Abstract
Malaria remains one of the most significant public health concerns in the world today. Approximately half the human population is at risk for infection, with children and pregnant women being most vulnerable. More than 90% of the total human malaria burden, which numbers in excess of 200 million annually, is due to Plasmodium falciparum. Lack of an effective vaccine and a dwindling stockpile of antimalarial drugs due to increased plasmodial resistance underscore the critical need for valid animal models. Plasmodium coatneyi was described in Southeast Asia 50 years ago. This plasmodium of nonhuman primates has been used sporadically as a model for severe malaria, as it mimics many of the pathophysiologic features of human disease. This review covers the reported macroscopic, microscopic, ultrastructural, and molecular pathology of P. coatneyi infection in macaques, specifically focusing on the rhesus macaque, as well as describing the critical needs still outstanding in the validation of this crucial model of human disease.
Malaria is one of the most critical public health problems in the world today, and the various responsible plasmodia are prominent causes of morbidity and mortality in a range of developing nations. Almost half the world’s population is at risk of malaria infection, and vulnerable populations in endemic areas are most commonly children and pregnant women. The human malaria burden worldwide in 2012 was assessed by the World Health Organization to be in excess of 200 million cases, 90% of which were caused by Plasmodium falciparum. 121 Of the estimated 627 000 fatalities, most occurred in sub-Saharan Africa, and about 77% were in children <5 years of age. 121 Although these numbers are high, they represent a worldwide decrease in malaria morbidity and mortality since the start of the current century. Between 2000 and 2012, an estimated 3.3 million deaths due to malaria were averted, with 90% among children <5 years of age. 121
Although phase 3 trials on the latest malaria vaccine RTS,S are promising, 93 the current lack of an effective vaccine and increasing plasmodial drug resistance in Southeast Asia to the artemisinin-based antimalarial drugs 6 threaten to reverse these positive trends. 1,6,95 Of the Plasmodium spp infecting humans, P. falciparum is responsible for the majority of severe disease and significant morbidity. Clinical syndromes of severe disease vary between semi-immune children and immune-naïve adults in nonendemic areas. Severe disease is commonly manifested as cerebral malaria, anemia, and metabolic disturbance, with additional complications, such as renal and hepatic dysfunction, frequent in adults. 72 There is a great need to develop and test preventive vaccines, new antimalarial drugs, and adjuvant treatments for severe disease, all necessitating the use of animal models of the disease.
Plasmodium coatneyi has several biological features in common with P. falciparum as well as a variety of distinguishing phenotypic traits. Both parasites show 48-hour cycles of intraerythrocytic development, followed by rupture (schizogony), reinvasion of uninfected erythrocytes, and a resultant fever spike in the host. They also both demonstrate microvascular cytoadherence, form rosettes of infected red blood cells with uninfected erythrocytes, and develop electron-dense knobs on the cell surface, which are theorized to play a role in cytoadhesion. 2,43,53,54,57,110,111 Both parasites undergo deep vascular sequestration of trophozoite- and schizont-infected red blood cells and demonstrate a consistent predilection for specific tissues. 1,25,26,77,99,102 P. coatneyi was first identified in 1961 in an anopheline mosquito from Malaya. It was experimentally inoculated into a rhesus macaque monkey and its unique life cycle described. 33 The species was subsequently found in the blood of a cynomolgus macaque (Macaca fascicularis) in the Philippines 32 and has since been used as a model for falciparum-like malaria in a variety of macaque species.* Foundational work on the parasite and the pathophysiology of infection in macaques was and continues to be conducted at the Armed Forces Research institute of Medical Sciences (AFRIMS) in Thailand in collaboration with scientists and research groups worldwide. † Recognizing 5 decades of research with this malaria model and its continuing potential utility, we review and summarize the features of this primate malaria model.
Primate Malarias
Several animal models of human severe and cerebral malaria have been established over the past 50 years using various Plasmodium spp in rodent and nonhuman primate (NHP) hosts. ‡ The histologic and ultrastructural pathology as well as the clinical features and pathophysiology of severe disease have been assessed, in comparison to key features of the human disease, such as sequestration-dependent pathology or immune activation. These animal models are proven platforms for the testing of experimental antimalarial drugs to determine safety and efficacy, as well as for the evaluation of potential adjuvant therapeutics. 18,69 There has, however, been significant controversy regarding the quality and relevance of these various models. The concern has focused primarily on murine models of malaria as not being representative of the human disease, 22,51,59,108,118 leading to renewed interest in malaria models in NHPs.
There is a recent and significant shift in the understanding of falciparum malaria due to the application of molecular techniques, but these have not yet been evaluated in the NHP models. With the increased parasite resistance to various established and newer antimalarial compounds in our arsenal, therapeutics are required that can be tailored to modulate the immune response to effectively combat plasmodial infection. But to fully investigate novel compounds, the pathophysiology of malarial infection in the animal model used needs to be more clearly understood.
More than 250 members of the genus Plasmodium have been described, and new species and novel host–parasite interactions are reported with regularity. 42,63,90 Unique plasmodium–vertebrate interactions continue to be discovered in reptiles and birds. 38,45,49,85,87,120
The earliest genetic evidence of a malaria plasmodium, dated at approximately 30 million years ago, was obtained through the extraction of DNA from mosquitoes fossilized in amber from the Paleogene period. 88 The Old World primate plasmodia themselves are polyphyletic and subdivide into 2 major groups. The first of these consists of Plasmodium ovale, Plasmodium malariae, and sublineages that infect Old World monkeys. Parasites of Cercopithecidae monkey species from Asia are more distantly related and comprise 10 species, including P. coatneyi. This group also includes Plasmodium gonderi, infectious to Cercopithecidae from Africa; Plasmodium hylobati, which parasitizes Hylobatidae monkeys from Asia; and Plasmodium vivax, the human plasmodium that is closely related to P. simium, a species of malaria affecting New World monkeys. 44 The closest relative P. coatneyi appears to be Plasmodium knowlesi, derived from a single-ancestor species, based on evaluation of 4 apicoplast genome–encoded genes in the 9 members of the Asian clade. These include a small subunit rRNA, large subunit rRNA, and caseinolytic protease C genes. 75 P. falciparum is separate from all of these and is found within the second major group of Old World malarias, paired with the chimpanzee parasite Plasmodium reichenowi. 9,44 Potentially relevant to the debate over models, the rodent malarial parasites are located in a grouping distinct from primate and all other plasmodia. 28,29,61
