Abstract
Pancreatitis has been described in cats with diabetes mellitus, although the number of studies currently available is very limited. In addition, ketoacidosis has been hypothesized to be associated with pancreatitis in diabetic cats. The aims of the present study were to investigate whether diabetic cats have pancreatitis and to determine if pancreatitis is more frequent with ketoacidosis. Samples of pancreas were collected postmortem from 37 diabetic cats, including 15 with ketoacidosis, and 20 control cats matched for age, sex, breed, and body weight. Sections were stained with hematoxylin and eosin, double-labeled for insulin/CD3, insulin/CD20, insulin/myeloperoxidase, insulin/PCNA, and glucagon/Ki67, and single-labeled for Iba1. A previously proposed semiquantitative score was used to characterize pancreatitis, along with counts of inflammatory cells. Scores of pancreatitis and the number of neutrophils, macrophages, and lymphocytes in the exocrine pancreas did not differ between diabetic and control cats or between diabetic cats with and without ketoacidosis. Of note, PCNA-positive acinar cells were increased (P = .002) in diabetic cats, particularly near islets (P < .001). Ki67-positive acinar cells were increased only near islets (P = .038). Ketoacidosis was not linked to proliferation. The results suggest that histopathologic evidence of pancreatitis may not be more frequent in diabetic cats and that ketoacidosis may not be associated with it at the time of death. Augmented PCNA-positive acinar cells might indicate increased proliferation due to chronic pancreatitis. The reason behind the prevalent proliferation of acinar cells surrounding pancreatic islets deserves further investigation.
Diabetes mellitus (DM) is one of the most common endocrinopathies in cats, and its incidence is increasing because of a rise in predisposing factors, such as obesity and physical inactivity. 2,15 Although DM is frequently diagnosed in cats, few studies have characterized the histopathologic changes occurring in the exocrine pancreas. Histopathologic evaluation of the exocrine pancreas in 6 diabetic cats that died with ketoacidosis revealed lymphoplasmacytic pancreatitis in 5 (83.3%), pancreatic adenocarcinoma in 1, and pancreatic nodular hyperplasia in 1 (16.7%). 4 Goossens et al examined diabetic cats postmortem and found chronic pancreatitis in 17 of 37 (46%; including 5 with ketoacidosis), acute to subacute pancreatitis in 2 (5.4%; both with ketoacidosis), pancreatic adenocarcinoma in 7 (18.9%), adenoma in 1 (2.7%), and multifocal pancreatic cysts in 1 (2.7%). 8 The high prevalence of pancreatitis led the authors to hypothesize that inflammation of the exocrine pancreas is associated with DM. An association between pancreatitis and ketoacidosis was also assumed because of the high prevalence of inflammation in the exocrine pancreas in cats with this diabetic complication. 4,8
The possible role of pancreatitis in the pathogenesis of DM in cats was investigated in several recent clinical studies. 7,18 Cats with long-standing DM had increased serum feline pancreatic lipase immunoreactivity (a marker of pancreatitis) as compared with that of control cats, 7 suggesting an association between pancreatitis and this endocrine disease. One of our studies revealed that about 60% of diabetic cats (18 of 30) had suspected pancreatitis at the time of initial diagnosis, based on increased activity of serum 1,2-o-dilauryl-rac-glycero-3-glutaric-acid-6′-methylresorufin ester lipase, increased feline pancreas-specific lipase activity, or the presence of ultrasonographic abnormalities of the pancreas. 18 In addition, diabetic cats with increased feline pancreas-specific lipase values were less likely to achieve clinical remission of DM, which suggests that glycemic control may be more difficult when pancreatitis is present, despite appropriate therapy. Histopathologic evaluation of the pancreas was not carried out in any of those clinical studies.
In light of the paucity of histopathologic analyses of the exocrine pancreas in cats with DM, the aim of the present investigation was to determine whether diabetic cats have histopathologic evidence of pancreatitis when compared with a control population of cats well-matched for age, sex, breed, and body weight. Furthermore, analysis was conducted to verify if diabetic cats with ketoacidosis are more frequently affected by pancreatitis than are those without the complication.
Materials and Methods
Cases and Controls
Cats that met the following criteria were included in the study: DM was diagnosed according to established clinical and laboratory findings 16 ; they died spontaneously or were euthanized at the Clinic for Small Animal Internal Medicine, University of Zurich (Switzerland), between 1997 and 2009; a postmortem examination was carried out; and pancreas samples of sufficient size were available. Randomly selected cats with a similar distribution of age, sex, breed, and body weight that died spontaneously or were euthanized because of any other disease during the same corresponding period at the same institution were used as controls, provided that pancreas samples were available. The present investigation on the exocrine pancreas is part of a previously published study. 17,19
Pancreas Histopathology
Pancreas samples, approximately 1.5 cm in length, were fixed in 10% neutral buffered formalin, embedded in paraffin, and cut to yield serial 3- to 4-µm-thick sections. The sections were stained with hematoxylin and eosin (HE) via an automatic staining system (Leica Autostainer XL, Leica Biosystems, Milan, Italy). Sections were deparaffinized and rehydrated in descending alcohol solutions. After washing in distilled water, sections were stained in a filtered solution containing 25% Mayer hematoxylin and 75% Carazzi hematoxylin (Sigma-Aldrich, Milan, Italy) for 7 minutes, rinsed in running tap water for 5 minutes, stained with eosin (Sigma-Aldrich) for 1 minute, dehydrated in ascending alcohol solutions, and cleared in xylene. All sections were mounted onto labeled glass slides with Eukitt (Bio-Optica, Milan, Italy). Sections were evaluated through light microscopy (Olympus BX-40, Milan, Italy). All samples were randomly selected; therefore, standardization of the pancreas section could not be performed.
