Abstract
In Switzerland, dicrocoeliasis is regarded as the most significant parasitic infection of llamas and alpacas. Fasciola hepatica infestation is also a problem but less common. The aim of the present retrospective study was to evaluate the lungs of New World camelids (NWCs) for evidence of arterial hypertension in association with liver changes due to liver fluke infestation. The lungs of 20 llamas and 20 alpacas with liver fluke infestation were histologically evaluated. The hematoxylin and eosin and van Gieson (VG)–elastica stains as well as immunohistology for the expression of α–smooth muscle actin (α-SMA) were used to visualize the structures of arterial walls. Parasitology of fecal matter (11 llamas and 17 alpacas) confirmed that most of these animals were infested with both Dicrocoelium dendriticum and other gastrointestinal parasites. In most cases (10/12 llamas, 4/6 alpacas), liver enzyme activity in serum was elevated. Histologically, arteries in the lungs of 9 of 20 llamas (45%) and 3 of 20 alpacas (15%) showed severe intimal and adventitial and slight to moderate medial thickening, which was confirmed with α-SMA and VG-elastica staining. All animals exhibited typical liver changes, such as fibrosis and biliary hyperplasia, in association with the presence of liver flukes. This study shows that liver flukes can induce proliferative changes in lung arteries in NWCs that resemble those seen with pulmonary arterial hypertension due to liver parasites in humans. However, the degree of liver fluke infestation was not correlated with the extent of liver damage, or with the amount of thoracic or abdominal effusion or pulmonary arterial changes.
New World camelids (NWC) have become increasingly popular in Switzerland, both as pets and for trekking. In 2009, around 5000 alpacas (Vicugna pacos) and llamas (Lama guanicoe glama) were registered in Switzerland (Federal Office for Statistic; www.statistik.admin.ch).
Parasitism in NWCs is well known and described worldwide, with lungworms (Dictyocaulus sp), intestinal parasites such as tapeworms (Monezia sp), Coccidia, Trichostrongylidae (eg, Ostertagia, Haemonchus, Trichostrongylus, and Trichuris sp), and liver flukes (Fasciola hepatica and Dicrocoelium dendriticum) as well as ectoparasites such as mites (Sarcoptes and Psoroptes sp) being reported. 1,20,,–23 In Switzerland, Trichostrongyles, Trichuris, Nematodirus, and Dicrocoelium sp are the most commonly found endoparasites. Clinical signs are mainly reported with dicrocoeliasis, which is regarded as the most relevant parasitic infection of llamas and alpacas. 14 In a recent study, 34% of examined herds were found infected with Dicrocoelium dendriticum and only 5% with F. hepatica. 14
F. hepatica infestation is a health problem for NWCs in Peru when they live at an altitude below 4000 m; above this altitude, climatic and environmental factors are inappropriate for the parasite and the intermediate snail host. 18 Animals infected with liver flukes show clinical signs, such as loss of body condition, reduced appetite, hypothermia, recumbency, and anemia. 1,7,25 F. hepatica is known to cause extensive fibrosis, bile duct proliferation and hyperplasia, granulomatous inflammation, and accumulation of large numbers of fluke eggs within affected bile ducts in the liver. 7,8,10,11 D. dendriticum induces similar liver lesions in NWCs, including abscesses, granulomas, and fibrosis, as well as bile duct proliferation. In contrast to the eggs of Fasciola, Dicrocoelium eggs are found in the liver parenchyma. 8,25
Pulmonary hypertension is well documented in humans in association with liver parasites. Around 200 million people are infected with parasites belonging to the Schistosoma sp, of whom about 4.6% to 10% have pulmonary arterial hypertension secondary to the hepatosplenic form of the disease. 5,9,20 Morphologically, this is represented by remodeling of the intima and media in lung arteries subsequent to portal hypertension. 5,9,20 Mice that are infected experimentally with Schistosoma mansoni also develop pulmonary arterial hypertension. 4
Extensive ascites and pleural effusion have been reported in 0.7% of human patients with liver cirrhosis and advanced portal hypertension, and up to 10% of patients with chronic liver disease develop hydrothorax. 16,17
Furthermore, severe medial hypertrophy of the small arterioles in the lung was observed in 11 of 28 Korean wild rats that had been experimentally infected with Capillaria hepatica and/or with Capillaria fasciolaris, indicating a strong correlation between parasitic infection and pulmonary lesion. 26
So far, 1 case of a llama with hepatic disease (macronodular fibrosis), ascites, and subsequent pulmonary hypertension with intimal fibrosis and medial hypertrophy of pulmonary arteries has been described. 21
The aim of the present study was to retrospectively examine the lungs of NWCs with liver fluke infestation for evidence of arterial hypertension and, if present, to correlate the proliferative arterial lesions in the lungs with the parasite-induced liver changes and with portal hypertension and subsequent thoracic and abdominal effusions.
