Abstract
“Shivers” is a progressive equine movement disorder of unknown etiology. Clinically, horses with shivers show difficulty walking backward, assume hyperflexed limb postures, and have hind limb tremors during backward movement that resembles shivering. At least initially, forward movements are normal. Given that neither the neurophysiologic nor the pathologic mechanisms of the disease is known, nor has a neuroanatomic locus been identified, we undertook a detailed neuroanatomic and neuropathologic analysis of the complete sensorimotor system in horses with shivers and clinically normal control horses. No abnormalities were identified in the examined hind limb and forelimb skeletal muscles nor the associated peripheral nerves. Eosinophilic segmented axonal spheroids were a common lesion. Calretinin-positive axonal spheroids were present in many regions of the central nervous system, particularly the nucleus cuneatus lateralis; however, their numbers did not differ significantly from those of control horses. When compared to controls, calretinin-negative, calbindin-positive, and glutamic acid decarboxylase–positive spheroids were increased 80-fold in Purkinje cell axons within the deep cerebellar nuclei of horses with shivers. Unusual lamellar or membranous structures resembling marked myelin decompaction were present between myelin sheaths of presumed Purkinje cell axons in the deep cerebellar nuclei of shivers but not control horses. The immunohistochemical and ultrastructural characteristics of the lesions combined with their functional neuroanatomic distribution indicate, for the first time, that shivers is characterized by end-terminal neuroaxonal degeneration in the deep cerebellar nuclei, which results in context-specific hypermetria and myoclonus.
Breeds of horses have been genetically selected and trained for hundreds of years to develop specific locomotor patterns that enhance their ability to perform within a wide range of athletic disciplines. Genetic or acquired gait deficits, even if minor, can have a major impact on performance. Shivers is a gait disorder affecting Warmbloods, Thoroughbreds, and other tall breeds, initially manifesting as hypertonic muscle contractions and protracted abduction and hyperflexion of the hind limbs when horses walk backward or hold up a hind limb for a handler while standing. 10 Forward gaits are typically normal initially. Horses with shivers have no evidence of proprioceptive or vestibular deficits and can compete successfully for many years in dressage and jumping. In >50% of cases, shivers slowly progresses, and in advanced cases, it causes intermittent hyperflexion and abduction of a hind limb when initiating forward walking or turning. 9,10 Excitement, transportation, or prolonged standing temporarily exacerbates muscle hypertonicity. 1,9
The underlying neuroanatomic or physiologic changes that lead to signs of shivers remain a mystery, but it has been postulated to be a muscular, skeletal, or nervous system disorder. 1,27 A genetic etiology is suspected based on its predilection for certain breeds. 1,9 Only 1 histopathologic study of the nervous system of 2 shivers-affected Belgian horses has been published. 27 No histologic lesions were identified in the nervous system. Thus, no clear cause for shivers was apparent. Both horses had polysaccharide storage myopathy, a disorder affecting 33% of Belgian horses. However, a subsequent study found that approximately 66% of Belgian horses with shivers do not have abnormal polysaccharide accumulation in myofibers. 12
The unique feature of shivers is its principal involvement of backward locomotion. Although much is known about central pattern generators and supraspinal circuits involved in forward walking, much less is known about the control of backward locomotion. 3,14 At this point, no clear pathophysiologic mechanism for the disease has been identified. No clear rationale has been provided for why the disease is especially manifested during backward locomotion. We undertook an extensive histopathologic, immunohistochemical, and electron microscopic study of the entire central nervous system (CNS), hind limb peripheral nerves, and hind limb musculature of shivers-affected Warmblood and Thoroughbred horses and age-matched control horses with the objective of localizing and characterizing the neuropathologic lesions associated with this disease.
Materials and Methods
Horses
Shivers-affected horses comprised 4 Warmblood horses (2 sharing the same sire) and 1 Thoroughbred horse. Control horses comprised 2 Warmblood horses and 1 Thoroughbred horse. All control horses were age matched to affected horses within 1 year, with the exception of one 18-year-old severely affected shivers horse (Table 1). All control and affected horses were >168 cm at the withers. Control horses were donated due to moderate chronic osteoarthritis causing lameness at a trot but not at a walk or horses were unsuitable for riding. A complete equine neurologic examination was performed—including mentation, cranial nerve function, and evidence of intention tremor and proprioceptive deficits—and results were normal for shivers and control horses. A video was made of a handler manually lifting each limb and leading the shivers and control horses forward at a walk, turning sharply in both directions and walking backward. When the horses walked backward, the presence of hyperflexion, hyperextension, and a normal contralateral quadrupedal gait pattern was noted. Hind limb movement was scored as normal, mildly abnormal (+), moderately abnormal (++), or very abnormal (+++) during forward walking, backward walking, and when a handler manually lifted the limbs.
Horse Signalment, Clinical Severity and Spheroid Count.a

Abbreviations: CM, castrated male; DCN, deep cerebellar nuclei; F, female; HE, hyperextension; HF, hyperflexion; NCL, nucleus cuneatus lateralis; TB, Thoroughbred; WB, Warmblood.
aAtaxia was absent for every horse.
b+, mild; ++, moderate; +++, severe.
Horses were euthanized on separate days by intravenous administration of a barbiturate and immediately transported to the necropsy facility. Tissue collection was completed within 4 hours of euthanasia. The study was approved by the University of Minnesota Institutional Animal Care and Use Committee.
Postmortem Examination
Central and Peripheral Nervous System
Gross abnormalities of the skull, vertebral column, brain, spinal cord, and peripheral nerves were evaluated, and the entire brain and spinal cord from cervical (C) vertebra 1 to lumbar (L) vertebra 1 were removed and placed into 20% buffered formalin within 2 hours of euthanasia. The spinal cord distal to L1 was collected en bloc within the lumbar vertebrae and sacrum after the femurs were disarticulated and the vertebral column severed between L1 and coccygeal vertebra 3. A lumbosacrococcygeal dorsal laminectomy exposed the terminal spinal cord and cauda equina, and the entire sacral block was placed intact in a tub containing 20% formalin to ensure rapid fixation. Nerve roots, ganglia, and peripheral nerves were dissected and preserved intact (see Supplemental Fig. 1). This in situ fixation technique allowed resampling of areas of interest with anatomic precision.
Neuropathology
Central and peripheral nervous tissue was fixed in formalin for a minimum of 7 days, and paraffin-embedded sections were stained with hematoxylin and eosin. Selected CNS sections were stained with Luxol fast blue and Bielschowsky to enhance visualization of myelin and neuronal processes. Sections were qualitatively evaluated for the presence and location of inflammatory, circulatory, and degenerative lesions and compared with the age-matched controls. Areas of the brain that were examined included telencephalon (coronal sections at 2 levels), basal nuclei (coronal sections at 3 levels), thalamus (coronal sections at 2 levels), and cerebellum (sagittal section at 1 level and horizontal sections at 2 levels), including lingula, central lobule, culmen, declive, tuber, folium, pyramis, uvula, nodulus, flocculus and paraflocculus lobules. Further regions examined were mesencephalon (coronal sections at 3 levels) and pons and medulla oblongata (coronal sections at 8 levels; exact regions shown in Supplemental Fig. 2). Cervical (C1, C2, C5, C6, C7) and thoracic spinal cord segments (T2, T10, T13) were examined. Cross sections of lumbar (L1, L3, L4, L5, L6), sacral (3 levels), and coccygeal segments (1 level) were removed from the in situ preparation (S1). All dorsal and ventral nerve roots of the lumbosacral segments and dorsal root ganglia were examined. In addition, transverse and longitudinal sections of the sciatic, peroneal, and tibial nerves were examined. The nerve branches to the biceps femoris and cranial tibial muscles were selected because they supply the primary abductor and flexors of the stifle and hock and these joints are abducted and hyperflexed during backward locomotion in shivers horses.
Neural centers and pathways associated with somatosensory and sensorimotor control were systematically evaluated for the presence of neuronal and axonal degeneration.
