Abstract
Borna disease is a progressive meningoencephalitis caused by spillover of the Borna disease virus 1 (BoDV-1) to horses and sheep and has gained attention due to its zoonotic potential. New World camelids are also highly susceptible to the disease; however, a comprehensive description of the pathological lesions and viral distribution is lacking for these hosts. Here, the authors describe the distribution and severity of inflammatory lesions in alpacas (n = 6) naturally affected by this disease in comparison to horses (n = 8) as known spillover hosts. In addition, the tissue and cellular distribution of the BoDV-1 was determined via immunohistochemistry and immunofluorescence. A predominant lymphocytic meningoencephalitis was diagnosed in all animals with differences regarding the severity of lesions. Alpacas and horses with a shorter disease duration showed more prominent lesions in the cerebrum and at the transition of the nervous to the glandular part of the pituitary gland, as compared to animals with longer disease progression. In both species, viral antigen was almost exclusively restricted to cells of the central and peripheral nervous systems, with the notable exception of virus-infected glandular cells of the Pars intermedia of the pituitary gland. Alpacas likely represent dead-end hosts similar to horses and other spillover hosts of BoDV-1.
Keywords
Borna disease virus 1 (BoDV-1; species Orthobornavirus bornaense; family Bornaviridae) causes a nonsuppurative meningoencephalitis known as Borna disease (BD) in spillover hosts such as horses and sheep.48,49 The disease is endemic in parts of central Europe including Southeastern Germany (federal states of Bavaria, Thuringia, Saxony, Saxony-Anhalt, and Brandenburg), the cantons Grisons and St. Gall in Switzerland, the principality of Liechtenstein, and the federal states Vorarlberg and Upper Austria in Austria.6,17,25,50,61 The only known reservoir host of BoDV-1 is the bicolored white-toothed shrew (Crocidura leucodon), which does not show any clinical or histopathological lesions.17,25,42,43,61 In its reservoir host, the virus infects neural as well as extra-neural cells and tissues including epithelial cells in the kidney, salivary glands, and skin, allowing for shedding of infectious virus via saliva, feces, urine, and dander.42,43,61 Thereby, the virus can be transmitted not only within its reservoir species but also to other mammals, including horses, sheep, New World camelids, and humans.8,34,41,47,52 In addition, singular cases of natural BoDV-1 infections have been reported and confirmed in cats, dogs, cattle, and goats.5,9,62 For these spillover hosts, an intranasal route of entry was suspected, followed by a spread to the central nervous system (CNS) in which high viral loads were detectable.20,32,37 In contrast to the reservoir species, the virus is described to be strictly neurotropic in spillover hosts and found almost exclusively in their CNS. Hence, these hosts do not further transmit the pathogen and act as dead-end hosts for the virus.46,52 The restriction of BoDV-1 to a rather immobile and territorially constrained small mammal reservoir, such as the bicolored white-toothed shrew, and the lack of an epidemiological role of more mobile erroneous hosts, such as domestic mammals and humans, likely adds to the remarkably restricted endemic area of this virus.16,28,50,52
BoDV-1 per se is not cytolytic and the elicited nonsuppurative meningoencephalitis is caused by T lymphocyte-mediated immunopathogenesis.23,45 Perivascular and intraparenchymal inflammatory infiltrates are dominated by CD4+ or CD8+ T cells, respectively.3,21,45 Pathognomonic, so-called Joest-Degen, inclusion bodies can be found in the nuclei of neurons and reflect the sites of bornavirus transcription and genome replication. 48
Recently, BD has gained increased attention as a zoonotic disease due to cases of fatal encephalomyelitis in humans. To date, at least 23 human infections have been published, of which 22 patients died.12,18,29,33,36,40,41,51,59 Three of these patients acquired the infection after receiving liver or kidney transplants from the same infected donor,33,51 while the majority of the cases appear to be the result of sporadic spillover events directly from the reservoir host.12,18,29,36,40,41,59 Viral antigen and RNA were localized mainly in the patients’ CNS, virtually identical to the other known dead-end hosts.41,51
Generally, BD occurs sporadically in spillover hosts and is usually restricted to a few individuals or animals of a herd.10,21,60,61,63 However, a mortality rate of roughly 40% was recently reported in an alpaca herd in a novel endemic area in Brandenburg, a federal state of Germany, 52 and New World camelids are discussed as a sentinel species for BoDV-1 infections. 34 To date, only single reports have described BD in New World camelids,26,27,34 and a comprehensive study of respective histopathological lesions of BD in this species is lacking.
