Abstract
Background and aim
Artisanal refining of crude oil is an illegal refining process that contributes to environmental pollution through the release of Polycyclic Aromatic Hydrocarbons (PAHs). PAHs compounds are known for their destructive effects on the environment as well as their harmful effects on human health. This study thus assessed the concentrations of PAHs in water, soil, and fish in communities where artisanal refining of crude oil is practiced in Bayelsa State.
Materials and methods
This descriptive, comparative study was conducted in Sampou (mildly exposed community), Gbarain, and Nembe (severely exposed communities) in Bayelsa State. Water, fish, and soil samples were collected using pre-existing environmental media collection guidelines and sent to the laboratory for GC-FID determination of the PAH concentrations. The data obtained were analyzed using the Statistical Package for Social Sciences (SPSS) software.
Results
Mean and (total) PAHs concentration in water samples obtained from Sampou was 3.50 ± 4.51 (59.59) μg/L; Gbarain 1.76 ± 4.35 (29.87) μg/L and Nembe 1.90 ± 4.20 (32.25) μg/L. A significant difference in the concentrations was also identified p-value: of 0.021. The mean concentration of PAHs in soil samples obtained from Sampou was 10.73 ± 15.53 (183.38) μg/kg; Gbarain 12.00 ± 19.57 (204.32) μg/kg and Nembe was 8.49 ± 10.07 (144.48) μg/kg. Finally, the mean concentration in fish samples obtained from Sampou was 5.62 ± 5.92 (95.43) μg/kg; Gbarain 3.81 ± 5.57 (64.75) μg/kg and Nembe 4.61 ± 5.33 (78.35) μg/kg. The difference in these concentrations was however not significant. Source diagnostic ratios of the PAHs in the water included Flt/(Flt + Pyr) ratio of 0.23, 0.16, and 0.21; Ant/(Ant + Phe) ratio of 0.87, 0.76, and 0.87 as well as BaA/(BaA + Chr) ratio of 0.43, 0.51 and 0.66 in Sampou, Gbarain and Nembe respectively.
Conclusion
Concentrations of total PAHs in water and fish samples obtained from the three communities exceeded the acceptable limits for ƩPAHs of 2.0 μg/L and 2 μg/kg in water and fish respectively stipulated by the United States Environmental Protection Agency and the Nigerian Petroleum Regulatory Authority. ƩPAHs concentrations from the samples obtained from Sampou were also higher than the other two communities. There is a need for regular environmental monitoring of PAH concentrations, especially in oil-producing communities, and a shift of focus toward the elimination of pyrolytic sources of PAH pollution.
Introduction
The Niger Delta region plays host to more than 80% of oil and gas activities that take place in Nigeria and there are increased tendencies for pollution by crude oil and its derivatives. 1 This tendency for pollution is traceable to the occurrence of accidental oil spills during oil exploration and transportation activities, gas flaring, fire outbreaks at exploration sites, and illegal oil bunkering activities. Illegal oil bunkering is the most common form of crude oil theft which usually involves the sabotage of oil pipelines and the diversion of crude oil from these pipelines for commercial gains.2,3
The unlicensed and illicit refining process of crude oil is known as artisanal refining. It is a highly inefficient means of refining crude oil as a result of its inability to refine as much as 80% of the heavy end of the crude and the tendency to produce refined petroleum products of low quality. Artisanal refining involves the use of primitive illegal stills (often a combination of metal pipes and drums that are welded together) in which the boiling of crude oil occurs. The distilleries are heated on open fires which are fueled by crude oil that is tipped into pits in the ground. As part of the oil burns away, some seep into the ground with the potential consequence of contaminating the underground aquifer. 4 During the course of the refining, dense clouds of soot, gaseous and particulate compounds are produced and released together with the unrefined portions into the environment. Carbon black and soot are thus potential air pollutants produced in large amounts as a result of artisanal refining activity. A major carcinogenic constituent found in black carbon and soot is polycyclic aromatic hydrocarbons (PAHs).4–7
Environmental and health risks can potentially emerge from the contamination of the environment with these pollutants.8–10 For several years now, the problem associated with the release of polycyclic aromatic hydrocarbons (PAHs) into the environment has been worsened by several factors. These include increasing levels of artisanal refining activities; intense oil exploration;3,11 the use of leaded petrol and biomass fuel; inappropriate dumping and burning of toxic waste12,13 and so on. Whenever PAHs are released into the environment, they travel long distances due to their persistent nature and accumulate in plants, animals, and humans either by direct inhalation or indirectly through the food chain.14,15
Artisanal refining activity has been linked with some adverse effects which are experienced by soil micro-organisms. 16 Studies have also blamed artisanal refining activities for several disastrous effects on the wetland ecosystem,10,17 degradation of the environment, farmland, and, forests; 2 severe depletion of biodiversity and aesthetic scenery of forests; destruction of wildlife habitat and the disruption of water cycles. 17 Aquatic organisms have also been shown to be adversely affected by exposure to PAHs with manifestations of altered endocrine functions, DNA damage, retardation of growth etc. 18
Although a lot of research has been published on the effects of PAHs, to the best of our knowledge, only scant literature addresses the contribution of PAHs, which are released from artisanal refining activities to environmental pollution in Bayelsa state, Nigeria. Artisanal refining of crude oil coupled with a myriad of activities that relate to crude oil theft remains a strong impediment to achieving sustainable development in aspects of environmental and human health in the Niger Delta region of Nigeria. Reports of the high incidence of crude oil theft and artisanal refining have been made within the Niger Delta region of Nigeria.1,2,4,19 Environmental health problems associated with illegal refining activities of crude oil may exist in Bayelsa State, one of the States located in the Niger Delta region. 2 This study thus set out to determine and compare the disproportionate environmental pollution due to exposure to PAHs, within the context of artisanal refining activities, between oil-producing Communities in Bayelsa State, Nigeria. This study was essential for improving the understanding of the wide-ranging impacts of artisanal refining practices and providing a guide for relevant future decisions and actions by the government. This guide is necessary to safeguard the physical environment and well-being of populations in the Niger Delta. Findings made from this study are believed to be capable of spurring stakeholders in environmental health and sustainability into action to ensure the achievement of a sustainable environment in the Niger Delta region of Nigeria.
