Abstract
Canine distemper virus commonly infects free-ranging, terrestrial mesopredators throughout the United States. Due to the immunosuppressive effects of the virus, concurrent opportunistic infections are also common. Among these, secondary systemic protozoal infections have been described in a number of species. We report an unusual presentation of necrotizing encephalitis associated with Sarcocystis sp in four raccoons and one skunk concurrently infected with canine distemper virus. Lesions were characterized by variably sized necrotizing cavitations composed of abundant mineral admixed with inflammatory cells and protozoa. Sarcocystis sp was confirmed via immunohistochemistry using a monoclonal antibody to Sarcocystis neurona. The pathologic changes are similar to lesions in human AIDS patients infected with Toxoplasma gondii.
Canine distemper virus (CDV) causes a systemic infection in dogs and has been reported in all families of carnivores. Among many other cellular targets, CDV is a lymphotropic agent, with resultant lymphopenia rendering affected animals highly susceptible to opportunistic infections. Systemic disease—as a direct effect of viral infection as well as from secondary infections—commonly results in pneumonia, neurologic disease, or a combination thereof. This report describes a unique histologic presentation of concurrent CDV and Sarcocystis infection in four raccoons and one skunk, all arriving within a 15-month period for postmortem analysis at the School of Veterinary Medicine, University of California, Davis. In general, animals presented with neurologic signs, including ataxia, circling, tremors, and passivity or unresponsiveness to human contact. The amount of time in rehabilitation ranged from four to 28 days, and all animals were eventually euthanized due to grave prognosis for recovery or release.
All five animals had few to dozens of grossly visible, discrete, soft to cavitary lesions (diameters, ≤1 cm) affecting the brain. Gross lesions were most commonly found within the cerebrum (Fig. 1) and thalamus and rarely located in the cerebellum, pons, and brainstem. These regions had a characteristic thin, soft, and yellowish rim that surrounded a central friable, chalky to gritty center that was easily removed by manipulation or gentle agitation of the fixed tissue.
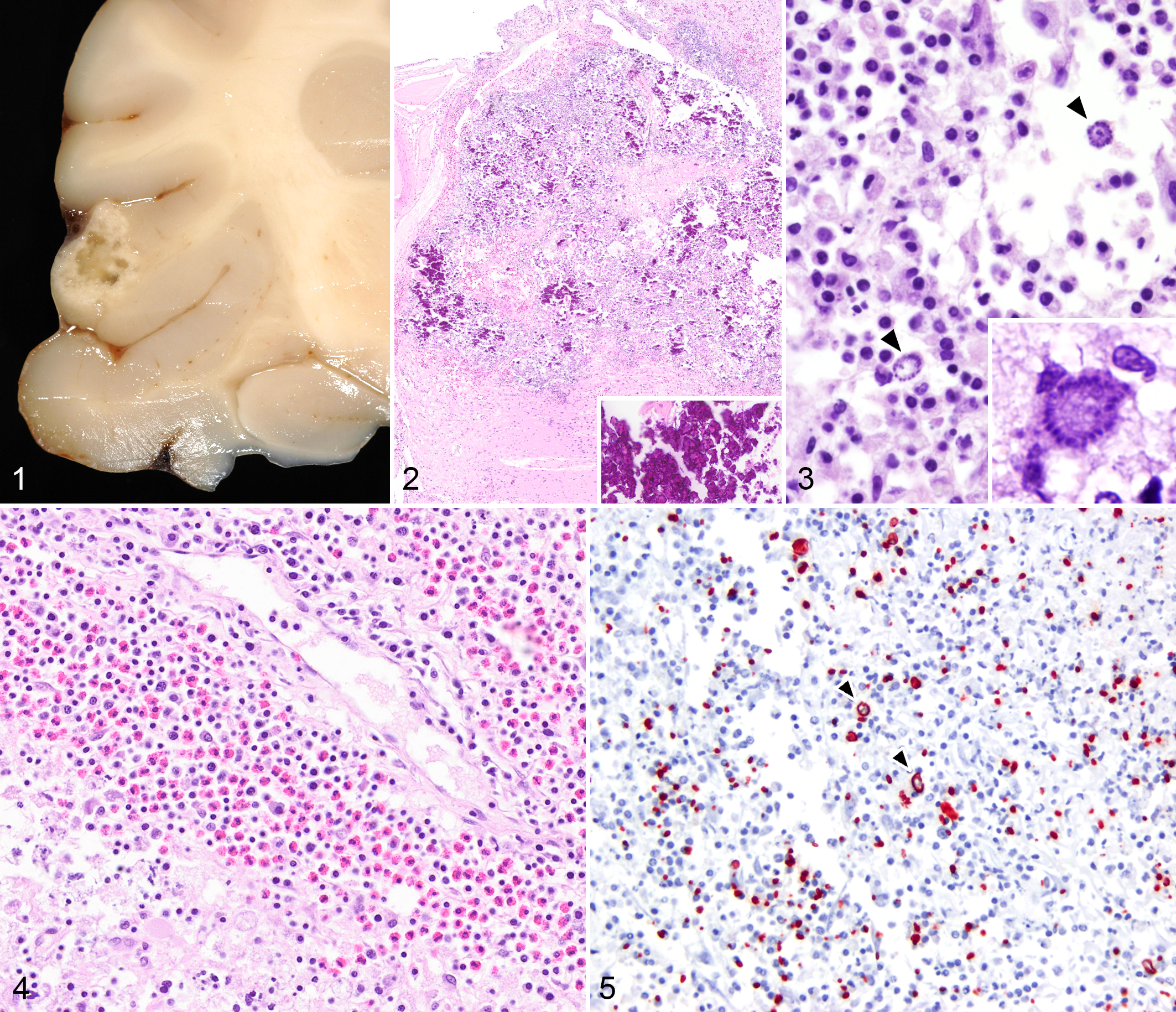
Necrotizing encephalitis with extensive mineralization associated with Sarcocystis sp and canine distemper virus, cerebral cortex, raccoon.