7,8
Initial qualitative evaluation included the following areas: cerebrum—including the gyri sigmoideus, marginalis, ectomarginalis, suprasylvius, ectosylvius, and cinguli; nuclei basalis, caudate, and putamen; thalamus, globus pallidus, capsula interna, externa and extrema, ventral thalamic nuclei, subthalamic nucleus, H fields of Forel, zona incerta, mesencephalic tectum, nucleus ruber, and substantia nigra; mesencephalon—oculomotor nuclei and reticular formation; brainstem—nuclei cuneatus lateralis, gracilis, oliva, vestibularis, and formation reticularis; cerebellum—all folia of the cerebellar vermis and hemispheres, nuclei lateralis, interpositus, and fastigii; and gray and white matter of the cervical, thoracic, lumbar, sacral, and coccygeal spinal cord.
Qualitative screening identified axonal spheroids in the CNS as a primary lesion. Subsequently, a quantitative assessment for the number of spheroids and digestion chambers was performed, in which the total number of spheroids in all levels of the cerebral cortex, basal nuclei, and thalamus was summed. Spheroid counts were also summed in neuroanatomic regions present in sections of the mesencephalon (2 levels, including oculomotor nuclei and reticular formation), brainstem (8 levels; nuclei cuneatus, gracilis, oliva, reticularis, and reticular formation), and cerebellum (1 sagittal and 2 horizontal sections; nuclei lateralis, interpositus, and fastigii). Spheroids and digestion chambers within the gray and white matter of the spinal cord were counted separately and summed for the cervical (4 levels), thoracic (4 levels), lumbar (6 levels), sacral (2 levels), and coccygeal (1 level) spinal cord segments. The number of spheroids was compared between shivers and control horses blocked for region of the CNS and disease state using a 2-way repeated measures analysis of variance and Bonferroni post hoc testing (GraphPad Prism 5.0 Software, San Diego, CA). Significance was set at P < .05.
Immunohistochemistry
Immunohistochemical staining was performed on selected sections from the cerebellum, medulla oblongata, and cervical, thoracic, and lumbar spinal cord. Staining for nonphosphorylated neurofilament (NF), calbindin, calretinin, glutamic acid decarboxylase (GAD), and glial fibrillary acidic protein (GFAP) was performed using monoclonal and polyclonal antibodies (details provided in Supplemental Table 1). An automated slide stainer (Dako, Carpinteria, CA) or manual benchtop staining was utilized with a peroxidase-labeled polymer conjugate system (Dako) as a secondary antibody (EnVision+ goat anti-mouse: GFAP, NF, calbindin; EnVision+ goat anti-rabbit: calretinin, GAD) using previously described methods. 20
Electron Microscopy
Formalin-fixed material from the 4 shivers horses and 3 control horses that had axonal spheroids present in the third sacral spinal cord segment, nucleus cuneatus lateralis, and the nucleus lateralis in the cerebellum were trimmed and processed for transmission electron microscopy. Additionally, areas of the cerebellar nucleus lateralis with axonal spheroids and matching areas in a control horse were excised from the paraffin block and examined for ultrastructural changes for a correlative lesion analysis. Approximately 80 blocks were examined. Fragments of 1 mm3 were postfixed in 2.5% glutaraldehyde (Electron Microscopy Sciences, Hatfield, PA) in 0.1M sodium cacodylate buffer (Electron Microscopy Sciences). Tissue samples were postfixed in 1% osmium tetroxide (Electron Microscopy Sciences) in 0.1M sodium cacodylate buffer, dehydrated, and embedded in resin as previously described. 20 Thin sections (60–70 nm) were stained with 5% uranyl acetate and lead citrate and observed under JEOL 1200 EX II transmission electron microscope (JEOL LTD, Tokyo, Japan). Images were obtained using a Veleta 2 K × 2 K camera with iTEM software (Olympus SIS, Munster, Germany).
Muscular System
Within 30 min of euthanasia, 2-cm cubes of muscle were obtained from the forelimb triceps and hind limb biceps femoris (abductor and flexor of stifle), vastus lateralis (extensor of the stifle), and cranial tibial (flexor of the hock) muscles. Muscle samples were available from 1 additional 7-year-old control Warmblood mare that was euthanized due to chronic lameness. A cross section of each muscle sample was mounted on cork, frozen in methylbutane chilled by liquid nitrogen, and then stored at –80°C until histologic analysis. Eight-micrometer cryostat sections of each muscle were stained with hematoxylin and eosin, adenosine triphosphatase (ATPase, preincubation pH 4.6), and periodic acid–Schiff stain and examined for lesions. 5 The percentage of type 1, 2A, and 2B fibers in each muscle sample was calculated. To assess muscle fiber recruitment, the total percentage of muscle fibers depleted of glycogen by approximately 50% was calculated from sequential ATPase and periodic acid–Schiff stains for each muscle. A general linear model analysis of variance blocked for muscle and disease was performed to determine if there were differences in muscle fiber–type composition and differences in the percentage of fibers depleted of glycogen between shivers and control horses.
Results
Clinical Assessment
When prompted by the handler to lift the limb, consistent (4 of 5 horses) or intermittent (1 of 5) hyperflexion of a hind limb occurred in shivers horses. A description for each horse is provided in Table 1. Shivers horses initially resisted lifting the limb when it was touched and would frequently flex the opposite hind limb before hyperflexing the hind limb being touched. Mild bilateral hind limb extension, trunk rigidity, and pivoting on the inside hind limb occurred in all shivers horses during sharp turns at a walk. Shivers horses exhibited moderate to marked resistance to backward walking when the handler firmly pressed on their pectoral muscles. During backward walking, 4 of 5 shivers horses showed protracted hyperflexion of the hind limb, and 1 of 5 horses showed protracted hyperextension of the hind limb (Table 1). All shivers horses lacked a normal contralateral gait pattern when walking backward, resulting in difficulty moving backward in a straight line. The 2 oldest shivers horses had an additional intermittent feature of protracted abduction and hyperflexion of a hind limb when initiating forward walking, turning, or changing surfaces (Table 1). The locomotor examinations yielded normal results in control horses.
Neuropathology
Sensory and motor nerve roots, dorsal root ganglia, and sciatic nerve branches were normal in shivers and control horses. No abnormalities were identified in the cerebral cortex, basal nuclei, and thalamus apart from mineralization of medium-sized arteries in the basal nuclei of 1 shivers horse. Nonspecific lesions—such as multifocal neuronal ceroid lipofuscinosis in the brainstem and spinal cord neurons (2 of 5 shivers, 2 of 3 controls), polyglucosan bodies in the cerebellum (2 shivers), and mild multifocal lymphoplasmacytic inflammation in the cerebellum, brainstem, and spinal cord (3 controls)—were present but not consistent features of shivers horses.
Some degree of axonal degeneration was present in the CNS of both shivers and control horses, characterized by the formation of pale to intensely eosinophilic granular to stratified axonal swellings or spheroids (Figs. 1–3). Secondary demyelination characterized by digestion chambers or vacuoles in the neuropil frequently accompanied axonal degeneration.