Materials and methods
This study utilized a descriptive, comparative design and was carried out in three communities in Bayelsa state. These communities included Ogbolomabiri in Nembe LGA, which has been impacted by artisanal refining activities, and Gbarain in Yenagoa LGA which has been impacted by both artisanal refining and gas flaring activities. These two communities were regarded as being severely exposed to the PAH pollutants in this study. The third study site was Sampou in Kolokuma/Opokuma LGA which has neither been impacted by artisanal refining nor gas flaring activities and is regarded as a community mildly exposed to PAHs pollutants. Sampou Community was chosen to serve as a reference group because it is also a community located in Bayelsa State in which artisanal refining has never been reported. It should be noted that this reference community was regarded as a mildly exposed site considering its downstream geographical relationship with communities in bordering Delta and Rivers States where artisanal refining activities are carried out as well. 10 The formula for the sample size for comparing two means was used in calculating the sample size for environmental samples used for this study. 20 The mean and standard deviation of the attribute of interest (PAHs concentration in water) in a PAHs non-exposed group was obtained from the study by Adekunle et al. at Ife north in Osun state. 21 Also, the mean and standard deviation of the attribute of interest (PAH concentration in water) in a PAHs exposed group was obtained from the study conducted by Aigberua 22 at Imiringi in Bayelsa state.
Environmental monitoring of the PAH levels of the water, soil, and fish was done and PAH levels were determined using the gas chromatography/flame ionization detector (GC/FID) method as described by Aigberua.
22
PAHs diagnostic ratios were used as a tool for the identification of the emission sources of the PAHs in the different environmental media.23–25 Water samples were collected from five points in each study community using previously cleaned 1-L capacity glass bottles. The geographic locations for these points (areas on the map with orange-colored pins) in Nembe (40N53l 32ll 60E 40l 31ll, 40N 521 95II 60E 391 82II 40N 531 04II 60E 391 26II), Gbarain (50N 021 79II 60E 281 22II, 50N 011 05II 60E 291 28II) and Sampou (50N 141 78II 60E 351 42II, 50N 14,146II 60E 351 04II) are shown in Figure 1. Geolocations of environmental sample collection points.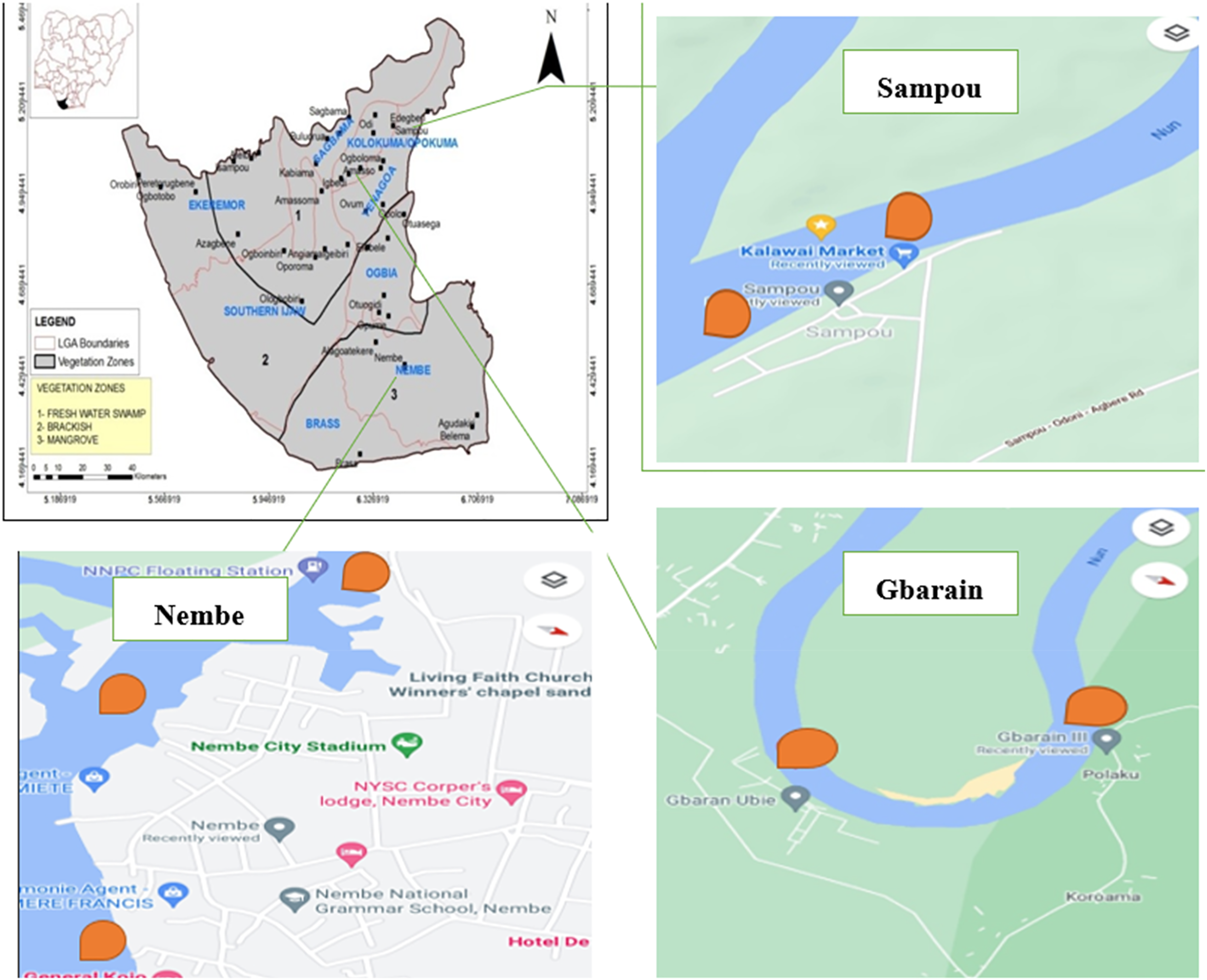
The soil samples were collected from the 3 different study sites. At the artisanal refining and gas flaring impacted sites, a line transect was set from the center point of the refining site in any accessible direction and soil samples were collected along the transect line at a distance of 200 m and 400 m. At every sampling point, 2 soil samples each were randomly collected from the depth of 0–15 cm (topsoil) and 15−25 cm (subsoil) using a stainless soil auger. Altogether, 8 soil samples (4 samples each of topsoil and subsoil) were collected from each site. Samples were collected using pre-labeled bags. Five fish samples of Tilapia fish (Oreochromis niloticus) harvested at the study sites were collected from each of the three sampling sites. Soil and fish samples were also retrieved from similar locations as the water samples as shown in Figure 1. These samples were immediately delivered to Analytical concepts Ltd, Elelenwo, Port Harcourt, Rivers State, Nigeria; for gas chromatographic analysis of 16 PAHs. This was done using the Gas Chromatographic System (6890 series and 6890 plus) version A.03.08 equipped with a dual detector (FID-ECD), dual column and TriPlus AS auto-sampler with a helium carrier gas and a quadrupole Mass Spectrometer (Agilent 5975 MSD) based on USEPA method 8100. 26
Extraction and clean-up of PAHs from water samples