While the various NHP species have far greater immunogenic homology to humans than do rodents, complications in the macaque and murine models include potential variation of the disease progression associated with the route of infection. In humans, the natural route of infection is via dermal injection subsequent to a mosquito bite, in comparison with the experimental murine models receiving predominantly intraperitoneal injections and NHPs inoculated intravenously. Furthermore, the macaque models of severe P. coatneyi malaria hitherto reported typically employ splenectomized animals, which are infected via an intravenous injection of parasitized blood and parasites that have been harvested from an animal with an intact spleen (hereafter referred to as spleen intact) or a splenectomized donor animal or infective-stage sporozoites dissected out from infected mosquitoes. Splenectomy results in much higher peripheral parasitemias and may modulate cytoadhesion and host immune reactions. Therefore, splenectomized experimental animals may be expected to suffer hyperparasitemia and more severe clinical pathologic abnormalities and clinical course of disease. Additionally, many of the studies to date have not had the luxury of using experimentally naïve animals but instead employed adult animals that had been previously used for studies involving antimalarial drugs or vaccine candidates in the relapsing malaria model (Plasmodium cynomolgi) or animals that had been previously infected with dengue virus, simian immunodeficiency virus, or enteric bacteria, all of which would have had some modulatory effects on the immune status in an individual animal. This presents a significant difference from the highly standardized murine experiments that use inbred mouse strains, but it is more in line with the genetic and environmental heterogeneity in affected human populations. §
Ecology of NHP Malarias
The fundamental life cycle of malaria in NHPs is quite similar to that of plasmodia-infecting humans. This has been underscored with the recent recognition of the simian malaria parasite P. knowlesi as a previously unrecognized but significant cause of malaria in humans. 16 Early taxonomy, based on microscopy, was often complicated by morphologic similarities among various species of Plasmodium and led to a degree of diagnostic confusion. 33,81,96,114,115
The ecologic and environmental requirements for the survival of any plasmodium depends on the parasite reaching a state of “balanced pathogenicity” with the host, whereby the majority of infections are nonfatal and allow a mosquito to acquire gametocytes through a blood meal and thereby propagate the life cycle. Experimentally, the NHP plasmodia of Southeast Asia can be transmitted by a wide range of mosquito species. However, the host–parasite combinations observed in the wild depend on the degree of pathogenicity within specific simian species. This is evidenced by the incompatibility of P. knowlesi and the rhesus macaque to cohabitate the same range of forest because P. knowlesi causes a rapidly fatal course of disease in this monkey, making malaria persistence impossible. 92,106 This degree of pathogenicity may be the result of recent evolutionary collision between the host and pathogen in which the malaria parasite maintained a high level of virulence due to insufficient evolutionary time to adapt to the host. This particular point is potentially important in selection and interpretation of a model, as differing species of macaques from diverse geographic origins may display differing susceptibility to various plasmodia. At present, only limited comparative studies have been conducted with P. coatneyi, examining the pathology in small numbers of animals from different geographic “strains” of either cynomolgus or rhesus macaques. 36,70,74 Studies in Malaysia in the 1960s examined multiple features of the ecology of the vector mosquitoes and described and categorized the malaria parasites found in the local populations of NHPs. It was discovered that an Anophelene mosquito was found to be a common vector for P. cynomolgi, Plasmodium inui, Plasmodium fieldi, P. knowlesi, and P. coatneyi. 117 These Anopheles mosquitoes primarily—and, apparently, preferentially—blood feed on primates. These mosquitos’ whole life cycle occurs within the forest, and they maintain a high level of endemic malaria within the NHPs coexisting in the same forested areas. This then translates to high rates of transmission to humans who enter these Southeast Asian rain forests for commerce, warfare, or habitation. 37
The epidemiology of transmission of disease within these border areas is especially critical in that the notable emergence of artemisinin-resistant falciparum malaria has come from such ecologic areas. The resistant plasmodia now appear increasingly established within the forests of the Thailand–Cambodian and Thailand–Myanmar borders. 8,12 The importance of this finding cannot be overstated since it holds the potential to reverse the worldwide trend of reduced P. falciparum–induced morbidity and mortality. 12,86 The arms race between malarial drug resistance and the limited arsenal of effective treatment options highlights the critical need for the development of a reproducible, reliable animal model that parallels the pathophysiology and tissue pathology of severe falciparum malaria in humans to act as a platform for future vaccine and drug development.
P. coatneyi is considered to be one of the more pathogenic plasmodia in nonadapted hosts, a fact that parallels P. falciparum infection in humans. The course of disease in splenectomized macaques is variable but typically culminates with severe anemia, renal dysfunction, metabolic acidosis, prostration, and death. 24 With P. falciparum, clinical presentation is highly heterogeneous, but the most frequently described complications, particularly in older patients, include dyspnea, respiratory distress, and eventual coma, with a relatively high mortality rate occurring within hours or days of admission. The literature suggests that the most significant factors leading to death in severe falciparum malaria in adults and children are metabolic acidosis and coma, as well as severely decreased renal function. 27,82,122
P. coatneyi has been reported as causing natural and experimentally induced disease in a variety of NHP species. However, only cynomolgus macaques (M. fascicularis), 33,34,62,74 pig-tailed macaques (Macaca nemestrina), 62 stump-tailed macaques (Macaca arctoides), 98 and Bornean orangutans (Pongo pygmaeus) 84 have been reported to be susceptible to infection in nature, with or without clinical evidence of disease. In addition to use of those species that are naturally infected, animals in which experimental P. coatneyi infection has been studied to various extents include the rhesus macaque (Macaca mulatta), || black-capped squirrel monkey (Saimiri boliviensis), 109 Japanese macaque (Macaca fuscata), 53,54,56,70 silvery lutung (Presbytis cristalus), 14 and white-handed gibbon (Hylobates lar). 14 The cynomolgus macaque appears to be one of the natural hosts of P. coatneyi and is thought to have coevolved with the parasite. Pathology elicited by P. coatneyi infection in this NHP species is variable and is likely dependent on the genetic heterogeneity of the infected population, since only a subset appears to develop severe disease. The rhesus macaque is far more susceptible to progression of disease than the cynomolgus, but the same heterogeneity appears to apply and may account for the spectrum of pathology and clinical syndromes observed experimentally. The ability to elicit severe disease appears to be more consistent in the rhesus than in the cynomolgus macaques. While both species are used extensively in medical research and their genomes have been fully sequenced, 123 it is the experimental reproducibility in the rhesus of the pathologic parameters of falciparum disease in humans that makes the P. coatneyi–rhesus macaque model of infection useful for the modeling of severe malaria in humans.