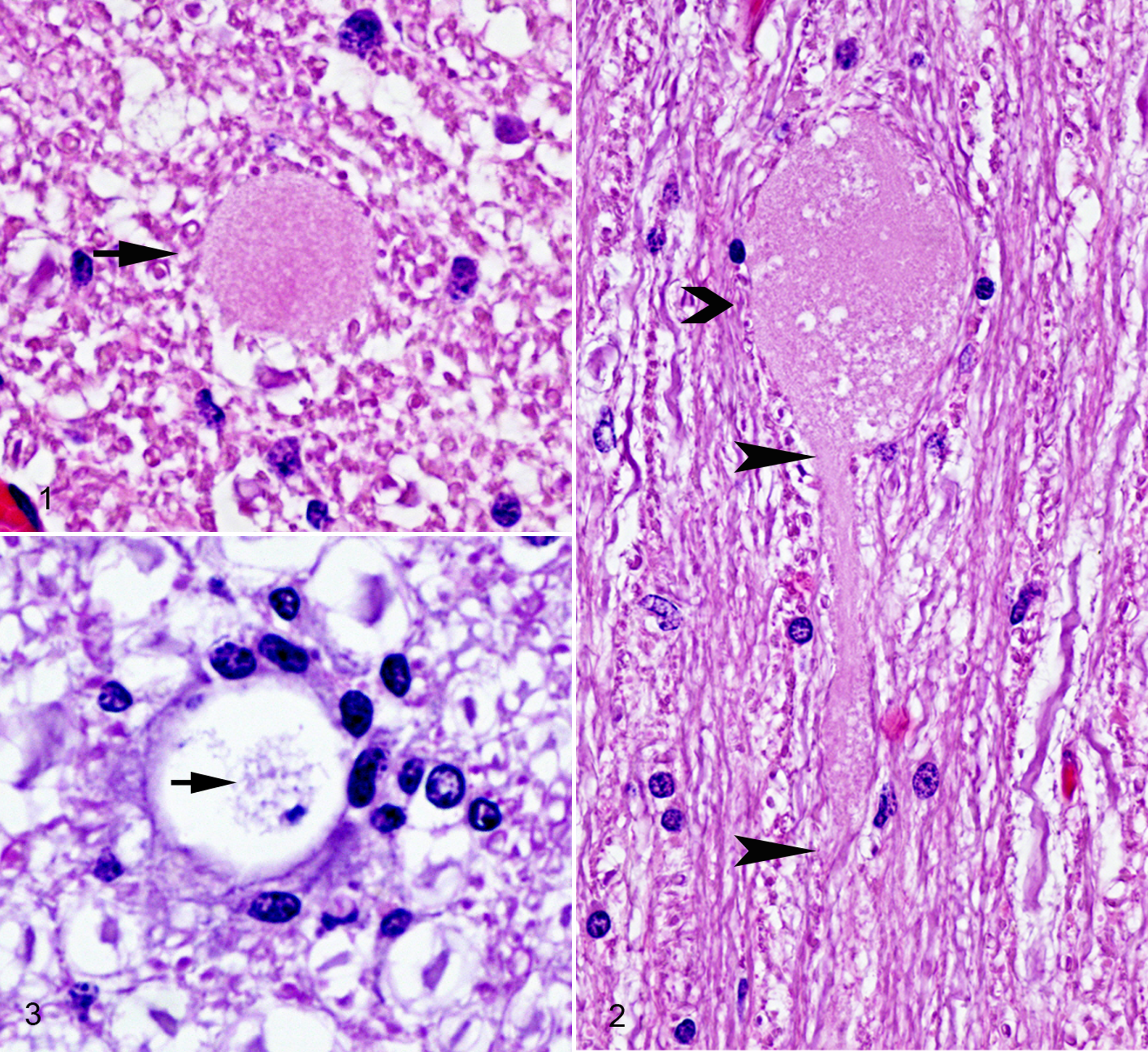
Cerebellum
Scattered axonal swellings in proximal Purkinje cell axons within the granular layer of the pyramids (Fig. 4), declive, and central lobules were highlighted by Bielschowsky stains of shivers but not control horses. A few axonal swellings were present in the white matter. No other abnormalities were apparent in the molecular or granular layer apart from a minor semiquantitative reduction in the number of Purkinje cells. Within the cerebellum, a segmental increase in GFAP was apparent, which varied from area to area within a folium and between folia. The regional variation in GFAP expression made it difficult to distinguish a clear difference between shivers and control horses. Trimming of the cerebellum was not performed in a fashion that would permit regional quantification and accurate comparison of Purkinje cell numbers between shivers and control horses.
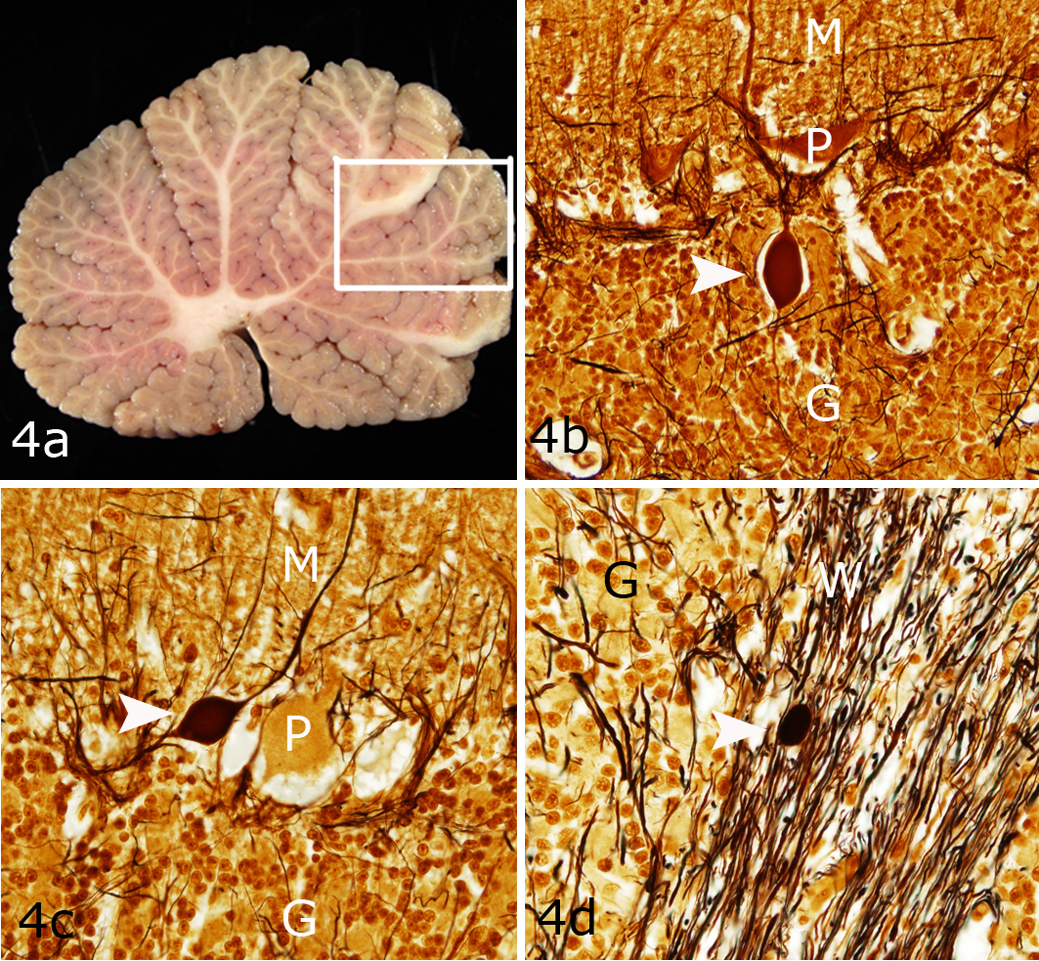
Shivers horse No. 4, cerebellar vermis. (a) Grossly normal cerebellum. The box depicts the region of the cerebellum shown in Figure 4b and c. (b,c) Axonal swellings (white arrowheads) in proximal Purkinje cell (P) axons within the granular layer (G) of the pyramids. M, molecular layer. (d) Axonal swellings (arrowhead) in the white matter (W). Bielschowsky.