Pancreas Immunohistochemistry
Single-labeling immunohistochemistry was carried out for the macrophage antigen ionized calcium-binding adapter molecule 1 (Iba1). 14 Double-labeling immunohistochemistry was carried out for insulin and the neutrophil antigen myeloperoxidase; insulin and the T-lymphocyte antigen CD3; insulin and the B-lymphocyte antigen CD20; insulin and the proliferating cell nuclear antigen (PCNA) expressed in G1 and S phases; and glucagon and the antigen Ki67 expressed in G1, S, G2, and M phases of the cell cycle. 20 Details of all specificities are provided in the supplemental material.
Morphology of the Exocrine Pancreas and Diagnosis of Pancreatitis
Sections stained with HE were evaluated to identify morphologic lesions of the exocrine pancreas. The frequency of nodular hyperplasia and neoplasia was calculated in 30 fields at 40× magnification. The semiquantitative scoring system developed by De Cock and coworkers was used to diagnose acute or chronic pancreatitis and to define its severity. 5
Analysis of the Number of Proliferating Cells in the Exocrine Pancreas and Inflammatory Cells: Description of Scoring Systems
To determine the number of proliferating acinar cells, PCNA-positive/insulin-negative cells in the exocrine pancreas were counted in 20 fields at 40× magnification. The number of proliferating acinar cells was also determined by counting PCNA-positive/insulin-negative cells in the 5 cell layers around the islets in 500 randomly selected cells of the exocrine pancreas. The same counting method was used to determine the number of Ki67-positive cells.
To determine the number of inflammatory cells in the exocrine pancreas, CD3- and CD20-positive lymphocytes, myeloperoxidase-positive neutrophils, and Iba1-positive macrophages were counted in 20 fields at 40× magnification, excluding those in large vessels. For either CD3- or CD20-positive lymphocytes, a semiquantitative score was developed that combined the degree and distribution of infiltrates. In particular, the degree of infiltration was subjectively defined as 0 (rare), 1 (mild), 2 (moderate), and 3 (severe), and their distribution was defined as 0 (focal), 1 (multifocal), 2 (scattered), and 3 (diffuse). The sum of the degree and distribution of infiltrates yielded the final score. The lymphocyte score and the count of neutrophils and macrophages in the exocrine pancreas were used in addition to the morphologic scoring system of De Cock and coworkers to improve the diagnosis of pancreatitis in cats. 5 The latter scoring system was developed to provide a standardized approach for histopathology of the pancreas in cats. The grading system is based on a point system (0–3, with the latter as maximum) for lesions that typically occur in acute and chronic pancreatitis. For acute pancreatitis, 2 lesions are considered, including interstitial edema/necrosis of mesenteric fat and neutrophilic inflammation. For chronic pancreatitis, 3 lesions are considered, including lymphocytic inflammation, interstitial fibrosis, and cystic acinar degeneration. The sum of the points for each criterion in acute and chronic pancreatitis was calculated, with a maximal score of 6 for acute pancreatitis and 9 for chronic pancreatitis. A total point score of 0 was considered normal pancreatic histopathology. A score of 1–3 total points was considered mild chronic pancreatitis, 4–6 moderate, and 7–9 severe. A score of 1–2 total points was considered mild acute pancreatitis, 3–4 moderate, and 5–6 severe. 5 All features described in the morphologic scoring system proposed by De Cock and coworkers were used for the present study. The number of fields and cells examined to complete the above analyses was arbitrarily chosen.
Statistical Analysis
To check the process of matching diabetic and control cats, the age, sex, breed, and body weight of cats of the 2 groups were tested with the Fisher exact test and the Mann-Whitney test. Differences in frequency of nodular hyperplasia and neoplasia in the exocrine pancreas between diabetic and control cats were analyzed with the Fisher exact test. The Mann-Whitney test was used to identify differences for counts of proliferating cells in the exocrine pancreas between diabetic and control cats and between diabetic cats with and without ketoacidosis. Differences in frequency and score of pancreatitis, based on the method proposed by De Cock and coworkers, 5 were analyzed with the Fisher exact test and the Mann-Whitney test, respectively. Scores of CD3- and CD20-positive lymphocytes in the exocrine pancreas were compared between groups through r × c contingency tables. Pancreas sections and images were assessed by 2 pathologists (L.C., F.L. or S.F., F.G.) in a blinded fashion, and interobserver discrepancies were resolved by consensus.
