Abstract
Avian chlamydiosis is a common disease found in domesticated and nondomesticated avian species caused by several species of chlamydiae including but not limited to Chlamydia psittaci, Chlamydia avium, Chlamydia gallinacea, Chlamydia buteonis, and Chlamydia ibidis. Generally, early in the disease course, birds present with mild nonspecific clinical signs associated with gastrointestinal and respiratory tract disease. During end-stage disease, birds may present in a severe state of emaciation, dehydration, and/or acute death with no known history of prior illness. Between 2000 and 2009, 14 unusual cases of avian chlamydiosis were submitted to the California Animal Health and Food Safety Laboratory System. Histologic lesions noted in the 14 birds included meningoencephalomyelitis (3 of 13, 23%), otitis media (3 of 8), bursitis (9 of 11, 81%), nephritis (8 of 13, 61%), and orchitis (1 of 8). Corresponding immunopositive chlamydiae intracytoplasmic inclusions were detected in all tissues. Positive immunolabeling was detected in optic nerves (5 of 10, 50%), meninges (5 of 13, 38%), and endothelial cells (14 of 14, 100%) in the absence of significant microscopic lesions. This study highlights unusual gross, histological, and immunohistochemical findings of chlamydiosis in psittacines and highlights the importance of a thorough diagnostic approach when confirming or excluding chlamydiosis in psittacine birds.
Avian chlamydiosis is a highly contagious systemic disease originally believed to be caused by the bacterium Chlamydia psittaci. Recent studies have identified several closely related species of Chlamydiaceae, which cause similar clinical disease to C. psittaci in infected birds including Chlamydia avium, Chlamydia gallinacean, Chlamydia ibidis, and Chlamydia buteonis. C. ibidis and C. buteonis are host specific and affect the African sacred ibis, and Accipitridae and Strigidae raptor families, respectively.11,13,16,19,22
Chlamydia is a genus belonging to the family Chlamydiaceae and encompasses 14 species including C. psittaci, C. avium, C. abortus, C. muridarum, C. suis, C. pecorum, C. trachomatis, C. felis, C. caviae, C. pneumoniae, C. gallinacea, C. ibidis, C. buteonis, and C. poikilothermis.10,13,19,22,24 Chlamydia are intracellular Gram-negative bacteria that maintain their life cycle in the host cell within a nonacidified vacuole called an inclusion. Unlike other bacteria that solely rely on the host cell’s cytoplasm for free nutrient access, chlamydia import host nutrients across the inclusion membrane.1,10,23,24 Traditionally, the life cycle of chlamydia was thought to involve three distinct morphological forms, specifically, the elementary body (EB), reticulate body (RB), and intermediate body. The EB is the infectious form that enters the host cell and converts to the RB, which is the intracellular metabolically active form. RBs replicate by binary fission, maturing into new EBs.10,17,20,21,23,24 In recent in vivo studies, RBs have been found to undergo stress-induced persistence, also known as “Chlamydia persistence” or “Chlamydia stress response.” 15 Chlamydia persistence allows chlamydia to evade stressful conditions (eg, nutrient deprivation, antimicrobial therapy) and persist within the host in a quiescent state as aberrant RBs. 15 The aberrant RBs prioritize cell functions to prepare for survival and will resume normal replication once the stressor is removed. 15 The process of survival requires RBs to remain within the host cell in a noninfective but viable aberrant form. After the cessation of the stressor, aberrant RBs will become active, reinitiating the life cycle with EB formation and reinfection of the host. 15
Historically, chlamydiosis in psittacines (psittacosis) was associated with infection with C. psittaci. The advent of molecular diagnostics and genomic sequencing has uncovered more species (eg, C. avium) within the Chlamydiaceae which induce clinical disease that is similar to psittacosis due to C. psittaci infection.11,13,16,18,19 C. psittaci genotypes A, B, C, D, E, F, and few reports of E/B have been documented in birds. Genotypes A, B, and E/B have been implicated in zoonotic disease.10,19,24 All avian genotypes should be considered to have the potential to cause disease in humans. 18 Serotype E is often associated with sporadic outbreaks in poultry slaughterhouses, primarily related to humans handling turkeys, ducks, and rarely chickens.7,21 Transmission most often occurs via respiratory secretions and occurs more frequently than reported due to asymptomatic infectious birds. Intermittent intestinal shedding through fecal material may also occur, resulting in the contamination of water, feedstuff, and equipment.10,26
Chlamydiosis in birds is associated with various clinical manifestations, including gastrointestinal signs (eg, greenish-yellow diarrhea), upper and lower respiratory signs, lethargy, ocular discharge, emaciation, dehydration, and rarely neurological signs.9,10,14,15 Lesions frequently observed after the commencement of clinical signs include airsacculitis, hepatitis, splenitis, pericarditis, and pneumonia.9,10,14,20 Diagnosis may often involve routine histopathology, serology, cytological examination of impression smears, immunofluorescence antibody (IFA), immunohistochemistry (IHC), and/or polymerase chain reaction (PCR). 23
This article describes atypical gross, histologic, and IHC findings in a select group of psittacine birds diagnosed with chlamydial infection over a period of 9 years.