Deep Cerebellar Nuclei
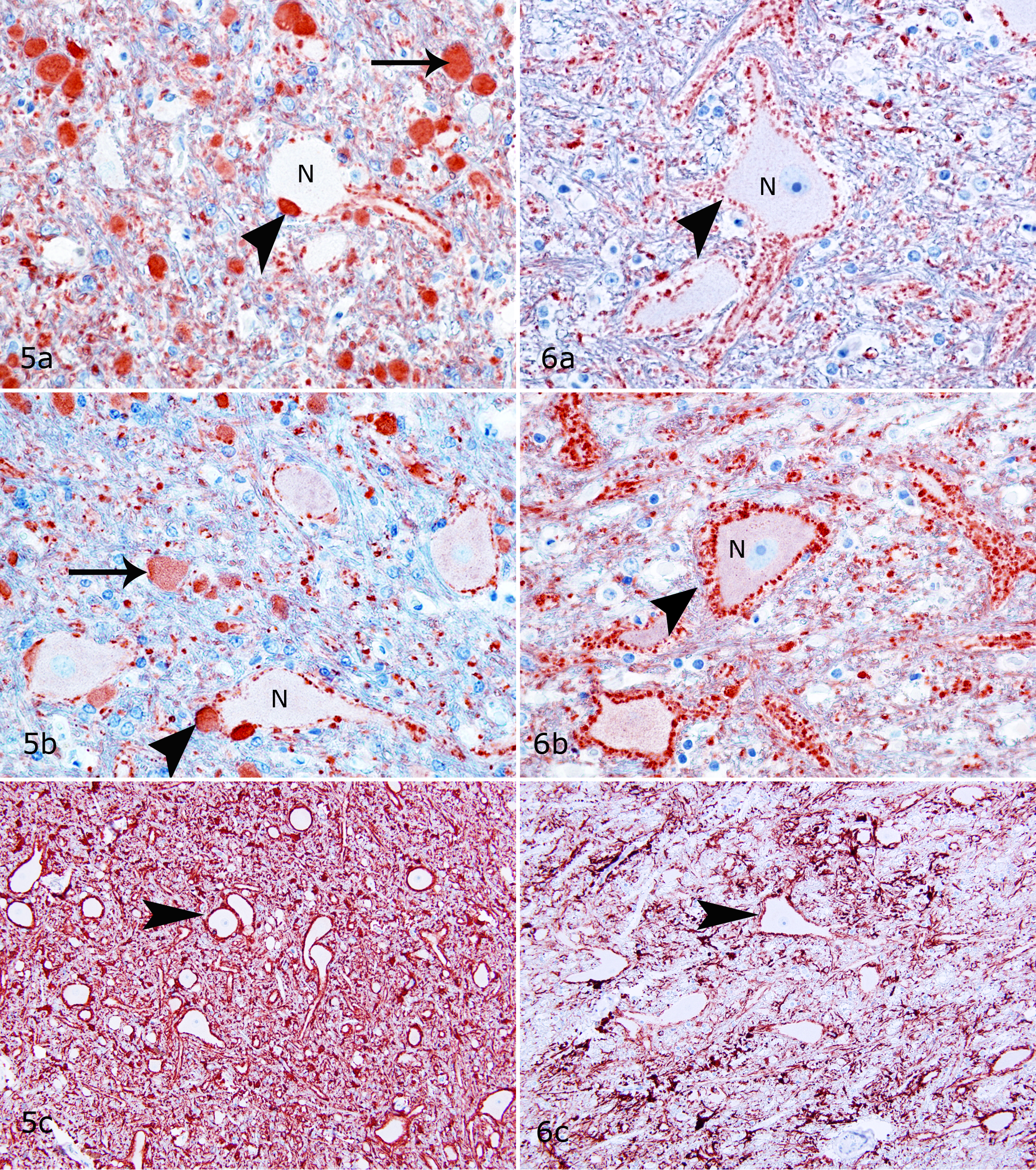
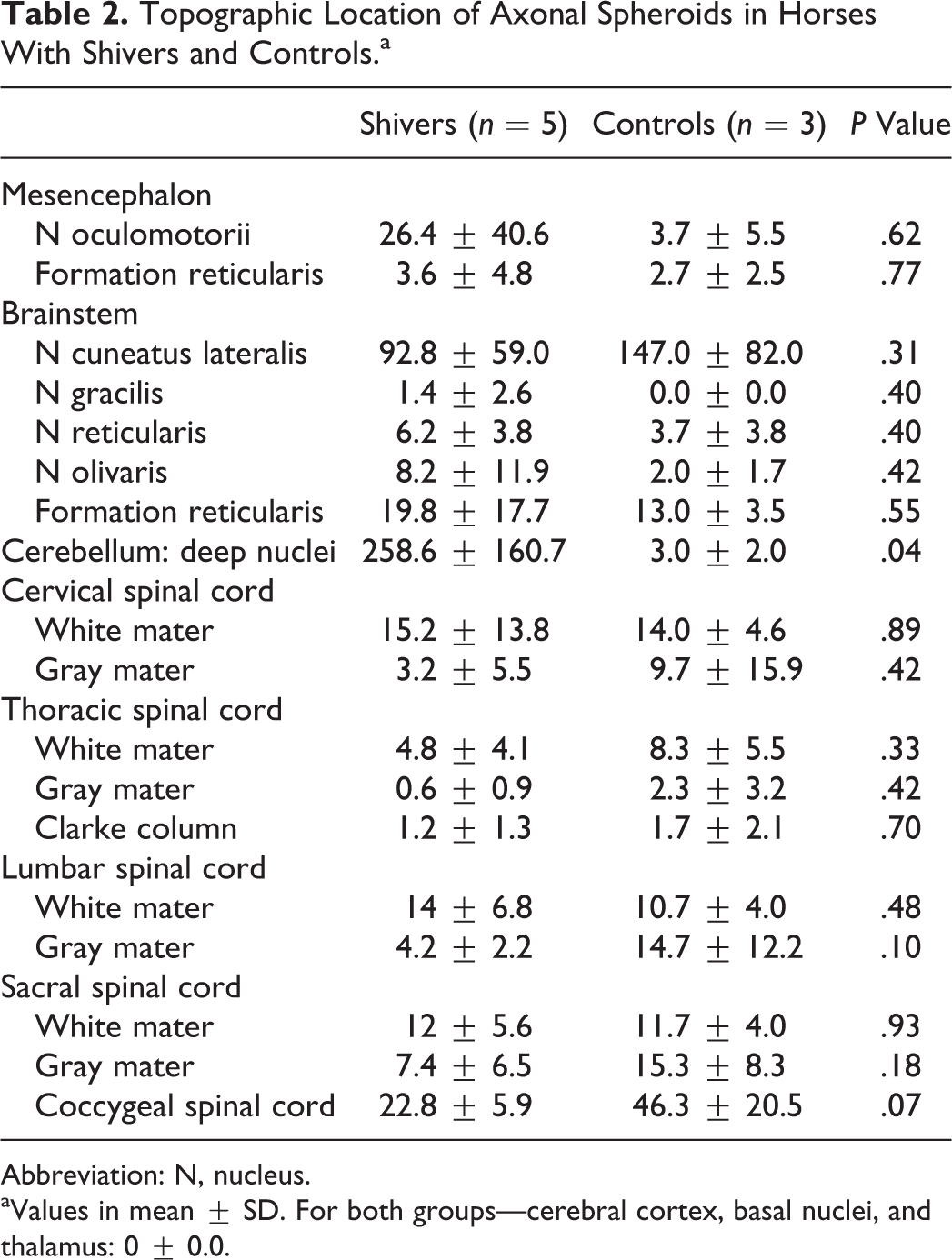
Spheroids were most prominent in the lateralis and interpositus nuclei and present to a lesser degree in the nucleus fastigii in shivers horses but rarely found in control horses. Spheroids in the nucleus lateralis were positive for calbindin (Fig. 5a), positive but less evident in GAD stains (Fig. 5b), and inconsistently positive in Bielschowsky stains. Spheroids did not stain for calretinin and nonphosphorylated NF. Calbindin-positive spheroids were located in Purkinje cell axonal end synapses (Fig. 5a), which appeared to be most prominent in the lateral regions of the nucleus lateralis. The number of synapses around neuronal cell bodies in the deep cerebellar nuclei (DCN) appeared to be reduced in shivers compared to control horses (Figs. 5a, b and 6a, b). Marked overexpression of GFAP by astrocytes occurred diffusely within the cerebellar nuclei of shivers compared to control horses (Figs. 5c and 6c). Astrocytes appeared slightly more numerous and their cell processes plump in shivers compared to control horses. More than 80-fold more spheroids were present in the DCN of shivers compared to control horses (F 6,21 = 5.68, P < .05; Bonferroni post hoc t = 10.63, P < .05; Table 2).
