Abstract
Two wild fledgling kestrels exhibited lack of motor coordination, postural reaction deficits, and abnormal propioception. At necropsy, the cerebellum and brainstem were markedly underdeveloped. Microscopically, there was Purkinje cells heterotopy, abnormal circuitry, and hypoplasia with defective foliation. Heterotopic neurons were identified as immature Purkinje cells by their size, location, immunoreactivity for calbindin D-28 K, and ultrastructural features. The authors suggest that this cerebellar abnormality was likely due to a disruption of molecular mechanisms that dictate Purkinje cell migration, placement, and maturation in early embryonic development. The etiology of this condition remains undetermined. Congenital central nervous system disorders have rarely been reported in birds.
Keywords
In contrast to mammals, congenital central nervous system (CNS) disorders have rarely been reported in birds.8,12,14 In mammals, CNS disorders have inherited, infectious, or toxic causes. 12 Cerebellar hypoplasia is one of the most common congenital diseases of the CNS in domestic animals. 12 The actively dividing granular cells in the developing cerebellum are especially sensitive to viral infections or chemical teratogenic agents.8,12 Selective necrosis of the granular cells has been recognized as an important cause of virus-induced cerebellar hypoplasia.8,12 The most prevalent and best-defined entities of cerebellar hypoplasia are those that follow viral infection of the fetal cerebellum by feline panleukopenia virus, canine parvovirus, bovine viral diarrhea virus, Border Disease virus, and classical swine fever virus.8,12 Recently, an outbreak of cerebellar hypoplasia and hydrocephalus in day-old broiler chickens was associated with a transovarial parvoviral infection. In affected chickens there were small cerebellar folia with loss of the granular cell layer and disorganization of the Purkinje cell layer.8,12 Cerebellar hypoplasia has also been described in chickens experimentally infected in ovum with either fowl glioma inducing retrovirus or Aino virus. 14
The morphogenesis of the cerebellum is highly preserved across species 13 and comprises four steps: (1) characterization of the cerebellar territory in the midbrain; (2) formation of two compartments for cell proliferation of, for example, GABAergic and glutamatergic neurons; (3) inward migration of the granular cells; and (4) formation of the cerebellar circuitry and further differentiation. 13 GABAergic cerebellar neurons, including Purkinje cells, stellate and basket cells, and a subset of deep cerebellar nucleus cells, arise from the ventricular zone of the mesencephalic alar plate, whereas glutamatergic neurons such as granular cells and a subset of deep cerebellar nucleus cells originate from the rhomboid lip.10,13
Purkinje cells cease proliferation within the ventricular zone and rapidly express numerous differentiation markers. Thereafter, they migrate along radial glia cells to their future position, making use of reelin secreted by external germinal cells.2,7 Purkinje cells group first in the cerebellar plate and, from there, disperse to form a monolayer and mature. Granular cell progenitors migrate to form the external germinal layer and further differentiate to form the granular cell layer. Granular cells form axons, known as parallel fibers, and migrate along the Bergmann glia fibers to their definitive site, the internal granular cell layer. Sonic hedgehog (Shh) protein that is expressed in migratory and settled Purkinje cells acts as a potent mitogenic signal to expand the granular cell progenitor population.10,13 The initial formation of the cerebellar fissures and folia, a process called “foliation,” is determined by physical forces provided by the granular cells. Cell maturation, characterized by changes in morphology of granular cells, Purkinje cells, and Bergmann fibers, occurs where fissures are emerging. Areas at the base of the fissures are known as anchoring centers. 11
Clinical History
Two wild fledgling American kestrels (Falco sparverius) presented to the veterinary clinic of the Wildlife Center of Virginia because of an inability to fly. The birds, a female and a male fledgling (kestrel Nos. 1 and 2, respectively), were estimated to be approximately 4 to 5 weeks old. Both birds were found within a span of approximately 2 weeks on a single private property in rural Augusta County, Virginia. On physical examination, the birds exhibited neurologic signs including severe ataxia, repetitive and exaggerated head bobbing, hock sitting, bilateral flexion of the digits into a tight ball, a broad-based stance, dysmetry, and a reduced ability to balance on a perch or flat surface. The two kestrels exhibited normal mentation, with appropriate responses to external stimuli, and good appetite. There was no physical or radiographic evidence of traumatic injury. The serum of the female bird was negative for antibodies against West Nile virus and St. Louis encephalitis virus by plaque reduction neutralization assay, avian influenza virus by agar gel immunodiffusion test, and Newcastle disease virus by hemaglutination inhibition. The disease did not progress but also did not improve despite supportive care so that both birds were euthanatized after a 4-week period.
