Abstract
Immunohistochemical study was performed on cerebellar Purkinje cells of two dogs with hypoglycemia using an antibody against the inositol 1,4,5-triphosphate receptor that is identical to the cerebellar Purkinje cell glycoprotein P400 (P400/InsP3R). In the cerebellar neocortex of an acute case of hypoglycemia, the P400/InsP3R staining of hypoglycemic Purkinje cells was heterogeneous: some peripheral dendrites, including spiny branchlets, were negative and others were stained with various intensities, although Purkinje cells were morphologically intact by hematoxylin and eosin (HE) stain. In a chronic case of hypoglycemia, almost all the dendrites of Purkinje cells of both the neo- and archicortex of the cerebellum were not stained with the P400/InsP3R antibody. This is in contrast to the normal dog where Purkinje cell bodies, axons, and dendrites, including spiny branchlets, are intensely stained by the P400/InsP3R antibody. These results suggest that P400/InsP3R immunolabeling of Purkinje cells decreased, despite their morphology being preserved by HE stain, and that the function of P400/InsP3R, especially in spiny branchlets that receive inputs originating from axon terminals of parallel fibers, may be impaired in hypoglycemia.
Keywords
Hypoglycemia refers to decrease in level of serum glucose and depressed cerebral energy metabolism. 5 In the brain, neuronal necrosis, with a predilection for cells in the superficial lamina of the cerebral cortex, has been described in hypoglycemia in humans and in experimentally induced hypoglycemia in animals. 1 Recently, we reported hypoglycemic brain lesions, characterized by an acute neuronal necrosis in the superficial layers of the cerebral cortex and spongy changes in the dentate gyrus of the hippocampus, in a dog affected with insulinoma. 21 On the other hand, the cerebellum has been considered to be less frequently affected than the cerebrum in hypoglycemia, 1 but others have described that cerebellar Purkinje cells are also vulnerable to hypoglycemia. 5 It has been reported that minimal loss of Purkinje cells is found in animals and humans with hypoglycemia, whereas granular cells are diffusely affected in humans with hypoglycemia. 8 There have been to date few reports of immunohistochemical examination of Purkinje cells in animals and humans with hypoglycemia.
Inositol 1,4,5-triphosphate (InsP3) is a well-known second messenger that initiates the release of calcium ions from intracellular storage sites. 2 The InsP3 receptor (InsP3R) is identical to cerebellar Purkinje cell–enriched glycoprotein P400 (P400/InsP3R). 12–15 Immunohistochemical analysis at the electron microscopic level demonstrated that P400/InsP3R is localized to the endoplasmic reticulum, at the plasma membrane, and at the postsynaptic density of Purkinje cells. 12 The receptor has been considered to be directly involved in calcium release from the endoplasmic reticulum and in calcium influx through the plasma membrane of Purkinje cells. The increase in the cytoplasmic concentration of calcium ions in the dendrites of Purkinje cells is necessary for long-term depression to occur. 9 Long-term depression is thought to be the physiologic basis for motor learning in the cerebellum. 6,7 Therefore, P400/InsP3R has been considered to be a characteristic functional marker of cerebellar Purkinje cells. 12–15
Glutamate may be a main neurotransmitter in the synaptic cleft between parallel fibers and the dendrites of Purkinje cells. 4 Extracellular levels of glutamate are regulated by non-vesicular transporting proteins (glutamate transporter) that reduce the concentration of glutamate at the synaptic cleft. 18 GLAST is a glutamate transporter enriched in cerebellar Bergmann glia and is considered to play an important role in the metabolism of glutamate in the synaptic cleft between parallel fibers and the dendrites of Purkinje cells. 4 The present study described morphologic abnormalities of Purkinje cells in two dogs with hypoglycemia during a short or long period, using a monoclonal antibody against P400/InsP3R. Furthermore, we carried out an immunohistochemical examination on canine GLAST and discussed the possible relationship between its expression and P400/InsP3R immunolabeling.