Topographic Location of Axonal Spheroids in Horses With Shivers and Controls.a
Abbreviation: N, nucleus.
aValues in mean ± SD. For both groups—cerebral cortex, basal nuclei, and thalamus: 0 ± 0.0.
Spinal Cord
A small number of axonal spheroids were present in the intermediate gray matter and white matter of the spinal cord of both shivers and control horses, with the highest numbers in the lumbosacral segments (Table 2). Spheroids in this region stained positively for calretinin and nonphosphorylated NF and negatively for calbindin and GAD. Slightly increased GFAP expression was found in the spinal cord of shivers compared to control horses.
Brainstem (Pons and Medulla Oblongata)
Numerous spheroids were present in the nucleus cuneatus, but their number did not differ between shivers and control horses (Table 2; Figs. 7, 8). Lesser numbers of spheroids were also randomly distributed within the formation reticularis, nucleus gracilis, nucleus reticularis lateralis, and nucleus inferior oliva of both shivers and control horses (Table 2). These spheroids stained positively for calretinin (Figs. 7, 8) and negatively for calbindin, GAD, and nonphosphorylated NF. Moderate to marked increase in GFAP expression was evident diffusely within nucleus cuneatus lateralis of shivers versus control horses.
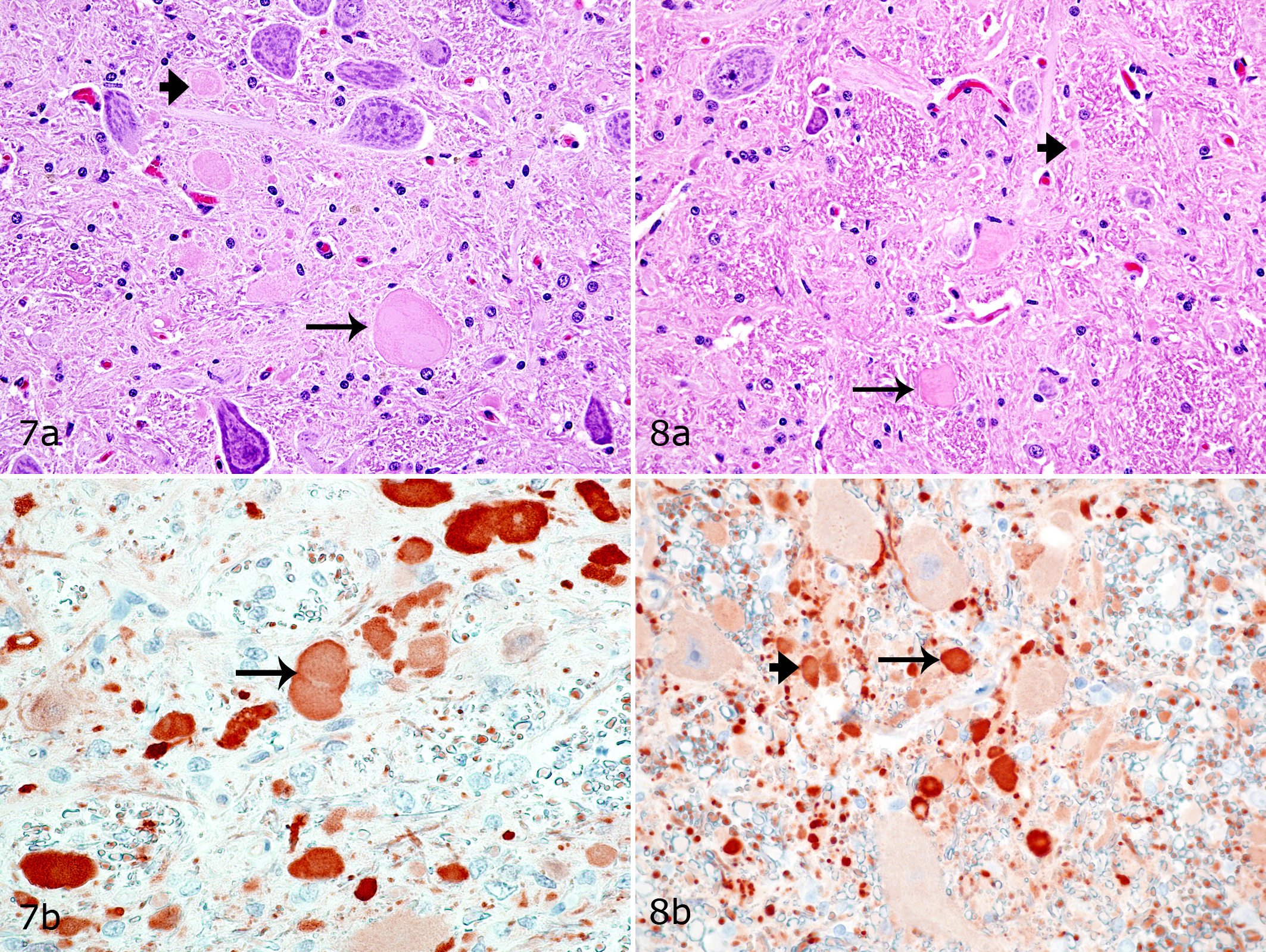
Muscle Histology
Myofiber necrosis or regeneration was not present in the 6 muscles evaluated from each horse. Blood vessels, connective tissue, and nerve branches appeared normal. Abnormal polysaccharide was not evident in muscle fibers of any of the shivers or control horses. Muscle fiber sizes, shapes, and distribution were within normal limits in all but 1 muscle sample evaluated. Cranial tibial muscle from 1 control horse showed focal areas of whole fascicular atrophy and other regions with fiber-type grouping that was not present in any of the other muscles obtained from that horse. Glycogen depletion patterns were not performed in this sample.
Pooled muscle fiber–type composition for the hind limb muscles showed no difference in type 1 fibers but a significantly higher proportion of type 2A fibers (P = .017) and lower proportion of type 2B fibers (P = .0009) in shivers compared to control horses (Table 3). A similar trend was found for fiber-type composition of the forelimb triceps muscle (Table 3). No significant differences were identified in muscle fiber recruitment patterns as evaluated by glycogen depletion patterns. The percentage of muscle fibers that showed 50% glycogen depletion in the hind limb or triceps muscles was not significantly different between shivers horses (hind limb pooled, 9.7% ± 9.5%; triceps, 14.8% ± 2.5%) and control horses (hind limb pooled, 5.6% ± 6.4%; triceps, 7.1% ± 12.0%).