Materials and Methods
Case Selection and Histology
This study was a retrospective study using archived postmortem reports and formalin-fixed paraffin-embedded (FFPE) tissues from multiple organs of psittacine birds with IHC-confirmed chlamydial infection. Psittacine bird postmortem reports finalized at the Fresno and Tulare branches of the California Animal Health and Food Safety Laboratory System during 2000–2009 were assessed for gross lesions and histologic findings that were considered atypical for chlamydia infection. Psittacine birds included in the study were required to have atypical lesions in addition to typical lesions such as hepatomegaly (hepatitis), splenomegaly (splenitis), renomegaly (nephritis), and airsacculitis. Fourteen psittacine birds including eight different species were included in the study. Affected species included cockatiel (Nymphicus hollandicus), double-yellow headed Amazon (Amazona oratrix), green-winged macaw (Ara chloropterus), blue-headed pionus (Pionus menstruus), sun conure (Aratinga solstitialis), hyacinth macaw (Anodorhynchus hyacinthinus), rosella (Platycercus spp), and umbrella cockatoo (Cacatua alba) (Table 1). From these cases, data were compiled into a spreadsheet (Excel 2016, Microsoft) detailing signalment (species, age, and sex), clinical history, gross summaries, ancillary testing, and histologic and immunohistochemical results. For light microscopic examination, tissues were fixed in 10% formalin and processed routinely for paraffin embedding. Paraffin-embedded tissue samples were sectioned into 4-µm-thick sections and stained with hematoxylin and eosin. Select cases were further stained with Giemsa stain using the Fresno and Tulare branches of the California Animal Health and Food Safety Laboratory System standard operating procedures. Histologic samples were evaluated and recorded by a pathologist. Select cases were chosen to undergo further diagnostics including bacteriology, reverse transcription (RT)-PCR, and fluorescent antibody test. The ancillary tests were performed at the Fresno and Tulare branches of the California Animal Health and Food Safety Laboratory System in accordance with methodologies described in Isolation and Identification of Avian Pathogens. 6
Signalment of 14 psittacine birds with chlamydiosis.
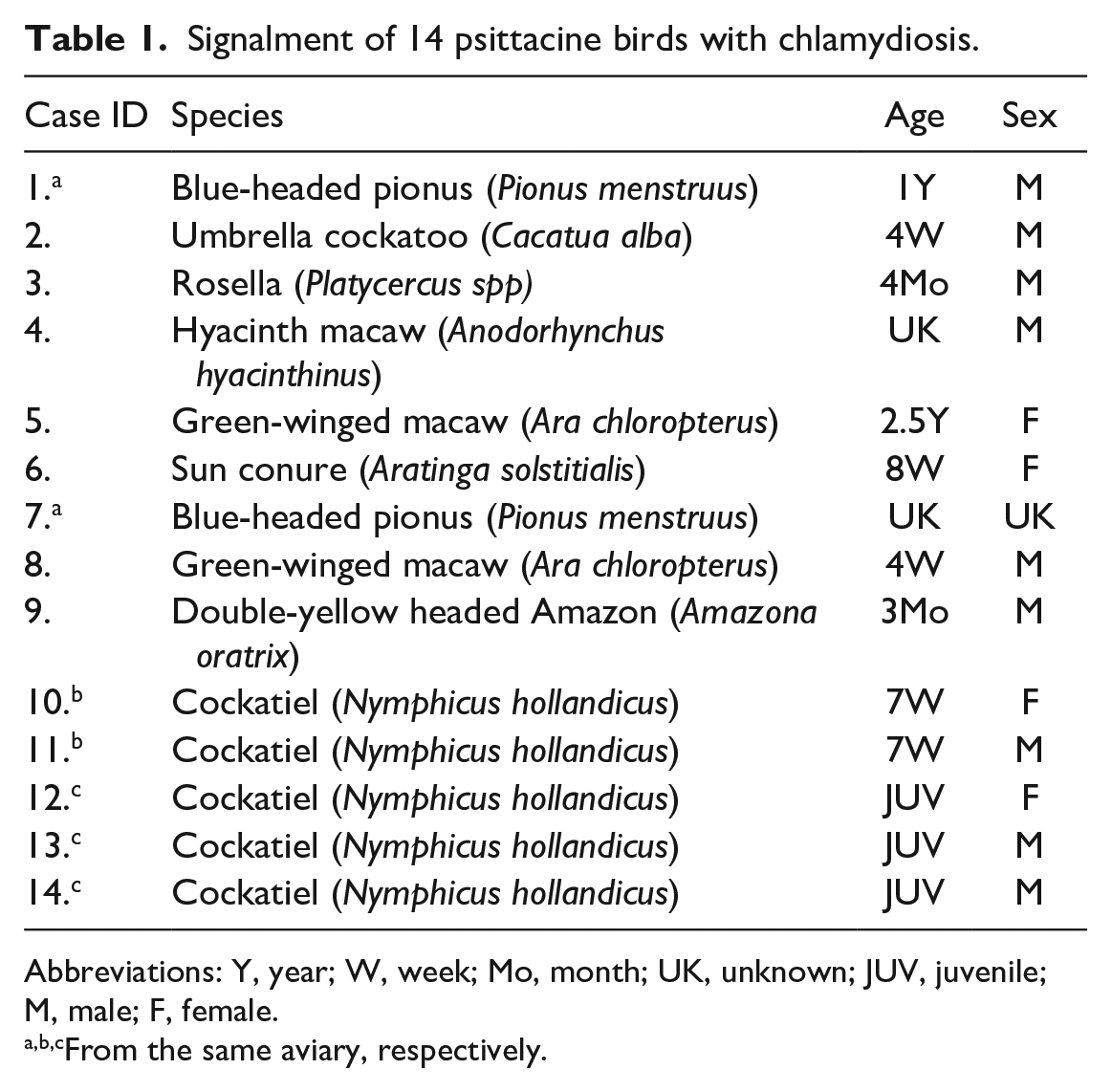
Abbreviations: Y, year; W, week; Mo, month; UK, unknown; JUV, juvenile; M, male; F, female.
From the same aviary, respectively.