A previous publication focused on the diagnosis, epidemiology, and phylogeographic analysis of the BD outbreak in alpacas. 52 The histologic analysis and the demonstration of BoDV-1 antigen in the nervous system had confirmed the diagnosis of BD. 52 However, a detailed and comprehensive histologic description of BD-related lesions in alpacas and cellular virus distribution was lacking to date. Therefore, this study aimed to systematically describe the pathology of BD and the BoDV-1 tissue and cell tropism in naturally infected spillover hosts, such as alpacas and horses, to reveal similarities or differences in BD pathology in spillover hosts. The degree of comparability could help to identify common pathomechanisms of this disease.
Material and Methods
Sample Retrieval
Six male alpacas (Vicugna pacos) that had died during a previously reported BoDV-1 outbreak were analyzed in this study (A1 to A6; Table 1). The outbreak had occurred in the state of Brandenburg, Germany, with a mortality rate of approximately 40%. 52 In addition, archived material of 8 horses from Brandenburg or Saxony-Anhalt with diagnosed nonsuppurative encephalitis (H1 to H8; Table 1) were analyzed. BoDV-1 infections were confirmed by BoDV-1-specific reverse transcriptase-quantitative polymerase chain reaction (RT-qPCR) following RNA extraction from formalin-fixed, paraffin-embedded (FFPE) brain tissues, as described previously. 52
Borna disease-affected alpacas and horses used in this study.
The identity and clinical course of alpacas A1 to A6 had previously been published in Schulze et al. 52
Abbreviations: F, female; M, male; MN, neutered male.
Representative samples of the CNS and other tissues were immersion-fixed in 4% formalin for 24 to 48 hours. The formalin-fixed brain tissue was cut into coronal sections and specific CNS regions, including the olfactory bulb, frontal lobe, basal nuclei, temporal lobe with hippocampus, occipital lobe, and cerebellum and brainstem, were analyzed in accordance with reports on human cases. 33 All formalin-fixed samples were subsequently embedded in paraffin and processed for histological and immunohistochemical examination.
Histopathology
Three-micrometer-thick tissue sections were routinely processed, dewaxed in xylene, rehydrated in decreasing ethanol concentrations, and stained with hematoxylin and eosin (HE). The severity of cerebral inflammation in terms of the extent of perivascular immune cell infiltration (PVI) in brain sections was evaluated according to a previously established grading system 8 with minor modifications: −: no perivascular infiltration; +, mild: few vessels with < 2 layers of PVI; ++, moderate: moderate number of vessels with mostly 2 to 3 layers of PVI; +++, severe: many vessels with > 3 layers of PVI. The immune cell infiltration at the transition of the neural to the glandular part of the pituitary gland was graded in a similar fashion: −: no infiltration; +, mild: < 2 layers of inflammatory cells; ++, moderate: mostly 2 to 3 layers of inflammatory cells; +++, severe: > 3 layers of inflammatory cells.
Cellular Detection of BoDV-1
For immunohistochemical detection of BoDV-1 antigen, all tissue sections were incubated with a rabbit polyclonal anti-BoDV-1 nucleoprotein (N; No. 201; 1:8000) and a rabbit anti-BoDV-1 phosphoprotein (P; No. 130; 1:4000) serum, as described previously. 52 A matched set of organs from an alpaca that died of other causes served as a negative control. This animal had no histopathological lesions consistent with encephalitis and tested negative for BoDV-1 RNA by RT-qPCR from FFPE brain material. In addition to this animal, the pituitary gland of another alpaca and a horse without any signs of BD were included as controls. Tissues with weak immunohistochemical signals from animal A6 including the eye and the nerve fibers were further analyzed via in situ hybridization (ISH) to detect viral RNA. ISH focused on the demonstration of RNA of the N gene and respective mRNA according to established protocols.6,64 The hippocampus of this animal served as a positive control.