Gross and Microscopical Findings
At necropsy, brain, spinal cord (only from kestrel No. 2), and tissue samples from major organs were fixed in 10% buffered formalin. On gross examination, both kestrels had a smaller than normal cerebellum and brainstem when compared to a hatch year control kestrel (kestrel No. 3), which died from non-neurological disease (Fig. 1). In addition, the diameter of the spinal cord of kestrel No. 2 was small compared to the control kestrel. In both diseased birds cerebellar folia were poorly defined and very thin.
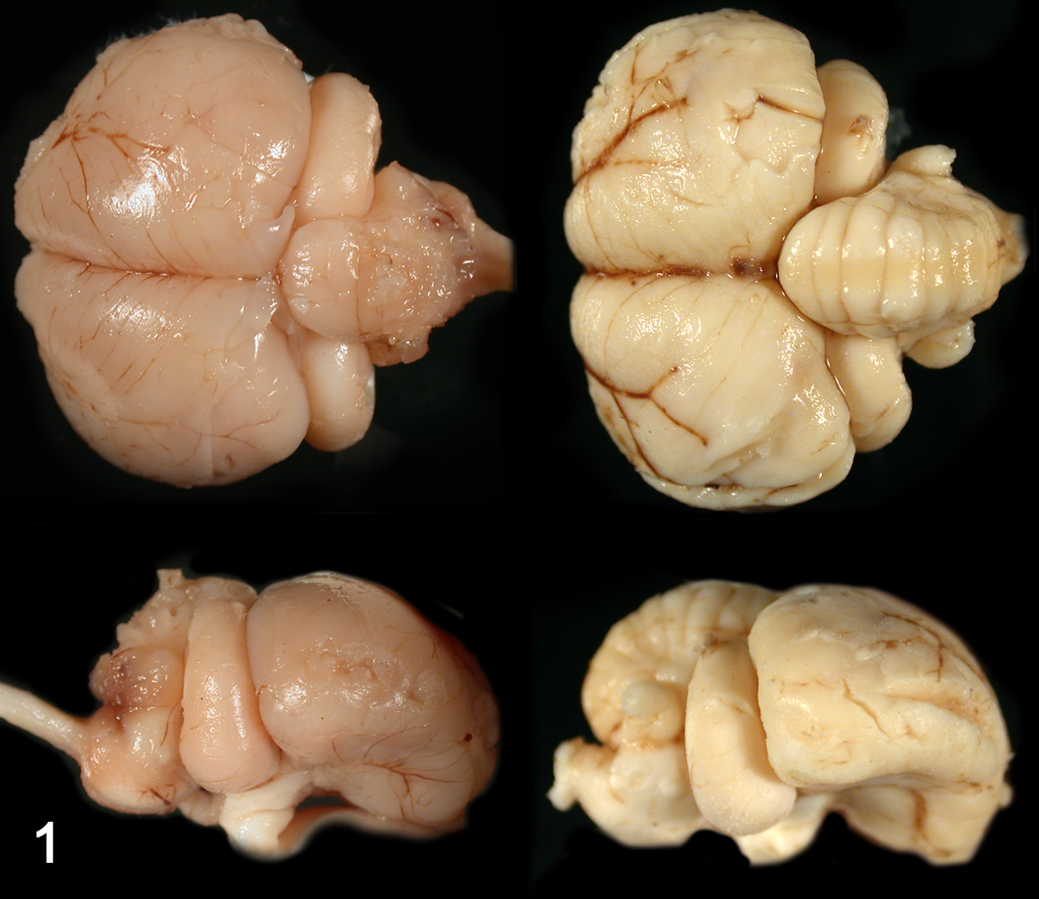
Brain. Dorsal and lateral view of the encephalon of Kestrel No. 2 (left) and a normal-sized, age-matched control (right). Note that the difference in color between the diseased kestrel and the control kestrel is a fixation artifact.