Immunohistochemistry
In all cases (14 of 14), FFPE tissues from multiple organs not originally assessed with IHC at the time of diagnosis were identified. The FFPE tissues were sectioned at 4 µm and mounted on charged slides already containing liver or spleen positive control tissue. Slides were deparaffinized and rehydrated using xylene and graded alcohols. Endogenous peroxidase activity was quenched with 3% hydrogen peroxide in methanol. The slides were treated with acidulated pepsin at 37°C for 15 minutes, rinsed in deionized water, and placed in tris-buffered saline (TBS)-Tween. All further incubations were at room temperature, and all rinses were with TBS-Tween. An anti-C. trachomatis lipopolysaccharide IgG1 mouse monoclonal antibody (ViroStat, 1681) specific to C. trachomatis, C. pneumoniae, and C. psittaci, diluted 1:50 with 1% casein in TBS-Tween, was applied for 30 minutes; detected with a horseradish peroxidase–conjugated anti-mouse polymer (DakoCytomation, K4001) for 30 minutes, and visualized by incubating with 3-amino-9-ethylcarbazole (AEC) (Dako, 3464) for 10 minutes. The sections were counterstained with Mayer’s hematoxylin and coverslipped with an aqueous mounting media. Nonspecific staining was evaluated with a duplicate slide receiving normal mouse whole IgG in place of the chlamydia-specific primary antibody.
Results
Clinical History
Thirteen of the 14 birds were from medium to large aviaries containing greater than 20 birds of varying species, cases 1, 2, 3, 4, 5, 6, 7, 8, 10, 11, 12, 13, and 14. Common antemortem clinical signs included diarrhea, weakness, and increased morbidity/mortality in the aviaries. Cases 12, 13, and 14 were noted to have weight loss, nondefined upper respiratory tract clinical signs, nondefined ocular changes, weakness, and lethargy (Table 2). Case 6 was from a large aviary with increased mortality/morbidity. The affected birds exhibited hematochezia without diarrhea (Table 2). Malodorous diarrhea was reported in cases 3, 8, 10, 11, 12, 13, and 14 (Table 2). Sudden death was reported in cases 1, 2, 4, 5, 7, 8, 9, 10, and 11. Cases 4 and 7 exhibited no clinical signs prior to death. Case 7 was submitted with limited clinical history.