In the case of dog No. 1, a 5-year-old female Collie showed sudden ataxia, excessive salivation, vomiting, hind-limb weakness, dullness, and subsequent recumbency with paddling movements of the limbs. The basal blood glucose concentration was 35 mg/dl. Glucose loading (glucose 500 mg/kg body weight) produced blood glucose concentrations of 320, 13, 6, and 40 mg/dl after 5, 25, 60, and 120 minutes, respectively. The blood insulin concentration was 70.0 µU/ml (normal range in our laboratory is 10–25 µU/ml). On day 6 after the onset of the clinical signs, the animal exhibited mild seizures and was euthanatized by an intravenous overdose of barbiturate because of poor prognosis. 21 In the case of dog No. 2, a 12-year-old castrated male Maltese showed sudden ataxia, muscular weakness and gradually dullness and blindness. The basal blood glucose was 20 mg/dl. Glucose loading (glucose 500 mg/kg body weight) produced blood glucose concentrations of 280, 11, 5, and 38 mg/dl after 5, 25, 60, and 120 minutes, respectively. The blood insulin concentration was 29.7 µU/ml (normal range in our laboratory is 10–25 µU/ml). On day 45 after the onset of the clinical signs, the animal developed seizures that progressed over a month and then developed coma until death. A complete necropsy was done immediately after death.
Complete postmortem examinations were performed on both dogs. The tissues were immediately immersed in 10% formalin. After fixation, the tissues were dehydrated in a graded series of ethanol and embedded in paraffin. In addition to routine histopathologic examination of the tissues, sagittal and transverse sections of the brain were serially cut at 4 µm from the cerebellar vermis and hemisphere. The sections were stained with hematoxylin and eosin (HE), and other sections were used for immunohistochemical analysis using the labeled streptavidin procedure (Dako, Glostrup, Denmark). Control tissues consisted of cerebellar vermis and hemisphere from six dogs (2.4–15 years old) with no clinical neurologic signs. Postmortem diagnoses for control cases were adenocarcinoma of mammary gland, cutaneous mast cell tumor, and no significant changes (control cases for other studies). We considered that they had conditions that did not affect the central nervous system. Primary antibodies used in this study were a rat monoclonal antibody to the InsP3R (P400/InsP3R) from mouse cerebellum, 13 a mouse monoclonal antibody against human glial fibrillary acid protein (GFAP) (Dako, dilution 1 : 300), and a rabbit polyclonal antibody against canine GLAST. 19 Endogenous peroxidase was quenched by incubating the slides in 0.3% hydrogen peroxide in phosphate-buffered saline (PBS) for 15 minutes. After rinsing in PBS for 15 minutes, sections were incubated with diluted normal serum for 30 minutes at room temperature. Primary antibodies diluted in PBS were applied, and sections were incubated overnight at 4 C. Negative-control sections were incubated with PBS. After rinsing in PBS for 10 minutes, biotinylated goat serum anti-rat, -mouse, and -rabbit secondary antibodies for each primary antibody were applied to both test and negative-control slides, and sections were incubated for 30 minutes at room temperature. Sections were washed in PBS for 10 minutes and incubated with streptavidin–biotin complex for 30 minutes at room temperature. After washing with PBS for 10 minutes, the reaction was revealed using diaminobenzidine tetrahydrochloride as the peroxidase substrate.
In dog No. 1, one reddish white mass, 3 cm in diameter, was found at the caudal edge of the pancreas, and another such mass, 10 cm in diameter, in the omentum as previously described. 21 There were no gross pathologic changes in the brain except for a moderate degree of ventricular enlargement. In dog No. 2, one reddish white mass, 2 cm in diameter, was observed at the middle part of the pancreas. The cerebrum showed laminar softening to a varying degree in the cortex at the level of the frontal lobe to occipital lobe, whereas no gross pathologic changes were apparent in other regions of the brain.
Histopathologic examination on systemic organs revealed pancreatic islet beta-cell carcinoma, which was confirmed by insulin immunohistochemistry in these two cases (not shown).
In control cases, Purkinje cells of six normal cerebellums were specifically stained by the antibody against P400/InsP3R (Fig. 1). Staining of the cell bodies, dendrites, and axons was readily seen, and primary, secondary, and tertiary dendrites, as well as spiny branchlets, were clearly demonstrable (Fig. 2). The glial cells, myelin, blood vessels, leptomeninges, and other neuronal cells did not react with the P400 antibody. There were a few GFAP immunoreactive Bergmann glial cell processes and astrocytes in the white matter (not shown).