P. coatneyi Life Cycle
The stage-specific morphology of P. coatneyi was beautifully illustrated in the original Eyles report 33 and again by Ms Gertrude Nicholson in The Primate Malarias text (see Fig. 1). 14
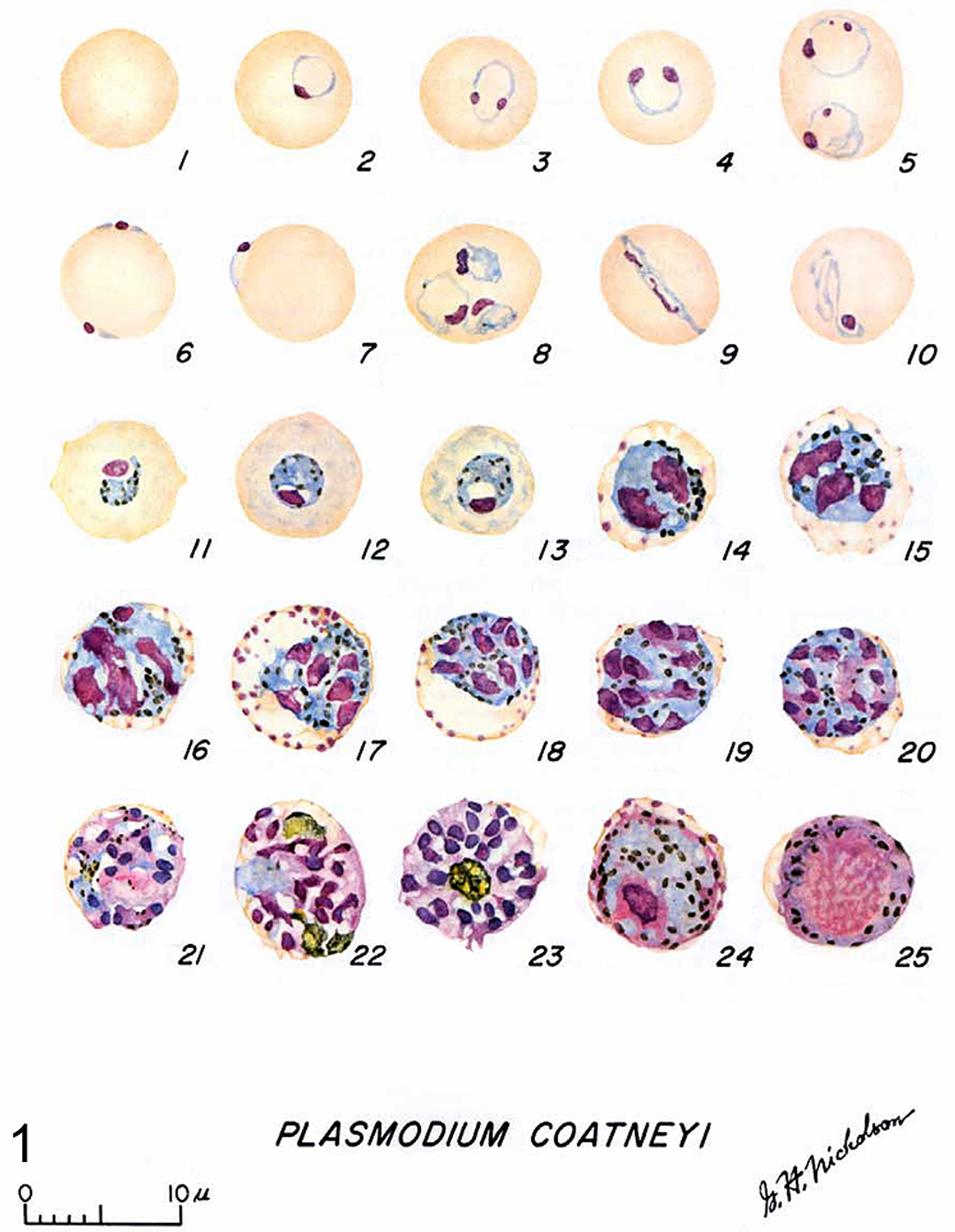
This plate is an illustration of the complex intraerythrocytic stages of Plasmodium coatneyi with clear demonstration of those diagnostic features that characterize the development of the trophozoites and schizonts. Reproduced with permission from Coatney et al. 14
The more immature stages of the species are indistinguishable from P. falciparum and, in fact, share some diagnostic features with the human malarial parasite. The trophozoite ring form of P. coatneyi is smaller than all other simian plasmodia, with the exception of P. knowlesi. 14 There are frequently 1 to 2 chromatin bodies or nuclei noted within the organism, which are either in close proximity to or at polar opposite sides of the ring form (Fig. 2). Significant morphologic variation was observed in the original description, but all of the shapes mimicked those that had been described in P. falciparum. As with the human parasite, the location of P. coatneyi trophozoites within the host erythrocyte varied. Common ring forms were observed as well as a high frequency of marginalized parasites along the periphery of the red blood cell. These forms have been described in P. falciparum as “rim” or “displaced’ types (Fig. 3). 14,35 The morphologic description that was the staple of pathologists and parasitologists before the advent of molecular technology remains crucial in malaria diagnostics. The variability of location within the host erythrocyte holds true with P. falciparum as well, with the peripheralized forms having been called “accolé” or “appliqué” and considered to be a diagnostic criterion for the human parasite. In these rim forms, the plasmodium typically appears as a continuous thickening of the cell wall, while the displaced form extends beyond the margin of the cell, creating an extruded plaque or bleb that is most noticeable at the point of the trophozoite nuclei (Fig. 4). 14
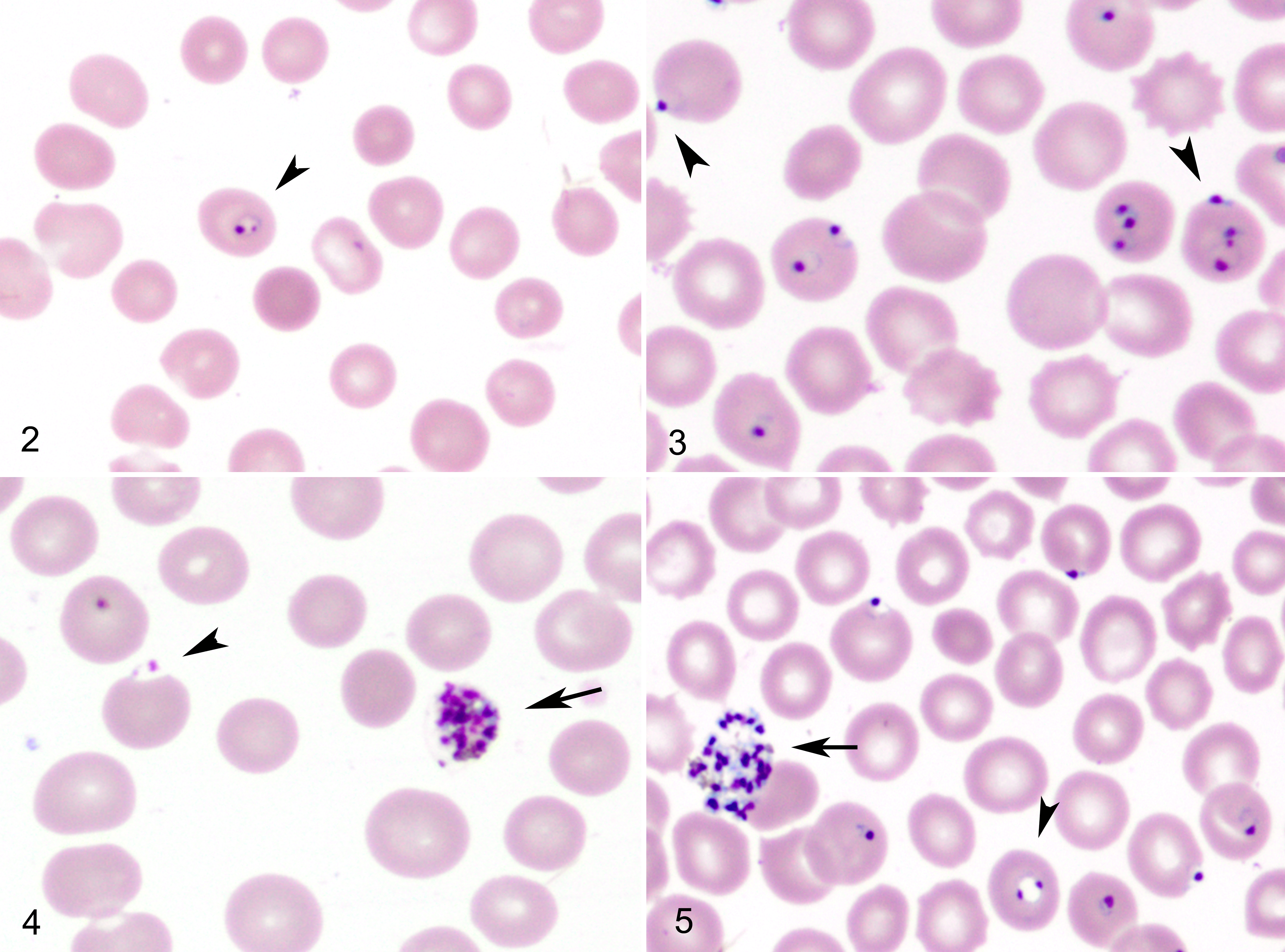
Intraerythrocytic Plasmodium coatneyi trophozoites, peripheral blood smears, rhesus macaque. Giemsa.