The liquid-liquid extraction technique was applied to extract PAHs in surface water samples using the method given by the United States Environmental Protection Agency 27 as well as the slight modification on the procedure used in another study. 28 Firstly, the 250 mL water sample was homogenized before emptying the entire volume into a 500 mL separating funnel. Afterward, PAHs were extracted by a three-batch extraction process using 20 mL of dichloromethane (DCM)/n-hexane (1:3 v/v) mixed solvents at each time. The sample-solvent mixture in the separating funnel was vigorously agitated with intermittent ejection of built-up pressure from the tap of the glass funnel. This was done to eliminate the risk of blowing up the glass material. Thereafter, the organic extract was dehydrated by filtering through anhydrous sodium sulfate. Organic contaminants in filtered extracts were cleaned by eluting through a 10 mm I. D (internal diameter) × 250 mm long chromatographic column packed with glass wool, a slurry of silica gel, and anhydrous sodium sulfate. The cleaned-up extract was reconstituted to about 1.0 mL, after being concentrated in a temperature-regulated water bath at 35–40°C. Finally, sample extracts were transferred into glass vials with rubber-crimped caps. Another 250 mL portion of water sample was transferred into a separating funnel and spiked with pre-deuterated PAHs mixture (naphthalened8, phenanthrene-d10, chrysene-d12 and perylene-d12) as internal standards, to establish the efficiency of the extraction protocol. The recovery rates ranged between 92% and 107%. Exactly 20 mL of dichloromethane (DCM)/n-hexane (1:3 v/v) mixed solvents were added to the sample mixture, thoroughly mixed and kept standing to allow for adequate phase separation before dehydration and filtration, followed by clean-up and elution through a chromatographic column. Afterward, the eluted extracts were concentrated to 1.0 mL volume and stored in air-tight rubber-crimp cap glass vials. 22
Quantification of PAHs from water samples
Exactly 1 μL portion of the reconstituted extract was injected into the gas chromatograph-flame ionization detector (GC-FID) using a hypodermic syringe. Nitrogen served as the carrier gas while a combination of hydrogen and air was used to create an ionization environment at the detector head. The various fractions of the aromatic compounds were automatically detected at the FID (whose response is dependent on the composition of the eluted vapor) as they emerged from the column. Results were expressed in μg/L units. Standard pre-set operating conditions of the GC-FID were ensured. 22 The instrument conditions above are based on manufacturer recommendations and PAHs method suitability for repeatability of analytical data on the HP 6890 Plus GC-FID, version A.03.08. Quality assurance/quality control (QA/QC) parameters applied during GC-FID analysis included the spike concentration, concentration obtained, percentage recovery, limit of detection (LOD), and limit of quantification (LOQ). 22 The surface water (SW) matrix was used to calculate extraction recovery efficiency for different PAHs. The instrument limit of detection (LOD) and limit of quantification (LOQ) were also estimated and ranged between 0.001–0.04 μg/mL and 0.004–0.10 μg/mL respectively. The acceptable recovery range of the equipment was stipulated between 90 and 110%. 22
Extraction and quantification of PAHs from soil samples
The extraction method that has been previously applied was adopted in this study. 29 They were air-dried at room temperature, crushed, and sieved through a 2 mm mesh sieve. Five grams of the well-homogenized sample was weighed, and this was transferred into 50 mL glass vials with Teflon crimp caps. They were then mixed with 2 g of anhydrous sodium sulfate and the mixture was vigorously agitated using 15 mL n-hexane/acetone (2:1 v/v) for 10 min. It was then left submersed in the mixed solvent for an hour. The supernatant was transferred into glass vials and the residue (soil) was re-extracted with 15 mL n-hexane/DCM (3:1 v/v). The recovered supernatants were combined into another 50 mL glass vial. Thereafter, the organic partition was made moisture-free by straining it through anhydrous sodium sulfate. The organic phase of the filtered extract was then rinsed by eluting in a 10 mm internal diameter × 250 mm long chromatographic column packed with glass wool, a slurry of silica gel, and anhydrous sodium sulfate. The extract which has now become cleaned up was reconstituted to about 10 mL before being concentrated to about 1.0 mL using a thermally regulated water bath at temperatures between 35°C and 40°C. Results were expressed in concentration units of μg/kg. 29