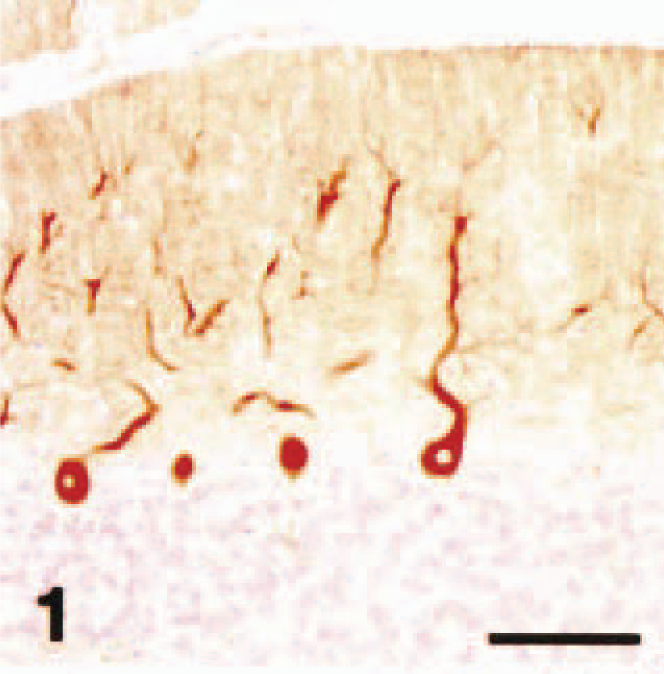
Cerebellar vermis; dog (control). P400/IP3R immunohistochemistry. Normal cerebellar Purkinje cells were intensely stained with the antibody. Streptavidin–biotin complex method, Mayer's hematoxylin counterstain. Bar = 100 µm.
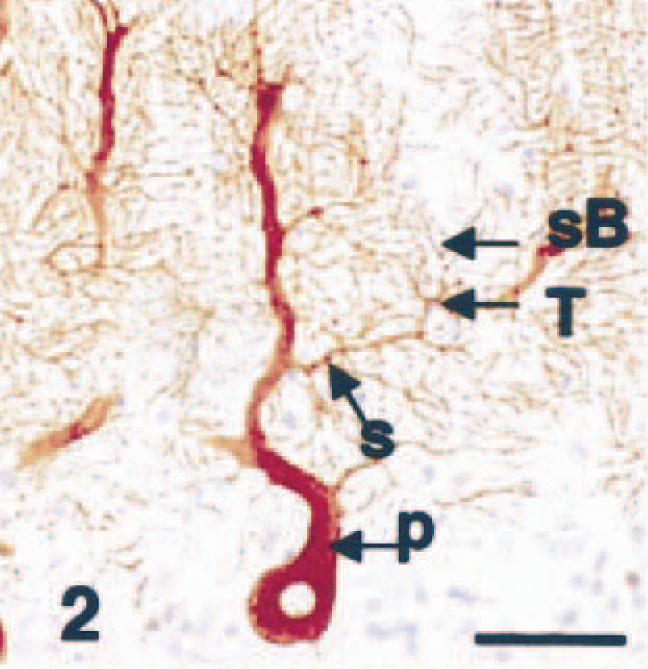
Cerebellar vermis; dog (control). P400/IP3R immunohistochemistry (IHC). Primary (P), secondary (S), and tertiary (T) dendrites as well as spiny branchlets (sB) are clearly stained. Streptavidin–biotin complex method, Mayer's hematoxylin counterstain. Bar = 50 µm.
In dog No. 1, acute neuronal necrosis was found exclusively in the superficial layers of the cerebral cortex, and spongy change was found in the dentate gyrus of the hippocampus. 21 In the cerebellum, there were no pathologic changes visible by HE stain including loss or necrosis of Purkinje cells or granule cells, proliferation of Bergmann glial cells, or astrogliosis. In the cerebellar neocortex, Purkinje cells that reacted with the anti-P400/InsP3R antibody did not have the same staining intensity as that seen in the control dogs. Among the P400/InsP3R-positive Purkinje cells, some Purkinje cell bodies and almost all the primary, secondary, and tertiary dendrites, as well as spiny branchlets, were devoid of immunoreactivity to varying degrees (Fig. 3), whereas the immunostaining of tertiary and spiny branchlets beneath the piamater was relatively preserved (Fig. 4). The immunoreactivity of Purkinje cells from cell bodies to spiny branchlets was well preserved in caudal lobules of vermis (Fig. 5). GLAST immunohistochemistry demonstrated a mild decrease in immunoreactivity in comparison with control cases (not shown). GFAP immunohistochemistry showed a mild increase in number of positive astrocytes in the granule cell layer (not shown).