With maturation, individual and global changes take place within the parasite. As the trophozoites reach more advanced stages, their numbers in the peripheral circulation decrease dramatically, while morphologically, the plasmodia assume a more ovoid form. As with P. falciparum, there is a single large vacuole that decreases in size as time progresses. It is at this stage in development that the highly visible and diagnostic malarial pigment begins to accumulate. In contrast to the pigment of P. falciparum, P. coatneyi hemozoin crystals—formed as part of hemoglobin consumption by the parasite—do not coalesce until schizogony. Instead, they remain as distinct individual granules that increase in both size and number. It is also at this point of growth that the parasitized erythrocyte begins to display the tinctorial stippling that has been coined Maurer’s spots. These require either Giemsa or a modified Leishman staining to properly assess by light microscopy. 73 As with the hemozoin crystals, the Maurer clefts increase in density with maturation. 10,33
At a certain point in the development cycle, the trophozoite will transform into a schizont and begin the process of asexual division, forming merozoites (Fig. 4). As with P. falciparum, at this stage the infected erythrocytes become infrequent in the peripheral circulation as they remain in the microvasculature, adhering to the endothelium in a process termed sequestration. This process is felt to underlie the pathophysiology of severe forms of human disease, such as cerebral malaria. The plasmodium parasite developing within the erythrocyte exports specific parasite-encoded adhesions 94 to the surface of the red blood cell. Here, these proteins—such as PfEMP-1 in P. falciparum and its homologs in other plasmodium species—are organized at the electron-dense knob protein structures. These mediate specific cytoadhesion to host receptor molecules, such as intercellular adhesion molecule 1, cluster of differentiation 36, and cell adhesion molecules. 3 This allows for adhesion not only to the vascular endothelium but also to host leukocytes, platelets, and uninfected erythrocytes (rosetting).
Developing schizonts are round with condensed cytoplasm that contains up to 20 merozoites. Unlike other species of protozoa, the merozoites in P. coatneyi do not assume a specific orientation or pattern. At this stage, the malaria pigment crystals begin to coalesce, forming larger irregular granules, and the parasitized erythrocyte enlarges and is eventually obscured by the schizont. 14 During this stage, parasitized erythrocytes can still be identified in peripheral blood, but their numbers are significantly decreased in spleen-intact animals. Once the schizont is fully mature, it ruptures, releasing the merozoites into circulation (Fig. 5). This occurs with relative synchrony and results in the periodicity of parasitemia eliciting the pyrogenic response in the host. In splenectomized monkeys, these late stages of the parasite are also found in peripheral blood, along with the ring forms.
In their initial study, Eyles et al 33 infected 6 rhesus macaques (spleen intact) with P. coatneyi. The animals all became clinically ill, displaying marked anemia and severe lethargy. The parasitemia in all of the animals persisted for almost 6 months postinoculation, with alternating peaks and troughs of parasite density that differed by 100- to 1000-fold.
A second study was conducted in which P. coatneyi–naïve animals were inoculated with blood passaged through the first experimental animals. This cohort developed significant malaria-associated mortality, causing the authors to question the possibility of multiple stains of P. coatneyi with varying levels of virulence. 32
There have been a variety of studies examining the suitability of P. coatneyi in macaques as a model for the cerebral or severe malaria caused in humans by P. falciparum, as well as for the evaluation of plasmodium-induced placental pathology. 20,21 Some of these experiments attempted to describe the pathophysiology of disease through histopathology, immunohistochemistry, and transmission electron microscopy, ¶ while others adopted the animal model as a route of drug assessment and testing. 58,69
Clinical Signs
Soon after the original identification of P. coatneyi, studies began at the US-Thai South East Asia Treaty Organization laboratories in Bangkok (now AFRIMS) to characterize the pathobiology of this infection in NHPs. # Although severe infections often led to death, coma and seizures were not typical of the clinical picture in macaques, differing from the common pattern associated with severe cerebral malaria in humans. Monkeys progressively lose appetite, develop malaise and lethargy leading to recumbency, and become dehydrated and tachypneic. 77,102 The most consistent pathologic effect noted in a P. coatneyi infection of rhesus and cynomolgus macaques was anemia, which paralleled the rise in parasite density. In many cases, this was followed by hemoconcentration and rapid deterioration secondary to a shocklike syndrome similar to that described in severe P. falciparum and the few reports of P. knowlesi malaria. 16,17 This exacerbation is theorized to result in part from a marked decrease in plasma volume concurrent with cytokine-associated increase in vascular permeability. 5,19,24,26,77,78 These findings are in no way unique to malaria. The infection is more severe in splenectomized animals. Anecdotal evidence from spleen-intact donor animals suggests that the parasitemia may even be cleared in immunocompetent animals.