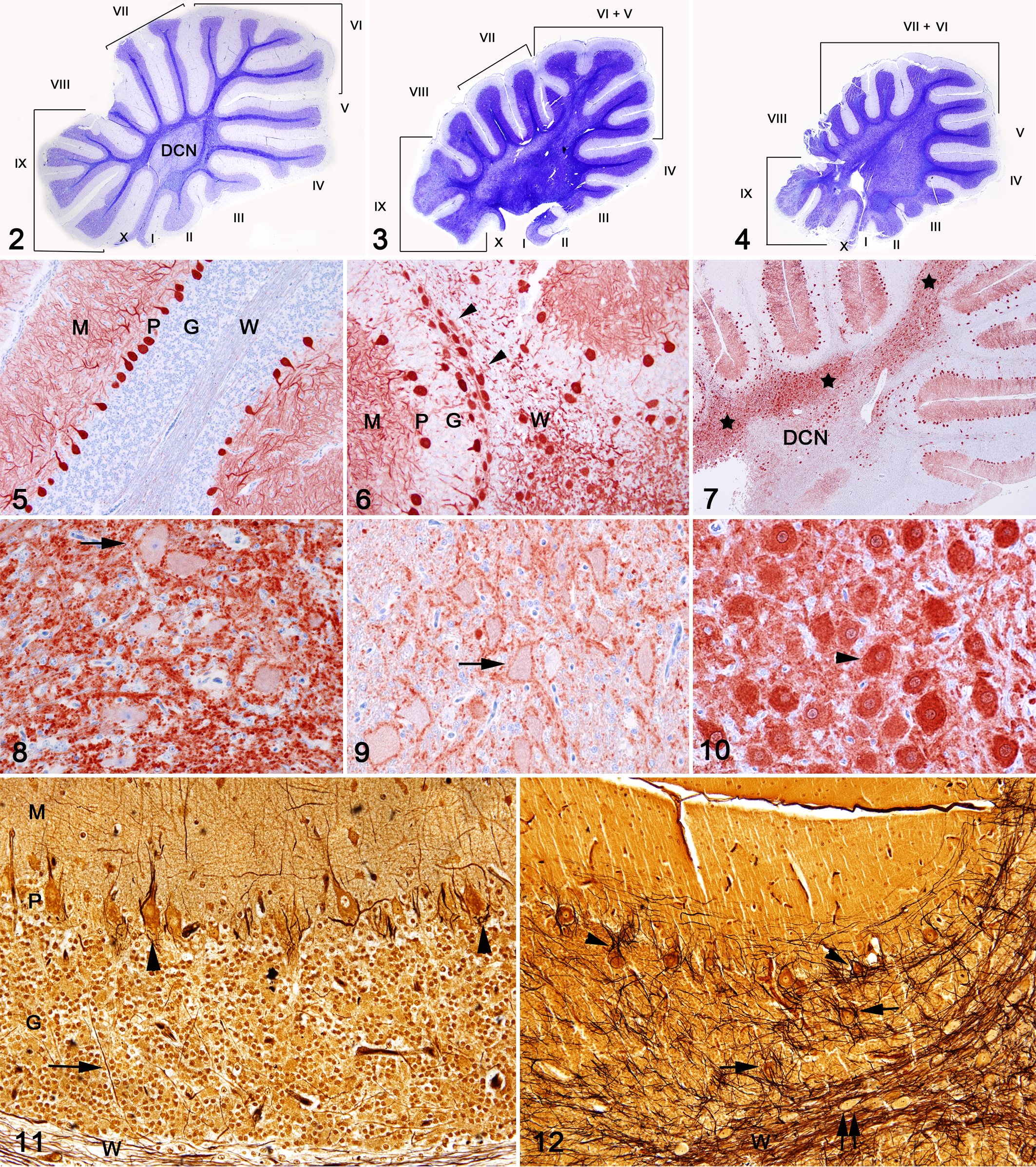
Formalin-fixed tissue samples were paraffin-embedded, sectioned at 5 μm, and hematoxylin and eosin (HE) stained. Select samples of brain and nerves were stained with Bielschoswky, Cresyl Violet, and Luxol Fast Blue (LFB). A peroxidase-based polymer system was used for immunohistochemical demonstration of Calbindin D-28 K protein (Swant, Bellinzona, Switzerland) using a mouse monoclonal antibody at a dilution of 1:2000. 1
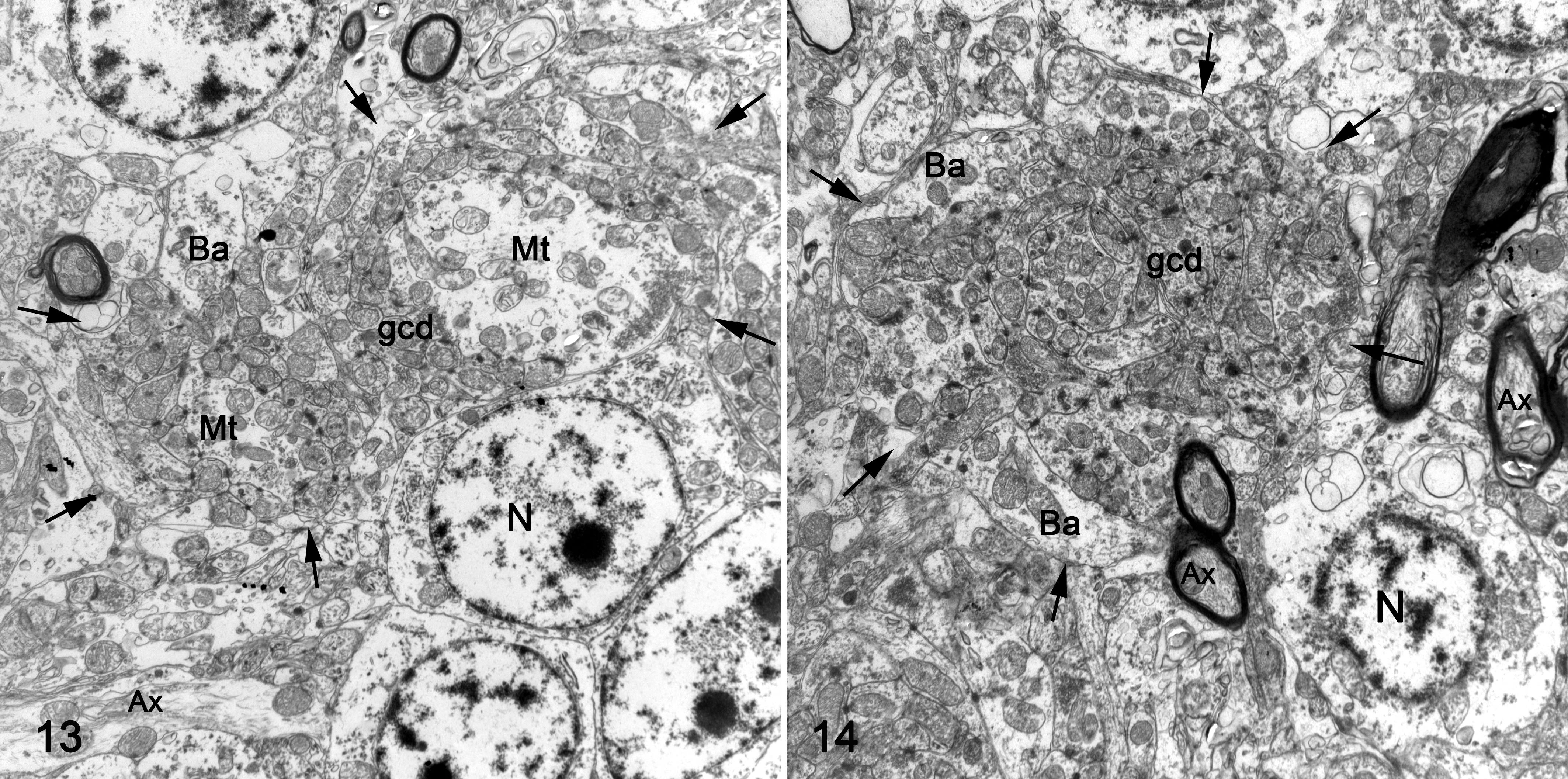
For electron microscopy, flocculus; cerebellar folia VI, VII, and IXcd; and pedunculus from kestrel No. 1 and the control bird were postfixed in 1% osmium tetroxide, embedded in Embed® (Electron Microscopy Sciences, Hatfield, PA, USA), and 80 nm ultrathin sections were stained with uranyl acetate and lead citrate (Electron microscopy sciences, Hatfield, PA, USA). Specific nomenclature of the cerebellar anatomy was based on previously published information on avian cerebellum. 6 Additionally, the liver of the female bird was tested by inductively coupled plasma spectroscopy for heavy metals and mineral elements.
