r, not reported; ref, reference; unp, unpublished.
The histologic changes seen in MNV-infected mice from endemic colonies with natural exposure (ie, via other infected animals) are summarized in Table 1. For these studies, the dose and route of inoculation are not controlled, and the MNV strain or isolate is often unknown or uncharacterized. Generally, MNV-associated liver lesions are significant only in immunodeficient
Controlled experimental MNV infections in wild-type mice allow for the evaluation of histologic lesions attributable to MNV without other confounding variables, such as immunodeficiency, unknown inoculating dose, viral strain, or timing of infection (Table 2). The majority of studies conducted in wild-type mice infected with MNV show no overt clinical signs of disease or histologic lesions. However, histologic changes were recently reported in wild-type 129 mice, including mild to moderate lesions in the liver (defined as 1–2 or 5–10 foci of inflammatory cells per 10 fields at 10× objective, respectively) and reactive changes in the lymphoid tissues, including enlargement of small intestinal Peyer patches with increased germinal centers, splenic red pulp hyperplasia, and activation of the white pulp. 45 Therefore, the strain or stock of the wild-type mouse and the particular infecting MNV strain or isolate are likely important determinants of whether histologic changes will be seen after infection.
In contrast to immunocompetent mice, experimental infection of mice with known immunodeficiencies results in significant disease (Table 3). Most of these reports involve mice that lack genes important in the type I and type II interferon response to viruses, such as
Since noroviruses are gastrointestinal pathogens, much of the research on the impact of MNV has been conducted on mouse models of gastrointestinal disease (Table 4), including dextran sodium sulfate (DSS)–induced colitis, mice with disrupted expression of the autophagy related 16–like 1 gene (
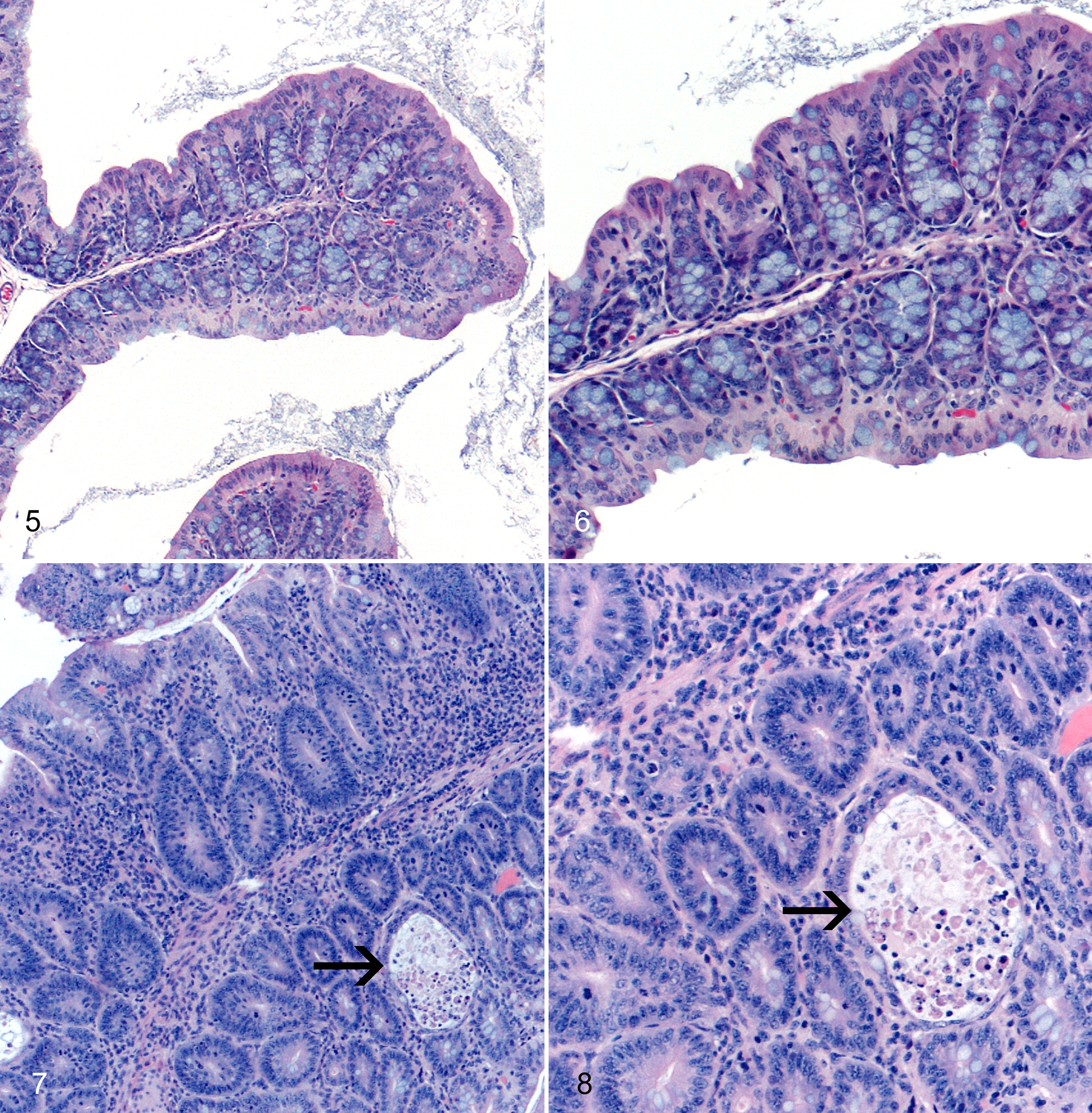