Materials and Methods
Animals, Clinical Examination, and Parasitology
The study was undertaken on a total of 40 animals: 20 llamas (7 male, 4 male castrated, 9 female; 3 mo to 14 y) with dicrocoeliasis (n = 19) or D. dendriticum and F. hepatica double infection (n = 1) (Suppl. Table S1) and 20 alpacas (12 females, 6 male, 2 male castrated; 8.5 mo to 10 y) with D. dendriticum (n = 18) or F. hepatica (n = 2) infestation (Suppl. Table S2).
The animals had been selected from a total of 156 alpacas and 79 llamas that had undergone a diagnostic postmortem examination at the Vetsuisse Faculty Zurich between 2000 and 2012. For comparison, the lungs of age-matched alpacas and llamas (20 each) without liver fluke infestation (randomly selected from the database) were examined histologically (uninfected control animals). These did not show any evidence of vascular changes. Similarly, the lungs of 23 animals (11 llamas, 12 alpacas) with liver fluke infestation did not exhibit any vascular alterations (infected control animals). Accordingly, a group of 12 animals (9 llamas, 3 alpacas) with liver fluke infestation and pulmonary vascular changes remained (infected animals with lesions).
All infected animals had undergone a full clinical examination, complemented in 2 llamas and 2 alpacas by an ultrasound investigation. In 12 llamas and 6 alpacas, liver enzyme activities (γ-glutamyltransferase [GGT], sorbitol dehydrogenase [SDH], glutamate dehydrogenase [GLDH], aspartate aminotransferase [ASAT]), albumin, alkaline phosphatase (AP), and the muscle enzyme creatine phosphatase (CK) had been measured (Suppl. Table S3) and interpreted based on published reference values. 13
A parasitologic examination of fecal samples had been undertaken either antemortem (9 llamas and 14 alpacas) or postmortem (remaining animals). For the detection of F. hepatica in fecal samples, the sedimentation method was used, and for D. dendriticum as well as for coccidia and Trichostrongylidae, the sedimentation-flotation method with zinc chloride was applied. 6
Gross and Histologic Examination, Immunohistochemistry
All animals underwent a full gross postmortem examination. When present, thoracic or abdominal effusion was scored: up to 1.5 L, mild (+); approximately 2 to 3 L, moderate (++); and more than 3 L, severe (+++). Samples from lung, liver, and most other parenchymal organs were fixed in 4% formalin and routinely embedded in paraffin wax. Sections (3–5 μm) were prepared and stained with hematoxylin and eosin (HE).
Further lung sections were stained with the van Gieson (VG)–elastica stain as described. 27 This highlights the internal and external elastic laminae, allowing a clear differentiation of all vascular wall components (tunicae intima, muscularis, and adventitia).
For immunohistochemistry, further lung sections were deparaffinized and rehydrated, then incubated for 30 min at room temperature with a prediluted mouse anti–human α–smooth muscle actin (α-SMA) antibody (clone 1A4; DAKO, Basel, Switzerland). A detection kit containing the secondary antibody and amino ethylcarbazole (AEC) as chromogen was subsequently applied according to the manufacturer’s protocols (Peroxidase/AEC Rabbit/Mouse Kit; DAKO).
Data Analysis
Based on sections stained with H&E and VG-Elastica, and for the expression of α-SMA, the thickening of the intima, media, and adventitia of arterial walls in the lungs was scored as follows: +, mild thickening (more than 1 layer); ++, moderate thickening (more than 2 layers); and +++, severe thickening (more than 3 layers) (Suppl. Tables S1 and S2).
