Abstract
An isolated population of beluga (Delphinapterus leucas) inhabits the St Lawrence Estuary, Quebec, Canada. This population has failed to recover despite the prohibition of hunting >30 years ago, suggesting the presence of other limiting factors. The authors summarize the reported causes of death and propose risk factors to explain the lack of recovery of this population. From 1983 to 2012, a total of 472 beluga were found stranded. Complete necropsies were carried out on 222 beluga, including 178 adults, 25 juveniles, and 19 newborn calves. Infectious diseases, the most prevalent cause of mortality in this population, accounted for the death of one-third of all beluga (32%). Verminous pneumonia was the cause of mortality of 13 juvenile beluga (52% of juvenile beluga). A total of 39 malignant neoplasms, diagnosed in 35 beluga, caused the death of 31 beluga (20% of beluga >19 years old). Median age at diagnosis of cancer was 48 years (range, 30–61 years). Dystocia and postpartum complications were the cause of death in 18 beluga, accounting for 19% of the females >19 years old examined. The occurrence of parturition-associated complications, as well as mortality of calves <1 year old, have increased recently in this population and may be the probable cause of the recent decrease in the size of this population. One of the hypotheses proposed to explain the unusually high occurrence of some of the pathologic conditions observed in this population is chronic exposure to environmental contaminants.
Keywords
An isolated population of around 900 beluga (Delphinapterus leucas), a medium-sized toothed whale, inhabits the St Lawrence Estuary (SLE), Quebec, Canada. Because the St Lawrence River drains the Great Lakes basin, one of the most industrialized regions in North America, this population has been exposed to anthropogenic contaminants and to various disturbances by human activities. SLE beluga, which were intensively hunted in the 19th and 20th centuries, 129 have been protected under the Fisheries Act since 1979 and were designated as endangered in 1983 by the Committee on the Status of Endangered Wildlife in Canada. This designation was revised to threatened in 2004 following a reevaluation of the population estimate. Reclassification to endangered was recently proposed in 2014. Despite reduction in hunting after 1945 and prohibition of hunting in 1979, this population has failed to recover toward its preexploitation abundance of around 8000 whales. 59,129 The goal of the current St Lawrence beluga recovery strategy is “to restore the beluga population to a level where its survival is no longer threatened by natural and anthropogenic disturbances.” 36 The long-term objective of the recovery program is to increase this population to 70% of its historical size. 36 Expected rates of growth for an unexploited population of beluga whose numbers do not exceed the environmental carrying capacity should be >2.5% with a maximum of 4%. 35 The current population growth, estimated to be <1%, 36 should therefore be considered suboptimal and potentially insufficient to restore this population.
Recent survey analyses suggest that this population has been decreasing since the early 21st century. 55 A population model estimates that in 2012, the total population was 889 whales (95% confidence interval: 672–1167). 110 The recent decline proposed by population models and surveys suggests that new threats are affecting reproductive success and recruitment in the SLE beluga population. Causes of death have been monitored since 1983. An ongoing necropsy program—which provides one of the longest uninterrupted data series on causes of mortality in a population of cetaceans—has identified various pathologic conditions and threats that could have significant effects on the survival of this population.*
In this review, we summarize the causes of death and other significant findings reported in this population from 1983 to 2012. Beluga carcasses were obtained through an ongoing, relatively consistent effort in monitoring stranding events. All confirmed or suspected strandings of beluga were investigated by a trained assessor in the field. The decision to transport the carcass to the pathology laboratory was based on the level of decomposition of the carcass and its accessibility by land or sea. Necropsies were performed in a laboratory setting under the supervision of 6 veterinary pathologists over the 30-year study period; 88% of these necropsies were supervised by 3 of these pathologists. Necropsies were conducted on each carcass with an evolving but relatively uniform standard protocol. 47 The level of decomposition was estimated via standard marine mammal carcass decomposition codes. 47 Intermediate scores (ie, 2.5, 3.5) were used when appropriate.
The primary cause of death was determined for each beluga by the supervising veterinary pathologist based on circumstances of the stranding and results of the postmortem examination. To increase consistency in the interpretation over the years, a complete review of each case, including evaluation of archived material as needed, was done in 2013 by the principal investigator (S.L.) leading to the reclassification of the primary cause of death in 9% of cases. In this review, each diagnosis was grouped into 1 of the following categories: infectious diseases, malignant neoplasia, dystocia/postpartum complication, mortality of dependent calves, ship/boat strikes, entrapment in fishing gear, primary starvation, intoxication, other noninfectious causes, undetermined (Suppl. Table 1).
Diagnostic Categories of Primary Causes of Death by Age Group of St Lawrence Estuary Beluga (1983–2012).
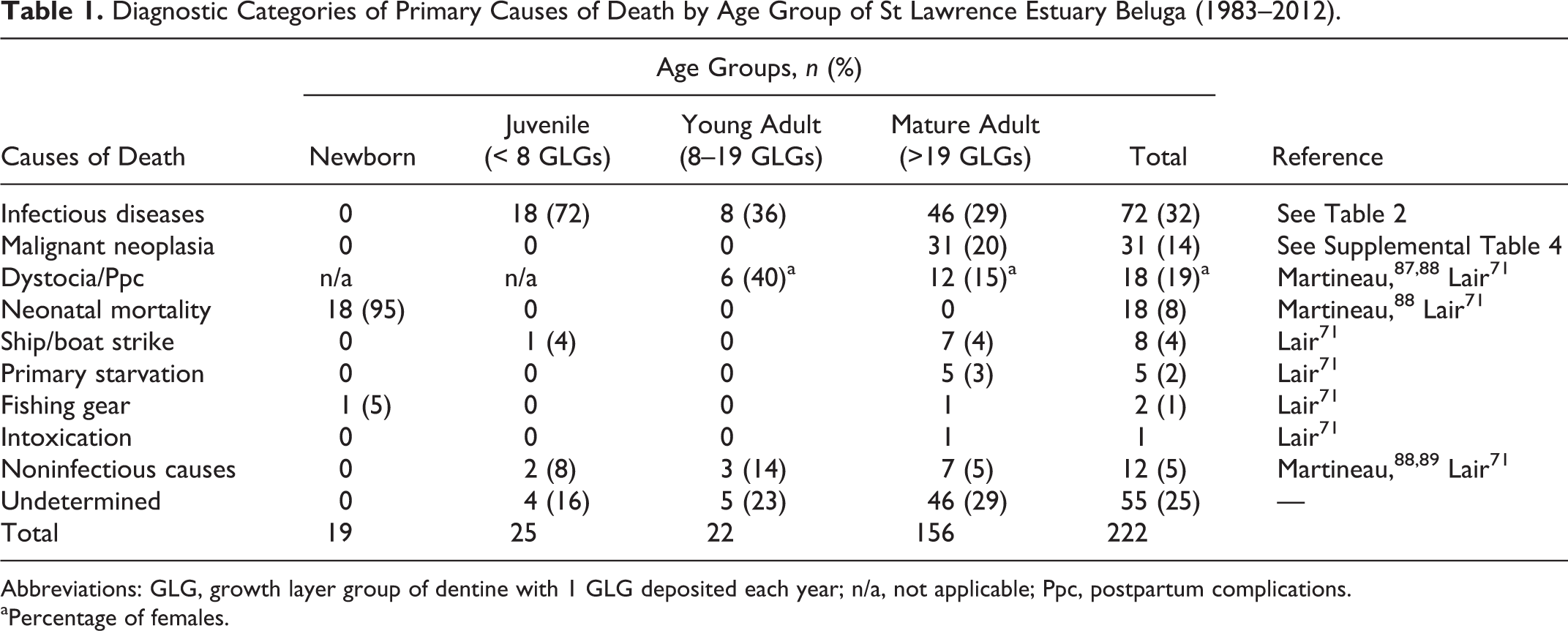
Abbreviations: GLG, growth layer group of dentine with 1 GLG deposited each year; n/a, not applicable; Ppc, postpartum complications.
aPercentage of females.
To examine association with age, frequencies of causes of mortality were evaluated according to age groups. Age was estimated by counting growth layer groups (GLGs) of dentine, through use of a longitudinal midline section of half tooth. 79 We consider that 1 GLG is deposited each year. 144 Despite the fact that the number of GLGs estimates minimum age owing to tooth wear, 79 the estimated number of GLGs was used as a relative estimation of age in years.