Another 5 g portion of the soil was ceded into a 50 mL glass vial with Teflon crimp caps. It was spiked with pre-deuterated PAH internal standards mix (naphthalene-d8, phenanthrene-d10, chrysene-d12 and perylene-d12). Thereafter, the sample mixture was extracted and analyzed. The recovery percentage of the internal standard mix was used to derive the extraction efficiency (88.0%–103.0%). With the use of a hypodermic syringe, exactly 1 μL portion of the reconstituted extract was introduced into the injection port of the gas chromatograph-flame ionization detector (GC-FID). PAH components were eluted through the capillary column based on their boiling points (BP) and molecular weights (MW). Standard pre-set operating conditions of the GC-FID included an initial oven temperature of 65°C, final oven temperature of 320°C, injector temperature of 275°C, an inlet pressure of 14.8 lbf/in2 as well as an inlet condition set to split-less. Others include setting the detector temperature at 310°C, nitrogen flow amount of 30 mL/min, hydrogen flow amount of 35 mL/min, and an airflow rate of 250 mL/min. 29
Extraction and quantification of PAHs from fish samples
Fresh fish samples were cleaned using distilled water to get rid of dirt and the tissues were dissected and cut up into smaller pieces, after which a sub-sample was taken from the homogenate. The blending of the samples was then done, and the blended components were kept in airtight containers before the extraction process. Two grams of samples were weighed into a 50 ml clean extraction container. 10 ml of dichloromethane serving as the analar grade extraction solvent was then added into the sample and these were mixed thoroughly and allowed to settle. The mixtures were then carefully filtered into a clean solvent-rinsed extraction bottle, using filter paper fitted on Buchner funnels. Transferred extracts were concentrated to 2 μl for cleanup/separation in gas chromatographic analysis. The manufacturer’s elution protocol was strictly followed as in high-pressure solvent extraction. Separation took place and was automatically detected as it eluted from the column (at a constant flow rate) by the FID detector. It was also ensured that analyte detection was not affected by the difference between the diluent used for PAHs extraction and the experimental sample matrix. 30
The Statistical Package for Social Sciences (SPSS) version 25 (IBM, Armonk, New York, USA) was used to perform both descriptive and inferential analyses. The One-way Analysis of variance (ANOVA) was used to compare the concentrations of PAHs in the water, soil, and fish samples obtained from the three communities. All analyses were conducted at a 95% confidence level and a p-value ≤ 0.05 was considered as being statistically significant. Ethics approval for the research was obtained from the Research Ethics Committee of the University of Port Harcourt (Approval number: UPH/CEREMAD/REC/MM72/097). Permission to collect the water, soil, and fish samples were also sought from the necessary authorities of the Communities involved. During the collection of samples from the environment, it was ensured that the appropriate techniques were applied and that no harm came to the environment in the course of doing so.
Results
Polycyclic aromatic hydrocarbons concentrations in water
Mean and total concentrations of PAHs from water.
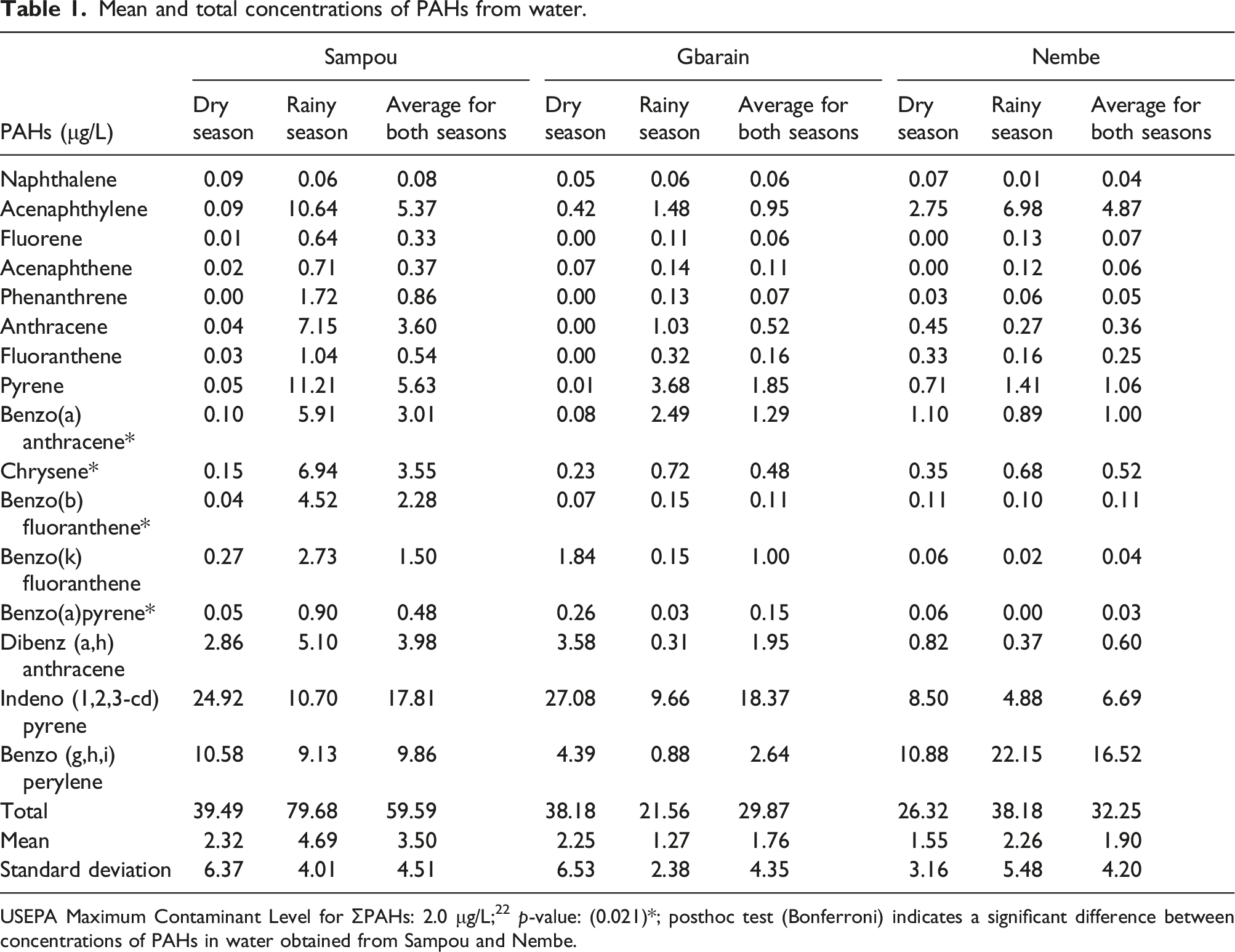
USEPA Maximum Contaminant Level for ƩPAHs: 2.0 μg/L; 22 p-value: (0.021)*; posthoc test (Bonferroni) indicates a significant difference between concentrations of PAHs in water obtained from Sampou and Nembe.