In the H&E-stained liver sections, the following parameters were evaluated: parenchymal degeneration and/or regeneration, proliferation of biliary ducts, and grade of fibrosis (slight = +, periportal; moderate = ++, periportal to porto-central; severe = +++, diffuse/sinusoidal); presence of granulomas, abscesses, and/or neutrophilic or lymphoplasmacytic inflammatory infiltrates; incidence of parasites and/or eggs of liver flukes in biliary ducts; and/or parenchyma. A combination of all criteria was used to score the severity of the changes as mild, moderate, or severe (Suppl. Tables S1 and S2).
Arterial thickening in the lung was semiquantitatively assessed in relation to the other established parameters (liver changes, thoracic and abdominal effusions, elevated liver enzyme activity, degree of liver fluke infestation) based on the applied grading scores (+ to +++) to identify any possible correlation between the different parameters (see Suppl. Tables S1 and S2).
Results
Clinical History and Signs
For all animals, similar clinical signs were reported. Most animals showed reduced body condition, became recumbent, and had died suddenly, showing muscular cramps and, sometimes, seizures.
Of the llamas, 10 were reported to be recumbent, of which 6 exhibited seizures and died; 7 had a history of sudden death, and each had nonspecific clinical signs, diarrhea, and abortion. In 2 llamas, ascites had been diagnosed by ultrasonography.
From 11 llamas, information was available regarding the potential contact with sheep. Of these, 5 had contact with sheep, 5 had no contact with sheep, and 1 llama had contact with camels. Information on antiparasitic treatment was available from only 5 llamas, of which 4 have been dewormed before euthanasia and 1 had not been treated. Deworming had been undertaken with doramectin and sometimes with ivermectin or praziquantel. Praziquantel is known to reduce the D. dendriticum burden by more than 90% in NWCs when given at 50 mg/kg per os. 25
In 12 llamas, the serum liver enzyme activities and albumin were measured, and in 10, the serum activity of liver enzymes, especially GGT (10/12), SDH (6/8), and ASAT (3/12), was increased (Suppl. Table S3). Albumin was decreased in 1 of the 3 tested animals. Four of the 11 tested animals showed a strong increase in CK activity, and another had a slight increase.
Of the alpacas, 7 were recumbent, 5 died suddenly, 4 showed nonspecific clinical signs, and 3 had diarrhea. In 2 animals, ascites was shown by ultrasonography. Information on the potential contact with sheep was available for 6 animals, of which 4 were known to have had no contact with sheep and 2 had contact with sheep and goats. Enzyme activities and albumin were measured in serum of 6 alpacas, of which 4 had a strong and one a slight increase in GGT and 5 an increased ASAT activity. All 3 animals tested for SDH showed increased activity of the enzyme. A decreased and 1 elevated albumin value was found in 2 out of 3 tested alpacas. The CK value was markedly increased in 3 of the 5 tested animals (Suppl. Table S3).
Parasitologic Findings
A parasitologic examination by fecal flotation and sedimentation was performed on 11 llamas and 17 alpacas either ante- or postmortem.
All tested llamas were infested with D. dendriticum. They also showed a high prevalence of gastrointestinal parasites—that is, Eimeria sp (9/11), Trichostrongylus sp (9/11), Trichuris sp (6/11), Strongylus sp (5/11), Nematodirus battus (4/11), Capillaria sp (3/11), and Ostertagia sp (2/11).
Most alpacas were also infested with D. dendriticum (15/17), with the remaining 2 tested animals exhibiting F. hepatica infestation. Gastrointestinal parasites were also frequently detected—that is, Eimeria sp (7/17), Trichostrongylus sp (7/17), Strongylus sp (6/17), Trichuris sp (4/17), and N. battus (2/17).
Pathologic Findings
The main macroscopic lesions in animals with liver fluke infestation were moderate to severe liver fibrosis with increased firmness and/or variably sized regenerative nodules (10/20 llamas, 10/20 alpacas). The liver of some animals showed calcified foci (4/20 llamas, 5/20 alpacas) or abscesses (5/20 alpacas); in the remaining (4/20 llamas, 7/20 alpacas), they appeared grossly normal. In 14 of 20 llamas and 6 of 20 alpacas, D. dendriticum specimens were grossly apparent, and in 2 alpacas, F. hepatica was found.