Clinical history and gross findings of 14 psittacine birds with chlamydiosis.
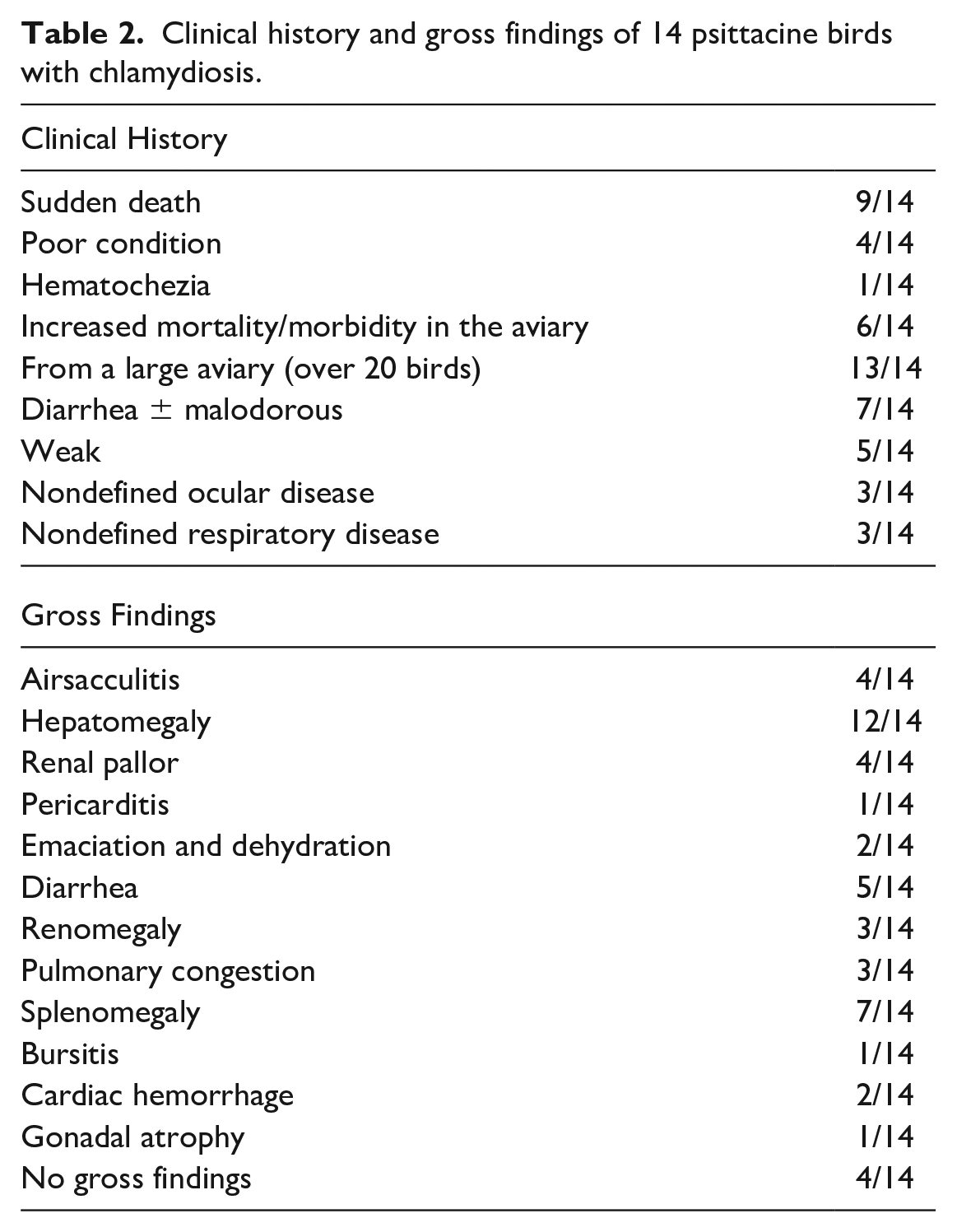
Gross Findings
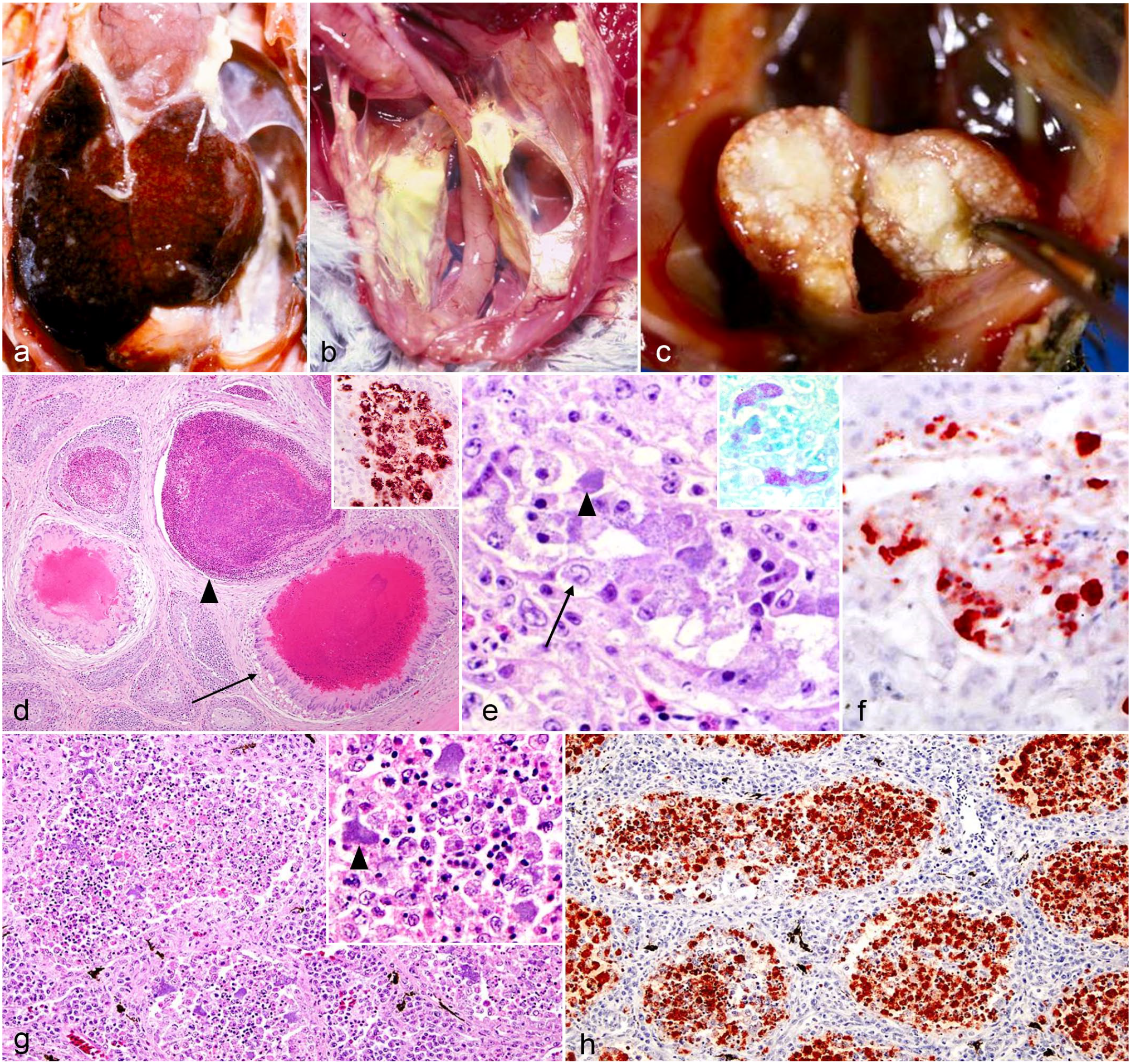
Eleven of the 14 cases were submitted as entire carcasses to the California Animal Health and Food Safety Laboratory System in Fresno or Tulare. Three of the 14 cases (cases 1, 2, and 4) arrived as external necropsies and were submitted with limited description of the gross findings. Postmortem findings were typical of chlamydial infection in most cases and included hepatomegaly (12 of 14), splenomegaly (7 of 14), and airsacculitis (4 of 14) (Table 2). Hepatomegaly was characterized by grossly enlarged livers encompassing and expanding the coelomic cavity. Liver margins were rounded and the parenchyma was often mottled red brown with regions of pallor and, in some instances, discolored yellow. Gross changes were consistent with those observed in the African grey parrot in Fig. 1a not included in this study. Cases with yellow livers were more frequently noted to be emaciated. Renomegaly was noted in cases 4, 5, and 9. However, renal pallor without evidence of renomegaly was found in cases 6 and 14 (Table 2). Airsacculitis was described in cases 4, 6, and 9. In these cases, air sacs were thickened by yellow-white, thick, friable exudate that extended into the thoracic and abdominal air sacs (Fig. 1b). Several cases exhibited unusual gross lesions infrequently reported in association with chlamydial infection. In case 8, there was enlargement of the bursa of Fabricius characterized by loss of normal bursal tissue with replacement by mottled white-tan soft tissue (Fig. 1c). Other unusual findings included gonadal atrophy (case 4), epicardial hemorrhage (cases 1 and 6), and pulmonary hemorrhage and congestion (cases 1, 2, and 8) (Table 2). Case 7 had no abnormal gross findings.