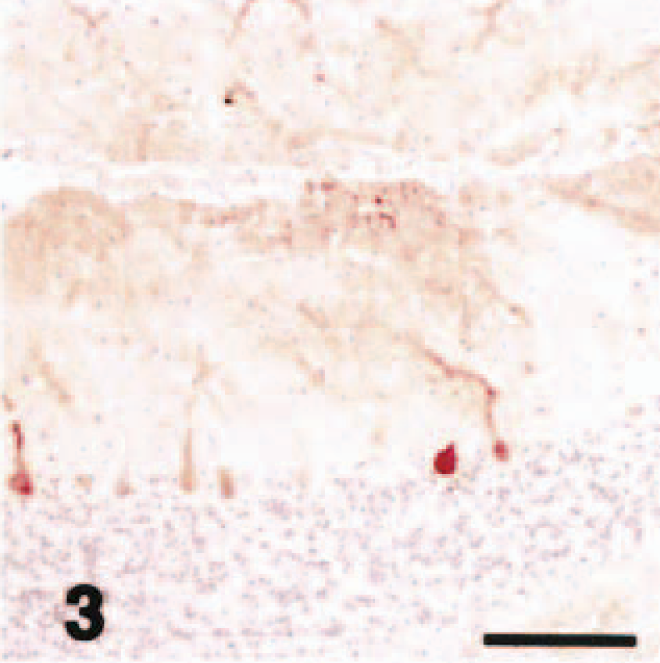
Cerebellar vermis; acute hypoglycemia case; dog. P400/IP3R immunohistochemistry. Some Purkinje cell bodies and almost all the primary, secondary, and tertiary dendrites as well as spiny branchlets were devoid of immunoreactivity to various degrees. Streptavidin–biotin complex method, Mayer's hematoxylin counterstain. Bar = 100 µm.
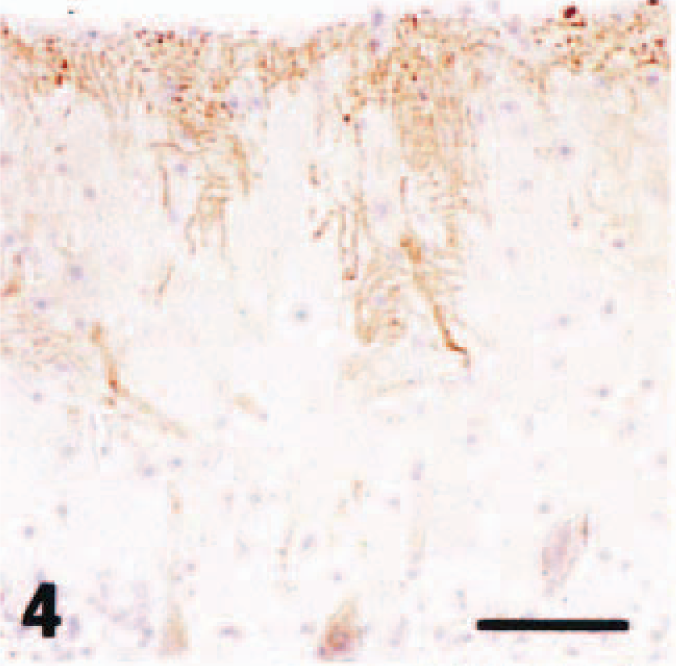
Cerebellar vermis; acute hypoglycemia case; dog. P400/IP3R immunohistochemistry. Immunostaining of tertiary and spiny branchlets beneath the piamater (arrowheads) was relatively preserved. Streptavidin–biotin complex method, Mayer's hematoxylin counterstain. Bar = 50 µm.