Thoracic and/or abdominal effusion was observed in 12 llamas and 14 alpacas. Moderate ascites was found in each llama and alpaca and severe ascites in 4 alpacas. In 1 llama and 4 alpacas, only a mild thoracic effusion was seen. Nine llamas and 5 alpacas had thoracic effusion and ascites in different degrees (Suppl. Tables S1 and S2).
In some animals, concomitant disease processes were recorded; these were forestomach compartment 1 (C1) acidosis, pulmonary tuberculosis due to Mycobacterium microti, bile duct carcinoma with lung metastases, and verminous encephalitis (consistent with Parelaphostongylus tenuis or Elaphostrongylus cervi) with secondary malacic foci in each llama. In alpacas, valvular endocarditis, old myocardial infarcts, lipid pneumonia, and an acute inflammation in the C1 forestomach were found in 2 animals, whereas an acute exudative peritonitis, mild acute fibrinous pericarditis, suppurative pneumonia, and end-stage kidney disease were diagnosed in each case.
Histologic and Immunohistochemical Findings
The histologic examination of the lungs revealed severe intimal and adventitial thickening and mild to moderate medial thickening of the arteries in 9 llamas (45%) and 3 alpacas (15%) (Figs. 1–5a,b,c). In none of the animals were there any changes that would indicate an early stage of proliferative and/or hypertensive arterial changes, such as slight thickening of arterial structures. The intimal and adventitial thickening was associated with the presence of spindle-shaped and variably sized activated cells that were strongly positive for α-SMA, indicating their myofibroblastic origin. 28 The normal arterial smooth muscle cells were also strongly positive for α-SMA (Figs. 1–5c).