Results
Cats
The 37 diabetic cats consisted of 26 (70.3%) neutered males and 11 (29.7%) spayed females, with a median age of 11 years (range: 2–18 years) and a median body weight of 4.9 kg (range: 2.2–10.0 kg). There were 29 (78.4%) crossbred cats and 8 (21.6%) purebred cats, which included Siamese (n = 2), Maine Coon (n = 2), Burmese (n = 1), Chartreux (n = 1), Oriental shorthair (n = 1), and Russian Blue (n = 1). Concurrent disorders at death were recorded in 21 (56.7%) diabetic cats and included chronic hepatitis, liver lipidosis, and severe chronic renal failure in each of 3 cats; chronic enteritis and chronic stomatitis in each of 2 cats; and acute enteritis, acute pancreatitis, asthma, hyperthyroidism, idiopathic epilepsy, idiopathic hypercalcemia, megacolon, and multicentric lymphoma in 1 cat each. Diabetic ketoacidosis was identified in 15 (40.5%) cats at the time of death.
The 20 control cats consisted of 14 (70.0%) neutered males and 6 (30.0%) spayed females, with a median age of 12 years (range: 2–17 years) and median body weight of 4.3 kg (range: 2.5–9.0 kg). Seventeen (85.0%) were crossbred cats, and 3 (15.0%) were purebred cats, which included 2 Chartreux and 1 British shorthair. Disorders recorded at death were chronic hepatitis in 3 cats; hypertrophic cardiomyopathy, meningioma, and severe chronic renal failure in 2 cats each; and acute enteritis, colon carcinoma, fibrosarcoma, hyperthyroidism, immune-mediated anemia and thrombocytopenia, liver carcinoma, liver lipidosis, lung carcinoma, multicentric lymphoma, pneumonia, and restrictive cardiomyopathy in 1 cat each. There were no significant differences between diabetic and control cats with respect to age, sex, breed, and body weight.
Morphology and Proliferation of the Exocrine Pancreas
Examination of HE-stained sections revealed pancreatic nodular hyperplasia of the acinar cells of the exocrine pancreas in 13 of 37 (35.1%) diabetic cats and in 7 of 20 (35.0%) control cats (P = 1.000). Nodules were scattered in the exocrine pancreas and did not contain endocrine tissue. Cats with nodular hyperplasia had ≥1 pancreatic nodules (diabetic cats: median, 1 in 30 fields at 40× magnification; range, 1–3; control cats: median, 1; range, 1–3). A pancreatic adenocarcinoma was diagnosed in 1 diabetic cat. In this cat, 2 adenocarcinoma nodules with diameters of 1.3 and 8 mm were documented in the pancreas; the former was not infiltrating, whereas the latter infiltrated the organ. Exocrine pancreatic tumors did not occur in control cats.
The number of scattered proliferating acinar cells positive for PCNA in exocrine tissue, excluding hyperplastic nodules, was significantly greater in diabetic cats than in control cats (diabetic cats: median, 0.7 per field at 40× magnification; range, 0–15.2; control cats: median, 0.5; range, 0–2.7; P = .002; Figs. 1–3). When the analysis was repeated by counting acinar cells within 5 cell layers around the islets, the median number of proliferating acinar cells was 4 times higher in diabetic cats than in controls (diabetic cats: median, 0.4 in 500 acinar cells; range, 0–9; control cats: median, 0.1; range, 0–2; P < .001; Fig. 4). The number of proliferating cells positive for Ki67 scattered in the exocrine pancreas tended to be higher in diabetic than control cats (diabetic cats: median, 0.4 per field at 40× magnification; range, 0–4.0; control cats: median, 0.3; range, 0–3.2; P = .083), and the number of proliferating cells positive for Ki67 in the 5 cell layers of the exocrine pancreas surrounding the islets was significantly higher in cats with DM compared with that of controls (diabetic cats: median, 0.3 in 500 acinar cells; range, 0–6; control cats: median, 0.1; range, 0–2; P = .038; Figs. 5, 6). Many cells of the hyperplastic nodules and adenocarcinoma of the exocrine pancreas were positive for PCNA and Ki67.