Immunofluorescence
For identification of BoDV-1-infected cell types in the CNS, slides from the cerebrum of BD-affected alpacas and horses were incubated with a mouse monoclonal anti-BoDV-1 (N; Bo18; 1:25) at 4°C overnight as described for IHC for double immunofluorescence with the antibody against the glial fibrillary acidic protein (GFAP). For double immunofluorescence with antibodies against CD3 or anti-ionized calcium-binding adapter molecule 1 (IBA-1), antigens were retrieved using microwave heating for 12 minutes in 10 mM citric acid, pH 6.0, containing 0.05% Triton X-100. Subsequently, an Alexa Fluor 568-conjugated goat-anti-mouse IgG antibody (1:200, Invitrogen) was added as a secondary antibody for 1 hour at room temperature. After repeated washing steps, the slides were incubated with a rabbit polyclonal anti-GFAP (1:300, Dako), anti-CD3 (1:40, Dako), or IBA-1 (1:50, FujiFilm Wako) overnight at 4°C. A secondary Alexa Fluor 488-conjugated goat anti-rabbit IgG antibody (1:200, Invitrogen) was added for 1 hour at room temperature. After repeated washing, the slides were mounted with Roti-Mount FluorCare DAPI (4,6-diaminidino-2-phenylindole, Carl Roth). Detailed information on the antibodies used and protocols of the immunofluorescence and immunohistochemistry are provided in the supplementary material (Supplemental Table S1). As controls, slides were analyzed without adding the anti-BoDV-1 antibody, the antibody for the respective cellular marker, and both primary antibodies.
Results
Alpacas Affected by BD Develop Lymphocytic Encephalitis With Interindividual Variation in Distribution and Severity
At necropsy, the brain tissue of all 6 alpacas lacked macroscopic lesions. However, multifocal, perivascular, lymphocytic meningoencephalitis of varying degrees was histologically diagnosed in all animals (Fig. 1a–e). In addition to lymphocytes, macrophages and a few plasma cells were also present. The perivascular inflammatory cells were admixed with neutrophils in alpacas A1 and A2 (Fig. 1d, e). Pathognomonic eosinophilic, intranuclear, Joest-Degen inclusion bodies were found exclusively in neurons of the hippocampal region of only 2 out of 6 alpacas (A2 and A5; Fig. 1c, inset).
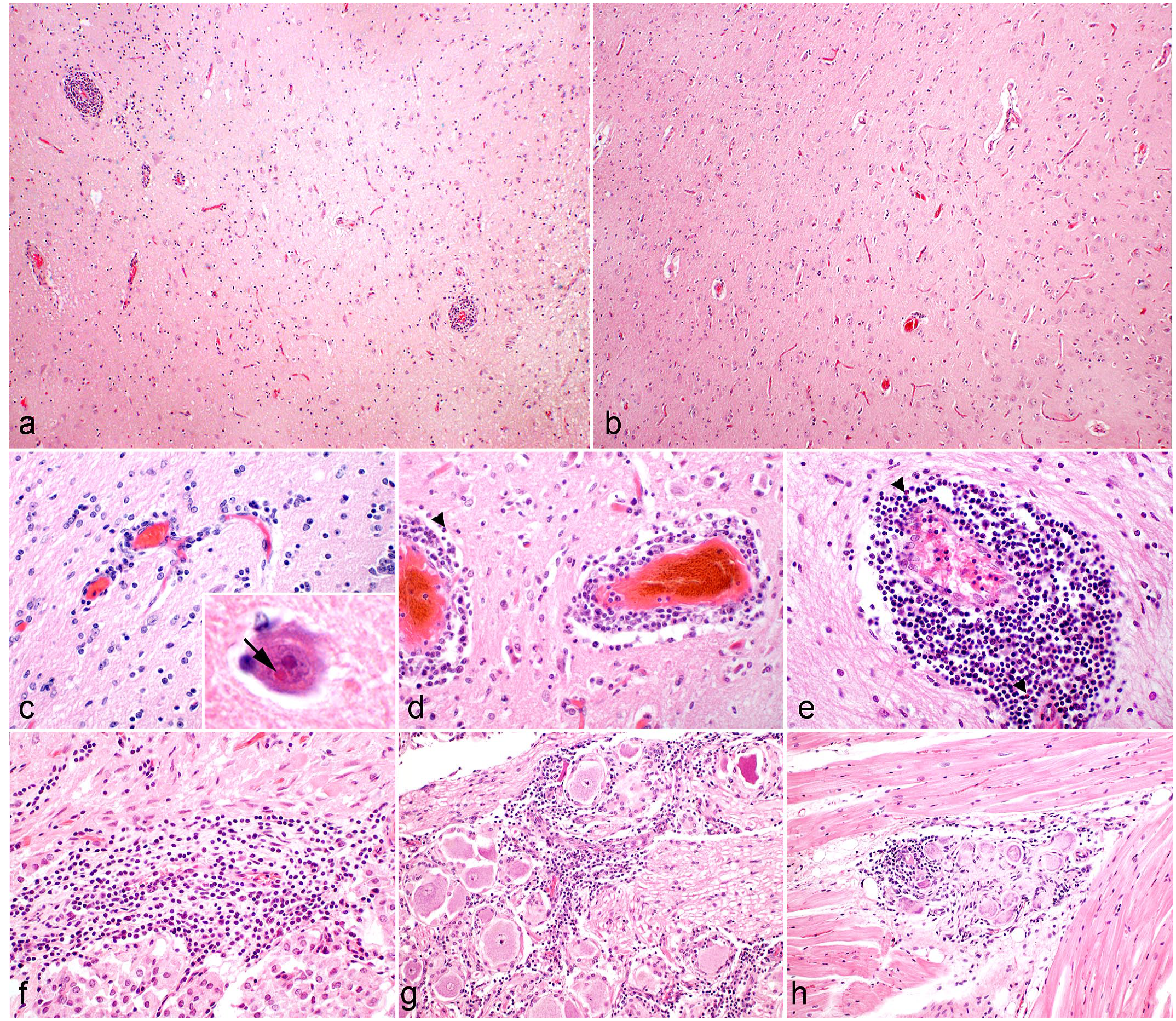
Severity of immune cell infiltration in nervous tissue of Borna disease-affected alpacas; brain, alpaca. Hematoxylin and eosin. The distribution and severity of the lesions varied between the alpacas. (a) Moderate to severe multifocal, perivascular, lymphocytic encephalitis was diagnosed in the cortex of alpaca A3, whereas (b) no inflammation was visible in alpaca A5. Multifocal, perivascular, predominant lymphocytic encephalitis with (c) mild (+, alpaca A5), (d) moderate (++, alpaca A1), or (e) severe (+++, alpaca A2) perivascular immune cell infiltration in the olfactory bulb was histologically