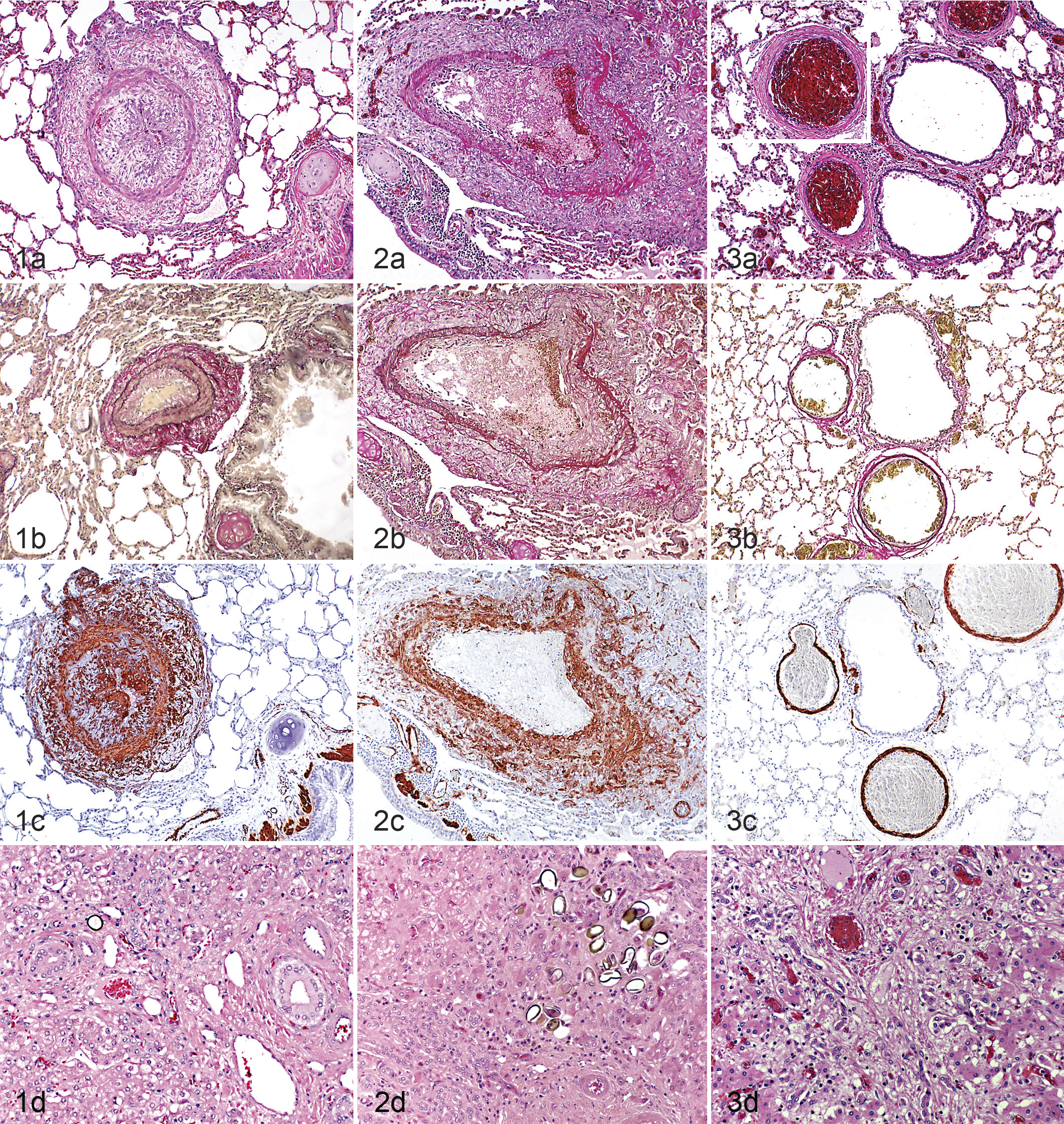
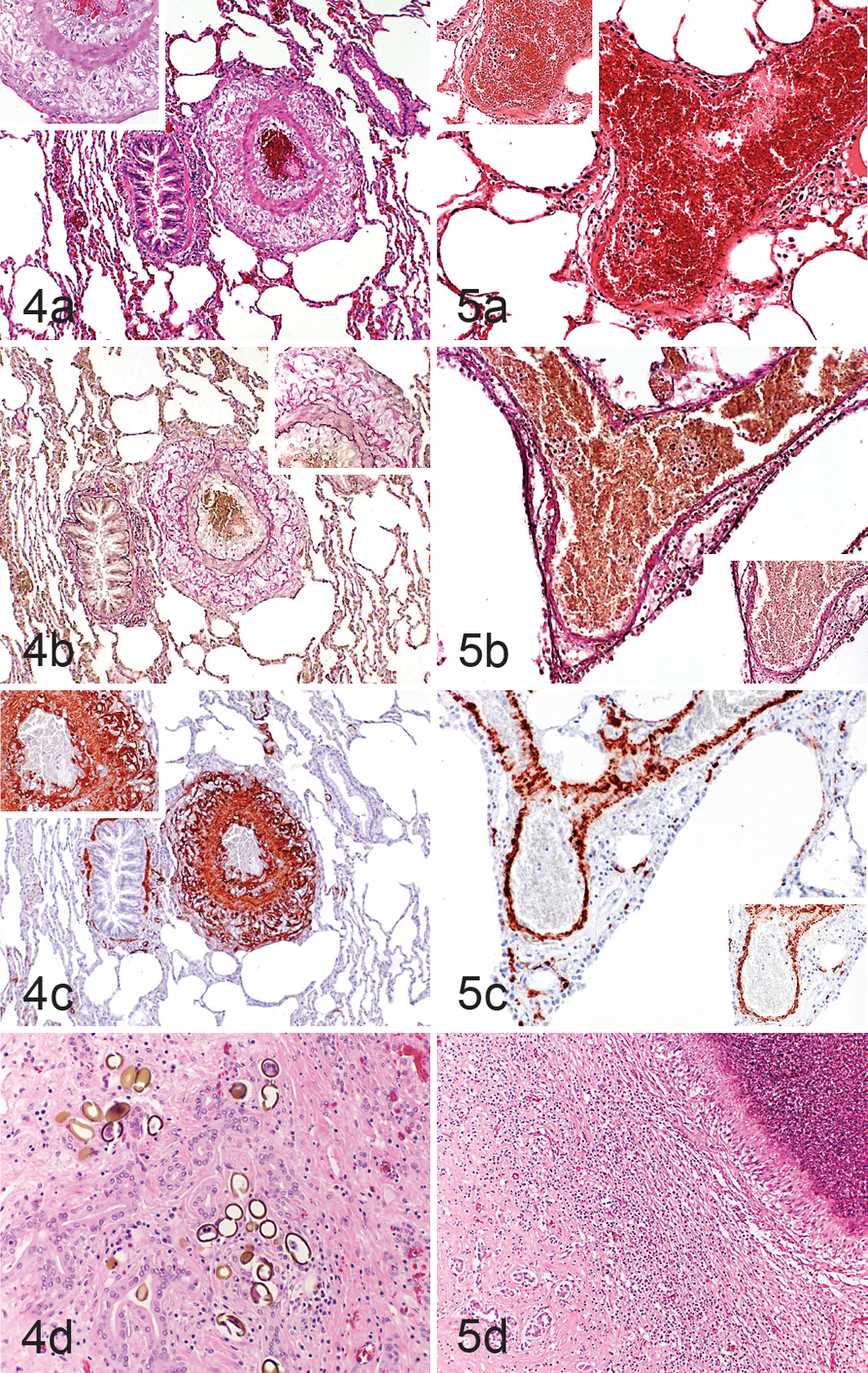
The liver of all animals exhibited changes attributable to liver fluke infestation (Figs. 1d–5d), which varied in their degree (mild to moderate in 7 llamas and 8 alpacas, moderate in 5 llamas and 6 alpacas, severe in 8 llamas and 6 alpacas). The main feature was a loss of hepatocytes with replacement by diffuse (sinusoidal) fibrosis (cirrhosis) and variably sized regenerative nodules as well as biliary hyperplasia. In most cases, neutrophils and/or lymphocytes and plasma cells were found randomly distributed in the parenchyma, around portal areas, and in the lumen of bile ducts (hepatitis and cholangitis). In some animals (llamas, n = 11; alpacas, n = 9), sections of flukes and eggs were found together with degenerate leukocytes in the lumen of dilated bile ducts. Also, a variable number of flukes and, more often, eggs were detected in the liver parenchyma in some animals (8 llamas, 6 alpacas). In 1 llama and 1 alpaca, cross sections of F. hepatica and D. dendriticum and F. hepatica alone, respectively, were found in the bile ducts. They were identified, since as trematodes, they have a tegument and no body cavity and are hermaphrodites. The differentiation of these flukes is based on their size (Fasciola are larger [cm vs mm]) and the presence of spines on the tegument of Fasciola. The eggs of D. dendriticum are oval and small (40 × 25 μm), have a gold brown wall, are operculated, and contain a miracidium, while the eggs of F. hepatica are larger (120–140 μm) and have no miracidium. 6