Fiber-Type Composition of Triceps Muscle, Individual Hind Limb Muscles, and Pooled Hind Limb Muscles in Horses with Shivers and Controls.a
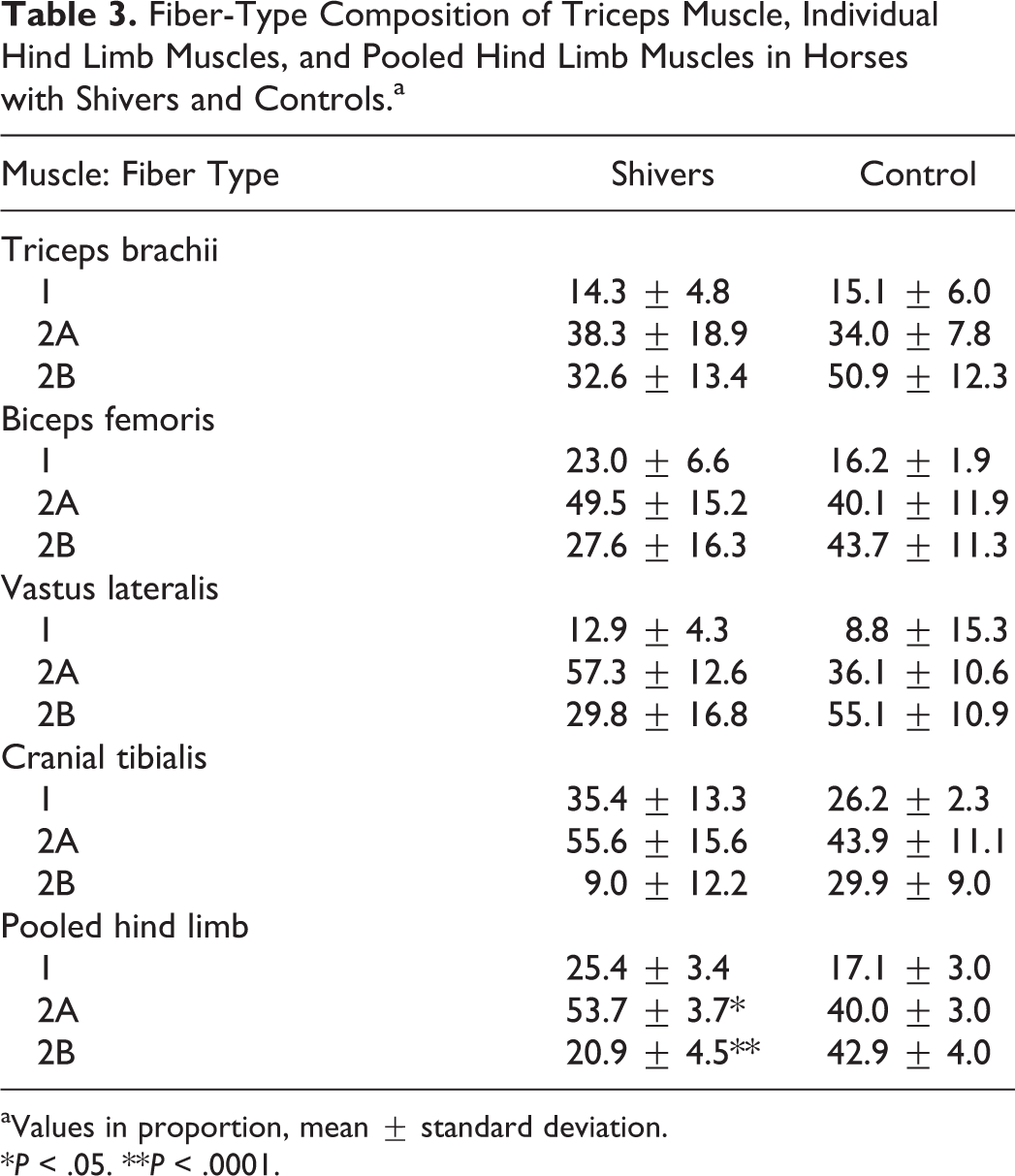
aValues in proportion, mean ± standard deviation.
*P < .05. **P < .0001.
Electron Microscopy
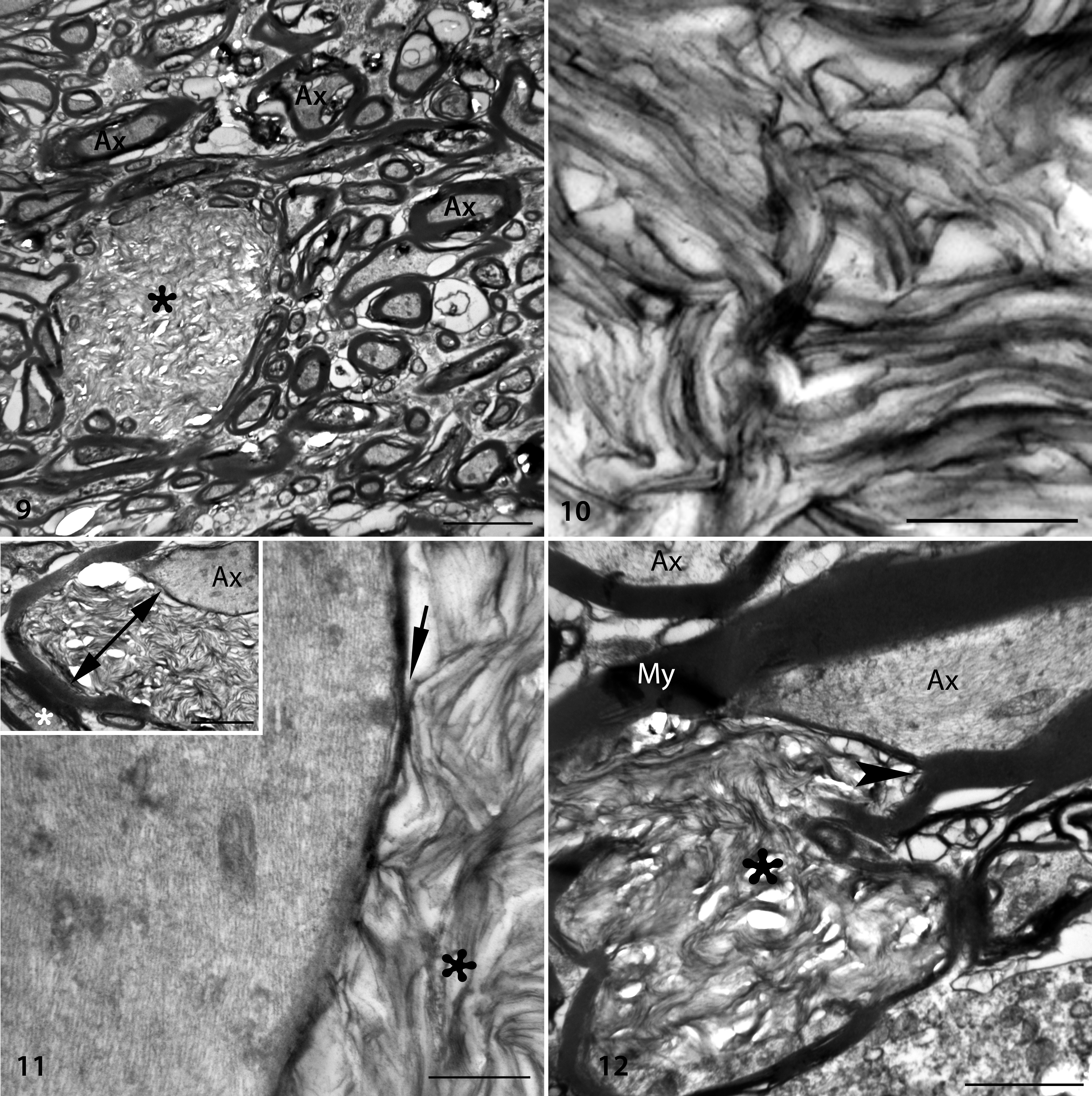
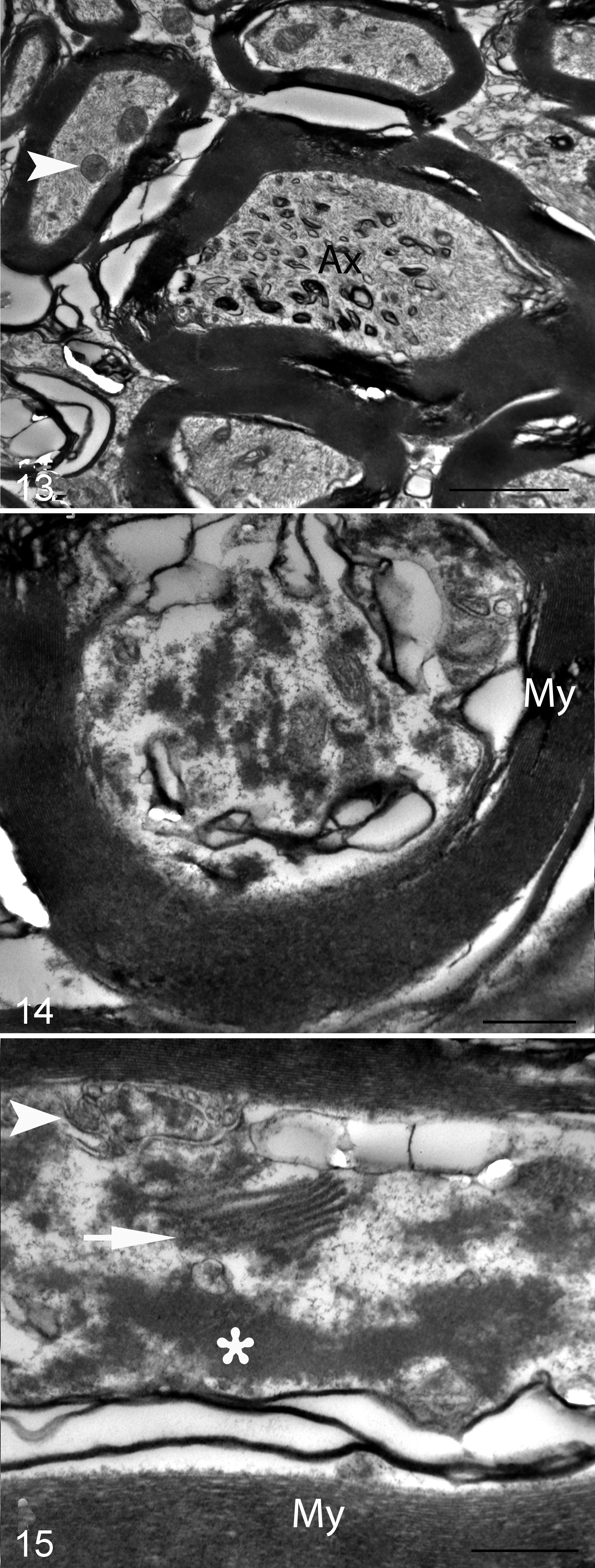
Highly unusual large elliptical (24 × 10 µm) accumulations of haphazardly arranged curvilinear to angular lamellar or membranous profiles were the most remarkable ultrastructural abnormality in the DCN of shivers but not control horses (Figs. 9, 10). The lamellae were at times contiguous with myelinated axons and present within the myelin sheath (Figs. 11, 12). On cross section, these accumulations reached larger dimensions and in some instances appeared physically separate from myelinated axons (Fig. 13). In other instances, lamellae were found between the inner and outer myelin sheath, giving the impression of severely decompacted myelin (Figs. 11, 12). Phagocytic cells or macrophages were present near some lamellar accumulations. In addition, axons in the deep cerebellar nuclei–presumptive Purkinje cell axons–commonly contained mild accumulations of residual bodies, tubular vesicular structures, and dense amorphous material (Figs. 13–15).