Histopathologic Findings
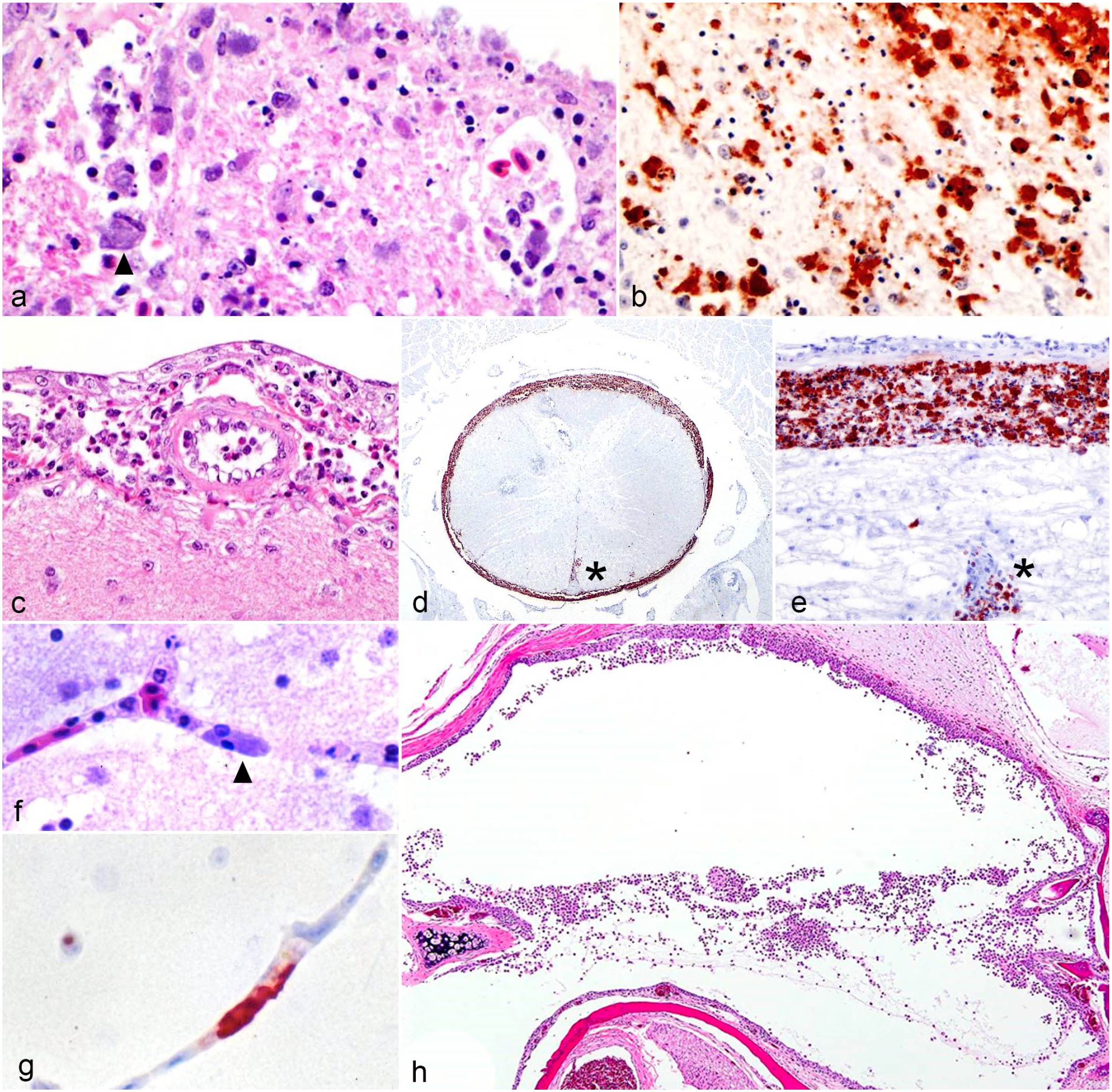
The 14 cases were initially evaluated by different pathologists from the Fresno and Tulare branches of the California Animal Health and Food Safety Laboratory System. Varied postmortem procedural techniques resulted in a lack of tissue standardization in most cases. Twelve of the 14 cases exhibited and/or were documented to have characteristic lesions associated with chlamydial infection including lymphohistiocytic hepatitis and splenitis with variable necrosis and heterophilic infiltrates (Table 3). Although airsacculitis was only noted grossly in 4 cases, 9 of the 13 cases evaluated have histological evidence of airsacculitis. In affected airsacs, there were interstitial and luminal aggregates of fibrin and infiltrates of histiocytes and heterophils. Nine of the 11 cases had histological evidence of bursitis (Tables 2, 3, Fig. 1c). Bursal changes included lymphoid depletion within medullary and cortical areas, and infiltration and replacement of bursal parenchyma by histiocytes and heterophils. Heterophilic infiltrates and fibrin were noted in the center of follicles. Histiocytes throughout the bursal lesions contained numerous amphophilic intracytoplasmic inclusion bodies (ICIBs) (Fig. 1d). Eight of the 13 cases had histological evidence of nephritis. Of these cases, only cases 4, 6, and 9 had gross changes described in the kidneys (Table 2). Microscopic renal changes were characterized by loss and replacement of nephrons by infiltrating plasma cells, lymphocytes, and macrophages. Remaining renal tubular epithelium contained multifocal necrosis with degenerate cells sloughing into the lumen (Fig. 1e). Multifocally, within the inflamed parenchyma, epithelial cells and macrophages contained numerous amphophilic ICIBs. The inclusions were highlighted by Giemsa staining (Fig.1e, inset). Orchitis was present in a single case (case 3) with multifocal lymphohistiocytic and heterophilic inflammation within the interstitium and seminiferous tubules (Fig. 1g; Table 3). Microscopic lesions were noted in the cerebrum, meninges, and optic nerve in select cases (Table 3, Fig. 2a, c). Cases 3, 4, and 6 had evidence of meningoencephalitis or meningoencephalomyelitis, and in cases 3 and 4, there was also evidence of optic neuritis. Cerebral lesions in case 6 included an infiltrate of lymphocytes and gitter cells separated by cellular debris and edema. Throughout the tissue, variable numbers of gitter cells contained amphophilic ICIBs (Fig. 2a). Similar infiltrates expanded the meninges of the spinal cord in this case (Fig. 2c). Other unusual microscopic findings included serositis (1 of 13), tracheitis (1 of 13), epicarditis (8 of 13), sinusitis/rhinitis (3 of 13), adrenalitis (3 of 13), pancreatitis (2 of 13), thyroiditis (2 of 10), myositis (2 of 13), and otitis media (3 of 8) (Table 3). Otitis media in 1 case (case 13) was characterized by the presence of numerous heterophils and histiocytes that were within the middle ear and often attached to the mucoperiosteal lining, which was attenuated and occasionally eroded (Fig. 2h).