Occasionally, multiple random necrotic foci (interpreted as coagulative necrosis) and small abscesses with centrally located, degenerating neutrophils and surrounding fibrotic tissue (3 llamas, 5 alpacas) were seen in the liver. Small granulomas with central necrosis admixed with neutrophils, surrounded by large numbers of epithelioid macrophages, moderate number of foreign-type giant cells, and lymphocytes and plasma cells, were also observed (2 llamas, 6 alpacas). Detailed results are listed in Supplemental Tables S1 and S2.
Correlation Between Thoracic and Abdominal Effusions, Liver Fluke Infestation, and Alterations in Lung and Liver
Llamas
Hypertensive pulmonary arterial changes were seen in 9 llamas with the typical liver changes (see also Suppl. Table S1). In 7 of these, D. dendriticum was confirmed as the causative agent, either by parasitologic examination (n = 5) or by histology (n = 2). Most of these animals (n = 7) exhibited thoracic or abdominal effusions, mainly moderate to severe (6/7) and in 1 case mild; these animals also exhibited elevated liver enzyme activity in serum when tested (6/7). Of the 2 llamas from this group without body effusions, 1 animal was tested for liver enzyme activity in serum, which was elevated.
Of the 11 llamas without hypertensive changes in lung arteries, 5 animals exhibited liver changes as well as thoracic and/or abdominal effusions. When tested (n = 3), these animals exhibited increased liver enzyme activity. All llamas were infested with Dicrocoelium, either confirmed by parasitology of fecal samples (2 with severe infestation, 1 each with moderate and mild infestation) and/or by histology (n = 3).