The following age groups were compared: newborn calves (determined by the standard length, the month of the stranding, and the presence of fetal structures—such as unhealed umbilical cord stump and presence of a cardiac foramen ovale), juveniles that are not sexually mature (<8 GLGs), young adults (8–19 GLGs), and mature adults (>19 GLGs). A GLG value of 8 was used as the upper limit for the juvenile age group based on the documented minimum age as an adult for females, 133 whereas a GLG value of 19 was determined to be the average age at which the asymptotic length is reached. 79 Three beluga that could not be aged were assigned to the mature adult category based on a total length higher than the asymptotic length. In some instances, young adult and mature adult groups were combined in an adult group (≥8 GLGs). The estimated year of birth was calculated by subtracting the estimated age from the year of stranding. Temporal changes over the study period were evaluated by comparing primary causes of mortality occurring during 3 decades: 1983–1992, 1993–2002, and 2003–2012. Disease findings reported in other stranded odontocete populations (ie, toothed whales) were compared with disease findings observed in SLE beluga to evaluate patterns and unusual occurrences of diseases.
A multivariate logistic regression model was used to evaluate the potential effect of the period of mortality (1983–2002 vs 2003–2012), the sex, and the age at death on the prevalence of bacterial infections in all age groups, as well as the relationship among the prevalence of neoplasia, the decade of birth, and the age at death in mature adults. The relationships between sex and prevalence of bacterial infections and between sex and neoplastic diseases were evaluated with an exact chi-square test in the adult group and mature adult group. The relationship between sex and prevalence of neoplastic diseases was evaluated with an exact chi-square test in the adult group. An exact chi-square was also used to evaluate the changes in the prevalence of mammary neoplasia and of digestive adenocarcinoma in mature beluga, as well as in the prevalence of dystocia in adult females over the different decades. The variation in prevalence of neoplasia in the different age groups was analyzed with the Cochran-Mantel-Haenszel test. Statistical analyses were performed by use of statistical software (SAS 9.4; SAS Institute Inc, Cary, NC). The level of statistical significance was set at 0.05 throughout.
Postmortem Examination of Beluga
From 1983 to 2012, a total of 472 SLE beluga carcasses were reported stranded or drifting. 79 A complete postmortem examination was conducted on 222 (47%) of the reported carcasses (Suppl. Table 2). The numbers of complete necropsies performed each year ranged from 3 to 12, with an average of 7.4. 71 The ratio of females:males was 1.14, with 117 females, 103 males, and 2 intersexes (Fig. 1; Suppl. Table 3, Suppl. Fig. 1). The majority (65%) of examined carcasses were found from March to August, with 31% in the fall (September to November) and 4% in the winter (December to February). 71 This seasonality is most likely due to the seasonal increase in the number of people (eg, tourists visiting the shorelines) and the presence of coastal ice during winter that decreased detection and limited access to carcasses. More than half (57%) of beluga carcasses were reasonably well preserved (eg, preservation codes <3). 47 Carcasses were more frequently classified with preservation codes <3 from October to April (67% of carcasses examined) as compared with carcasses examined from May to September (54%).
Infectious Diseases as Cause of Death by Age Group of St Lawrence Estuary Beluga (1983–2012).
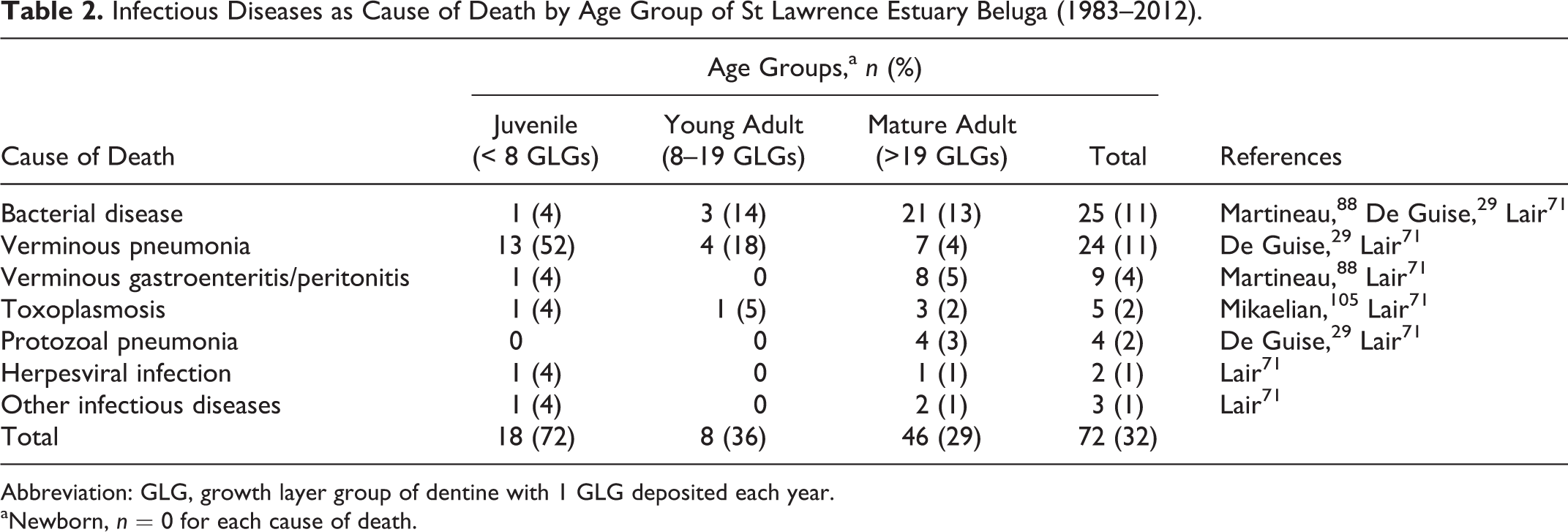
Abbreviation: GLG, growth layer group of dentine with 1 GLG deposited each year.
aNewborn, n = 0 for each cause of death.
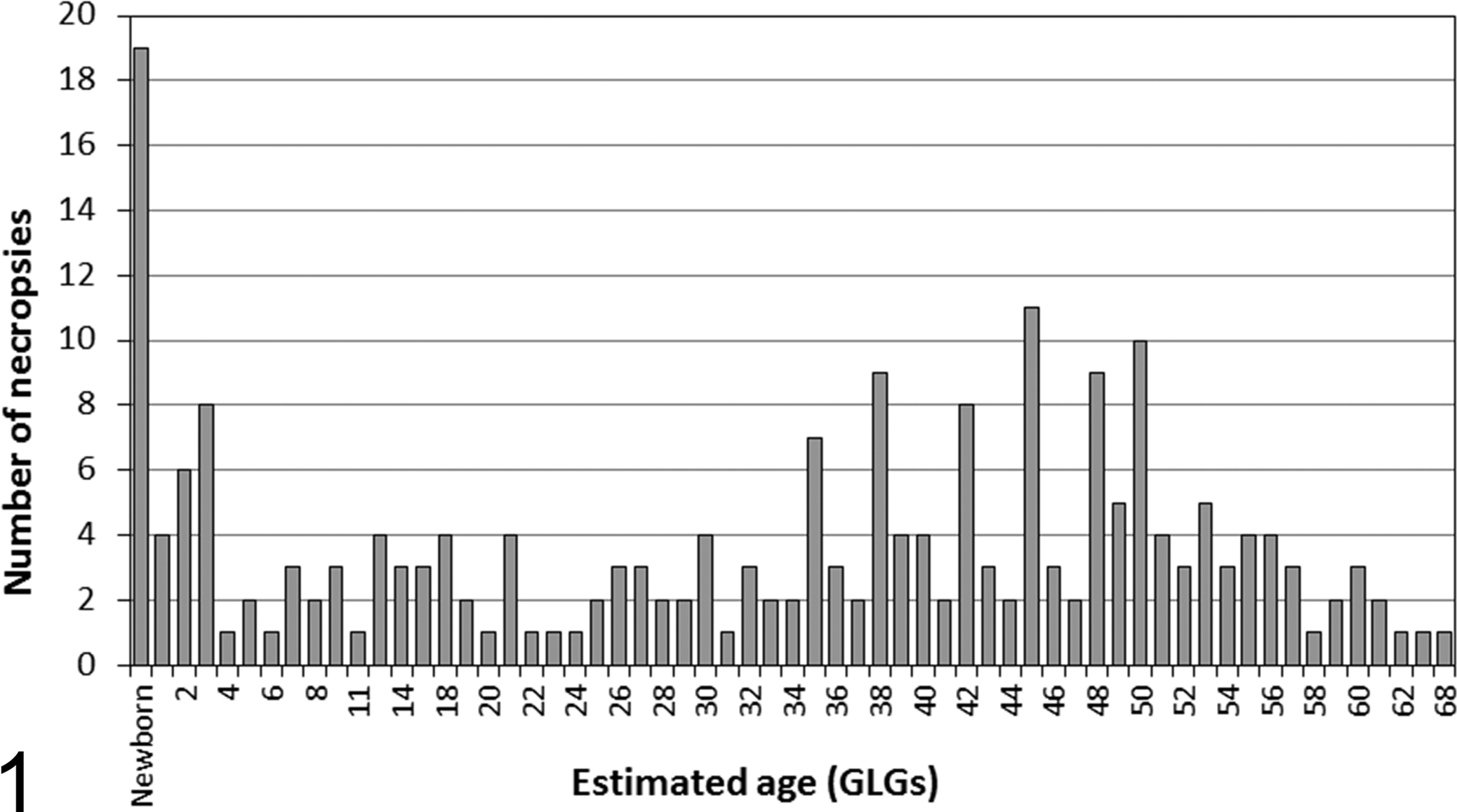
Estimated age distribution of St Lawrence Estuary beluga examined at necropsy by a veterinary pathologist in a laboratory setting, 1983–2012. N = 219. GLGs, growth layer groups of dentine with 1 GLG deposited each year. Age was not determined in 3 beluga.