Histologically, lesions were confined to the cerebellum. Brain stem and spinal cord were devoid of significant microscopic architectural changes despite the macroscopically noted smaller size. In both kestrels there was an abnormal and marked reduction of the size of the folia compared with those of a control kestrel (Figs. 2–4). Primary and secondary fissures were indistinct compared with the control. Abnormal folia were present, including folia V, VI, and IX in kestrel No. 1 and folia VI, VII, and IX in kestrel No. 2 (Figs. 3, 4). In kestrel No. 1, folium V appeared subdivided, whereas folia VI and IX exhibited few secondary subdivisions (Fig. 3). Kestrel No. 2 had a smaller cerebellum and relatively uniform-sized folia VI to VII, which made it difficult to distinguish a secondary subdivision of folium VI (Fig. 4). The peduncular area was expanded and highly cellular compared to the control kestrel (Figs. 2–4). Cells in these areas were large and were considered to be misplaced neurons. Purkinje cells in the control kestrel as well as Purkinje cells and misplaced neurons in both diseased birds strongly expressed Calbindin D 28 k protein (Figs. 5–7, 10). Therefore, these misplaced neurons were considered to be heterotopic Purkinje cells. Heterotopic Purkinje cells were aggregated within the white matter and formed a large and irregular lamina between the deep cerebellar nucleus and the base of the folia (Figs. 6, 7). In the control kestrel and both diseased birds, neurons of the deep cerebellar nucleus were Calbindin D-28 k protein negative (Figs. 8, 9). In kestrel No. 1, the irregular lamina with heterotopic Purkinje cells extended from folium VI to folium IX (Figs. 3, 7). In kestrel No. 2, this lamina extended from folium III to folium IX (Fig. 4). In both birds folium IX was markedly disorganized with poorly defined cortical layers and a large number of heterotopic Purkinje cells within the granular cell layer and white matter (Fig. 6). The granular cells surrounded small, poorly defined glomeruli. The Purkinje cell layer at the interface of granular and molecular layers lacked Purkinje cells. The molecular layer was thin and hypocellular. Folium III in kestrels No. 1 and No. 2 and folium VI in kestrel No. 2 were moderately affected. In these folia, there was a moderate to marked decrease in the number of Purkinje cells. The folia I, II, and X in both birds were mostly devoid of Purkinje cells and myelinated axons. In kestrel No. 1, folia VI, VII, and VIII and in kestrel No. 2, folia IV, V, VII, and VIII displayed multifocal clusters of heterotopic Purkinje cells. In these folia there was a moderately to markedly decreased number of Purkinje cells, small cerebellar glomeruli, and a thin, highly cellular molecular layer (Fig. 6). In sections stained with Bielschowsky and LFB, folia revealed a diffuse and dense framework of disorganized cell processes when compared to the folia of the control bird (Figs. 11 and 12). Bizarre, dense, and sometimes empty baskets were present in the Purkinje cell layer. Abnormally arranged cell processes also surrounded heterotopic Purkinje cells within the granular cell layer.