Polycyclic aromatic hydrocarbons concentrations in soil
Mean and total concentrations of PAHs from soil.
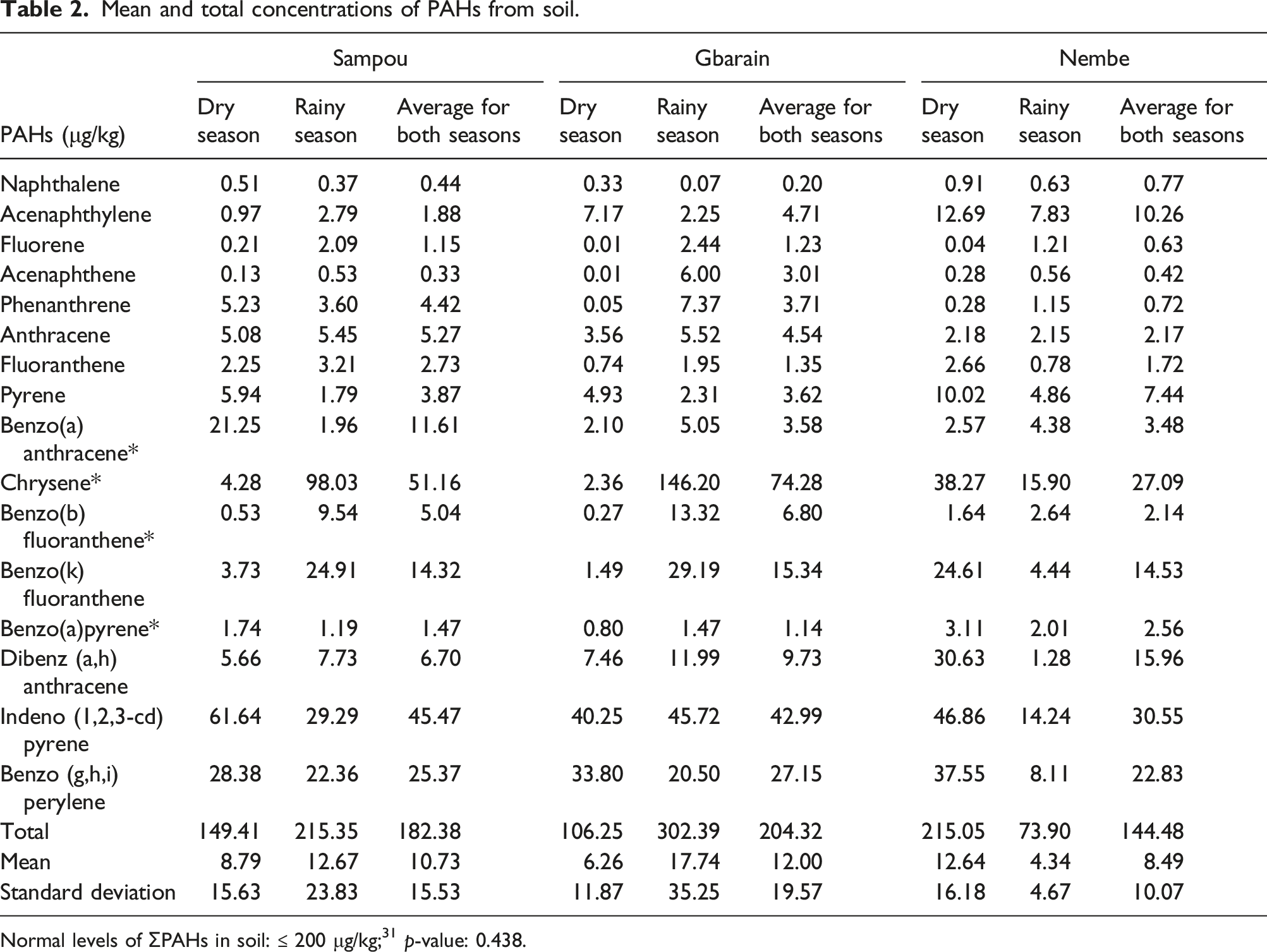
Normal levels of ƩPAHs in soil: ≤ 200 μg/kg; 31 p-value: 0.438.
Polycyclic aromatic hydrocarbons concentrations in fish
Mean and total concentrations of PAHs from fish.