Despite the fact that noroviruses are gastrointestinal pathogens, MNV may infect research mice used to study a wide array of diseases beyond the gastrointestinal system. Since MNV is highly prevalent in many research institutions and has been reported to infect macrophages, dendritic cells, and B cells, MNV has the potential to alter research outcomes in diseases for which these cells are important, including atherosclerosis, diabetes, and obesity (Table 5). Our laboratory has evaluated the impact of MNV infection on atherosclerosis in B6.129P2-
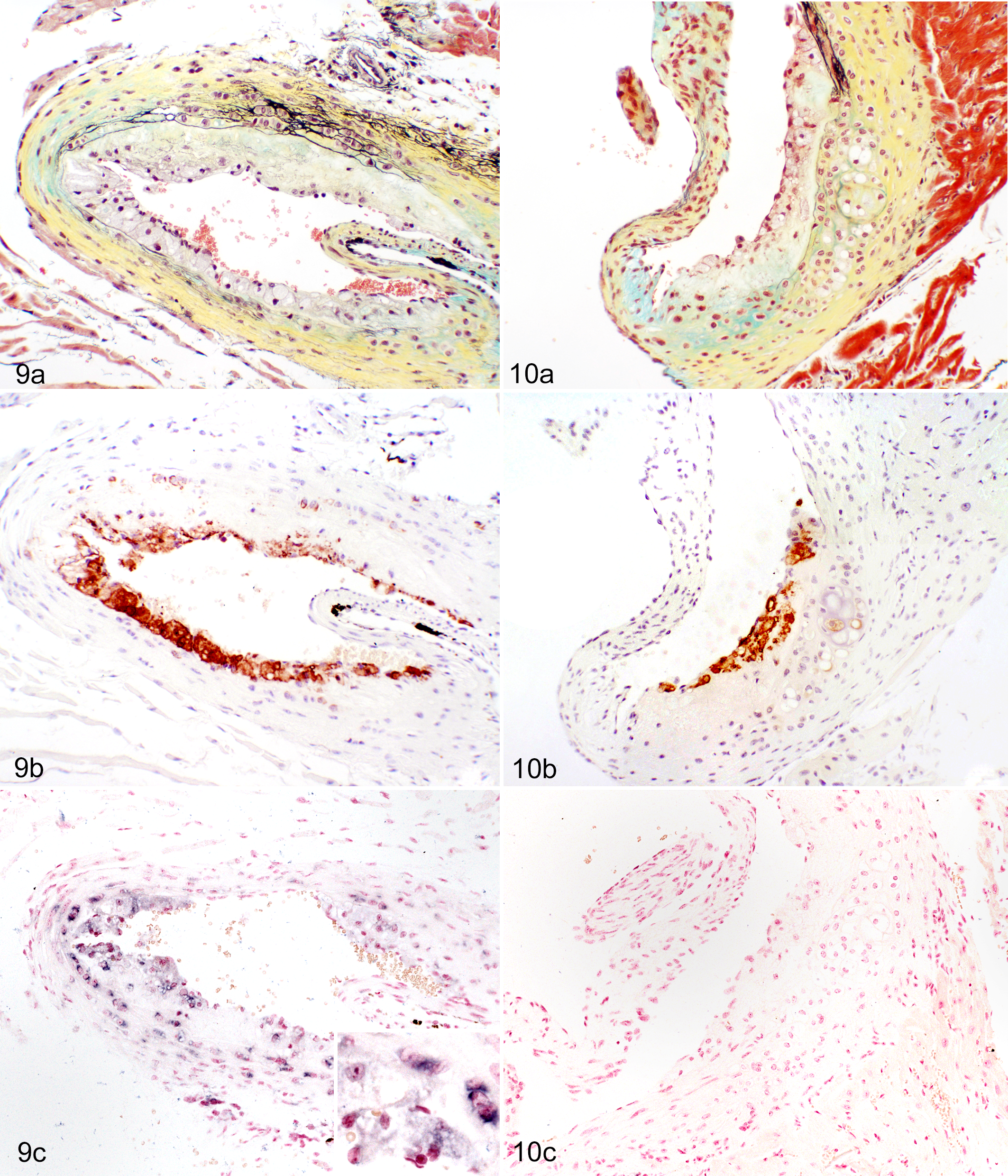
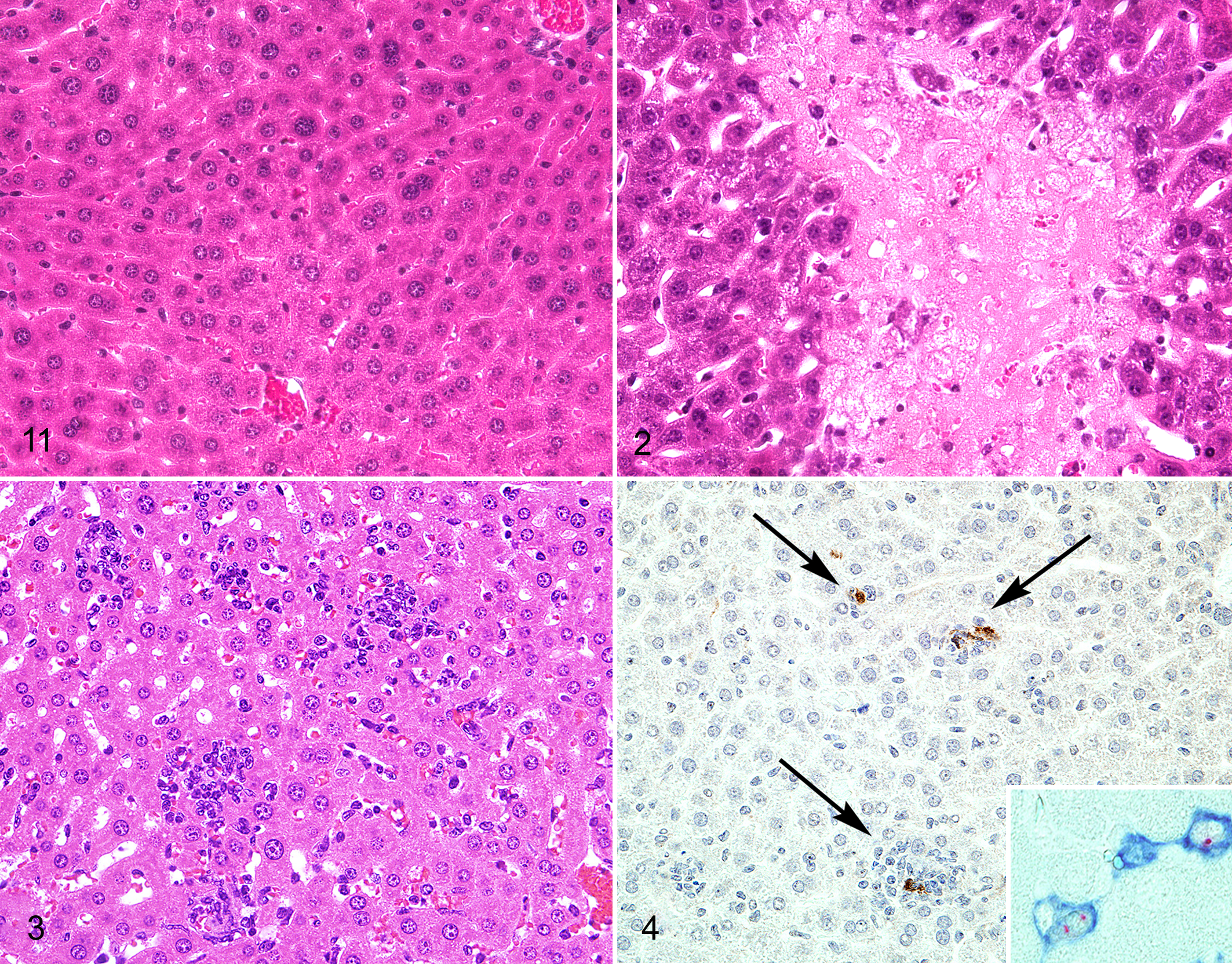
Many studies have employed immunohistochemistry, immunofluorescence, in situ hybridization, and in situ reverse transcription polymerase-chain reaction to localize virus in tissue sections derived from MNV-infected mice (Figs. 4, 9c). These studies confirm that MNV can infect macrophages and dendritic cells in a variety of tissues, including liver, lung, mesenteric lymph node, peritoneum, small intestine, spleen, and aorta, indicating that systemic spread of the virus may occur in both immunocompetent and immunodeficient mice (Table 6). In the liver, MNV antigen was detected in Kupffer cells 38,47,50 and inflammatory cells, including F4/80+ macrophages. 49 MNV antigen was detected in mesenteric lymph node DC-like cells, F4/80+ cells in the medullary cords, and CD40+ paracortical cells in alveolar mononuclear cells and pleural inflammatory cells, peritoneal macrophages, and mesothelium. 49 Studies examining the spleen demonstrated MNV antigen in macrophages and macrophage-like cells, 47 red pulp and marginal zone, 50 and nonlymphoid cells in the white pulp. 50 Although the majority of MNV+ cells in the small intestine are inflammatory or antigen-presenting cells within the lamina propria or gastrointestinal-associated lymphoid tissues, enteric epithelium was also positive in some studies. 3,33,49
Localization of MNV by Immunohistochemistry, Immunofluorescence, In Situ Hybridization, or In Situ RT-PCR.