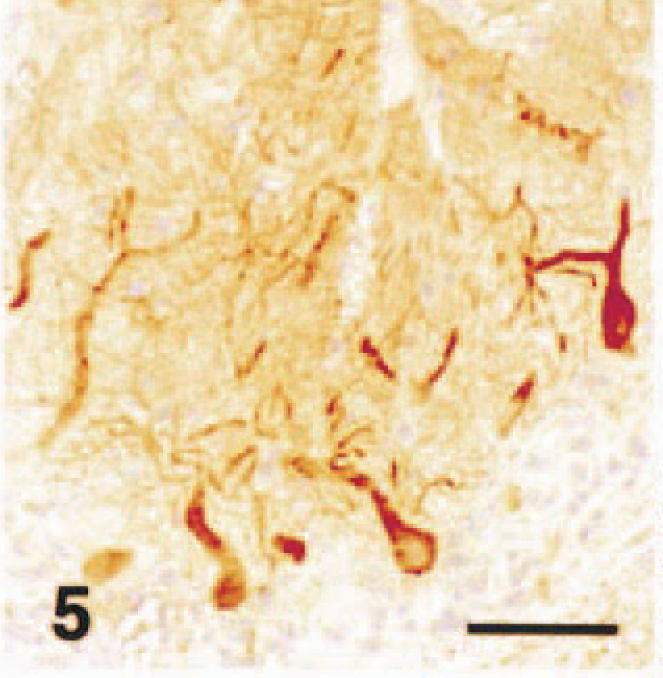
Cerebellar vermis; acute hypoglycemia case; dog. P400/IP3R immunohistochemistry. Immunostaining of Purkinje cells from cell bodies to spiny branchlets was well preserved in caudal lobules. Streptavidin–biotin complex method, Mayer's hematoxylin counterstain. Bar = 50 µm.
In dog No. 2, cerebrocortical laminar necrosis was found in the superficial to middle layers, and lesions consisted of neuronal cell necrosis, marked infiltration of foamy macrophages, marked gliosis (gemistocytic astrocytes), hypertrophy of vascular endothelial cells, and vacuolation in neurophil. In the most severely affected areas, necrotic foci extended to deep layer of the cerebral cortex, and the hippocampus was also involved. In the cerebellum, acute necrosis and patchy loss of Purkinje cells, accompanied by loss of basket cell soma and their descending axons (“baskets”), were occasionally seen. There was proliferation of Bergmann glial cells and mild loss of granule cells. Almost all the secondary and tertiary dendrites as well as spiny branchlets showed no reaction for P400/InsP3R antibody with or without morphologic changes by HE stain in all the lobules including caudal lobules (Fig. 6). The molecular layer showed a weak fine-granular immunoreactivity for GLAST antibody (Fig. 7). GLAST immunohistochemistry revealed a marked decrease in immunoreactivity in comparison with control cases (Fig. 8). GFAP immunohistochemistry showed a moderate increase in the number of positive Bergmann glial processes in the molecular layer and astrocytes in the granule cell layer (not shown).
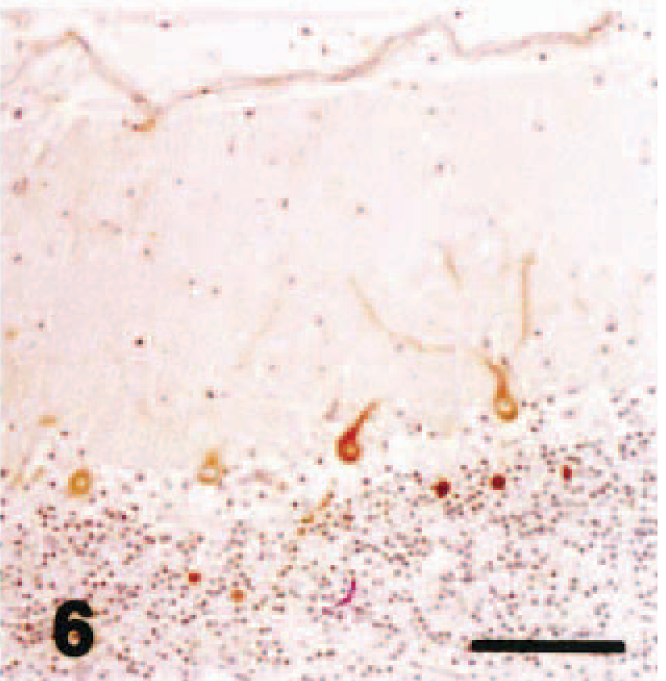
Cerebellar vermis; chronic hypoglycemia case; dog. P400/IP3R immunohistochemistry. Almost all the secondary and tertiary dendrites as well as spiny branchlets showed no reaction for P400/InsP3R antibody. Streptavidin–biotin complex method, Mayer's hematoxylin counterstain. Bar = 100 µm.