Determining Causes of Mortality
The primary cause of death was determined by the supervising pathologists in 75% of the cases, a figure similar to that reported in other studies of stranded cetaceans: for instance, 71% of 38 stranded beluga examined at necropsy from the Cook Inlet population in Alaska 19 and 77% of 405 stranded marine mammals in Cape Cod 11 received a diagnosis. Other studies reported higher rates (83% to 94%). 1,32,98 Differences in the state of preservation or decomposition of carcasses and in the proportion of cases that died of anthropogenic causes associated with readily observed traumatic lesions, such as entanglement in fishing gear or ship/boat strike, could account for the differences in diagnostic rate. Failure to identify a cause of death of SLE beluga was proportional to the degree of decomposition; the cause of death was not determined in 14%, 29%, and 36% of the relatively fresh carcasses (code 2), moderately decomposed carcasses (codes 2.5 and 3), and markedly decomposed carcasses (codes 3.5 and 4), respectively. 71
Infectious Diseases
Infectious diseases were the most frequent cause of death of stranded beluga (32% overall), with 72%, 36%, and 29% of the cases in juvenile, young adult, and mature adult age groups, respectively (Tables 1 and 2). The rate of infectious diseases documented in stranded beluga from this population was higher than that reported in stranded beluga from the Cook Inlet population in Alaska (8%). 19 The relative frequency of mortalities due to infectious diseases was especially high in juvenile beluga, accounting for 72% of the mortalities, compared with adult beluga from this population.
Two fatal cases due to infection with a herpesvirus were documented. 71 One case, a 25-GLG male, had extensive gingival and palatine ulcerations, and the other case, a 2-GLG male, had systemic necrotizing lesions in multiple organs. A multifocal severe dermatitis associated with viral particles typical of herpesvirus was also seen in a 3-GLG female. 88 The dermatitis contributed to the death of this beluga, but the primary cause of death was diagnosed as gastric perforation. A novel herpesvirus has been documented in penile ulcerative or proliferative lesions in 3 male beluga and in ulcerative vulvar lesions in a female. 9
Bacterial diseases were the cause of death of 25 beluga from different age groups and included abscesses (7 cases), septicemia (5), mastitis (3), peritonitis (3), cystitis (2), endocarditis (2), lymphadenitis (2), and nephritis (1). 29,71,87,88 These infections were associated with Edwardsiella tarda (5 cases), Edwardsiella hoshinae (2), Edwardsiella sp (2), Aeromonas sp (2), Shewanella putrefasciens (2), Klebsiella oxytoca (1), Acinetobacter baumanii (1), Vibrio fluvialis (1), Vibrio alginolyticus (1), Kingella kingae (1), E. coli (1), Nocardia sp (1), and Streptococcus phocae (1). Most bacterial diseases were in mature adults; 21 of the 25 cases diagnosed in animals with >19 GLGs. The occurrence of bacterial diseases was not statistically associated with sex (P = .52) or with age (P = .41).
Verminous pneumonia was the cause of death of 24 beluga involving all age groups except newborn and accounting for over half (58%) of the mortalities caused by infectious diseases. 29,71 Parasitic pneumonia is the most common cause of mortality of juvenile beluga (n = 13; 7 females and 6 males). No sex predisposition is observed in lungworm infections (13 females, 11 males).
Based on morphologic characteristics, 2 genera of nematodes cause fatal verminous infections in SLE beluga: Halocercus monoceris and/or Halocercus taurica and Stenurus arctomarinus. 101 While vertical transmission of Halocercus sp has been reported in dolphins, 25,44,149 this mode of transmission does not appear to be significant, as Halocercus was seen only rarely and in small numbers in beluga <1 year of age. Nematodes from the genus Halocercus and Stenurus have been reported in the lungs of many species of odontocetes. † A relationship between intensities of infection with lungworms and poor body condition has been documented in by-caught harbor porpoises (Phocoena phocoena). 161 However, the significance of lungworm infections in the health of odontocetes is often difficult to assess. 19,44,100,149 In this review, a diagnosis of fatal verminous pneumonia is assigned by the severity of the inflammatory reaction, the extent of affected pulmonary parenchyma and airways, the poor body condition of the animal, and the absence of other significant findings. The relatively high occurrence of fatal verminous pneumonia in the juvenile cohort of SLE beluga suggests that infections by Halocercus sp and S. arctomarinus have a negative effect on recruitment and may contribute to the lack of recovery of this population. Burek-Huntington et al reported that the majority of Cook Inlet beluga with lesions due to mild lungworm infections were incidental findings but that moderate lungworm infections were likely contributory to death. 19 Abiotic and biotic factors affect survival, development, and transmission of parasitic nematodes. For instance, higher water temperature may favor development and transmission of marine lungworms. 100 Biotic factors potentially involved in higher rates of nematode infections are abundance and availability of intermediate and paratenic hosts in the marine environment or host factors such as immunosuppressive pathogens or chemical contaminants.
Parasitic infections in the abdominal cavity (verminous gastroenteritis and peritonitis) were the cause of death in 9 animals, including cases of perforated ulcerations of the first gastric compartment (3 cases) and the intestine (3 cases) associated with anisakid nematodes. 71,88 Anisakid nematodes are found in the gastrointestinal tract of most beluga from this population. 71,88 These parasites often cause low-grade ulcerative gastritis of the first compartment. Large fibrotic, often ossified, mesenteric masses also caused the death of 2 beluga. 71,88 These masses were associated with entrapment of an intestinal segment or with torsion of the mesentery. Similar but smaller mesenteric masses were observed in 11 other beluga as incidental findings. Debris of degenerated parasites, occasionally observed in the centers of some of these masses, were highly suggestive of chronic inflammatory reactions subsequent to the abdominal migration of helminths from the gastrointestinal tract. 71,88 With the exception of a perforated gastric ulcer in a 3-GLG juvenile, all cases of fatal verminous gastroenteritis and peritonitis were seen in beluga >20 GLGs, and neither sex predisposition nor temporal variation was observed.