In contrast, segmental distention of axons—up to 5 μm in diameter and containing aggregates of cytoskeletal elements and organelles—was identified in the nucleus cuneatus lateralis and spinal cord. Microtubules and intermediate filaments were localized at the periphery, whereas vesicles, autophagic organelles, dense bodies, myelin figures, and mitochondria were in general localized at the center of the axonal dilation (Fig. 16). The myelin sheath was regionally thin or discontinuous (Fig. 16). Astrocytic processes were more numerous in the areas of degenerate axons, and microglia were also present. Numerous astrocytes and microglia contained residual bodies and lipofuscin inclusions.
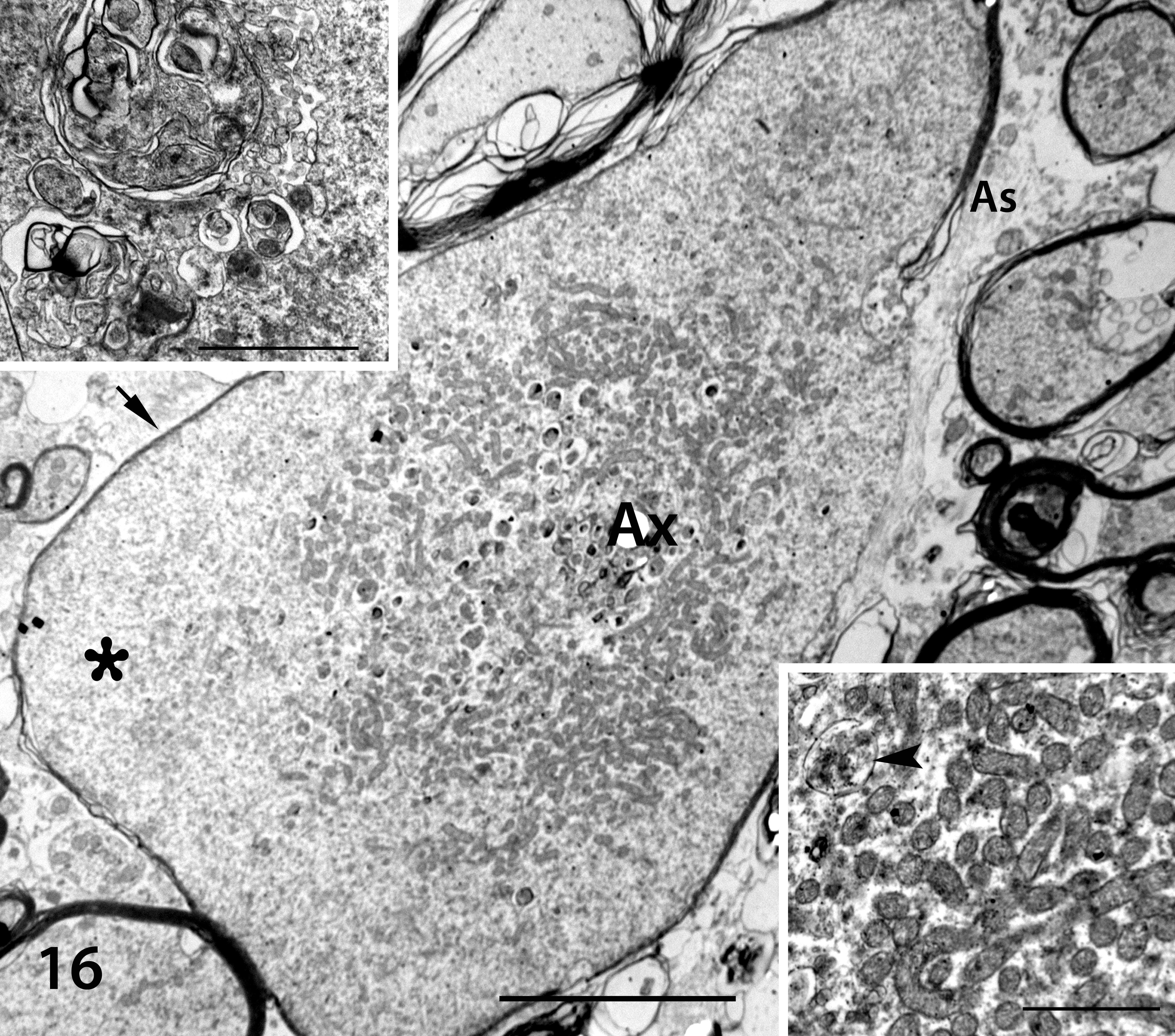
Shivers horse No. 1, nucleus cuneatus lateralis. Transmission electron microscopy. Tangential section of a 3.2-µm diameter axonal spheroid (Ax), central aggregation of vesicles and mitochondria, and presumed peripheral aggregations of microtubules and intermediate filaments (asterisk). Note a relatively thin myelin sheath (arrow) and astrocytic processes (As) partially surrounding the axon. Scale bar = 5 µm. Insets: Top left, autophagic vacuole containing membranous profiles. Scale bar = 2 µm. Bottom right, mitochondria and a residual body (arrowhead) within the center of a spheroid. Scale bar = 1 µm.