Postmortem Findings
There is only scant information in the literature regarding autopsy findings in P. coatneyi–infected animals. Macaques in one particular study consistently were dehydrated and either pale or icteric. There was serous atrophy of fat in multiple organs, and a mild dilation of the ventricles of the heart was noted. Petechiae and coalescing ecchymoses were described in multiple tissues but most significantly within the epicardium. Concentrated within the kidneys and the liver, much of the viscera was described as having a diffuse brown discoloration (Fig. 6) as well as concurrent organ enlargement. This was characterized by splenomegaly (2 to 3 times normal), hepatomegaly, and enlarged lymph nodes. 1,19,55,102
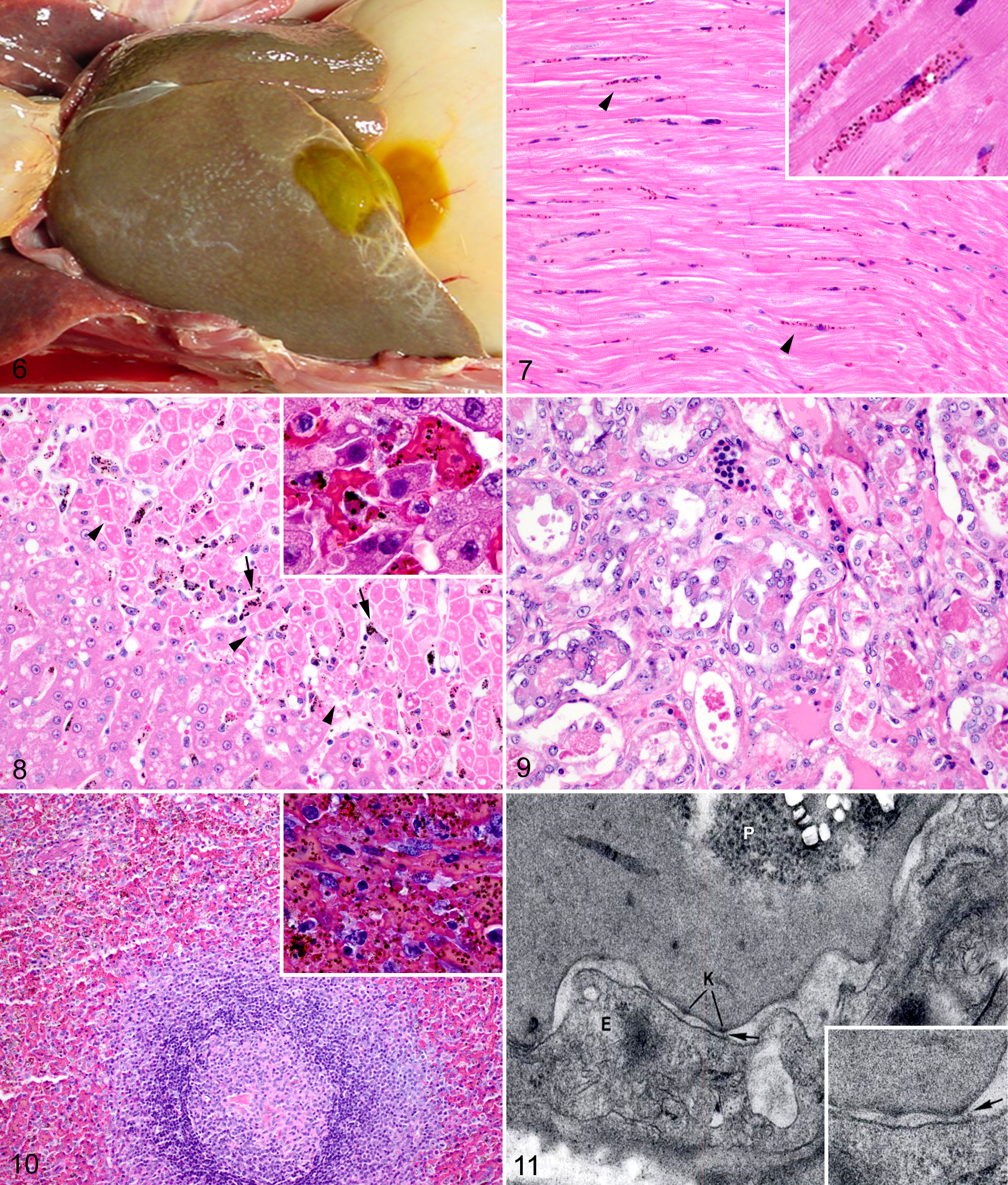
Rhesus macaque, Plasmodium coatneyi infection.
Clinical records of upward of 60 splenectomized rhesus macaques at AFRIMS provided the foundation of the previously reported pathologic data 20,24,25,53,55,60,78,102 and corroborated the clinical findings and autopsy observations in this current study. With parasitemias of ≥20%, the animals’ clinical condition rapidly deteriorated around the seventh day postinfection. Clinical signs included hematuria, hemorrhage within the perineum and scrotum, severe depression, recumbency and eventual prostration, progressively worsening anorexia, occasional vomiting, and severe dyspnea and tachypnea. Once the animals began to display signs of decreased mentation, death followed rapidly, and only rare animals survived beyond the eighth day postinfection. Postmortem findings were consistent with the few descriptions in the literature. Animals with higher parasitemias in and around the time of euthanasia or sudden death were observed to have brown discoloration of the liver, lungs, kidneys, heart, and brain. 19 The discoloration of the organs is due to vascular congestion and the abundance of malaria hemozoin pigment in erythrocytes and monocytes, both circulating and sequestered. The relationship between malaria infection and the brown discoloration of organs was established by Rudolph Virchow and Friedrich Theodor von Frerichs in the mid-19th century. 46 Additional macroscopic findings in this retrospective review conducted at AFRIMS included hepatomegaly with increased friability, heavy congested lungs, and petechiae in the epicardium.
There is a single report in the literature in which a rhesus macaque experimentally infected with P. coatneyi developed progressive cutaneous necrosis of the extremities secondary to disseminated intravascular coagulopathy. This resulted in dermal infarction of the digits and the tail due to thrombosis of the superficial vasculature. 78 A similar unpublished case occurred in Thailand in a rhesus macaque also experimentally infected with P. coatneyi. In that instance, the animal developed a necrotizing rhinitis and cheilitis that progressed to erosion and ulceration.
There are occasional reports of gangrenous necrosis secondary to disseminated intravascular coagulopathy in P. falciparum but not in any other forms of human malaria. 13,66,101 While reports of disseminated intravascular coagulopathy vary in frequency in different human populations, progression to peripheral cutaneous gangrenous necrosis appears to be a rare complication. However, when it does occur, P. falciparum is implicated as the causative plasmodium in 95% of human cases, and typically the presentation is of ischemia and subsequent necrosis within peripheral tissues. 11,39,71,76
Clinical Pathology
Microscopy remains the gold standard of malaria diagnosis in humans and NHPs, clinically and under experimental settings. Examination is conducted using blood prepared as thin films (in which the sample is prepared with the same methodology as a routine blood smear) and thick films (involving a process of erythrocyte lysis). The latter is more sensitive in plasmodium detection, especially earlier in infection when parasite density is lower, whereas the former allows for speciation and parasite staging. Although the World Health Organization suggests that microscopic slide evaluation in cases of malaria is at best 75% effective based on a range of clinical, geographic, and species-specific factors, microscopy has significant advantages over other diagnostic modalities that may be more accurate. 119 The benefits of microscopy for malaria diagnosis are linked to the geography and epidemiology of the various plasmodial diseases. Falciparum malaria is endemic circumferentially along the equator and extends significant distances to the north and south but still covers a large swath of the poorer and less developed portions of the planet. As such, the most tangible benefits of microscopy are the low cost and the minimal need for infrastructure. However, the technique is also crucial in rapid speciation of parasites, identification of mixed infections, and monitoring treatment. Microscopy has been the mainstay of NHP malaria research as well, and the bulk of the publications evaluating models of malaria employs microscopic examination to quantitate parasitemias in experimental animals. By using skilled microscopists and associated clinical symptomology, hematologic, and blood chemistry data, it is possible to closely map the course of disease.