Summary of histologic and immunohistochemical findings of psittacine birds with chlamydiosis.
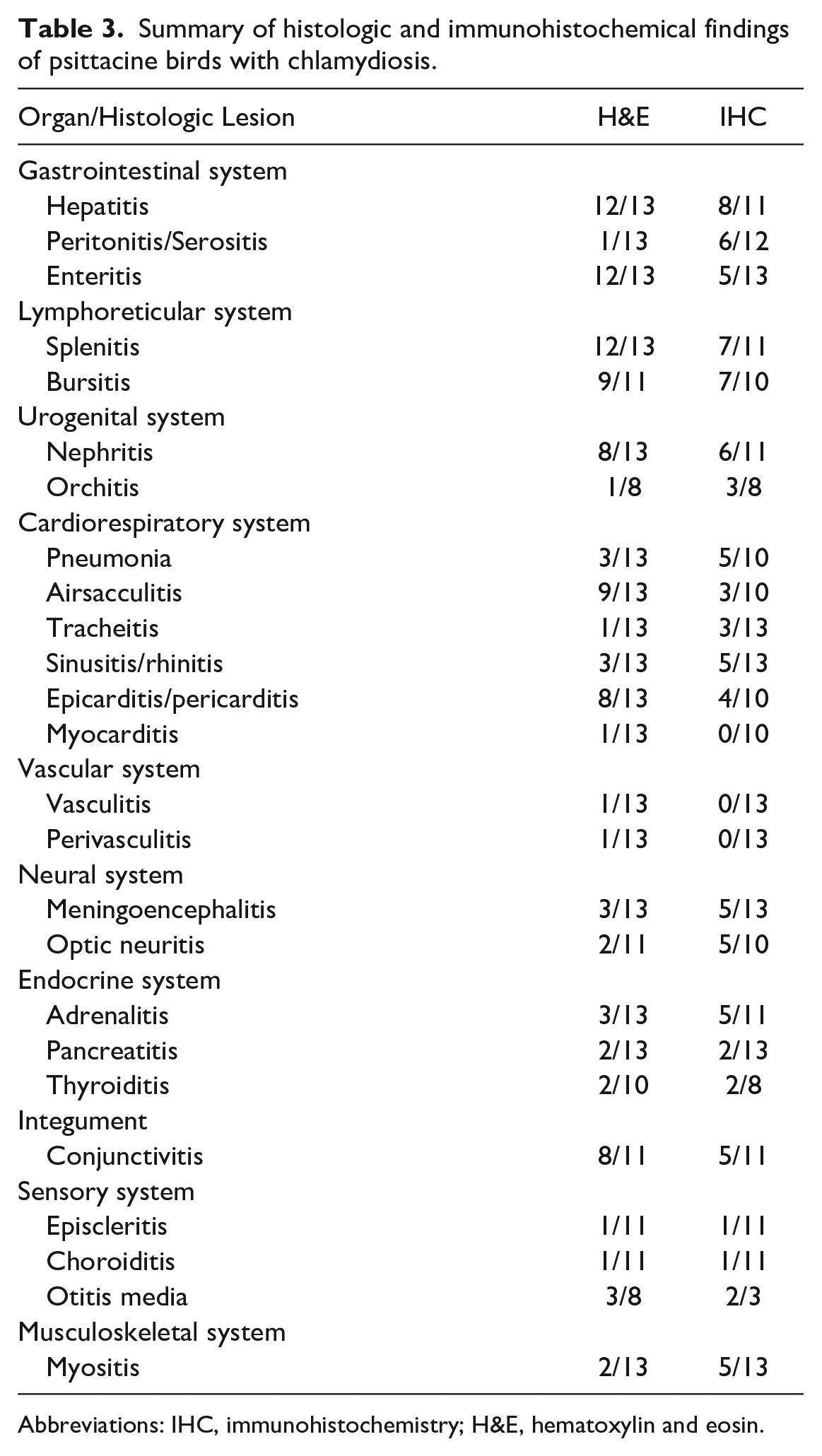
Abbreviations: IHC, immunohistochemistry; H&E, hematoxylin and eosin.
Immunohistochemical Findings
All available tissues from the initial postmortem for each case were evaluated with IHC. A detailed list of immunohistochemical findings is presented in Table 3. Eight of the 11 livers exhibited strong hepatocellular and histiocytic cytoplasmic staining for chlamydia. Livers from cases 2, 4, and 9 were not assessed with IHC. Similar findings were noted in the spleen of 7 of the 11 cases. Spleens from cases 4, 6, and 9 were not assessed with IHC. Nephritis was noted histologically in 9 of the 13 cases. Of the 9 cases, only 3 had gross renal changes, and 6 exhibited strong positive intraepithelial and intrahistiocytic positive labeling for chlamydia (Tables 2, 3 and Fig. 1f). Renal tissues from cases 4, 6, and 9 were not assessed with IHC. In 9 cases, there was no gross evidence of orchitis, and histologically, only case 3 demonstrated evidence of orchitis. Three of the 8 cases evaluated had intraepithelial and intrahistiocytic immunolabeling despite the lack of inflammation in 2 of the 3 cases (Fig. 1h).
Cerebral cortex and meninges were evaluated with IHC in 13 cases. In 5 cases (cases 1, 3, 6, 10, and 11), strong intracytoplasmic labeling for chlamydia was detected in the meninges and neuropil of the cerebrum (Fig. 2b, 2d, 2e). Cases 2, 3, 5, 6, and 12 exhibited positive endothelial and histiocytic immunolabeling despite all cases but case 3 lacking corresponding histologic lesions. A sixth case, case 4, lacked positive immunolabeling despite having microscopic lesions.