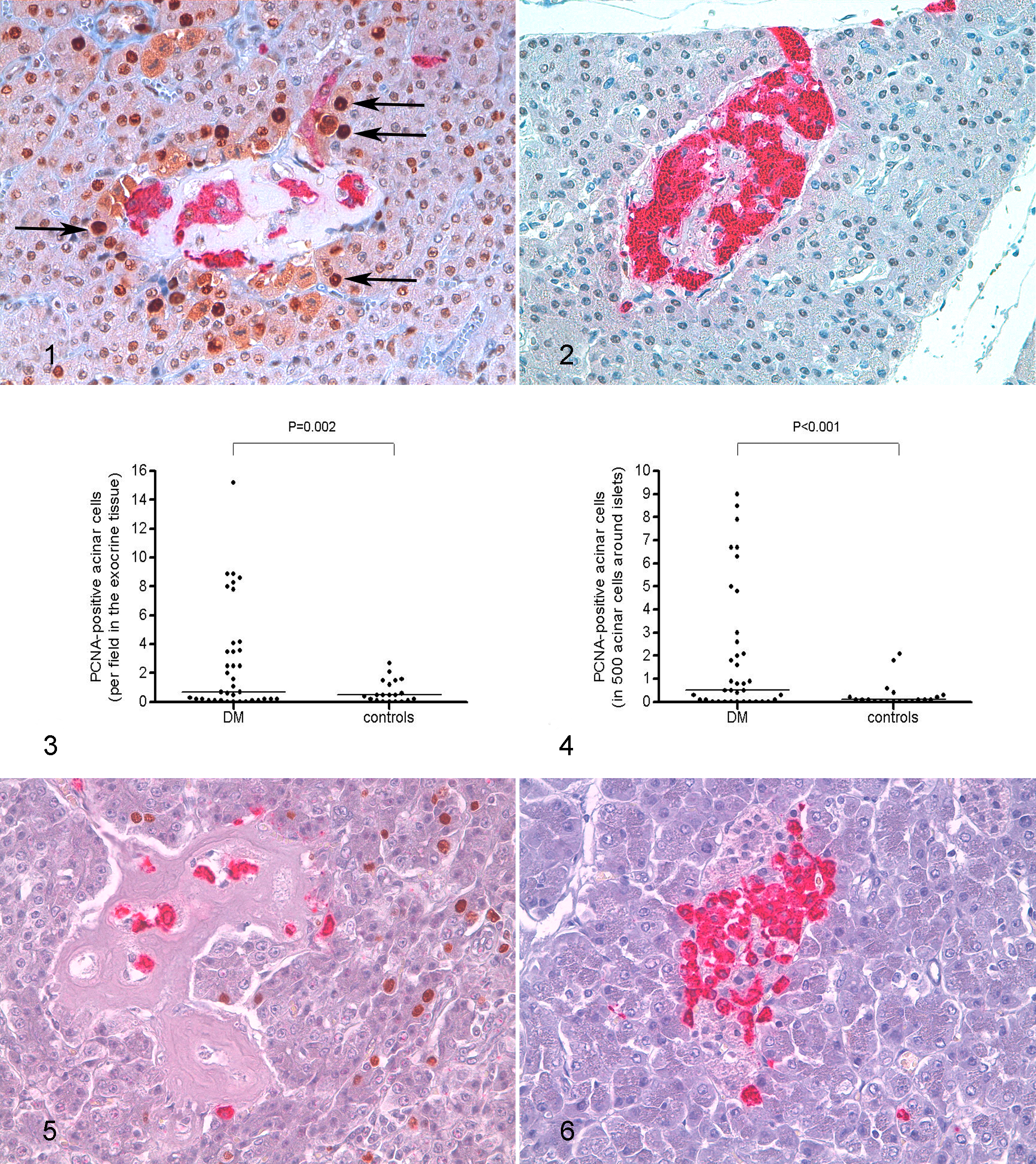
Pancreas, cat. Immunohistochemistry for proliferating cell nuclear antigen (PCNA) and insulin.
Inflammation of the Exocrine Pancreas in Diabetic and Control Cats
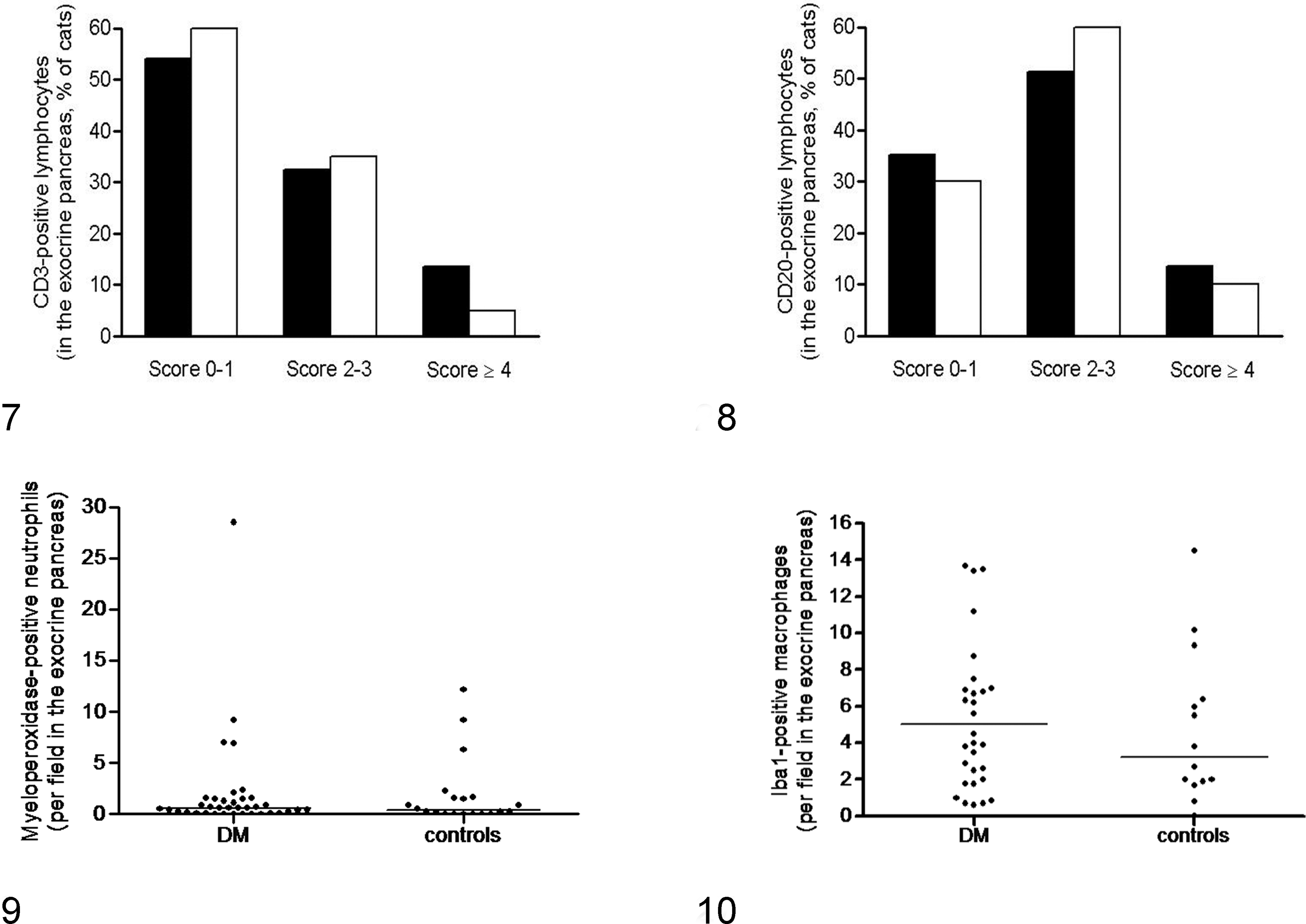
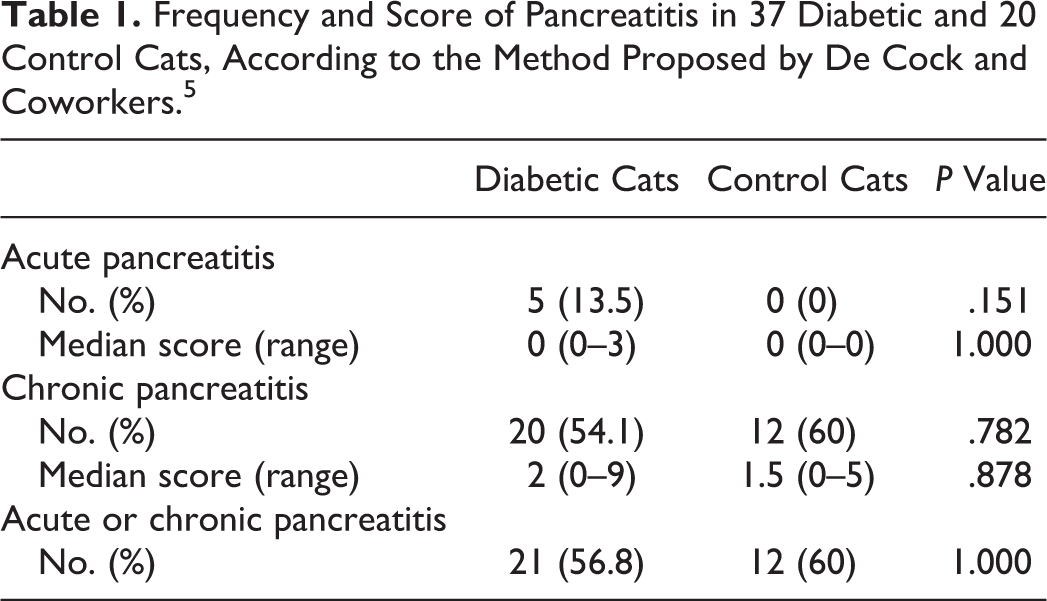
The score of CD3-positive lymphocytes in the exocrine pancreas did not differ between diabetic and control cats (diabetic vs control cats: score 0–1, 54.1% vs 60.0%; score 2–3, 32.4% vs 35.0%; score ≥4, 13.5% vs 5.0%; P = .606) nor that of CD20-positive lymphocytes (diabetic vs control cats: score 0–1, 35.1% vs 30.0%; score 2–3, 51.4% vs 60.0%; score ≥4, 13.5% vs 10.0%; P = .814; Figs. 7, 8). The median number of myeloperoxidase-positive neutrophils was similar between groups (diabetic cats: median, 0.6 per field at 40× magnification; range, 0–28.5; control cats: median, 0.4; range, 0–12.2; P = .951; Fig. 9). The median number of Iba1-positive macrophages was also similar between groups (diabetic cats: median, 5.1 per field at 40× magnification; range, 0.6–49.2; control cats: median, 3.3; range, 0–14.5; P = .340; Fig. 10). Based on the morphologic method developed by De Cock and coworkers, 5 the frequency and score of acute and chronic pancreatitis, as well as the frequency of either type of pancreatitis, did not differ between diabetic and control cats (Table 1). With regard to acute pancreatitis, edema or fat necrosis was observed in 4 of 37 (10.8%) diabetic cats, which was mild in 3 