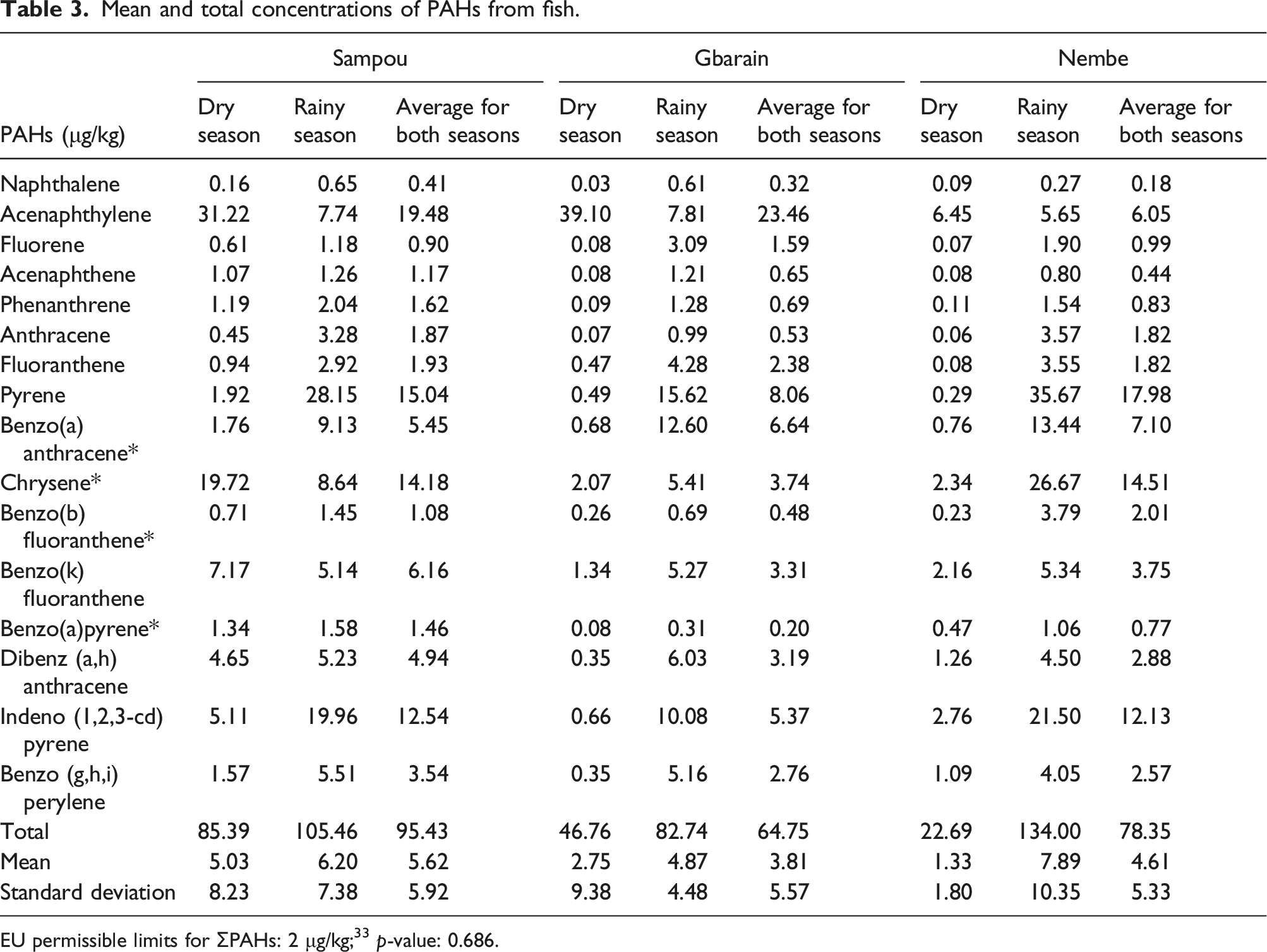
EU permissible limits for ƩPAHs: 2 μg/kg; 33 p-value: 0.686.
Source diagnostic ratios of the polycyclic aromatic hydrocarbons concentration in water, soil, and fish samples
In the water samples obtained from Sampou, diagnostic ratios used in characterizing the PAHs concerning their sources gave an Flt/(Flt + Pyr) ratio of 0.23; Ant/(Ant + Phe) ratio of 0.87, and BaA/(BaA + Chr) ratio of 0.43. In samples obtained from Gbarain, Flt/(Flt + Pyr) ratio was found to be 0.16; Ant/(Ant + Phe) ratio was 0.76 and BaA/(BaA + Chr) ratio was 0.51. In samples obtained from Nembe, Flt/(Flt + Pyr) ratio was found to be 0.21; Ant/(Ant + Phe) ratio was 0.87 and BaA/(BaA + Chr) ratio was 0.66.
Also, in soil samples obtained from Sampou, diagnostic ratios gave an Flt/(Flt + Pyr) ratio of 0.46; Ant/(Ant + Phe) ratio of 0.55, and a BaA/(BaA + Chr) ratio of 0.43. In samples obtained from Gbarain, Flt/(Flt + Pyr) ratio was found to be 0.29; Ant/(Ant + Phe) ratio was 0.70 and BaA/(BaA + Chr) ratio was 0.25. In samples obtained from Nembe, Flt/(Flt + Pyr) ratio was found to be 0.17; Ant/(Ant + Phe) ratio was 0.77 and BaA/(BaA + Chr) ratio was 0.14.
Source identification of PAHs in environmental media.