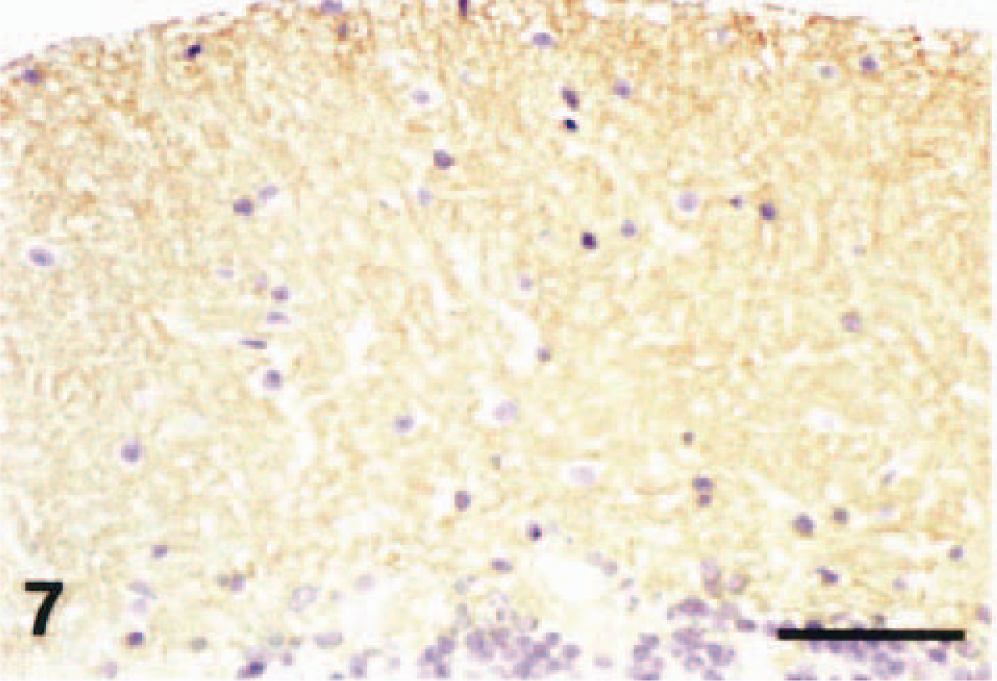
Cerebellar vermis; dog (control). GLAST immunohistochemistry. Note a weak fine-granular immunoreactivity for the antibody in the molecular layer. Streptavidin–biotin complex method, Mayer's hematoxylin counterstain. Bar = 50 µm.
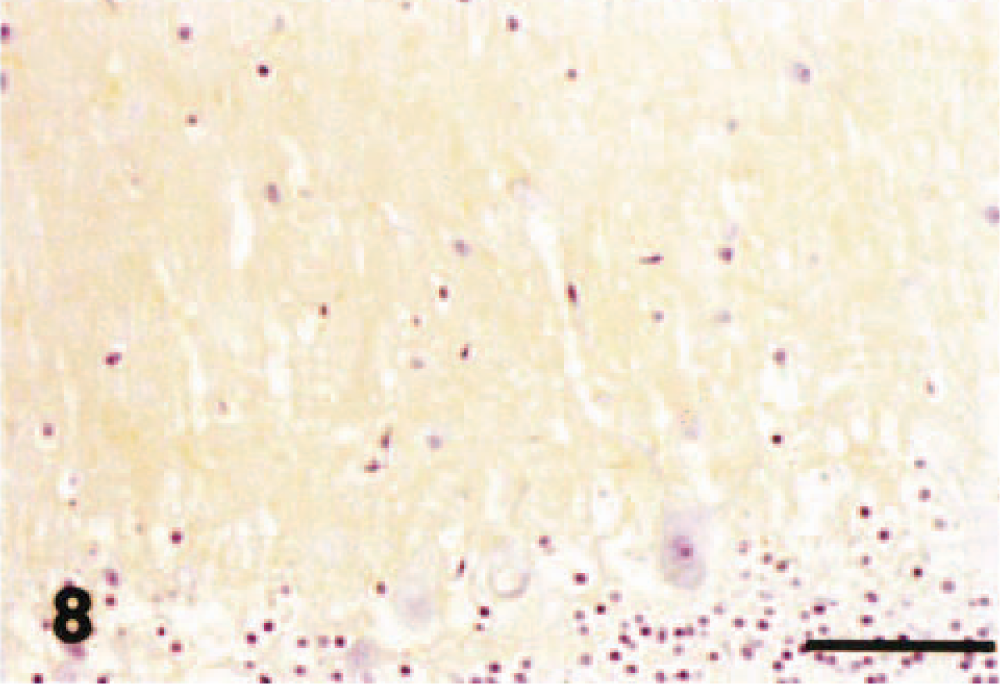
Cerebellar vermis; chronic hypoglycemia case; dog. GLAST immunohistochemistry. Note a decrease in GLAST immunoreactivity compared with control. Streptavidin–biotin complex method, Mayer's hematoxylin counterstain. Bar = 50 µm.