Other parasitic diseases included 5 cases of toxoplasmosis 71,105 and 4 cases of protozoal pneumonia. 29,71 Fatal infections by the protozoan Toxoplasma gondii were characterized by systemic infections affecting numerous organs, including lung, spleen, and brain, and are observed in different age groups (2–51 GLGs). Single cases of fatal toxoplasmosis are reported in several species of odontocetes. ‡ While infections of T. gondii were not a frequent cause of mortality in SLE beluga, these infections indicate that pathogenic infectious pollutants (or biological contaminants) can have an effect on susceptible cetaceans, such as SLE beluga. 105,153 Risk factors proposed for toxoplasmosis in cetaceans may include coinfections by immunosuppressive pathogens such as morbillivirus, 38 exposure to immunosuppressive contaminants such as polychlorinated biphenyls (PCBs), 105 and coastal freshwater runoff contaminated with T. gondii oocysts. 15
Protozoal pneumonia was caused by ciliated protozoa of undetermined species and was diagnosed only in mature adults; 3 of the 4 cases were in males. These cases are noteworthy since deep respiratory infection by ciliated protozoa is not reported in other cetacean species. Several species of protozoa have been described in the nasal cavities of odontocetes where they do not cause apparent disease. 146 Ciliated protozoa are associated with dermatitis in dolphins infected with immunosuppressive morbillivirus. 137 As for T. gondii infections, risk factors for tissue invasion by these saprophytic protozoa may include immunosuppressive pathogenic infections, generalized debilitation, and exposure to immunosuppressive chemical contaminants. Fatal infectious diseases in wildlife indicate imbalance between pathogen and host. This disequilibrium can be due to overwhelming exposure of the host to pathogens, to exposure of naïve hosts to pathogens (some of which may be emerging), and/or to a decrease in host defense mechanisms against pathogens. Risk factors for immunosuppression include social and reproductive stress, 138 malnutrition, immunosuppressive infectious pathogens (eg, morbillivirus), and chemical contaminants (eg, PCBs). 74,140 Exposure to and active infection with morbillivirus have not yet been documented in SLE beluga. 108,117
Beluga from the SLE are highly contaminated with PCBs. 31,74,87,94,97,112,127 Numerous studies documented an association between exposure to PCBs and immunosuppression or increased risk of infectious diseases in marine mammals. One of these studies demonstrated an epizootiologic association between levels of PCB contamination and infectious diseases in harbor porpoises (P. phocoena). 68 Leucocytes from Arctic beluga exposed to PCB concentrations similar to those measured in the tissues of SLE beluga showed altered functions in vitro. 30 Van Loveren et al 154 showed diminished immune function in captive harbor seals (Phoca vitulina) fed herring highly contaminated with PCBs as compared with seals fed noncontaminated herring. High levels of PCBs are associated with a decrease in production of antibodies following immunization in free-ranging polar bear, 80 and immunosuppression is associated with PCB exposure in Atlantic bottlenose dolphins (Tursiops truncatus). 139 PCBs or other immunosuppressive chemical contaminants found in SLE beluga might increase their susceptibility to infectious agents. 29,31,84,86,87,88 Adipose tissue (blubber) collected between 1982 and 1987 showed that PCB levels in SLE beluga were 25 times higher than in Arctic beluga. 112 PCB and PCB metabolite concentrations measured in the liver of SLE beluga (2000–2003) were 16-fold higher than in Arctic beluga sampled over the same period, whereas total OH-PCB and total MeSO2-PCB concentrations were approximately 22- and 49-fold higher, respectively, in SLE beluga. 97 Almost all the SLE beluga sampled before 1987 had values of PCBs higher than the threshold value that has been established to be associated with a 50% increase in risk of suffering an infectious disease in harbour porpoises (Fig. 2). 57,69 Afterward (1987–2002), total PCB concentrations in blubber lipids—but not in liver—of SLE beluga decreased about 2-fold (Fig. 2). 74,127 Despite this decrease in concentrations of PCBs over the years, 51% of the SLE beluga sampled between 1987 and 2007 still had levels of PCBs higher than this threshold value in porpoises (Fig. 2). 57,69
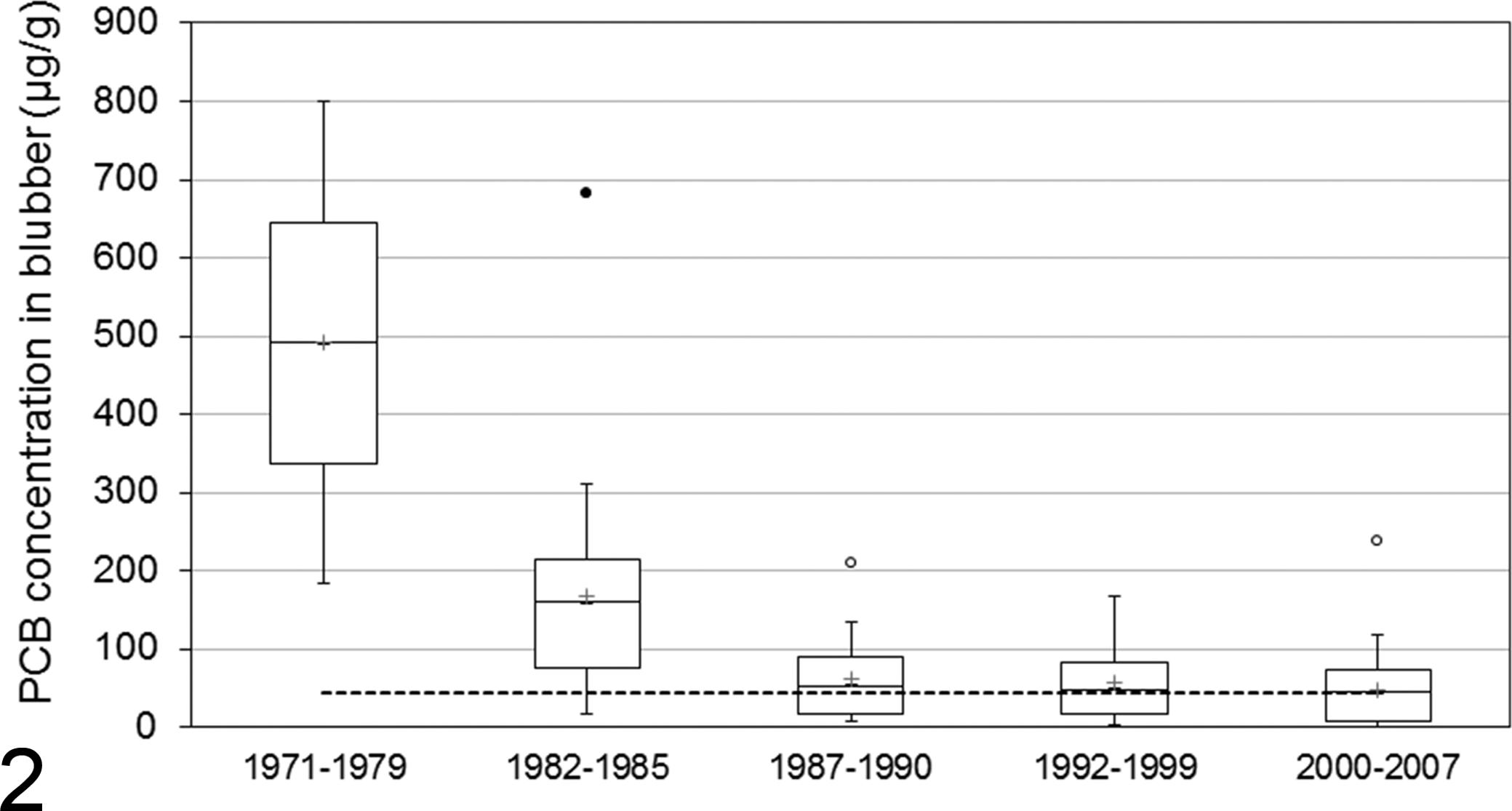
Polychlorinated biphenyl (PCB) concentrations found in blubber (µg/g; lipid weight basis) of dead beluga stranded on the shoreline of the St Lawrence Estuary. Adapted from Martineau. 84 Periods indicated correspond to when carcasses were recovered and sampled: 1971–1979 (n = 2), 141 1982–1985 (based on aroclor mixtures; n = 21), 86 1987–1990 (based on specific PCB congeners; n = 25), 112 and 1992–1999 (n = 59) 74 and 2000–2007 (n = 60). 75 The dotted line corresponds to 45 µg/g (lipid weight basis), which represents a 50% increase in risk of suffering an infectious disease in porpoises. 57,69 The box shows the 25th and 75th percentile; bars = median; asterisks = mean; circles = outliers.
If it is assumed that these PCBs have linear dose-dependent immunosuppressive effects, then a decrease in these compounds in beluga tissues over the years should be associated with a decrease in the occurrence of fatal diseases caused by infectious agents in this population. The odds of fatal infectious diseases decreased by a factor of 3.8 (95% confidence interval: 1.08–13.37) in the 2003–2012 period (average cases, 0.40/y) compared with the 1983–2002 period (average cases, 1.05/y; P = .02). This temporal trend supports the hypothesis that anthropogenic immunosuppressive contaminants may have a negative effect on the health of SLE beluga.
Adult male beluga carry in general a higher burden of PCBs than that of adult female beluga since females transfer a portion of their load of these lipophilic contaminants in the placenta and milk. 73,74,86 Consequently, if PCBs play a key role in the development of bacterial diseases in this population, adult males should be overrepresented in this category. Even though more adult males (average cases, 0.40/y) died of bacterial diseases compared with adult females (average cases 0.27/y), this difference was not statistically significant (P = .27).