Discussion
Axonal spheroids are a common feature of aging and neurodegenerative diseases and often precede the loss of neuronal cell bodies. 4 The accumulation of NFs, mitochondria, and organelles in axonal swellings is believed to represent a disruption in anterograde or retrograde transport along the axon that can have a major impact on neuronal function. 4 Determining the significance of spheroids in subpopulations of neurons within the CNS of shivers and control horses required immunohistochemical and electron microscopic evaluation combined with an understanding of functional neuroanatomy. We selected (1) immunohistochemical stains for calcium-binding proteins such as calbindin and calretinin because they stain either Purkinje cells (calbindin) or other CNS neurons (calretinin) and (2) GAD stains to represent an inhibitory neurotransmitter. 16 In addition, stains for NFs (nonphosphorylated) were selected to highlight axonal degeneration.
Axonal spheroids were present throughout the spinal cord and in the nucleus cuneatus lateralis of both shivers and control horses. These axonal spheroids were calretinin positive and calbindin negative in both shivers and control horses and contained accumulations of NFs and organelles similar to those of many reported axonal spheroids. 4 Remarkably, however, 1 region in the CNS of shivers horses—the cerebellar nuclei—contained 80-fold more spheroids than that of control horses. These axonal spheroids were calretinin negative, calbindin positive, and GAD positive localizing axonal degeneration to Purkinje cells of shivers but not control horses. Furthermore, end terminals of Purkinje cells within the nucleus lateralis appeared to be reduced in shivers compared to control horses. Typical axonal spheroids, such as those found in the nucleus cuneateus lateralis, were not found in the DCN. Rather, ultrastructural analysis revealed that the region containing calbindin-positive spheroids contained highly unusual accumulations of membranous lamellar structures. These structures were frequently located between myelin sheaths of presumed Purkinje cell axons and appeared to represent severely decompacted myelin. 18
Axons of Purkinje cells are the sole efferent output from the cerebellum, and their pattern of connections in the cerebellar nuclei roughly maintains the temporal and spatial features conserved within the cerebellum itself. 15 The dorsal portion of the lateral cerebellar nucleus sends projections to the primary motor and premotor areas of the cerebral cortex, which have a key role in modulating voluntary movement of the extremities. 11,19 Lesions in the lateral dentate nucleus in human patients (roughly corresponding to the nucleus lateralis in animals) are characterized by decomposition of multijoint movement manifested by errors in the direction and rate of slow movements. 15 Of note, shivers horses have the greatest difficulty with slow learned movements, such as holding up a limb for a handler and walking backward. Thus, focal axonal degeneration within the lateral cerebellar nuclei in shivers horses could explain the specificity of the gait disorder.
Shivers horses lack evidence of ataxia, and the disorder has been characterized as a form of myoclonus due to the presence of quivering hind limb muscles during hyperflexion. 8 The increased ratio of type 2A to 2B muscle fibers in shivers horses is a finding seen in horses undergoing training. 23 In shivers horses, the increased ratio could result from constant muscle fiber recruitment induced by myoclonus. Myoclonus is a feature of some human cerebellar disorders. Degeneration of the dentate nucleus in a case of Gaucher disease led to action- and stimulus-sensitive myoclonus of the limbs and face, which was exacerbated by movement. 28 Focal damage to the cerebellum in human patients is also reported to cause nonepileptic myoclonus without cerebellar ataxia. 15 Such cases provide evidence that cerebellar damage can lead to aberrant muscle contraction, as seen in shivers horses without the more severe signs of cerebellar ataxia.
A unique clinical feature of shivers is that in the early stages of the disease, backward but not forward locomotion induces hind limb myoclonus. When healthy horses walk backward or forward, they have a smooth arc to the swing phase. 10 When walking forward, the swing phase of shivers horses is usually similar to that of healthy horses; however, when walking backward, they have a higher arc, as well as a pause at the height of the arc with abduction before rapidly placing the limb on the ground. 10 We speculate that in horses, the learned behavior of backward walking or lifting a limb from a manual cue necessitates cerebellar involvement in a manner that is different from the natural gait of forward walking. Supporting this, in humans there appear to be separate functional networks controlling forward and backward locomotion, and acute reactive locomotor adjustments are not dependent on cerebellar input, whereas learned motor behaviors and predictive locomotor adjustments are dependent on cerebellar input. 3,21 In addition, leg hypermetria is more severe in patients with cerebellar lesions when lifting the leg over an object from a standstill versus walking over the object. 22 Thus, cerebellar hypermetria appears to be context specific, with certain movements more likely to induce hypermetria than others. This may partly explain why shivers horses can perform complex motor tasks when moving forward, such as advanced dressage and jumping, while displaying marked clinical signs when moving backward or manually lifting the hind legs. 9
Unlike shivers, cerebellar abiotrophy in Arabian horses—the only previously described equine cerebellar disorder—is characterized by cerebellar ataxia, intention tremor particularly affecting the head, and variable hypermetric ataxia. 6 Lesions are much more severe, including marked degeneration, apoptosis, and loss of Purkinje cells. 2 In shivers horses, the focal nature of the lesion within the cerebellar nuclei, relatively normal numbers of Purkinje cells, and normal granular layer is entirely consistent with the absence of prominent signs of cerebellar ataxia. Purkinje cell numbers were only subjectively evaluated in a semiquantitative fashion in the present study, and it is possible that shivers horses had a regional loss of Purkinje cells. Furthermore, we cannot rule out that with progression of shivers, a greater loss of Purkinje cells or Purkinje cell axons could occur. Horses in the present study were in differing phases of the disease.
The pathogenesis of shivers is unknown, but the strong breed predilection suggests a genetic basis. 1,9 Of the many spontaneously inherited cerebellar disorders in mice, very few have a primary lesion within the DCN. 17,24 The closest homology based on DCN lesions would be mutations in Cacna1a, which encodes the α1A subunit of P/Q-type Ca2+ channel. Several spontaneous mutations have been identified in this gene, and depending on the site of the mutation, the severity of clinical signs varies from mild ataxia and paroxysmal dyskinesia (tottering) to severe ataxia (leaner). 16 Mice with a mutation in this gene can develop paroxysmal myoclonus in the hind limbs that gradually spreads to the front limbs and eventually the head and neck, often in response to changes in terrain, the environment, and stress. 10,13 The stimuli for hind limb myoclonus in shivers horses also include changing terrain and stress, as well as walking backward and lifting the limb. 8 Lesions vary across mutations and temporally, with early lesions of axonal spheroids in the DCN resembling those of shivers horses. 25,26 Later lesions include a variable loss of Purkinje cells. The end terminals of Purkinje cells in tottering Cacna1a mutation mice are enlarged and have an increased number of vacuoles, whorled bodies, and mitochondria. 16
Another progressive neurodegenerative disorder of Purkinje cell axons occurs in ataxic vacillator mice, which have a mutation in N-myristoylated, calcineurin-like EF hand protein (CHP-1). 18 CHP-1 homozygous mutant mice develop focal calbindin-positive swelling of Purkinje cell axons that resemble the lesions in shivers horses. Myelin sheath decompaction and detachment and accumulation of organelles in swollen neuronal fibers are reported in vacillator mice. 18 CHP-1 is part of a complex system that regulates calcium and cellular pH, which is especially important at nerve terminals. The exact means by which the CHP-1 mutation causes end terminal axonal degeneration is still under investigation. Thus, there are several potential pathologic mechanisms for Purkinje cell end terminal axonal degeneration in shivers horses, and genomic approaches could prove a key to identifying the etiology of this disorder.
In conclusion, the discovery that shivers is associated with Purkinje cell axonal degeneration is an important first step in better understanding an unusual disease that presents with difficulty walking backward and/or lifting the limb in response to manual cues. Further investigations using surface electromyography (to characterize decomposition of multijoint movement in shivers horses) and genome sequencing will be required to define the pathophysiology and etiology of this highly unusual movement disorder.
Footnotes
Acknowledgements
We wish to acknowledge the contributions of Dr Anne Nicholson, Dr Carrie Finno, Dr Steve Reed, Dr Joe Mayhew, Dr John Baird, Edgar d’Almeida, and Dean Muldoon.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this projected was provided by the United States Equestrian Foundation and the University of Minnesota Equine Center, with funds provided by the Minnesota Racing Commission and Minnesota Agricultural Experiment Station.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