On electron microscopy, Purkinje cells properly located at the molecular and granular cell layer interface (Purkinje cell layer) exhibited a cytoplasm rich in rough endoplasmic reticulum, which was composed of small cisterns forming a closely packed lamellar structure, mitochondria, and prominent Golgi complex. Heterotopic neurons had a similar density of these organelles. Segmentally, myelinated axons within the Purkinje cell layer varied in diameter and contained aggregation of intermediary filaments and numerous abnormally large mitochondria. In contrast with the control bird, the granular cell layer of affected folia (flocculus, cerebellar folia VI, VII, and IXcd) and pedunculus from kestrel No. 1 showed a variable density of granular cells and variable degree of glomerular disorganization on thick sections. Granular cells did not exhibit any significant ultrastructural changes. The architecture of cerebellar glomeruli was disorganized (Figs. 13, 14). Compared to the control bird, mossy fiber terminal density in the affected folia of kestrel No. 1 varied. While the overall density was reduced, there were areas in the granular cell layer in which mossy fibers cluster close together. These mossy fiber terminals appeared irregular in size and shape and were surrounded by an abnormally large number of granular cell dendrites and more peripherally with axons and/or dendrites of interneurons such as unipolar brush cells and Golgi cells (Figs. 13, 14). In the cerebellar folia IXcd, the vast majority of glomeruli had indistinct mossy fiber terminals and were instead composed of large conglomerates of disorganized granular cell dendrites. Numerous large, presumably unmyelinated processes (axons) of Purkinje cells, and small and poorly myelinated axons were found throughout the granular cell layer. The molecular layer exhibited disorganized parallel fiber fascicles surrounding underdeveloped Purkinje cell dendrites. Parallel fibers contained abnormally large mitochondria.
Inductively coupled plasma spectroscopy of liver tissue (kestrel No. 1) revealed that the concentration of arsenic, cadmium, and lead were below the detection level of 1 ppm (wet weight).
Discussion
This report describes a developmental disorder of the central nervous systems of two wild fledgling kestrels that was characterized macroscopically by an underdeveloped cerebellum, brainstem, and spinal cord and microscopically by cerebellar hypoplasia, abnormal cerebellar foliation, and Purkinje cell heterotopy. Based on the similar morphology of the lesions in both birds and the fact that both birds were found within a short period of time in the same location, we propose that the etiology of the condition was identical in both cases. The etiology of this disease was not determined, but possible etiologies may include viral infection, nutritional deficiency, toxic insult, or an inherited genetic defect. Serological testing of both birds failed to detect antibodies against a variety of neurotropic viruses including influenza virus, Newcastle disease virus, West Nile virus, and St. Louis encephalitis virus. Brain malformations have been associated with methyl mercury exposure in mallards. 5 Unfortunately, mercury concentrations, including methyl mercury concentrations, were not tested in these birds. Given the history, it appears possible that both animals were from the same clutch. Hence, a primary genetic disorder may be considered.
In both kestrels, the hypoplastic cerebellum presented three major histologic features, which included neuronal heterotopy, abnormal circuitry, and defective folia formation.
In both diseased kestrels, Purkinje cells as well as heterotopic neurons expressed Calbindin D-28 k protein, which supports the hypothesis that these misplaced cells were in fact Purkinje cells. In the cerebellum, Calbindin D-28 k protein is exclusively expressed by both migrating and mature Purkinje cells of mammals and birds. 1 In addition to the calbindin D 28 k protein expression, several observations support the Purkinje cell identity of these neurons: (1) the size range of the misplaced neurons matched with Purkinje cells. (2) Purkinje cells were absent in large stretches in the Purkinje cell layer at the molecular and granular cell layer interface. (3) An extensive laminar aggregation of heterotopic cells (cerebellar plate) within the white matter above the deep cerebellar nucleus and below the cerebellar folia was present. (4) Ultrastructurally, ectopic neurons displayed a similar density of organelles including small lamellar packed rough endoplasmatic reticulum (RER), which resemble RER arrays found only in mature Purkinje cells.