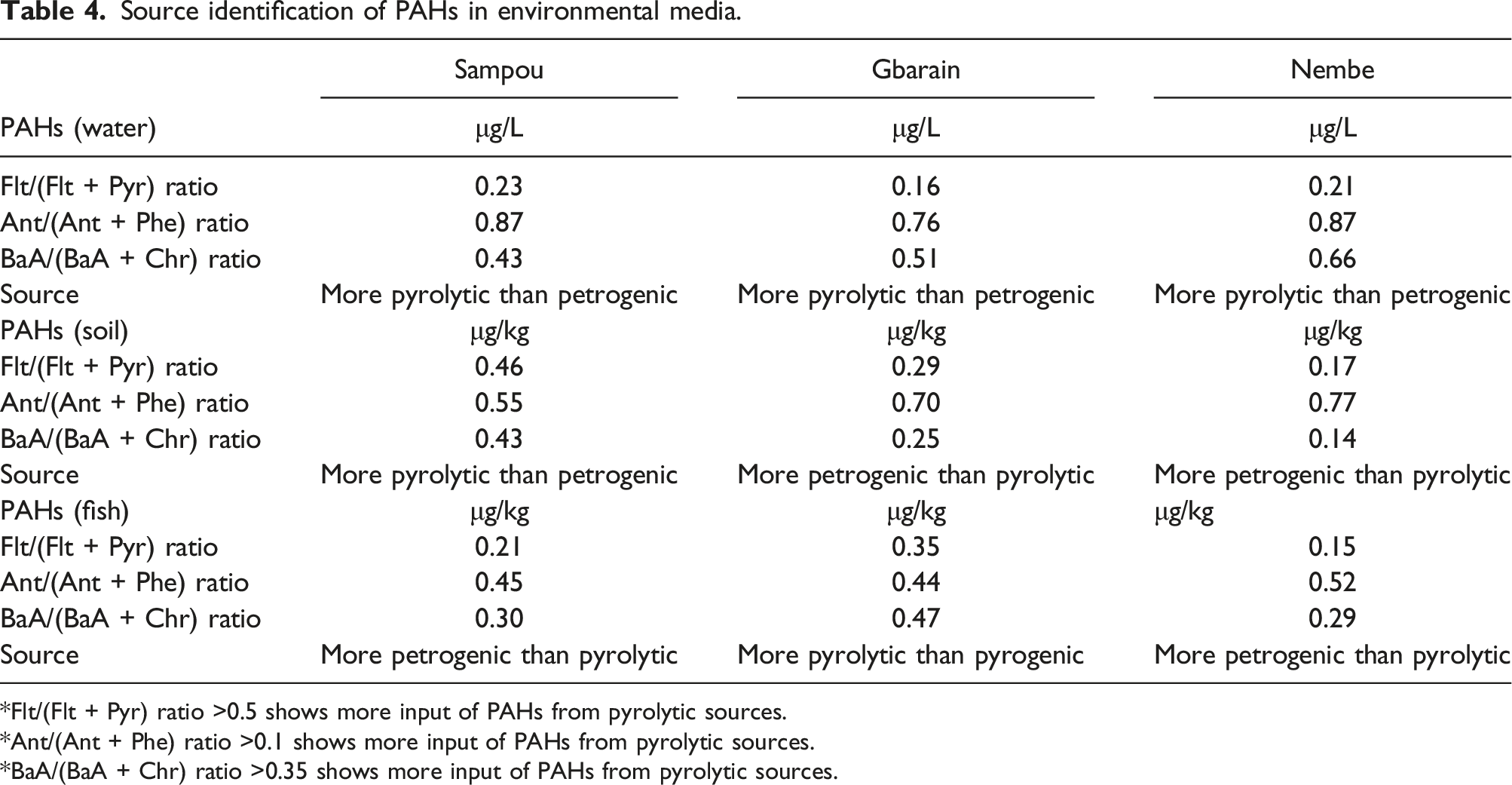
*Flt/(Flt + Pyr) ratio >0.5 shows more input of PAHs from pyrolytic sources.
*Ant/(Ant + Phe) ratio >0.1 shows more input of PAHs from pyrolytic sources.
*BaA/(BaA + Chr) ratio >0.35 shows more input of PAHs from pyrolytic sources.
Discussion
In this study, it was found that varying concentrations of the PAHs occurred in these environmental media with a higher proportion of samples exceeding the guideline limits of PAHs for each medium as stipulated by various regulating agencies.22,32–34
Regarding the pollution of the water bodies in this study, the total concentration of PAHs obtained from water samples in the three communities was found to exceed the required maximum contaminant level (MCL) of total PAHs to be found in water as given by the United States Environmental Protection Agency and the Nigerian Petroleum Regulatory Authority (NPRA).22,27 It was also found that the concentrations of total PAHs in water obtained from Sampou (mildly exposed community) were higher than the concentrations from other communities where artisanal refining, as well as gas flaring activities, are carried out. This difference was however not statistically significant and the finding can be related to the source of the PAHs using their source diagnostic ratios. Water samples in all three communities showed more pyrolytic than petrogenic sources of PAHs which could have been obtained from the incineration of waste, bush-burning, dumping unsegregated waste along river banks as well as biomass combustion.22,35,36 This finding is in line with the findings of studies conducted to survey concentrations and source characteristics of PAHs in surface waters in Imiringi, Bayelsa State, and in Shitou Koumen, China where vegetation, wood, and coal combustion were identified as the primary sources of PAHs in the surface water.22,35 This has also been predominantly said to occur among households in rural areas of developing countries as a result of their exclusive reliance on burning wood for their cooking and heating needs. 37 The implication of this finding points to the fact that pollution of the environment from pyrolytic sources is just as important as those from petrogenic sources considering that the end-result is environmental pollution and its attendant problems. It is thus recommended that as efforts are put into mitigating environmental pollution by PAHs from petrogenic sources, environmental regulatory bodies should also ensure that pyrolytic sources of these compounds are controlled as much as possible. Another possible explanation for the higher PAH concentrations in Sampou, is its proximity with bordering Delta State which has been reported as an area where artisanal refining activities are carried out as well. 10 Considering that PAHs compounds can travel distances as a result of their environmentally persistent nature in combination with meteorological factors temperature, turbulence, windspeed, and so on; there could have been a spread of PAHs compounds through the atmosphere from artisanal-refining/gas flaring sites from surrounding States to the mildly exposed community in this study.14,15,38
This notwithstanding, petrogenic PAH sources were identified in Gbarain and Nembe which show that when artisanal refining or gas flaring activities are carried out, they have the capacity of causing the release of PAHs into the environment. The distilleries where artisanal refining of crude oil is done are heated on open fires which are fueled by crude oil that is tipped into pits in the ground. This can seep into the ground with the potential consequence of contaminating the underground aquifer 4 and the destruction of aquatic and marine life. 2 Also, most of these oils are hydrophobic, firmly adsorbing to bottom sediments, and end up destroying the food chain of aquatic ecosystems, causing a distortion of natural biological cycles as well as impaired growth of marine organisms. 39 Aquatic-sourced foods from this kind of environment might also have objectionable odor or flavor, thereby adversely impacting their acceptance as food and marketability. 5
In the soils, it was found that the mean concentration of the 16-priority PAHs obtained from Gbarain, had the highest concentration of PAHs in soil when compared with the other two communities and was classified as having soil that was weakly polluted by PAHs. 33 Concentrations of PAHs in soil samples obtained from Sampou and Nembe communities were however found to lie within safe limits. 33 Nembe however presented with the highest levels of naphthalene in the soil although it did not exceed normal limits. This is relevant because naphthalene has been reported as the most water-soluble and bioavailable PAH in the environment. 40 Applying source diagnostic ratios also showed that the source of PAHs in the soil from the “mildly exposed” community was more pyrolytic e.g., bush burning, coal and combustion than petrogenic sources. This was however not the case for the other communities as the ratios suggested that the PAHs were more from petrogenic than pyrolytic sources. 22 This finding of raised PAH concentrations in areas impacted by artisanal refining activities is similar to findings made by other authors.10,41,42 Very high concentrations of PAHs were reported to be found in crude oil-polluted soils obtained from an artisanal refining site in Tombia, Rivers State. 41 Likewise, total hydrocarbon content in artisanal-refining impacted sites was found to be higher than the acceptable limit of environmental standards and guidelines in studies conducted on assessing the impact of artisanal petroleum refining on the vegetation and soil quality.10,42 There are certain environmental and epidemiological implications of this level of pollution of the soil by PAHs. This is especially relevant in areas where artisanal refining of crude oil as well as other crude oil exploration activities such as gas flaring are carried out. These implications could include the adverse effects on soil biodiversity, fertility, and vitality with consequent drastic depletion of vegetation in affected areas.10,16 This is a result of atmospheric contaminants produced which have the capacity of acidifying the soil and reducing soil nutrients. 43 The heat emitted into the environment at gas flare sites is capable of causing temperature changes that result in stunted growth, scorched plants as well as withered young crops.44–46 Furthermore, in areas where the major source of livelihood is agriculture; the pollution of the soil by activities that promote the pollution of the environment with crude oil including artisanal refining of crude oil, can result in the loss of livelihoods as well as reduction of food production.37,47