Until recently, the clinical pathology of P. coatneyi infection in macaques had been poorly elucidated in the literature. A 2013 report by Moreno et al provided more systematic data on the clinicopathologic findings of P. coatneyi malaria in malaria-naïve, spleen-intact rhesus macaques. 77 The animals were cured and reinfected at a later date to examine the role of specific acquired immunity in macaques. The study noted that blood chemistry data from the primary infection showed marked azotemia, hypertriglyceridemia, hypoalbuminemia, and increased levels of creatinine phosphokinase in comparison to preinfection levels. There was a marked decrease in clinical signs in the second episode of infection, which was reflected in the clinical pathology. Triglyceride levels, while elevated, were not significantly different between the first and second P. coatneyi infections, suggesting that the release of triglycerides was not directly correlated to the severity of the malaria infection. 77
Furthermore, the authors observed thrombocytopenia and severe coagulopathy. This was associated with an increase in fibrin degradation product levels and concurrent drops in protein C and protein S, suggesting increased consumption of inhibitors of coagulation. 69
There are several proposed reasons for severe thrombocytopenia occurring in animals during the acute phase of malaria infection. As with the erythrocytic clearance, immune-mediated clearance of platelets may occur as a bystander effect of the presence of the parasitized erythrocytes, stimulating antibody-mediated clearance and upregulation of phagocytic activity in histiocytic cells. 41,52 The thrombocytopenia may result from splenic sequestration, peroxidation of the platelet lipid membranes, or platelet consumption from formation of systemic microthrombi. 78
Metabolic disturbances were evaluated in the rhesus macaque model. 19 Animals infected with P. coatneyi demonstrated variably increased serum and cerebrospinal fluid glucose levels, as well as elevated lactate that increased with parasitemia. This was discussed as being associated with progressive insulin resistance and a late tendency to lactate acidosis in infected animals, processes observed in human patients with P. falciparum infections.
In an early study consisting of a small mixed population of cynomolgus macaques as well as Indian- and Thai-origin rhesus macaques, the renal and hepatic clinical pathology associated with severe P. coatneyi infection was followed through the course of disease and compared with spleen-intact infected animals as well as with uninfected controls. The authors noted concurrent increases in blood urea nitrogen and serum creatinine, both of which initially resolved, only to increase a second time with a concurrent rise in parasite density that continued until death. Furthermore, a slight increase in hepatic alanine transaminase was observed immediately following the initial parasitemia peak, but it subsequently normalized. Alkaline phosphatase remained within reference ranges, although a minor decrease was observed in some of the animals. The animal numbers in the study were insufficient for the purpose of drawing general conclusions to clinical pathology. The study underscores the absence of adequate data within the literature as a whole. 24
Noteworthy among these various studies is the significant variability in the level of parasitemia achieved and sustained. Often the degree of acquired immunity is either uncertain or is not reported. This then translates to a degree of variation in the clinicopathologic changes observed and reported. There are also significant gaps in the P. coatneyi literature regarding the impact of the parasite on separate organ systems within the rhesus, much less any of the other NHPs evaluated as models. An example of this can be seen in the absence of adequate studies of the metabolic changes associated with the renal and the hepatic dysfunction.
Histopathology and Immunohistochemistry
The literature contains scant systematic information on the histologic lesions resulting from P. coatneyi parasitism in any species. However, a few of the experiments did involve partial necropsies of the infected macaques and subsequent histopathologic analysis, which is summarized here.
Central Nervous System
Sequestration patterns in the infected animals have been the mainstay of the proposal that P. coatneyi be used as a model for cerebral malaria in humans. 25,80,99 In P. falciparum infections in humans, the sequestration (adherence to endothelium) of parasitized erythrocytes in the microvasculature of the brain often results in partial or complete occlusion of the vessels, as characterized by significant congestion with increased margination due to cytoadhesion of infected erythrocytes. These microscopic lesions were correlated with hemorrhage and signs of tissue hypoxia and theorized to be a significant contributing factor in the development of the symptomology associated with cerebral malaria—namely, coma and seizure activity. 72 Histologic degrees of vascular sequestration differ among regions of the brain in humans and NHPs. 79,89,102 Reports based on microscopic examination of human brains with cerebral malaria noted a significantly higher degree of parasitized red blood cell sequestration in the cortical gray matter than in the white matter. 79
As a result of the human data and to build on previous experiments that evaluated the sequestration patterns in the brains of P. coatneyi–infected rhesus macaques, 24 –26,99 a histopathologic and electron microscopic study was conducted in Thailand to examine the distribution of sequestration within the brains of experimentally infected rhesus macaques. 102 Animals (5 spleen intact, 1 splenectomized after infection) were inoculated with a bolus of parasitized blood consisting of 77% trophozoites and 23% schizonts. Parasitemia was assessed daily and parasite staging conducted. Between days 7 and 9 postinfection, all animals were euthanized, necropsied, and evaluated by a veterinary pathologist. At the time of euthanasia, parasitemias ranged from in excess of 200 000 to almost 900 000/mm 3 and consisted primarily of trophozoites. The highest parasitemia was observed in the animal that had been splenectomized at day 5 postinoculation. The presence of sequestration was quantified in the microvasculature of the cortex, the midbrain, and the cerebellum, based on histologic examination of 200 microvessels per section. Sequestration was observed in 25% of microvessels in the cerebrum, 24% in the midbrain, and 37% in the cerebellum. While not specific to any of the 3 regions evaluated, the histopathologic observations within the central nervous system consisted of congestion, functional obstruction, as characterized by evidence of perivascular edema, hemorrhage, and peripheral necrosis as well as increased cytoadhesion of parasitized cells (sequestration) of the microvasculature with parasitized and uninfected erythrocytes. In addition, frequent intraerythrocytic and intramonocytic hemozoin pigment and occasional microhemorrhages within the neuropil were described. 102
A window to events within the central nervous system that is of great clinical interest and potential histopathologic importance includes changes within the retina. Based on recent descriptions of retinal findings in children in East African with severe malaria, retinal hemorrhages are being used as a diagnostic criterion for cerebral malaria in children. 30,47,64,65 Preliminary unpublished histopathologic evaluation of 2 cohorts of animals in prospective studies and the large body of retrospective tissues from various studies conducted at AFRIMS have failed to show similar hemorrhaging within the retinas of P. coatneyi–infected animals. However, based on the diagnostic significance in humans, malaria-induced retinopathy should be evaluated in greater depth in animal models.
Liver
The liver of infected NHPs occasionally has a diffuse centrilobular hepatocellular necrosis accompanied by mild increases in serum levels of transaminase. 24 In addition to the necrosis, mononuclear hepatitis was frequently noted, accompanied by a marked accumulation of birefringent malaria pigment observed within Küpffer cells. 60,78
It is significant that while increases in inducible liver enzymes are a common feature of severe malaria in adult humans, there are only rare reports of centrilobular hepatic necrosis. 20,24
Kidney
Kidneys from P. coatneyi–infected rhesus macaques in one study had varying degrees of acute tubular necrosis, accompanied by hyaline droplet degeneration characterized by expansion of the cytoplasm of the tubular epithelium with eosinophilic globules that are interpreted as possible protein resorption (Fig. 7). Furthermore, tubules contained abundant hemoglobin and cellular casts, and the urinary space contained proteinaceous filtrate with a relative absence of inflammation. 24 Hemoglobinuria has been observed clinically, but a formal correlation of clinicopathologic and histopathologic findings of renal disease in this model remains to be done.