Conjunctivitis was not evident grossly within tissues evaluated; however, 8 of the 11 cases had histologic evidence of lymphohistiocytic, plasmacytic, and heterophilic conjunctivitis (cases 3, 5, 9, 10, 11, 12, 13, and 14). Only 5 of these cases were immunohistochemically positive on sections of conjunctiva (cases. 2, 3, 5, 7, and 13).
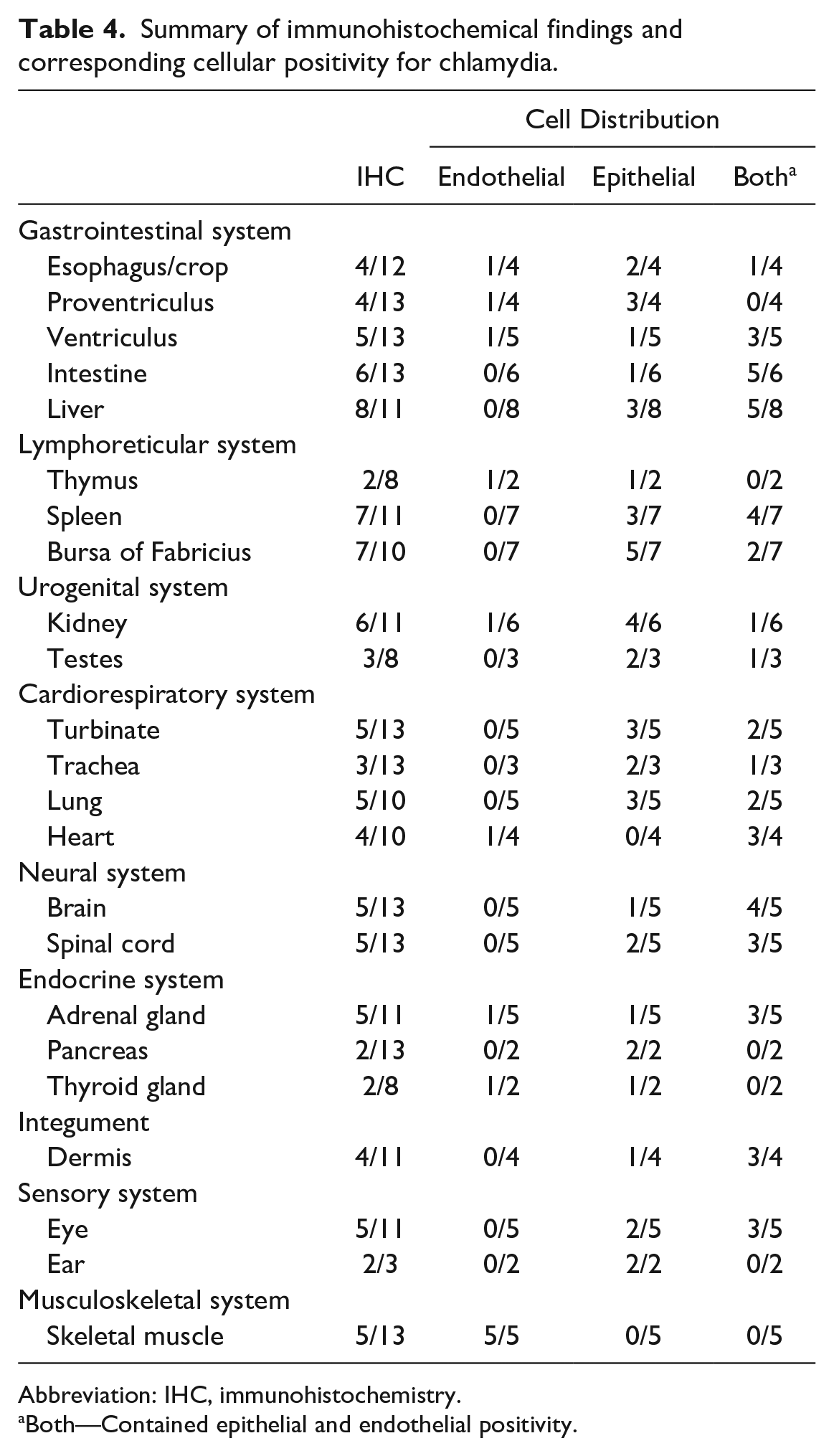
In multiple cases, amphophilic ICIBs within endothelial cells were identified with routine histologic staining (Fig 2f). Cytoplasmic IHC labeling was present in endothelial cells in skeletal muscle, and at a lower frequency in the kidney, heart, adrenal gland, thyroid gland, thymus, and gastrointestinal tract in multiple cases (Table 4). Many tissues exhibited positive immunolabeling in both endothelial and epithelial cells, including the intestine, liver, spleen, heart, brain, spinal cord, adrenal gland, dermis, and eyes (Table 4, Fig. 2g).
Summary of immunohistochemical findings and corresponding cellular positivity for chlamydia.
Abbreviation: IHC, immunohistochemistry.
Both—Contained epithelial and endothelial positivity.
Ancillary Diagnostics
Twelve of the 14 cases underwent ancillary diagnostic testing in conjunction with IHC for chlamydia. Cases 1 and 2 did not have any ancillary diagnostics performed. Bacterial culture yielded variable bacterial growth of unknown significance on a range of tissues in most cases. PCR testing for avian influenza, West Nile virus, exotic Newcastle disease, and avian paramyxovirus was performed on pharyngeal and/or cloacal swabs in several cases; all yielded negative results. 6
Chlamydia fluorescent antibody testing was performed in several cases prior to IHC. Multiple tissues yielded positive results, including liver, spleen, conjunctiva, and air sac. A comprehensive table detailing microbial and virology results can be found in Supplemental Table 1.