and moderate in 1, and in none of the 20 control cats. With regard to chronic pancreatitis, fibrosis was observed in 19 of 37 (51.4%) diabetic cats, which was mild in 11 and moderate or severe in 8, and in 11 of 20 (55.0%) control cats, which was mild in 7 and moderate or severe in 4; cystic degeneration was observed in 9 of 37 (24.3%) diabetic cats, which was mild in 6 and moderate or severe in 3, and in 3 of 20 (15.0%) control cats, which was mild in all. The frequency of morphologic features did not differ between groups. Representative pictures of acute and chronic pancreatitis are provided in Supplemental Figures 1 and 2.
Exocrine pancreas, inflammatory cells, cat.
Frequency and Score of Pancreatitis in 37 Diabetic and 20 Control Cats, According to the Method Proposed by De Cock and Coworkers. 5
Inflammation and Proliferation of the Exocrine Pancreas in Diabetic Cats With and Without Ketoacidosis
Regarding inflammatory cells distributed in the exocrine pancreas, differences were not observed between diabetic cats that died with and without ketoacidosis for the score of CD3-positive lymphocytes (ketoacidosis vs no ketoacidosis: score 0–1, 60.0% vs 50.0%; score 2–3, 26.7% vs 36.4%; score ≥4, 13.3% vs 13.6%; P = .893) or for the score of CD20-positive lymphocytes (ketoacidosis vs no ketoacidosis: score 0–1, 26.7% vs 40.9%; score 2–3, 60.0% vs 45.5%; score ≥4, 13.3% vs 13.6%; P = .804; Figs. 11, 12). The median number of myeloperoxidase-positive neutrophils was