Malignant Neoplasms
A total of 39 malignant neoplasms were diagnosed in 35 adult beluga (median age at diagnosis, 48 GLGs; range, 30–61 GLGs), causing the death of 31 beluga, all mature adults. Four animals were affected concurrently by 2 types of malignant neoplasms (Suppl. Table 3). § Cancer is the second-most frequent category of cause of death after infectious disease, accounting for 31 of 222 (14%) of all beluga and 31 of 156 (20%) of mature adult beluga. This percentage (14%) is the highest occurrence of cancer reported in a population of cetaceans. Some studies reported a frequency approximately 10 times lower, between 0.7% and 2%, 1,10 in cetaceans while others did not report any cancer. 11,32,67,98,121 For instance, no case of cancer has been documented in stranded beluga (N = 38) from the Cook Inlet population in Alaska. 19 Over half (54%) the malignant neoplasms reported in free-ranging odontocetes worldwide ‖ are diagnosed in SLE beluga. In California sea lions, a higher occurrence (26%) of a specific type of cancer, urogenital carcinoma, is reported in stranded adults but also in subadults. 17,21
The percentage of examined beluga affected by cancers increases with age (P < .0001; Fig. 3). Malignant neoplasms seem to be more frequent in females (28% of mature adult females examined for an annual average of 0.77 cases) than in males (16% of mature adult males examined for an annual average of 0.40 cases), but this association with sex was not statistically significant (P = .09). Adenocarcinoma of the gastrointestinal mucosa is the most commonly observed neoplasm, with 11 cases (7.1% of mature adults examined). 71,92,93 Two other cancers, a salivary gland adenocarcinoma 52 and a cholangiocellular carcinoma, 71 involved organs closely associated with the digestive tract. These cancers of the digestive system, which were seen in beluga >30 GLGs, increased in frequency with age, reaching 57% of stranded beluga >57 GLGs. There is no obvious sex predisposition for cancers of the digestive system. The occurrence of gastrointestinal adenocarcinomas in the SLE beluga is significant, as only 1 case of gastric adenocarcinoma and 1 case of intestinal adenocarcinoma have been reported in other populations of cetaceans. 5,121
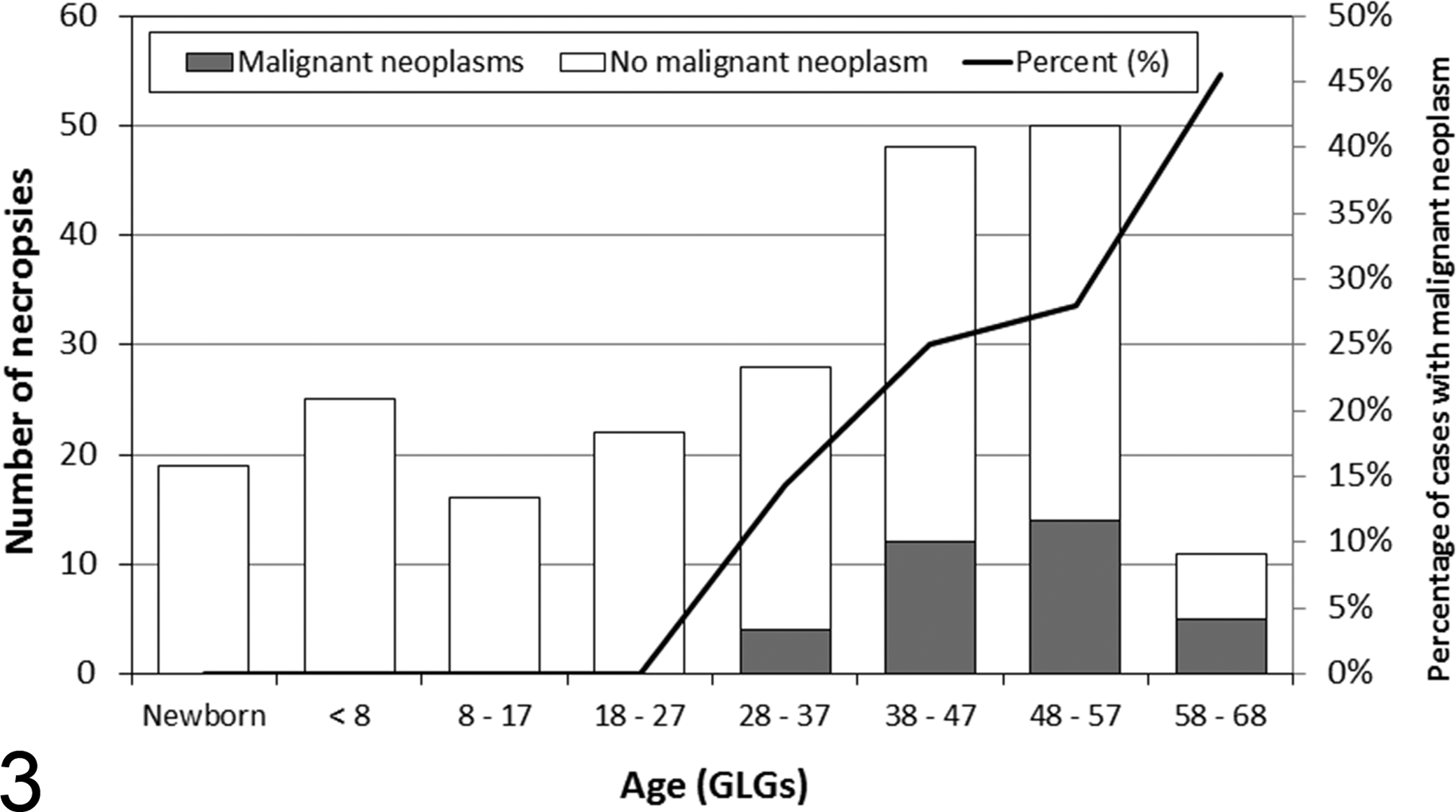
Estimated age distribution of St Lawrence Estuary beluga with at least 1 malignant neoplasm (n = 35) and beluga without any neoplasm (n = 184) examined at necropsy by a veterinary pathologist in a laboratory setting, 1983–2012. GLGs, growth layer groups of dentine with 1 GLG deposited each year. The age was not determined for 3 beluga without neoplasms.
Second to the gastrointestinal tract, the mammary gland is the organ most commonly affected by neoplastic changes in SLE beluga; a total of 8 cases of mammary carcinomas affected 9.9% of mature adult females examined. 28,71,107 These cancers, often metastatic, caused the death of 6 of these females. Mammary cancers have not been diagnosed in other populations of odontocetes. Affected females were born during the period 1939 to 1957, with an age ranging from 42 to 59 GLGs and a median of approximately 50 GLGs. The occurrence of these cancers did not change over the years (P = .60). Many organochlorine compounds (eg, PCBs) have estrogenic effects; many studies have reported organochlorine-induced stimulation of hormone sensitive tissue, such as mammary glands. In women, a causal relationship between breast cancer and PCB contamination remains controversial. 3 Studies have reported diverse associations between higher risk of breast cancer, high PCB tissue levels, induced cytochrome P450 (CYP) levels, and specific CYP1A1 alleles. 12,49,111 Interestingly, some of these studies deal with Inuit women, highly contaminated with PCB because their diet includes contaminated marine mammal meat. 12,49
The occurrence of cancers in SLE beluga versus other populations of odontocetes indicates that this population of beluga has an unusually high occurrence of malignant neoplasms. This difference is especially obvious for cancers of the gastrointestinal tract and mammary glands. Cancer is a complex multifactorial disease. Hypotheses that may explain the higher occurrence in SLE beluga include exposure to chemical contaminants or oncogenic pathogens, genetic susceptibility, or a combination of factors. The hypothesis most frequently proposed to explain this high frequency of cancer is the exposure to carcinogenic contaminants, such as polycyclic aromatic hydrocarbons (PAHs). 31,83,85,91,92 The role that contaminants may have in the occurrence of neoplasia and the relative importance of cancer as the cause of death in stranded animals from this population have caused controversy. 41,58,147 It has been argued that stranded beluga are not representative of the live population, as stranded animals examined at necropsy are primarily adults (median age, 37 GLGs). It has been also suggested that the life expectancy of SLE beluga may be longer than that in other populations due to the lack of predators and absence of hunting, accounting for the high occurrence of cancer. 58 However, animals as old as those observed in the SLE population 79 have been reported in other populations of beluga, 20,81,144 suggesting that beluga from the SLE do not have an unusual long life expectancy. In addition, the relatively young age of SLE beluga at which some of these cancers are observed (1 beluga was as young as 30 GLGs) suggests that a sampling bias toward older animals in a somewhat geriatric population cannot fully explain the clustering of cancers documented in this population. However, in the absence of hunting and predators, either neoplastic or infectious disease is clearly playing a large role in this population. 59,99