Based on the clinical course and pathologic findings of the cerebrum, we classified dog No. 1 as an acute case with hypoglycemic brain damage and classified dog No. 2 as a chronic case also with hypoglycemic brain damage. Immunohistochemical findings of hypoglycemic Purkinje cells observed in this study are summarized as follows: 1) Purkinje cells, which appeared to be morphologically intact by HE stain, showed a decrease in P400/InsP3R immunoreactivity of peripheral dendrites including spiny branchlets except for caudal lobules in dog No. 1 (acute case); 2) the hypoglycemic Purkinje cells were devoid of P400/InsP3R expression in almost all the cell bodies to peripheral branchlets in dog No. 2 (chronic case); 3) GLAST immunoreactivity apparently decreased, especially in dog No. 2 (chronic case).
The spiny branchlets are the main fields that receive the inputs originating from axon terminals of parallel fibers, 3 and P400/InsP3R has been considered to play an important role in the regulation of calcium ions in cytoplasm of cerebellar Purkinje cells. 12–15 Therefore, the results suggest that hypoglycemia reduces P400 expression, and that regulation of calcium ions by P400/InsP3R may be at least impaired, leading to disturbance of long-term depression (physiologic basis for motor learning) and thereby to the appearance of ataxia that was present in both cases of hypoglycemia in this study, which is a common clinical sign associated with hypoglycemia in animals and humans.
In the acute case, the P400/InsP3R immunostaining of tertiary and spiny branchlets beneath the piamater was relatively preserved. In human cerebellar degeneration, such as Menke's kinky hair disease, 11 olivopontocerebellar atrophy (OPCA), and granule cell–type cerebellar degeneration, 10 the disappearance of P400/InsP3R immunoreactivity starts from the peripheral portions of the Purkinje cell dendrites and extends to the cell body. Similar immunohistochemical features in Purkinje cell spot 35 protein (Ca-binding protein specific for Purkinje cells 23 ) expression have been reported in OPCA and hereditary cerebellar atrophy of Holmes type. 16 In these human diseases, there are few reports describing preserved P400/InsP3R immunoreactivity beneath the piamater as seen in the dog with acute hypoglycemia studied here. Therefore, this finding may be a characteristic change that is related to a decreased level of glucose metabolism in Purkinje cells due to hypoglycemia.
In the dog with chronic hypoglycemia studied here, almost all the remaining Purkinje cells lacked P400/InsP3R staining in the cell bodies and dendrites. On the other hand, in the acute case, the immunoreactivity of Purkinje cells from cell bodies to spiny branchlets tended to be well preserved in caudal lobules, which are classified in archicerebellum. 3 Caudal lobules in cerebellar vermis have been considered to be most preserved in cerebellar degeneration of animals and humans. 17,22 Therefore, Purkinje cells in caudal lobules may be relatively less affected in the early stage of hypoglycemic conditions as well as in other degenerative disorders. The variability in P400/InsP3R staining between the archicerebellum and other areas of the cerebellar vermis would prompt us to consider that the immunohistochemical staining intensity was related to hypoglycemia and not to technical differences such as postmortem interval and storage conditions.
GLAST is a glutamate transporter enriched in cerebellar Bergmann glia and is considered to play an important role in the metabolism of glutamate in the synaptic cleft between parallel fibers and Purkinje cells dendrites. 4 The reduced GLAST immunoreactivity in the present hypoglycemic cerebellums might reflect a reduced concentration of glutamate in synaptic clefts between parallel fiber axons and Purkinje cell dendrites in the molecular layer. It has been reported that neurons regulate expression of glial GLAST protein; mixed cultures of neurons and astrocytes caused GLAST protein to increase. 20 In the present study, Purkinje cells showed a decrease in P400/InsP3R immunoreactivity in peripheral dendrites including spiny branchlets, therefore, the reduced immunoreactivity of P400/InsP3R might be functionally related to the decreased expression of GLAST protein.