also similar (ketoacidosis: median, 0.7 per field at 40× magnification; range, 0–6.9; no ketoacidosis: median, 0.5; range, 0–28.5; P = .526) as well as that of Iba1-positive macrophages (ketoacidosis: median, 4.0 per field at 40× magnification; range, 0.7–13.4; no ketoacidosis: median, 6.5; range, 0–49.2; P = .259; Figs. 13, 14).
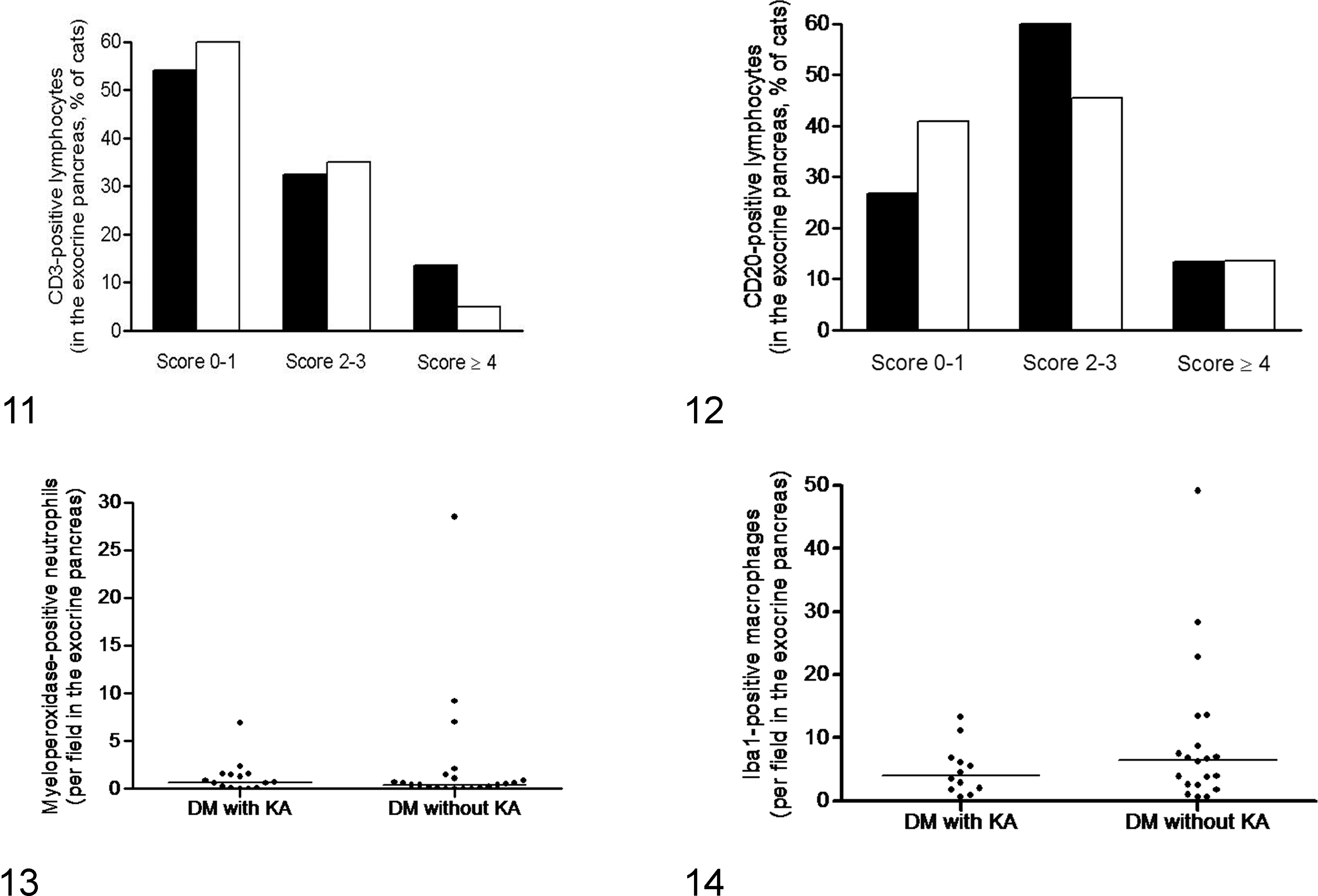
Exocrine pancreas, inflammatory cells, cat.
In addition, the frequency and scores of pancreatitis based on the method developed by De Cock and coworkers 5 did not differ between diabetic cats with and without ketoacidosis. In particular, chronic pancreatitis was observed in 8 of 15 (53.3%) diabetic cats with ketoacidosis and in 12 of 22 (54.5%) diabetic cats without ketoacidosis (P = 1.000); the median score of chronic pancreatitis was 2.0 (range, 0–7) in the former and 1.5 (range, 0–9) in the latter (P = .791). Three (20.0%) diabetic cats with ketoacidosis had acute pancreatitis and 2 (9.1%) diabetic cats without ketoacidosis (Fig. 9).
The number of scattered proliferating acinar cells positive for PCNA in the exocrine tissue did not differ between diabetic cats with and without ketoacidosis (ketoacidosis: median, 1.5 per field at 40× magnification; range, 0–8.9; no ketoacidosis: median, 0.7; range, 0–15.2; P = .926); differences were not documented when the analysis was repeated by counting acinar cells within 5 cell layers around the islets (ketoacidosis: median, 0.5 in 500 acinar cells; range, 