The remaining 6 llamas showed the typical histologic liver fluke-induced lesions but only in 3 cases could an infestation with Dicrocoelium be proven, 1 of them with concomitant Fasciola infestation and increased liver enzyme activity when tested (n = 2) but no effusion.
Alpacas
In contrast, hypertensive pulmonary arterial changes were seen in only 3 alpacas with the typical liver changes, as well as thoracic and/or abdominal effusions of variable degree (see Suppl. Table S2). In 2 of 3 cases, Dicrocoelium was found in moderate or high amount respectively; in 1 case, Fasciola but no Dicrocoelium was found. Liver enzyme activity was not measured in these animals.
The remaining 17 alpacas showed no hypertensive pulmonary arterial changes. From these 17 animals, liver enzymes were measured in serum of 6 alpacas and showed a marked activity elevation, mostly of GGT (5/6). Fecal parasitology by flotation and sedimentation methods was done in 5 of 6 of these animals, showing a severe (n = 2) or mild to moderate Dicrocoelium infestation (n = 3). These 6 animals showed mild to marked liver lesions, and histologically, Dicrocoelium eggs in 4 animals and Dicrocoelium flukes in 1 animal could be identified. Of these animals, 3 had no thoracic and/or abdominal effusions, 2 had severe thoracic and/or abdominal effusions, and 1 had only a severe abdominal effusion.
The remaining 8 animals showed no (n = 1), mild (n = 1), or moderate (n = 5) infestation with Dicrocoelium, and 1 animal had moderate infestation with F. hepatica that was confirmed by parasitology. All 8 animals showed liver changes of variable degree, but only 6 had thoracic and/or abdominal effusions.
Discussion
The present study investigated the lungs of NWCs with liver fluke infestation for evidence of arterial hypertension and aimed to correlate these with the parasite-induced liver changes as well as portal hypertension and subsequent thoracic and abdominal effusions. We found that 45% of llamas and 15% of alpacas with chronic fluke-associated liver lesions exhibited pulmonary changes consistent with hypertension, indicating a correlation between these lesions. However, the high proportion of animals with liver fluke–associated liver lesions but without any arterial lesions in the lung indicate that additional factors play a role in the development of the arterial lung lesions.
Marked thickening of the intima and adventitia but not the media represented the hypertensive changes in the lung. The changes were observed in different lung lobes, suggesting that there is no predilection site in the lungs. Similar changes were also detected in other natural parasitisms of the liver, such as the infestation of rats with C. hepatica, in which a strong association between liver fibrosis and pulmonary arterial remodeling (portopulmonary hypertension) was observed. 26 In animal models and humans with pulmonary hypertension, the main histologic features are arterial medial hypertrophy, cell proliferation in the intima, and, in the later phase, concentric laminar fibrosis, fibrinoid necrosis, plexiform lesions, and recanalization. 5,20,22 Different from liver flukes in animals, the eggs of Schistosoma sp (eg, S. mansoni, S. haematobium, and japonicum) can directly colonize the lung parenchyma and induce pulmonary lesions (ie, vascular pulmonary remodeling and finally pulmonary hypertension). 4,5
However, it has also been suggested that the pulmonary arterial changes are a direct consequence of the host inflammatory response in the liver that induces fibrosis and the destruction of the portal venous system and, finally, portopulmonary hypertension. 9,16,20 Furthermore, humoral substances with vasoactive properties, including serotonin, interleukin 1 (IL-1), endothelin 1, glucagon, secretin, thromboxane B2, and vasoactive intestinal peptide, that are normally metabolized by the healthy liver, are suggested to induce the arterial pulmonary remodeling. 17 Therefore, the pathogenesis of the pulmonary arterial remodeling in pulmonary hypertension remains poorly understood. 4,5,9,16,20,22
The degree and severity of the liver fluke–induced lesions in the NWCs in our study suggest that these animals are particularly prone to infection and generally develop a strong response. Such an increased susceptibility of NWCs to D. dendriticum in contrast to sheep and cattle could partly be explained by anatomic and physiologic differences between these species. 8 Also, F. hepatica is widespread in South America, but D. dendriticum is not autochthonous in the Andean region, which would suggest that the severe pathologic liver changes are the result of an inadequate host immune response. 14 However, we also found pulmonary hypertensive changes in 1 alpaca infected with F. hepatica, which suggests that Fasciola must also be regarded as a potential cause of pulmonary arterial proliferative lesions and hypertension in NWCs in Switzerland.