Histologically, these areas of malacia contained dense aggregates of mineral and necrotic debris, a scant network of small arterioles and capillaries, and numerous free merozoites and meronts (Figs. 2, 3). Blood vessels within regions of malacia were cuffed and infiltrated by eosinophils and lymphocytes with fewer histiocytes and plasma cells (Fig. 4). The adjacent meningeal vessels contained similar infiltrates. In addition, small-caliber arterioles and capillaries within the areas of malacia contained Von Kossa–positive mineral, which appeared within the lumen, adherent to the endothelium, or within the vascular wall (Suppl. Figs. 1–3). In these same locations, occasional neurons and astrocytes displayed cytoplasmic aggregates of similar deposits (Suppl. Figs. 1–4). Neighboring areas exhibited large numbers of reactive gemistocytic astrocytes and gitter cells containing abundant cellular debris, mineral, and cytoplasmic merozoites. A diagnosis of Sarcocystis was presumed on the basis of the morphology of the immature schizonts. These were typical of Sarcocystis in that their nuclei became multilobed and eventually gave rise to palisading merozoites arranged as a rosette-like structure around a residual body (Fig. 3). Merozoites were strongly immunoreactive to an anti–S. neurona monoclonal antibody, 2G5, 4 and not immunoreactive to anti-Toxoplasma or anti-Neospora antibodies, supporting the diagnosis (Fig. 5). More typical histologic lesions of protozoal infection were present in three of the five animals and included scattered nodular foci of gliosis and nonsuppurative encephalitis. In all cases, there were perivascular cuffs that included eosinophils.
Additionally, all examined brains had lesions typical of canine distemper virus that included multiple foci of astrogliosis and glial nodules, lymphoplasmacytic perivascular cuffing (Suppl. Fig. 5), occasional neuronal necrosis, and spongiosis of the white matter (interpreted as demyelination). Astrocytes and adjacent ependymal cells in these areas occasionally had small, round, glassy, eosinophilic intracytoplasmic or intranuclear inclusions (Suppl. Fig. 6). These regions and similar areas of gliosis without identifiable inclusions showed strong immunoreactivity with monoclonal anti-morbillivirus antibody DV2-12 (Custom Monoclonals International, West Sacramento, CA; Suppl. Fig. 7).
While dual CDV-protozoal infection has been reported in many species, cavitary lesions with extensive mineralization have not been described. A similar clinical presentation of immunosuppression and protozoal coinfection occurs in HIV-infected humans that develop encephalitis associated with Toxoplasma gondii. 3,10 Large regions of “abscessation” termed “target” lesions based on their radiographic findings typify such cases. The gross appearance of those lesions was similar to those described in this report, with notable histologic differences including mineralization and significant numbers of eosinophils.
In all of our cases, the amount and distribution of mineral in these lesions were striking. No evidence of concurrent renal disease was present in affected animals, though circulating calcium and phosphate levels at the time of death are unknown. Treatment protocols among rehabilitation centers vary, but most are limited to supportive fluid and nutritional therapy. There was no history of these animals receiving drugs associated with tissue mineralization. The pathogenesis of mineralization in these cases is speculative. In intermediate hosts, Sarcocystis spp undergo schizogeny in endothelial cells, 1,8 which may lead to vasculitis and ischemic damage. 1,6 The discrete nature of the malacic lesions observed in this case series suggests an interruption of the blood supply in the specific areas most likely caused by necrosis of the blood vessel in association with the parasitemia, rather than by an embolic process resulting in a sharply demarcated infarct. Thereafter, the area of necrosis may have undergone dystrophic mineralization. The mineral in vessels adjacent to and distant from the cavitations may have complicated ischemic damage in these areas. Interestingly, mitochondrial disorders of humans and at least 1 report in dogs have been associated with neuronal and vascular mineralization. 2 However, it would be highly speculative to suggest this as a contributing factor to pathogenesis of this infectious lesion.
Sarcocystis neurona is an apicomplexan parasite, best known in veterinary medicine as a causative agent of equine protozoal myeloencephalitis. The encephalitis in these raccoons and skunks may have resulted from reactivation of latent bradyzoites within the brain or other tissues or from an acute infection with dissemination to the brain
We assume from the natural history of these pathogens that immunosuppression due to CDV infection predisposed these animals to secondary disease caused by Sarcocystis infection. Relatively little is known about the circulation of virus among susceptible, free-ranging mesopredators, but it has been proposed that CDV genotypes of different virulence and cell tropism are involved. 5,7,9 It is unknown if the lesions that we attribute here to Sarcocystis infection could also be sequelae of the diverse immunopathology among CDV strains.
Footnotes
Acknowledgments
We thank the veterinarians and staff of Wildcare (Marin County, California) for the care of these animals and for providing the samples evaluated in this study. We would also like to thank Boehringer Ingelheim Vetmedica, Inc for their general support to our lab.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