0–9; no ketoacidosis: median, 0.7; range, 0–7.9; P = .653). Also, the number of proliferating cells positive for Ki67 scattered in the exocrine pancreas and surrounding the islets was similar between diabetic cats with and without the complication (data not shown).
Discussion
In the present study, diabetic cats had no evidence of more severe inflammation of the exocrine pancreas than that of control cats. Among diabetic cats, inflammation of the exocrine pancreas was not more frequent in those with ketoacidosis. Diabetic cats had an increase in the number of proliferating acinar cells when compared with controls.
Morphologic assessment of the exocrine pancreas revealed nodular hyperplasia in approximately one-third of diabetic and control cats. Hyperplastic nodules did not appear to contain endocrine cells and were scattered in the exocrine tissue. Hyperplastic nodules are a frequent finding in the exocrine pancreas of older cats at necropsy, but their significance is unknown. 10 It has been suggested that the nodules represent age-related hyperplastic foci rather than a response to previous injury or an early stage of progressive pancreatic epithelial neoplasia. 9 Indeed, pancreatic adenocarcinoma was diagnosed in only 1 diabetic cat in our study. A recent study showed that tumors of the exocrine pancreas can predispose cats to DM. 12 It is therefore possible that the pancreatic adenocarcinoma seen in our study caused DM by infiltrating and disrupting viable islets.