In general, cerebellar folia in both kestrels in our study were shortened and abnormal. Small and misshapen cerebellar folia are features of hypoplastic cerebella due to parvovirus and avian leukosis virus infections in chicken.8,14 In these infections, the disfiguration of the cerebellum has been associated with a primary depletion of the granular cell layer and disorganization (misalignment) of the Purkinje cell layer.8,14 In the kestrels, the absence of an external germinal layer indicates that migration and maturation of the internal granular cells had been accomplished. The cerebellar folia, however, were narrow, which may indicate a lower number of granular cells possibly due to lack of mitogenic stimulation by properly located Purkinje cells, rather than a primary granular cell depletion. Molecular signals exchanged in the nerve tissue are crucial for providing positional identity to cells that will form different populations in the brain. Spatiotemporal modifications of the signaling during neurogenesis can result in profound changes and can be the origin of brain malformations. 4 The changes in these kestrels indicate a disruption of the early cerebellar development after the Purkinje cell proliferation took place in the ventricular zone and suggest that the migration was affected more specifically after the Purkinje cells reached the cerebellar plate.7,10 Furthermore, the large number of misplaced Purkinje cells strongly suggests that a defective migration of a large number of these cells may be the main abnormality that triggered the subsequent developmental errors such as abnormal foliation and neuronal maturation. Silver impregnation (Bielshowsky), Calbidin immunohistochemistry, and ultrastructural analysis demonstrated abnormal cell interconnection and circuitry even in folia that had normal architecture based on HE-stained sections.
The mechanisms that command cell migration, placement, and maturation seem to be genetically controlled and complex. We suggest that the Purkinje cell heterotopy may be associated with: (1) a defect of the Purkinje cells (e.g., receptors for migration factors), (2) a defect in the production of trophic factors (migration factors), and/or (3) a defect in radial glial fiber support.7,10,13 In mice with mutation of the reelin gene, most Purkinje cells fail to disperse and migrate to form a mature monolayer.7,10,13 Alternatively, the number of folia seems to be regulated by the Shh secretion by Purkinje cells via its influence on granular cell precursors in the external germinal layer. 3 The size and shape of the folia appear to be determined principally by the time and position at which anchoring centers are formed. 11 Further, Purkinje cell heterotopy and altered foliation defects have been identified in “Cerebellar deficient folia” (cdf) gene mutation. 9
The term neuronal heterotopies specifically applies to neuronal migration disorders described in humans, but these conditions have not been recognized in animals. In humans, cerebellar heterotopies are common in children with trisomy 13, cerebellar hypoplasia, and brainstem dysplasia and in conjunction with other migratory disorders. Cerebellar heterotopies are characterized by large clusters of Purkinje cells surrounded by a thin corona of neuropile and islands of cortex. 4
Clinically, both kestrels demonstrated severe incompetence to modulate the range, force, and rate of the muscular tonus necessary to stand and execute postural reaction. They also were unable to execute and coordinate the complex movements associated with walking or flying. Thus, the nature of these neurological deficits is consistent with a Purkinje cell deficit and subsequent dysfunction of the vestibulocerebellar and spinocerebellar systems. 12 Overall the most severely affected cerebellar folia were V, VI, and IX. These folia have been associated with the walking and flying functions in birds and damage to these folia conceivably may result in lack of motor coordination and agonist-antagonist control of wings and leg movement. 6 Despite their small size, the brainstem in both kestrels and spinal cord in one kestrel did not show any remarkable histological changes indicating dysplasia. Therefore, the reduction in size of the brainstem and spinal cord is considered to be hypoplasia as a consequence of the cerebellar lesions, resulting in a decreased number of axons. Primary pontocerebellar hypoplasia has not been recognized in animals to date. 12
In summary, a unique cerebellar malformation with Purkinje cells heterotopy, abnormal circuitry, and hypoplasia with defective foliation in two possibly related free-ranging kestrel hatchlings is described. Microscopical features of the lesions indicate a primary Purkinje cell defect. We suggest that the pathogenesis of the hypoplasia and defective foliation is likely due to a disruption of molecular mechanisms that dictate Purkinje cell migration, placement, and maturation.
Footnotes
Acknowledgements
We thank Jan Shiver, Don Ariyakumar, Rhonda Aho, and Dean Muldoon for outstanding histology, immunohistochemistry, and electron microscopy preparations.