The St Lawrence River ecosystem receives effluents from much of northeastern North America, one of the most industrialized regions of the world. In addition, a large complex of aluminium smelters developed as early as 1926 is located near the Saguenay River, 100 km upstream of the SLE beluga habitat. Electrolysis of alumina necessary for the production of aluminium produces large quantities of PAHs, which were released into the atmosphere. 82 From 1956 to 1976, liquid effluents from smelters using the Söderberg process contained large amounts of PAHs and were discharged without treatment directly into the Saguenay River, resulting in a significant contamination of the sediments. The heavy contamination with PAHs of land mammals and fish from the Saguenay region and Saguenay River has been documented by others (reviewed by Martineau 84 ). PAH DNA adducts were detected in brain tissue of 3 SLE beluga, whereas adducts were undetectable in the brain and liver of 4 beluga from the Arctic. 88 The diving pattern during feeding suggests that beluga ingest sediment through suction feeding on benthic prey (R. Michaud, personal communication). This is supported by regular observations of sediments in the stomach of beluga. 71,156 Direct ingestion of potent carcinogens—which have been documented in sediments of the Saguenay Fjord 82 as well as in benthic worms (Nereis sp), 123 found in the stomach of beluga 78,156 —has been hypothesized in the development of these cancers in SLE beluga. 71,92,93 Beluga with cancers were alive in the 1956–1976 period, during which contamination of the Saguenay River with PAHs was at its maximum; liquid industrial effluents containing PAHs were released into the Saguenay River until 1976. 82
With changes in the aluminum production process and stricter environmental regulations, the concentrations of PAHs in the upper layers of sediments of the Saguenay River have markedly decreased since 1976. 143 Consequently, if most cancers in SLE beluga are caused by exposure to PAHs, this decrease of PAHs in beluga habitat should be associated with a decrease in the overall occurrence of cancers in this population. The number of diagnosed cancers reached a maximum in mature adults that were born in the 1940s and 1950s and decreased progressively in animals born after that period (Fig. 4). However, there is no statistical association between the occurrence of cancer in mature adults and the decades of birth (P = .06). Nevertheless, the fact that malignant neoplasia has not been observed in beluga with an estimated year of birth after 1971, including 12 mature adults at death, does suggest a decrease in the occurrence of cancers in beluga born after the abrupt decline of the PAH concentrations in the surface sediments. Even though a statistical association between cancers of the digestive system in mature adults and the decade of the examination could not be demonstrated (P = .06), the occurrence of cancers of this body system seems to have decreased over the years; malignant neoplasia of the digestive system has not been observed in beluga with an estimated year of birth after 1958, and no case of digestive system cancer has been diagnosed in stranded beluga since 2004. These observations support a possible association between exposure to industrial PAHs and cancers in SLE beluga. 71,93
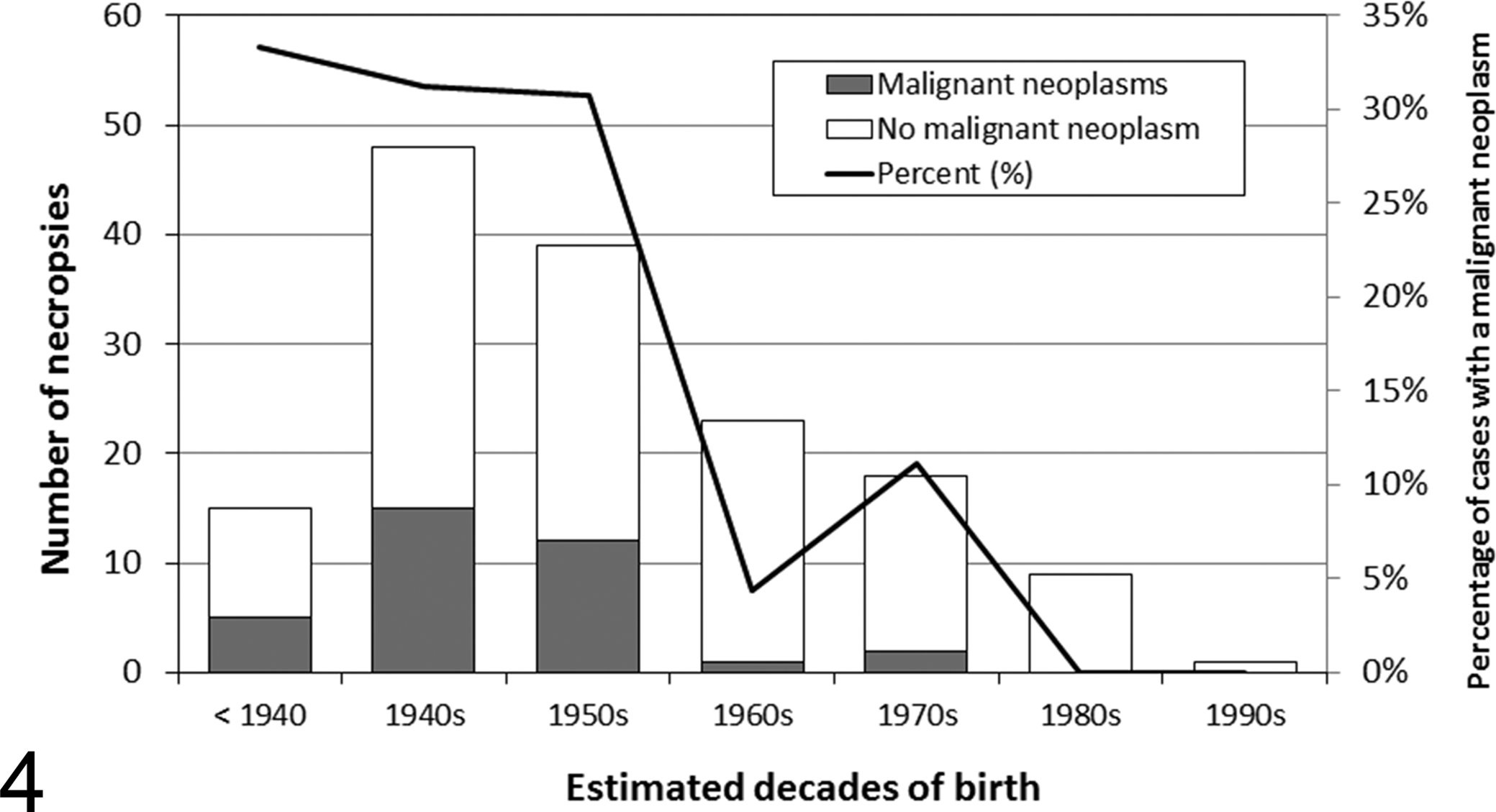
Occurrence of mature adult (>19 GLGs) St Lawrence Estuary beluga with at least 1 malignant neoplasm (n = 35) and beluga without neoplasms (n=118) examined at autopsy by a veterinary pathologist in a laboratory setting (1983–2012) by decade of their estimated year of birth. The estimated year of birth of 3 beluga without neoplasms could not be determined. GLGs, growth layer groups of dentine with 1 GLG deposited each year.
The link between PAH exposures and the development of cancers is also supported by the occurrences of pre-neoplastic and neoplastic lesions in the liver of fish exposed to PAHs from the St Lawrence River. 23,106
Alternatively, by enhancing the CYP-mediated degradation of PAHs into carcinogenic metabolites, contamination with PCBs could play a role in the occurrence of neoplastic disease in this population. 93 A decline in the levels of PCBs in beluga tissues has been documented in recent years. 74 Accordingly, the decrease in PCB contamination of SLE beluga could also contribute to the decrease in the prevalence of cancers in SLE beluga.
The proposed link between the unusually high occurrence of cancers in SLE beluga and exposure to industrial contaminants is of interest in a public health perspective. Associations between occupational exposures to PAHs and neoplasia (of the lung, bladder, and stomach) have been documented in workers of aluminum plants located on the Saguenay River upstream of the beluga habitat. 2,50,151
An increasing number of viruses, bacteria, and parasites are known to be oncogenic in animals and humans. 96,120 As examples, papillomavirus has been associated with gastrointestinal tract cancer in cattle along with the ingestion of carcinogenic and immunosuppressive bracken fern. 66 In marine mammals, otarine herpesvirus-1 and papillomaviruses (TtPV-1, TtPV-2) have been associated with urogenital carcinomas in California sea lions (Zalophus californianus) 18 and oral squamous cell carcinoma in Atlantic bottlenose dolphins, respectively. 14,130 Exposure to anthropogenic contaminants may be etiologic cofactors in both conditions. 13,162 Since papillomaviruses have been detected in papillomas of the first gastric compartment in SLE beluga, 26 it is conceivable that these viruses could be oncogenic or act as a cofactor in gastrointestinal tract cancer in SLE beluga exposed to carcinogenic contaminants. Nevertheless, infectious agents are not likely the major reason for the development of cancers in SLE beluga, because of the wide range of neoplastic phenotypes present in this population.