Cardiovascular System
Two studies conducted in Thailand examined the histopathologic sequestration patterns of P. coatneyi in the viscera of rhesus macaques with microscopic examination of tissues from multiple organ systems by a veterinary pathologist. 25,102 Overall findings in multiple tissues consisted of congestion and occlusion of small vessels with parasitized and uninfected erythrocytes. There was also an abundance of phagocytic monocytes containing malaria pigment and varying degrees of lymphocytic inflammation. Frequently, the erythrocytes formed rosettes of uninfected cells bound to a parasitized cell. While this is typically an ultrastructural diagnosis, the authors of this study were able to microscopically identify the rosettes within larger vessels. The study evaluated the degree of sequestration within different organ systems and noted that the myocardium had the highest degree of histologic sequestration, with individual animals having 84% to 95% of the myocardial microvasculature showing sequestered parasitized red blood cells (Fig. 8). This was particularly pronounced within the interventricular septum and the left ventricular free wall. Furthermore, the pericardial adipose tissue had a high degree of sequestration and congestion, as did the microvasculature of the pulmonary interstitium and the mesenteric adipose tissue, with an average microvascular sequestration of 85% and 76%, respectively. 102 Other studies in macaques report sequestration of P. coatneyi–infected erythrocytes in the coronary vasculature, adipose tissue of the omentum, subcutaneous tissues and other sites of fat deposition, the intestinal tract, and the skeletal musculature. 53,63 This site partiality is consistent with those described in P. falciparum infections. 69
Lymphatic System
An experimental infection of Japanese macaques observed splenomegaly in all P. coatneyi–infected animals. Microscopically, the spleens were characterized with lymphoid follicular hyperplasia, activated macrophages, and red pulp sequestration with parasitized erythrocytes accompanied by hemozoin pigment (Fig. 9). The splenic lesions were considered similar to those noted in humans infected with acute P. falciparum infections. 55
Reproductive System
Studies of placental pathology in the rhesus macaque model were conducted at Tulane University using primiparous and multiparous macaques. The histopathologic features of the disease in macaques mirrored that seen in humans infected with P. falciparum. The most severe placental lesions consisted of infarction, fibrinoid necrosis, deposition of fibrin, and mononuclear and polymorphonuclear fibrinoid-necrotizing placentitis. A few of the experimental animals aborted, and fetal death was correlated with the presence of severe placental infarction. The endometrium was typically infiltrated with significant numbers of mixed inflammatory cells, and the uterine microvasculature contained parasitized erythrocyte sequestration that increased with proximity to the placental tissues. Furthermore, there was typically infarction of the basal plate with small numbers of neutrophils admixed with pigment-laden macrophages. 20
Other Systems
Although other organ systems, such as the respiratory and hematopoietic systems, are involved in the pathophysiology of severe malaria in humans, the histopathologic examination of P. coatneyi infection in any species of NHP is significantly lacking.
Only a few immunohistochemical studies of the pathophysiology of P. coatneyi in macaque models have been conducted. In 1992, Aikawa and colleagues examined rhesus macaques in which the cerebral microvasculature containing sequestered parasitized red blood cells was examined for immunohistochemical reactivity for the endothelial sequestration receptors: cluster of differentiation 36, TSP (thrombospondin), and intercellular adhesion molecule 1. The cerebral microvasculature of infected animals demonstrated immunoreactivity to all 3 molecules, while that of cerebral endothelial cells in the cerebral microvasculature of uninfected control macaques was negative. 1 This study mirrored work that had been conducted in P. falciparum–infected tissues. The positive immunoreactivity of P. coatneyi–infected tissues suggests that there is a similar mechanism of cytoadherence in humans and NHPs. 83,91,104 Subsequently, in 1996, noncerebral macaque tissues were evaluated using the same molecular markers. All of the experimentally infected animals displayed positive immunohistochemical staining in the heart, lungs, small intestine, and spleen in contrast to the control animals. Additionally, deposition of immunoglobulin G and complement component 3 within the vascular wall (immune complexes) was evaluated using immunofluorescence in all harvested tissues. All of the tissues examined in the P. coatneyi–infected animals were positive for both markers. In contrast, in the uninfected animals, there was immunoreactivity in the spleens and faint immunofluorescence in the kidneys of 2 of the animals, but the brain, heart, lungs, and small intestine were universally negative. This microvascular damage in the kidneys is consistent with studies conducted in falciparum malaria–infected tissue. 69
Electron Microscopic Findings
In contrast with the dearth of histopathologic analysis described in the literature, the bulk of the early studies of P. coatneyi included transmission electron microscopic examination of the pathologic changes in the rhesus macaque. Concentrating on changes associated with the process of sequestration, which is best observed and confirmed using ultrastructural examination to identify specific cytoadhesion, a variety of pathologic observations have been reported.
Rosettes—consisting of a central infected erythrocyte, cytoadherent or bound to a variable number of surrounding uninfected erythrocytes—have been described as contributing to the pathogenesis of P. coatneyi, 112 P. fragile, and P. falciparum infections. They have been observed with cultured P. falciparum and in occasional instances with fresh isolates of the human cerebral malaria agent. 43,116 Rosette formation has been examined ultrastructurally, and the theorized mechanism of occurrence posits that the uninfected erythrocytes are bound by receptors to the electron-dense knobs found on the cell surface of a centrally located, parasitized red blood cell that contains either a trophozoite or a schizont. 110 Rosettes can subsequently begin to recruit and bind to other rosettes or uninfected erythrocytes, forming larger aggregates. P. vivax, P. cynomolgi, P. knowlesi, and the ring stage of P. falciparum (and P. coatneyi) lack the electron-dense knobs that mediate cytoadhearence and, as such, are not generally associated with high rates of rosetting. Thus, erythrocyte sequestration, which is considered important for the pathogenesis and clinical outcome of cerebral malaria, is thought to result from a combination of rosette formation between parasitized and uninfected erythrocytes, interactions between distorted erythrocytes and induced pseudopodia of endothelial cells, and direct adhesion between erythrocytes and endothelial cells. 43,48,113
Packed infected erythrocytes are characterized by distortion and prolongation of the cytoplasm, generating irregular forms. These appear to intricate themselves into invaginations and pseudopod-like projections emanating from the adjacent endothelium. Furthermore, the surface of the affected endothelium is crenated, with thickening of the endothelial basal lamina, especially in proximity to the parasitized red blood cells. The infected erythrocytes themselves develop the conical, membrane-delineated, electron-dense, evenly spaced “knobs” that are involved in binding to unparasitized erythrocytes and potentially to the endothelium as well (Fig. 10). 2,57 Within the red blood cells, in addition to the malaria hemozoin crystals, the plasmodia were described at various stages of development, ranging from trophozoites to schizonts, with schizonts predominant. 99 Furthermore, infected erythrocytes were noted to be bound to uninfected cells at the point of the electron-dense knobs and, in doing so, formed the described rosette patterns. 110
The descriptions in the literature suggest that the observed endothelial pseudopodia occur with greater frequency within the microvasculature of the cerebellum than in other regions of the brain. 99 Cerebral endothelial pseudopodia have only rarely been described in the literature in diseases other than malaria. Chickens fed a diet high in linoleic acid and deficient in vitamin E were reported with similar changes, 125 as were cats with induced endotoxic shock. Rats experimentally manipulated to generate or decrease fluid shear in skeletal muscle microvessels induced formation of endothelial pseudopodia. 50 Furthermore, pigs with experimental Mycoplasma suis infections were assessed ultrastructurally and were noted to have similar changes in endothelium throughout multiple organ systems, as described with P. coatneyi. 105