It has been suggested that pancreatitis may be a predisposing factor for the development of DM in cats, 8 possibly because of involvement of the islets of Langerhans in the inflamed exocrine tissue, which leads to injury of the β cells. In the present study, the number of T and B lymphocytes, neutrophils, and macrophages scattered in the exocrine pancreas did not differ between diabetic and control cats. Furthermore, the frequency and score of acute or chronic pancreatitis were similar in both groups. Acute pancreatitis was uncommon, whereas more than half of the diabetic and control cats had evidence of chronic pancreatitis; these results are in agreement with another study in which histopathologic evaluation of the pancreas showed similar prevalence to ours for acute and chronic pancreatitis in diabetic cats. 8 Therefore, pancreatic inflammation may not be specifically associated with DM in cats at necropsy. However, in a recent clinical study, diabetic cats had significantly higher feline pancreatic lipase immunoreactivity levels than those of controls, suggesting an association between pancreatitis and DM. 7 In addition, we recently observed that sustained hyperglycemia for 10 days in healthy cats increases the number of neutrophils infiltrating the exocrine pancreas. 21 It is therefore possible that pancreatitis occurs in some diabetic cats soon after disease onset and that hyperglycemia contributes to its pathogenesis. Furthermore, in diabetic cats, the number of T and B lymphocytes, neutrophils, and macrophages scattered in the exocrine pancreas did not differ between cases that died with and without ketoacidosis; differences were also not observed with regard to frequency and score of pancreatitis between the 2 groups of diabetic cats. Previous studies hypothesized that ketoacidosis is associated with pancreatitis because of the high prevalence of inflammation documented in the exocrine pancreas of cats with the complication. 4,8 In humans with diabetic ketoacidosis, acute pancreatitis is diagnosed in at least 10% to 15% of cases and is more likely to occur if marked acidosis and hyperglycemia are documented, clearly suggesting a link between the 2 disorders. 13 The present results in diabetic cats suggest that ketoacidosis might not be associated with inflammation of the exocrine pancreas in this species, although prospective studies should be performed to clarify this issue.
An unexpected finding was the increased proliferation rate of acinar cells in the exocrine pancreas of diabetic cats, especially near the islets, as shown by either PCNA or Ki67 staining. In humans with chronic pancreatitis, acinar cells proliferate at a higher rate because of increased epithelial cell turnover, 3 but it is not known whether the proliferation is more prominent around the islets. The reason for elevated numbers of proliferating cells in this particular location of the exocrine pancreas is unclear. One explanation might be that the cycling cells represent exocrine tissue cells undergoing transdifferentiation, possibly into endocrine cells. Transdifferentiation from acinar to endocrine cells has been demonstrated in rodent models of DM by transfecting acinar cells with transcription factors (eg, neurogenin 3). 1 However, in the present diabetic cats, double-labeling immunohistochemistry for Ki67 and glucagon or for PCNA and insulin did not yield acinar cells concurrently positive for cycling and endocrine antigens, making the hypothesis of transdifferentiation questionable. Further studies are therefore needed to clarify the origin of these cells in diabetic cats. Of note, although PCNA is expressed in fewer phases of the cell cycle than Ki67, 20 in the present study more acinar cells were immunopositive for PCNA. Similar results were recently achieved in human pancreatic tissue. 11 A possible reason to explain their distinct expression might be nonspecific labeling of other processes within the nuclei independent of replication. To support this hypothesis, PCNA has been shown to be involved in processes of DNA repair in hamsters. 6 If DNA repair is partly responsible for the increased immunopositivity of acinar cells to PCNA in diabetic cats, the cause remains unclear. Differences were not observed for the number of PCNA-positive acinar cells between diabetic cats with and without ketoacidosis, excluding this complication as a possible source of DNA damage.
Our study had some limitations. The morphologic scoring system 5 used in the present investigation led to diagnose acute or chronic pancreatitis in more than half of the diabetic and control cats, suggesting that pancreatic inflammation is very common in this species. However, identification of pancreatitis is often difficult in living cats, 18 and its clinical diagnosis was achieved in only 1 diabetic cat. Therefore, in light of the large discrepancy observed between the frequency of histopathologic and clinical diagnosis of pancreatitis, the significance of the adopted scoring system 5 remains uncertain, and the term pancreatitis might have been misleading for some cases. Samples of pancreas were collected postmortem and did not include biopsy specimens obtained during the course of the disease. It is therefore possible that some of the results would have differed if pancreatic biopsies had been obtained at the time of initial diagnosis. For instance, some diabetic cats with ketoacidosis at initial diagnosis might have had a concurrent episode of pancreatitis that later became chronic, after recovery from the complication of DM. Because pancreatitis was compared in consideration of ketoacidosis at death and not at first admission, these cats could have partly biased the results. In addition, pancreatitis may not be evenly distributed in the left and right lobes or body of the pancreas in cats. 5 Because of the retrospective nature of the study, it was possible to obtain only a single sample from the pancreas. Absence of standardization in preparation of samples and collection from every part of the organ might have therefore caused failure to detect pancreatitis. However, the effect of this potential bias was expected to be minor because the location of sampling was random in both groups of cats.
In conclusion, pancreatitis was not associated with DM in cats, although the increased proliferation of acinar cells suggested increased turnover, which is seen in humans with chronic pancreatic inflammation. Furthermore, pancreatitis was not more frequent in diabetic cats with ketoacidosis as compared with those without the complication at the time of death.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Policlinico di Monza, Italy, for the work represented.