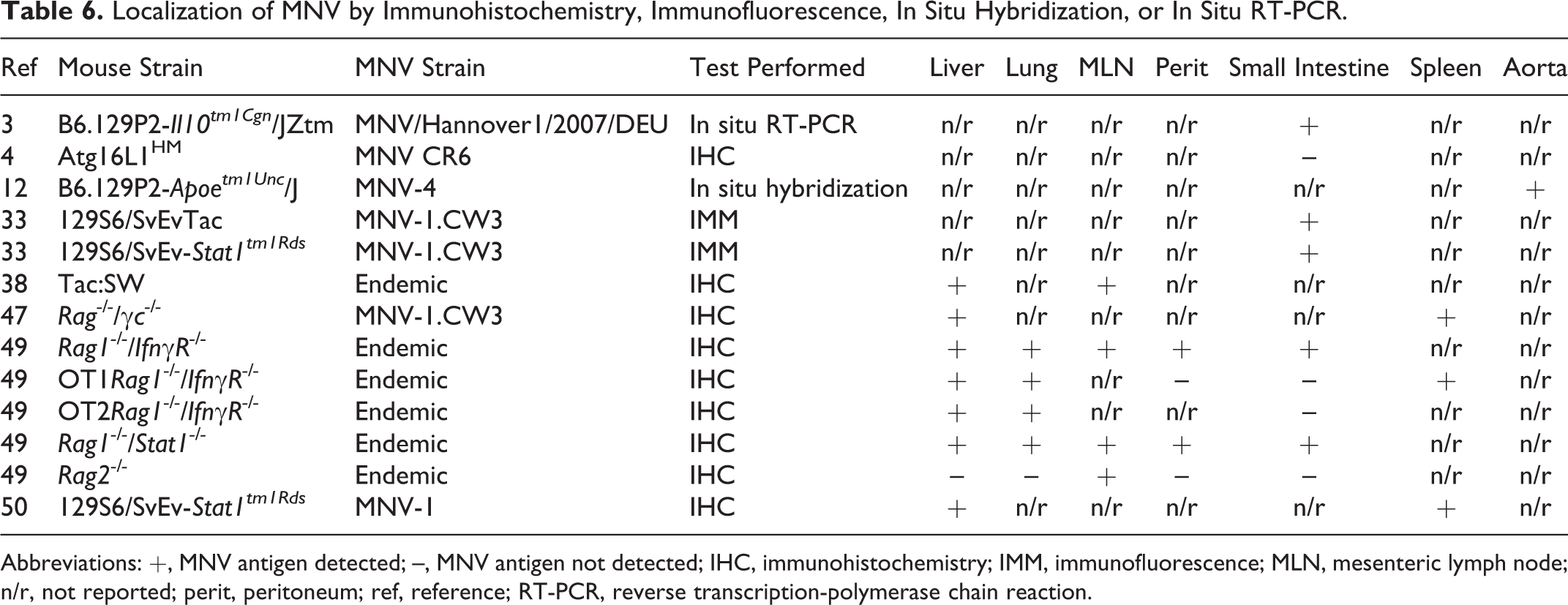
Abbreviations: +, MNV antigen detected; –, MNV antigen not detected; IHC, immunohistochemistry; IMM, immunofluorescence; MLN, mesenteric lymph node; n/r, not reported; perit, peritoneum; ref, reference; RT-PCR, reverse transcription-polymerase chain reaction.
Conclusion
MNV infection in laboratory mice continues to be endemic in many research colonies. Therefore, determining the impact that MNV infection may have on various mouse models of disease is important to ensure that research results are valid. While it is impractical to test whether MNV infection alters all mouse models of disease, more and more studies are being performed to answer the following question: “Will MNV infection alter and confound my research results?” MNV is detected and may induce lesions in a wide variety of tissues, principally liver, spleen, and gastrointestinal tract, from immunocompetent and immunodeficient mice where it infects mononuclear cells, including macrophages and dendritic cells. Importantly, MNV has a variable impact on mouse models, in some cases increasing lesions while in other cases having no impact. Consequently, it is important to recognize reported MNV lesions to help discern the possible influence of MNV infection on data generated in mouse models.
Footnotes
Acknowledgements
We thank Claudia Monaco from the University of Oxford for sharing
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health (grants R21-OD011135, R01-OD011149) and the University of Washington’s Nutrition Obesity Research Center (grant P30-DK035816).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