Discussion
This cohort of psittacine birds demonstrated unusual gross, histologic, and immunohistochemical findings that were attributed to chlamydial infection. Evaluation of the literature documenting typical lesions of chlamydia in psittacine birds highlights that these cases are unique. Generally, lesions resulting from systemic chlamydiosis in clinically unwell psittacine birds are commonly reported to affect the lower and upper respiratory tract, gastrointestinal tract, liver, and spleen.5,8,9,10,14,19,20,25 Urogenital, cardiovascular, and musculoskeletal infections were documented in a single report, but other similar changes have not been documented in the literature to the best of the authors’ knowledge. 5 A passeriform (Gouldian finch; Erythrura gouldiae) with C. psittaci was found to have histologic inflammatory changes within the nasal and Harderian glands, brain, spinal cord, kidney, and the serosa of the testes with coinciding positive immunolabeling for chlamydiae. 5 Curiously, the same finch was also determined to have coinfection with circovirus. 5
Recent studies detailing lesions associated with C. avium infection describe gross and histologic presentations similar to infections with other chlamydiae species. 25 Few reports in the literature document atypical gross and histologic lesions associated with C. avium infection. A single report detailing 2 cases of C. avium in a Bolivian military macaw (Ara militaris boliviana) and red-fronted macaw (Ara rubrogenys) described interstitial nephritis, glomerulonephritis, and myocarditis. 25
In comparison with traditional descriptions of avian chlamydiosis, our investigation revealed a range of unusual findings. Several cases had no significant immunolabeling for chlamydiae in commonly affected organs such as the liver and spleen (cases 2, 4, 7, 9, 12, 13, and 14), while several cases had histologic lesions and/or positive IHC immunolabeling in unusual organs such as the bursa of Fabricius, testes, kidney, brain, and middle ear. Ten of the 14 cases also had an increased prevalence of multiorgan and visceral endotheliotropism.
Several possible reasons for the unusual distribution of the lesions in these cases were considered. Reports of dual infection with C. avium, C. gallinacean, and C. psittaci have been documented.5,18 The lack of species specificity in the chlamydial IHC antibody used in our cases precludes determination of dual chlamydial infection.
Dual infection could explain the unusual pathology described in this cohort and may account for the unusually high degree of endotheliotropism noted in most cases. Endotheliotropism is described more frequently in cases of C. pneumoniae infection, and therefore, it is possible that coinfection could explain this finding.2,3,4
The signalment and husbandry were also considered as contributing factors in the severity and unusual lesion distribution noted in this cohort. Most cases in our study were young juvenile psittacine birds from large aviaries. Age-related infection may be associated with reduced immunity. Large aviaries have the potential for higher incidence of disease due to proximity, stocking rates, and animal movement.
The possibility of coinfection with other infectious agents was also considered. Many of the cases demonstrated bacterial growth from samples of intestine, liver, lung, and in 1 case the bursa of Fabricius. As with any intestinal bacterial culture results, careful consideration of the results and correlation with clinical findings are important. Several bacterial species cultured (eg, Escherichia coli, Enterobacter spp, Enterococcus spp) are known to be pathogenic, but also to represent part of the normal enteric microbiome; therefore, growth of such an organism may not be significant in the absence of lesions or appropriate clinical signs. The severity of lesions and wide distribution of chlamydiae within affected tissues in these cases suggest chlamydial infection was the primary cause of death. However, comorbidities such as bacterial enteritis or undetermined viral infection cannot be entirely ruled out as contributing factors to death in these cases.
Endemic environmental chlamydial infection is a possibility, especially given the vast majority of these cases were from large aviaries. Recent studies have found environmental infection may not be the result of respiratory secretions and/or fecal material, but associated with a symbiotic relationship with intracellular chlamydia infection in protists. 3 Another possibility is that the birds in the study had subclinical chlamydial infection.
As with any retrospective study, there are several historic, diagnostic, and financial limitations. Several cases were submitted with limited clinical history. Therefore, routine psittacine bird diagnostic testing was performed initially based on the presentation and gross findings. As a result, several cases lacked initial IFA tissue assessment for chlamydia. The IFA results were reported in the ancillary diagnostics; however, detailed discussion of this information was not included in this report due to low case numbers.
Currently, PCR testing for chlamydia identification is considered the gold standard diagnostic modality. However, during 2000–2009, IHC and IFA were primarily used in the diagnostic laboratory setting for the identification of chlamydial infection. The reliance on IHC to confirm chlamydiosis in these cases is a limitation of this study. The antibodies initially used in the study were thought to be specific for C. psittaci and C. pneumoniae. However, more recently, the antibodies used in this study have demonstrated positive immunolabeling in C. buteonis, a newly discovered species of chlamydia.12,11 C. avium was also recently discovered through molecular analysis; it is unclear whether the antibodies used in this study would also detect this species of chlamydia. 19 Due to the recognized limitations of IHC, the cases in this study were collectively grouped and presented as chlamydial infection. 16
In conclusion, this report describes a range of unusual lesions associated with chlamydial infection in psittacine birds. The study highlights the importance of a thorough diagnostic workup when investigating avian disease events. These findings in this cohort emphasize the importance of investigating unusual lesions in birds. Recording and describing unusual lesions associated with chlamydiosis or any other classically described avian disease is paramount as our diagnostic knowledge grows with technological advancement. The recent discovery of a number of new species of chlamydiae further supports the need to document similarities and differences in diagnostic findings in affected birds.11,13,16,19,22
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231162905 – Supplemental material for Unusual cases of chlamydiosis in psittacine birds
Supplemental material, sj-pdf-1-vet-10.1177_03009858231162905 for Unusual cases of chlamydiosis in psittacine birds by Audra L. Walsh and H. L. Shivaprasad in Veterinary Pathology
Footnotes
Acknowledgements
We thank E. J. Hurley, K. Sverlow, and the staff of the California Animal Health and Food Safety Laboratory System—Tulare and Fresno branches for their technical contributions. We also thank Dr Jason Stayt (Novavet), Mr Shae Watmore, and Dr Megan Curnow (Vetpath Laboratory Services) for comments on the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