Also, total PAHs concentrations in fish samples obtained from the three communities exceeded the normal limits of fish contamination by PAHs of 2 μg/kg. 34 Concentrations of total PAHs in fish obtained from Sampou were also found to be higher than the concentrations from other communities where artisanal refining as well as gas flaring activities were carried out. Fish samples in Sampou and Nembe communities showed the source of the PAHs to be more petrogenic than pyrolytic, however samples from Gbarain revealed the PAHs sources to be more pyrolytic than petrogenic. This finding is contrary to the expectation that fish sample PAHs concentration in Sampou would have been sourced from more of pyrolytic than petrogenic sources. An explanation for the more petrogenic source of the PAHs found in fish samples in Sampou could be the migration of fish from polluted tributaries of surrounding oil-producing areas sharing close boundaries with Sampou and are located further upstream in Delta and Rivers states.34,48,49 It has also been reported that the levels of PAHs found in unprocessed foods in rural settings could be a reflection of the background contamination originating from long distance transportation of contaminated particles in the atmosphere. 50
The more petrogenic source of PAHs in Nembe where artisanal refining of crude oil is practiced which exceeded normal limits could be a pointer to the nature and extent of environmental pollution subsequent to local artisanal refining activities.30,34 This finding is corroborated by results of a study which was conducted to determine the PAHs levels of fish from polluted Niger Delta coastal waters. This study demonstrated that oil spills due to pipeline sabotage and artisanal refining of the crude oil could increase mean PAHs concentration to as high as 35.800 ± 0.100 μg/kg which exceeds normal limits.30,34 This finding implies that these PAHs-polluted fish are harvested for food by humans who ingest them in different amounts and frequencies. 36 When ingested, these compounds become metabolized in the body and could end up being retained in the body where they cause some acute or chronic health abnormalities. These abnormalities are of course dependent on the intensity, duration, and frequency of exposure to the PAHs compounds.31,51–54 Considering the harmful properties of many polycyclic aromatic hydrocarbons, their capacity to travel long distances and bioaccumulate, as well as their persistent nature in the environment, extensive exposure to these compounds is of great public health concern.55–57
In conclusion, concentrations of total PAHs in water and fish samples obtained from the three communities in this study exceeded normal limits stipulated by regulatory agencies. Soil samples in the Gbarain community were also found to be weakly polluted by PAHs and ƩPAHs concentrations from the fish and water samples obtained from Sampou were higher than the other two communities. Recommendations made for policymakers included the strict adherence to regulations for ensuring environmental health and safety during the conduct of oil and gas activities within the oil-rich region of the Niger Delta. There is also the need for policymakers to shift focus towards the reduction of other sources of PAH contamination within the Niger Delta region of Nigeria including bush burning and other anthropogenic activities. This can be done by formulating policies that promote the use of environmentally-safe methods for cooking and waste management. For practice, it was recommended that constant environmental monitoring of environmental media including soil, water, and so on; in oil-producing regions should be regularly done. It is also necessary that illicit activities such as artisanal refining which result in environmental degradation should be continually dissuaded through unwavering enforcement of environmental health laws. Finally, it was recommended that as efforts are put into place for mitigating environmental pollution by PAHs from petrogenic sources, environmental regulatory bodies should also ensure that pyrolytic sources of these compounds are controlled as much as possible. This can be done through the provision of effective waste collection mechanisms that pools the waste from the populace and disposed of using environmentally-safe methods. Others include the promotion of catalytic converters in car exhausts that converts harmful exhaust gases to harmless substances among others.
Footnotes
Acknowledgements
We would like to thank Zeelicious foods and FeyiKola foundation for their assistance in this research.
Author contributions
BE-E and BO researched literature and conceived the study. BE-E, BO, and EO were involved in protocol development, gaining ethical approval, patient recruitment and data analysis. BE-E wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: BE-E is a Doctoral student of the World Bank, Africa Centre of Excellence for Public Health and Toxicological Research, University of Port Harcourt.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the World Bank.
Ethical approval
Ethical approval for this study was obtained from the Research Ethics Committee of the University of Port Harcourt (Approval number: UPH/CEREMAD/REC/MM72/097).
Informed consent
Informed consent was not applicable to this study. This study assessed the concentrations of Polycyclic Aromatic Hydrocarbons in environmental media in Bayelsa.