MacPherson et al suggested that the endothelial pseudopodia within the cerebral microvasculature, which has also been described in human cases, is a possible pathologic manifestation of cerebral vascular insult and a response to endothelial damage. Since these were observed in regions with the slowest blood flow, it was proposed that there was a higher likelihood of trophozoite- and schizont-infected erythrocyte deposition. 68 Ultrastructural lesions in endothelial infections other than malaria have been described in detail, including the cytokine-mediated transendothelial migration of leukocytes as well as ligand-receptor connections, suggesting that electron microscopic findings seen in cerebral malaria are multifactorial and not necessarily specific to the plasmodium pathogens. 40,97
Pathophysiology
While there have been remarkable advances, the pathophysiology of P. falciparum and P. coatneyi infections remain an area of significant scientific debate. Erythrocytic sequestration and cytoadherence have been definitively shown to occur with both parasites through in vitro studies and electron microscopy, 53,56,102,112 and it is this pathologic commonality that promoted the use of the P. coatneyi–rhesus macaque as a good model of P. falciparum cerebral malaria in humans. However, although sequestration is described in the brain, there is a wide range of secondary neuropathologic features of the disease that have been recently described in the human disease. Examples include cerebral edema, breakdown of the blood-brain barrier, axonal injury, and leukocyte and fibrin/platelet thrombus formation. All of these topics need to be examined in the macaque model to determine the conformance of the macaque model with the human disease. 73
The role of cytokines in the immunopathogenesis of plasmodium-induced disease has been extensively studied in murine models; however, there have been few reports of similar work in NHPs. One of these studies, undertaken at the US Centers for Disease Control and Prevention, utilized the rhesus macaque–P. coatneyi model to evaluate the correlation between serum levels of inflammatory cytokines and prostaglandin with parasitemia levels. Serum interferon γ levels rose earlier and mirrored the periodicity of the parasitemia, while interleukin 1β and tumor necrosis factor α increased late in infection. The authors theorized that the change in interferon γ was associated with early activation of T lymphocytes, while the elevated levels of the latter 2 cytokines correlated with monocyte activation paralleling increased parasitemia. Serum prostaglandin E2 levels were also elevated. Considering the microvascular insult associated with severe malaria in human disease, the report theorized that the progressive increase in prostaglandin E2 observed in the rhesus was a corollary to the rise in inflammatory cytokines. Finally, the study also evaluated ciliary neurotropic factor, a molecule involved in the maintenance of calcium homeostasis in glial cells and neurons, as a means of assessing neural injury in the rhesus. By the later stages of disease, ciliary neurotropic factor had risen in all 4 experimental animals. 124
A more recent study evaluated erythropoietin (EPO) levels in serum and compared the findings with microscopic evaluation of core bone marrow samples. EPO levels in P. coatneyi–infected macaques reached a peak of 6 times that recorded in the uninfected control animals, and the EPO zenith coincided with the nadir in hemoglobin levels in the affected monkeys. The increase in renal production of EPO strongly suggests a functional hyperplastic response in the bone marrow to the massive erythrocyte destruction. The bone marrow had a significant shift toward immaturity in both the myeloid and erythroid lineages during the acute stage of infection. Furthermore, concurrent dyserythropoiesis was morphologically observed, characterized by karyorrhexis and increased nuclear lobulation. Within a week following cure, the nuclear changes in the bone marrow had normalized. 77
Erythrocyte longevity was also assessed in the infected macaques. A severe anemia was observed approximately 10 days following infection and was correlated with parasitemia. The highest degree of erythrocyte turnover occurred at relatively low parasitemias, suggesting that infected erythrocytes have a bystander effect resulting in concurrent destruction of uninfected red blood cells. Greater than 90% of the cleared erythrocytes were uninfected. 77 Suggested mechanisms of this bystander effect relate to changes in erythrocyte morphology, decreased longevity, and immune-mediated clearance through upregulated erythrophagocytosis. 103,107
Conclusions
It has been more than 50 years since the discovery of P. coatneyi in the forests of Malaysia, but there remains much to accomplish in describing this potentially valuable model of severe human malaria. Considering the continuing debate regarding the suitability of rodent models of experimental severe and cerebral malaria, there remains a significant need to more fully describe the rhesus macaque–P. coatneyi model.
Fundamentally, the question remains, what represents a substantiated and scientifically valid model of severe or cerebral malaria in humans? Is the macaque model able to replicate falciparum malaria in children as well as adults? Will it be useful for preventive and therapeutic studies? The sequencing of the genome of P. coatneyi will open up a wide range of new possibilities for critical examination of the parasite and its role as an animal model of human disease. Since the majority of the studies to date have been conducted in very small cohorts, the observed pathology in those animals may not be representative of the scope and variation of the infection and resulting disease. Molecular pathology techniques have enormous potential for investigating the pathophysiology of severe malaria in animal models. They have been applied to murine models but barely used in the macaque models.
The macaque model has the potential advantage of a high degree of homology to the human immune system. NHP models could address problems involving metabolic and physiologic changes (eg, the pathogenesis of coma and neurologic dysfunction) that are difficult to study in human patients. 7 Functional magnetic resonance imaging, intracranial pressure monitoring, and laser video microscopy of flow and electroencephalogram under controlled settings (and in response to potential adjuvant therapies) are difficult to perform in human patients with malaria. In comparison with the nonsequestration-dependent, immune-mediated encephalitis of experimental murine cerebral malaria, the similarities of the pathophysiologic tapestry of P. coatneyi to human disease should make its results more medically relevant. Thus, a catalogue of the molecular markers of the infection needs to be documented and assessed, to determine the true validity of the P. coatneyi–infected rhesus macaque as a model for severe or cerebral malaria in humans. The arms race that is ongoing with this disease is all the more heightened by the parasite’s broadening drug resistance. The search for a strong model of human disease is all the more crucial.
Footnotes
Acknowledgements
We would like to state our sincere appreciation to the generation of researchers and scientists who have labored toward a greater understanding of Plasmodium coatneyi malaria, to the staff of the Department of Veterinary Medicine of AFRIMS, and to constructive discussions and advice of Professor Nick White, Mahidol Oxford Tropical Medicine Research Unit and University of Oxford, and grant support from the Wellcome Trust of Great Britain (grant 089275/Z/09/Z).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. E. D. Lombardini is a lieutenant colonel in the US Army. The opinions or assertions herein are those of the authors and do not necessarily reflect the view of the Department of the Army or the Department of Defense.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
*References 1, 19–21, 25, 32, 53, 54, 69, 70, 77, 78, 80, 99, 100, 102, 109–112, 124
†References 1, 19, 24–26, 69, 99, 102, 110, 112
‡References 1, 15, 18, 21, 23, 54, 59, 67, 69, 77, 118
§References 11, 18, 22, 27, 51, 59, 79, 103, 122
||References 1, 4, 5, 19–21, 25, 32, 69, 70, 74, 77, 78, 80, 100, 102, 110–112, 124
¶References 1, 24, 31, 33, 53, 56, 57, 70, 74, 77, 78, 80, 102, 110–112
#References 1, 2, 19, 24–26, 69, 99, 102, 110, 112