Because SLE beluga have relatively low genetic diversity, 113,122 it has been proposed that this population may have a genetic susceptibility to cancer. 41 Examples of neoplasms in small populations of free-ranging mammals include facial tumors due to transplanted neoplastic cells in Tasmanian devils (Sarcophilus harrisii), 142 lymphomas in koalas (Phascolarctos cinereus) caused by a retrovirus, 33 and papillomavirus-associated carcinomatosis in western barred bandicoots (Perameles bougainville). 160 Neoplasms in these small populations may have a genetic component, as decreased genetic diversity, including major histocompatibility complex (MHC) diversity, is suspected and/or documented in most of these populations. The loss of MHC diversity probably hampers antiviral immunity. Genetic factors and contaminants may both be involved in viral oncogenesis, as in the large and increasing population of California sea lions where specific MHC alleles and a high burden of PCB may contribute to viral oncogenesis. 16,17,162
The epizootiology and phenotypes of cancers observed in the SLE beluga population greatly differ from what is described in familial cancers in people. Familial cancers usually affect individuals at an early age and involve rare types of neoplasms in multiple sites (reviewed by Philippe 124 ). None of these characteristics are observed in SLE beluga—for example, most cancers arise late in life; single sites are affected; and cancers common in other mammals are observed (inasmuch as one considers proximal intestinal cancers common in other mammals). Familial predisposition to common sporadic neoplastic diseases, such as lung, breast, and colon cancers, has been described frequently in people. 124 Similar predispositions seem to occur in animals, where the occurrence of neoplasia also varies from species to species. Variation in tumor occurrence is also observed among breeds of dogs, 61 and loss of genetic heterogeneity in these breeds may be a factor predisposing to neoplasia. 152 In these situations, increased risk of cancer may be due to many specific and common genetic features associated with normal polymorphism. As an example, these features may be located in genes such as CYPs, which again code for enzymes that bioactivate contaminants such as PAHs into carcinogenic metabolites. 115,131 There is much evidence that CYP genes contribute to the genetic susceptibility to cancer (as low penetrance genes). 115,148 Interestingly, CYP genes are induced in SLE and Arctic beluga, 118,159 and SLE beluga are contaminated by PAHs (reviewed by Martineau et al) 83,93 and PCBs, 74 which are strong inducers of CYP. 150 Consequently, genetic susceptibility to cancer in SLE beluga, if present, could belong to this category.
Productivity and Parturition-Associated Mortality
Female beluga give birth to a single calf in summer over a 3-year reproductive cycle. 110 Gestation lasts 14 to 15 months, and calves nurse for up to 24 months with progressive weaning. 133 Critical habitats for this population have been identified on the basis of field observations of areas of high use or concentration of SLE beluga, particularly for females with calves and juveniles. This habitat is likely vital for birth and survival of calves and juveniles. 34 Suboptimal calf production, suboptimal survival of calves and juveniles, or a combination thereof may account for lack of recovery 36 and explain the recent decline of this population. 110 Results from photographic surveys indicate that the proportion of calves in relation to the total number of beluga counted in surveys decreased from 15% to 18% for the 1990–1997 period to 3% to 8% for the 2000–2009 period. 55 This observation suggests either a decrease in calf productivity or survival. Before 2008, the number of documented newborn deaths varied annually from 0 to 3, with a median of 1 calf per year, whereas from 2008 to 2012, 1 to 16 calves were found dead annually, with a median of 7 calves per year. 79 This observation suggests that the decrease in summer abundance of calves is likely due to decrease in calf survival.
With the exception of 1 by-caught calf, none of the stranded calves examined over the years presented any pathologic changes that could have accounted for their death. Dead calves were recovered from June to September, with a peak in August (37 of the 61 documented mortalities). 79 Lung inflation, a sign that the calves were born alive and breathed, was observed in most cases. Five calves live stranded. All examined calves had passed meconium, another sign that they were born alive, and had an empty stomach. The presence of various amounts of amniotic squamae and meconium in the pulmonary alveolar lumens in most calves is suggestive of fetal distress. 109,157 No other pathologic findings were observed. A circumstantial diagnosis of dehydration and electrolytic imbalances subsequent to the inability to nurse and thrive—due to exhaustion, lack of maternal care, or mortality/morbidity of the dam—was assigned to these calves. 71,88
Signs suggestive of fetal distress indicate possible prolonged or difficult birth. It is possible that increases in calf mortalities are a consequence of an increase in parturition complications. This possibility is supported by the concurrent increase in the occurrence of parturition-related mortality of pregnant females. 71 Dystocia caused the death of 11 females and was characterized by the presence of a full-term fetus in the uterus or its engagement in the birth canal with signs of active labor, such as dilated cervix or uterine contusions, and by the absence of other significant pathologic findings. 71,87,88
In addition to the confirmed dystocia, postpartum complications were suspected as causing the stranding of 7 females. 71 These females were found dead with evidence of recent parturition (within days) based on the appearance of the genital tract (signs of infection or trauma centered on the grossly asymmetrical uterus) combined with the absence of other significant pathologic findings. Consequently, the deaths of 19% (18 of 96) of the adult females examined were attributed to problems occurring during birth or shortly after. Age of affected females ranged from 9 to 45 GLGs, with a median age of 29 GLGs at diagnosis. Dystocia and/or postpartum complications were the most frequently documented causes of mortality in young adult (8–19 GLGs) females (40%). Even if there was no statistical association between the occurrence of dystocia/postpartum complications in the adult females and the decade of the study, dystocia and postpartum complications were especially prevalent during the last 3 years of the study (2010–2012), accounting for the death of 47% (7 of 15) of adult females examined over that period. The age composition of the carcasses found stranded indicated an increase in the mortality of young adult females during the 2000–2012 period as compared with the 1990s, 79 which is likely related to the increase in reproductive-associated mortalities. The uneven female:male ratio (favoring females at 1.45) in stranded adult beluga between 2000 and 2012 also indicates an increase in sex-linked mortalities due to dystocia/postpartum complications.
Although dystocia and signs of parturition complications have been occasionally reported in wild cetaceans, they are relatively rare in most populations. 5,53,60,145 Two (6.3%) of the 32 finless porpoises (Neophocaena phocaenoides) found stranded in the territorial waters of Hong Kong showed prolapsed uteri suggestive of parturition complications. 121 Between 2005 and 2010, 7 (1.4%) cases of dystocia of 478 harbor porpoises examined and 1 (0.8%) of the 129 short-beaked common dolphins (Delphinus delphis) examined with signs of postpartum complication were reported in British waters. 32 The frequency of dystocia/postpartum complications (19%) reported in stranded adult female SLE beluga is the highest recorded in wild cetaceans. None of the 11 carcasses of adult female beluga examined from the Cook Inlet population showed signs of dystocia or postpartum complications. 19 Dystocia has been documented in captive beluga (3 of 25, or 12% of pregnancies). 133 Comparison between captive beluga and beluga in a natural setting is limited since several factors inherent to captive settings could favor dystocia. The cause of this elevated occurrence of obstetric complications in SLE beluga remains unknown at this time.
Risk factors for dystocia include disturbance of parturient females, nutritional stress, and exposure to endocrine-disrupting chemicals, such as polybrominated diphenyl ethers (PBDEs). Periparturient environmental stresses, such as stress induced by the continuous presence of a human observer, increases the risk of dystocia in cattle. 102 Little is known regarding stress experienced by SLE beluga from recreational or commercial marine traffic. Given the increase in some of these activities in areas identified as critical habitat for beluga, 103 a possible relationship between the apparent increase in frequency of dystocia in recent years and the disturbance of prepartum females should be investigated.
Nutritional restriction during the last trimester can increase the risk of dystocia in cows. 56 Negative energy balance subsequent to either shortage of food resources or increased energy expenditure during the winter preceding parturition may also be a potential risk factor for dystocia in beluga.
Evaluation of ecosystem variability in the Gulf of St Lawrence between 1990 and 2012 showed a shift in several ecosystemic indicators. 125 For instance, increased surface water temperature, decreased winter ice volume, and decreased biomass of large demersal fish and spring herring are correlated with increased documented mortality of beluga calves. 125 These observations suggest that changes in the ecosystem could play a role in decreased calf survival. Changes in diet composition may also be associated with deficiencies in some nutrients. For instance, thiamine deficiencies may be the cause of reproductive failure in fish from the Great Lakes 45 and birds in the Baltic Sea. 6 Data on thiamine in prey species and in SLE beluga are lacking.