It is also possible that the high cardiac output seen as a consequence of hepatic cirrhosis exposes the pulmonary vascular bed to increased shear stress, thereby inducing vasoconstriction, hypertrophy, and proliferation of pulmonary arterial endothelial cells and finally portopulmonary hypertension. 17 However, myofibroblasts arising from arterial smooth muscle cells and adventitial fibroblasts also play an important role in the thickening of arteries in hypertension. 15,19,28
In vascular injury models, pronounced adventitial changes are seen, and they develop early. 28 In response to multiple pathophysiologic stimuli, such as atherosclerosis and hypertension, adventitial fibroblasts become activated and can differentiate into myofibroblasts that express α-SMA and migrate to the media and intima. 19,28 In rats, the activation of the adventitial fibroblasts precedes the neointimal hyperplasia in experimentally transplanted vessels. Transforming growth factor β, matrix metalloproteinase 7, monocyte chemoattractant protein 1, and tumor necrosis factor α and IL-1β are expressed in the adventitia prior to the neointimal formation, suggesting that the activated adventitial (myo)fibroblasts contribute to the neointimal proliferation. 19
In NWCs with pulmonary hypertension, the adventitia and intima of arteries were predominantly affected, showing marked thickening and distinct α-SMA expression. There was no evidence of medial thickening. These findings would be in line with the hypothesis that activated adventitial fibroblasts differentiating into myofibroblasts are important for neointimal thickening. Considering that adventitial and intimal changes are seen early and prior to the medial hypertrophy in pulmonary hypertension in humans and animal models, it is possible that the affected NWCs died or were euthanized before the medial hypertrophy had developed. 19,28
In up to 10% of human patients with liver cirrhosis, extensive body effusions develop due to increased venous pressure resulting from sodium retention and subsequent extracellular volume expansion, coupled with low plasma oncotic pressure resulting from hypoabuminemia. 16,17 We observed body effusions in 60% of affected llamas and 70% of alpacas. As albumin levels were decreased in only a few of these animals (one-third tested of each species), it appears likely that this developed mainly due to increased venous pressure.
In our cohort, liver enzyme activity was elevated in serum of the majority of tested animals. Serum levels of GGT and GLDH, the activity of which is elevated in hepatocellular damage, are postulated to be of value in diagnosing Dicrocoelium infestation in NWCs. 13,24 In contrast, ASAT activity can be increased with hepatocellular or muscular damage. 24 CK activity is elevated (eg, in rhabdomyolysis). 24 In our study, several llamas and alpacas exhibited increased CK activity, which could be explained by the (prolonged) recumbency and/or muscular cramps that were observed in most animals.
We did not find evidence of a direct correlation between the degree of liver fluke infestation and liver damage, elevated liver enzyme activity, and the amount of pleural effusion or ascites with the hypertensive pulmonary arterial changes. However, when arterial lesions were present, they showed a strong association with the typical liver changes.
In NWCs, praziquantel has been described as being effective against D. dendriticum infestation, with an effectivity of over 98% when used at a dosage of 50 mg/kg. 25 However, only a few animals from the present study had been dewormed and rarely with praziquantel.
In summary, this report demonstrates that liver fluke infestation of NWCs has the potential to induce pulmonary proliferative arterial changes and hypertension as a consequence of the associated chronic liver changes, similar to Schistosoma sp infection in humans.
Footnotes
Acknowledgements
We thank the technical staff of the Institute of Veterinary Pathology, Vetsuisse Faculty Zurich, for excellent technical support and the staff of the Institute of Parasitology for the parasitological examinations.
Author Contribution
Conception or design: MH; data acquisition, analysis, or interpretation: NR, CG; drafting the manuscript: MH. All authors participated in critically revising the manuscript, gave final approval, and agree to be accountable for all aspects of work to ensure integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