PBDE concentrations in blubber of female SLE beluga increased rapidly until 1998, then stabilized over the 1999–2007 period. 75 Levels of contamination with PBDEs of SLE beluga are approximately 20- to 30-fold higher than levels reported in beluga from the Canadian Arctic. 72 Several PBDE-homolog groups interfere with thyroid activities in several species, including mink 163 and rat. 62 A negative correlation between blood levels of PBDE-homolog and thyroid hormones was documented in beluga from Svalbard, suggesting that these contaminants could cause disruption of thyroid function in beluga. 155 Epidemiologic associations have been reported between hypothyroidism in pregnant women and increased risk of fetal distress. 64,158 In addition, pregnant hypothyroid bitches have been shown to have longer and weaker uterine contractions and produce puppies with lower viability scores, lower weight, and lower periparturient survival. 119 In utero exposure to PBDEs could also decrease calf survival since these compounds have been shown to induce developmental neurotoxicity following prenatal exposure. 22 Fetal exposure to PBDEs is associated with disruption of motor activity in animal models, and an inverse epidemiologic association has been documented between PBDE concentrations in umbilical cord blood and adverse neurologic development test scores in children. 63 Beluga calves exposed in utero to PBDEs may have decreased cognitive abilities leading to suboptimal nursing, possible separation from the dam, and ultimately decreased survival. However, the increase of calf mortalities do not coincide exactly with levels of contamination by PBDEs; concentrations of PBDEs in SLE beluga have been stable since 1998, whereas increased mortality of dependent calves was first documented in 2008. 75 The possible association between exposure to relatively new contaminants such as PBDE and the frequency of parturition complications and neonatal mortalities needs to be investigated further.
Saxitoxin Intoxication
Nine SLE beluga mortalities along with high numbers of dead birds, marine mammals, and fish were reported in August 2008 during a massive bloom of Alexandrium tamarense that coincided spatially and temporally with the distribution of the multispecies mortalities. 136 Unfortunately, only a single beluga, a 50-GLG female, was completely examined during that month, leading to a diagnosis of saxitoxin intoxication based on the detection of the toxin in tissues, signs of acute death (very good body condition, the presence of food in gastric compartments), co-occurrence of multispecies mortalities, location, chronology and drift of the algal bloom, and the absence of other significant pathologic findings that could explain death. The effect of these harmful algal blooms on the SLE beluga population remains uncertain, 136 but an increase in the frequency, intensity, and duration of these events could become a serious threat to this population especially in light of climate variability and ecosystemic regime shifts. 125
Other Causes of Mortality
Ship/boat strikes were confirmed or suspected as the cause of death in 8 beluga. 71 These cases presented with fractures of the ribs, vertebrae or mandible, deep skin lacerations, and/or pulmonary lacerations with no other significant pathologic findings. Although causes of these traumatic lesions could not be determined with certainty in all cases, ship/boat strikes are likely involved. No information is available with respect to the type of vessel involved. However, collisions with midsized motor boats are more likely since large ship collisions are likely to lead to complete sectioning of the body. Entrapment in fishing gear caused the death of 2 beluga: 1 newborn animal drowned or asphyxiated following entanglement in a herring net, and a 38-GLG male drowned or asphyxiated after entanglement in longline fishing gear. 71 Direct effects of commercial fishing activities and marine traffic on the SLE beluga population appear to be low, as only a few cases of fishing gear entanglement and ship/boat strikes have been documented.
Five females apparently died of primary starvation. 71 Primary starvation mostly affected old beluga—all but 1 of the beluga were >50 GLGs. The low occurrence of primary starvation observed suggests that access to food resources is likely not a limiting factor in the growth of this population. Interestingly, all but 1 of the cases in which starvation was believed to be primary occurred in the last decade of the study, a period that coincided with the ecologic shift documented in the Gulf of St Lawrence in the 21st century that was associated with increased surface water temperature and decreased availability of potentially important prey for beluga. 125
Other documented noninfectious causes of mortality include dissecting aneurysm of the pulmonary trunk (2 cases), 71,89 intestinal torsion, 8 wooden foreign body in the blow hole, dilated cardiomyopathy, segmental aplasia of the vagina with hydrometra, idiopathic brain necrosis, aspiration pneumonia, cardiac tamponade, and idiopathic renal thrombosis. 71
Biases and Limitations of Data Obtained From Stranded Cetaceans
Sampling stranded marine mammals is biased, as stranded animals are not fully representative of the live population. 128 Some bias is inevitable due to problems in the detection and reporting of dead animals. As an example, due to their dark color and small size, carcasses of young beluga are probably less likely to be found and reported than large white adult beluga. As a consequence, newborn and juvenile beluga are probably underrepresented in our stranding data set. Few carcasses are reported during the winter months when there are few people walking on the shores. Carcasses may simply not be reported should they drift out of the SLE, strand in areas with little human habitation, or be consumed by scavengers. 59 Despite these limitations, since the intensity of the monitoring of SLE beluga strandings was relatively constant over time, examination of carcasses from all age and sex groups provides a reasonable evaluation of temporal changes in the causes of death in these groups.
Similar sampling biases and effort constraints are documented in other marine mammal stranding programs. In addition, since the determination of cause of death often relies, to a certain extent, on the professional judgment of the supervising pathologist, interstudy variations in the criteria to assign causes of mortality are likely to occur. Consequently, comparisons of populations with respect to causes of death, while informative, should be undertaken with caution. For the above reasons, stranding data should be used with caution in estimating mortality indices or in demographic modeling—but, at the very least, they provide minimum estimates. Despite these limitations, an unusually high prevalence of a disease (or diseases) in a declining population of cetaceans, as compared with other populations of cetaceans, suggests that this disease (or diseases) may play a role in the failure of this population to recover.
Conclusion
Causes of mortality of the SLE beluga have been documented over 30 years. Complete necropsies of >200 stranded carcasses have provided valuable information on causes of death and have identified significant threats to the recovery of this endangered population. Changes in the frequencies of certain causes of mortality over the years show that new threats face this population while “old” threats appear to be diminishing. For example, gastrointestinal cancers have not been observed in this population for several years, whereas dystocia, suspected postpartum complications, and deaths of dependent calves have become more frequent over the past few years. In addition, new threats—such as infectious diseases (herpesvirus) and toxic algal blooms (saxitoxins)—have recently been documented.
Postmortem data are informative with respect to causes of death in a given population, but this is not a measure of the rate of mortality. Consequently, the effect of these diseases on the growth of the SLE beluga population remains difficult to determine. Mortality owing to disease occurs in many populations of wild animals and does not, in the majority of cases, prevent population growth or sustainability. Nevertheless, the apparent suboptimal growth of the SLE beluga population and its failure to recover indicate the presence of some limiting factors. Understanding causes of mortality and their etiology can provide some insight on these factors. Obviously, the deaths of reproductive-age female beluga due to infectious diseases, dystocia, and cancers, as well as the death of dependent calves and juvenile beluga due to parasitic diseases, affect recruitment and recovery of this population.
Comparisons with other populations of marine mammals and inferences from controlled laboratory studies 126,140 as well as robust epidemiologic 95,114,134 and epizootiologic 7,42 studies in public and veterinary health suggest hypotheses to test regarding risk factors that might account for disease prevalence in SLE beluga. One of these hypotheses focuses on chronic exposure to industrial contaminants. Although a cause-and-effect relationship for an observed disease in SLE beluga and chemical contamination may never be conclusively demonstrated, a precautionary approach is advisable given the known effects that some of these contaminants have on the health of animals and humans.
Research and monitoring of this population are critical and should be continued and expanded to support or refute hypotheses that may explain the lack of growth and recovery of this population in a degraded ecosystem. To preserve this population of beluga as well as the biodiversity of the SLE, it is essential to mitigate the effect of detrimental factors and detect new threats by continuing research and monitoring, by protecting critical habitat, by decreasing the production and release of environmental toxic contaminants, and by promoting actions to minimize anthropogenic climatic changes.
Footnotes
Acknowledgements
We thank the staff of the Réseau québécois d’urgences pour les mammifères marins, Pierre Béland, Carl Guimond, and Richard Plante for the collection and transport of carcasses, as well as all the veterinarians and veterinary students involved in necropsies, including Sylvain De Guise, Christiane Girard, Igor Mikaelian, André Dallaire, and Sylvain Larrat. We also thank Robert Michaud and Pierre Béland for their long-lasting involvement and significant contribution to this program.