diagnosed in all animals analyzed, occasionally admixed with few neutrophils (arrowheads). Inset c) Distinct, eosinophilic, intranuclear viral inclusions (Joest-Degen inclusion bodies, arrow) in neurons of the hippocampal region (alpaca A5) were rarely diagnosed. Additionally, other neural structures inconsistently showed lymphocytic infiltrations: (f) the Pars intermedia at the transition to the Pars nervosa of the pituitary gland (alpaca A3), (g) the trigeminal ganglion (alpaca A5), and (h) the sinoatrial node (alpaca A5).
Comparative analysis revealed remarkable inter-individual differences of immune cell infiltration in the cerebrum, brain stem, and pituitary gland. For animals A1 to A3, which had died 0 to 25 days after the onset of clinical signs, moderate to severe inflammation was identified in the cerebrum and pituitary gland (Supplemental Table S2, Fig. 1a, d, e, f). The distribution of the inflammation was virtually identical in animal A4, which died 26 days after onset of disease; however, the severity was classified as mild to moderate (Supplemental Table S2). In contrast, for both animals that died after chronic disease progression (A5, 102 days, and A6, 192 days), the inflammation was absent or a mild inflammation was observed in parts of the brain (Supplemental Table S2, Fig. 1b, c). The cerebellum was affected in only one alpaca (Supplemental Table S2).
Other parts of the nervous system also showed signs of inflammation in some alpacas. A mild, multifocal, perivascular, lymphocytic myelitis was diagnosed in alpacas A1 and A3 to A5, but not in A6. The trigeminal ganglions of alpacas A5 and A6, but not A4, were mildly or moderately infiltrated by lymphocytes, respectively (Fig. 1g). Peripheral nerves showed no signs of inflammation, except for the sinoatrial node of alpaca A5, which was mildly infiltrated by lymphocytes (Fig. 1h). Histologically, no other lesions were identified in any of the tissues analyzed.
Horses Affected by BD With a Peracute to Subacute Course of Disease Had Moderate-to-Severe Inflammation in the Cerebrum and Pituitary Gland
In addition to alpacas, 8 BD-affected horses were analyzed in this study. The horse showed a peracute to subacute course of disease with 1 to 18 days from disease onset to death (Table 1). Similar to those alpacas with a shorter course of disease of up to 25 days, all horses analyzed had moderate to severe, lymphocytic encephalitis in the cerebrum (Fig. 2a, Supplemental Table S3). Lymphocytes were the predominant inflammatory cell type, admixed with macrophages and few plasma cells (Fig. 2a). In addition, a moderate or severe, lymphocytic hypophysitis of the Pars intermedia was also observed in horses affected by BD (Fig. 2b, Supplemental Table S3). The PVI in the brain stem varied from mild to severe and the cerebellum was affected in only 5 of 8 horses (Supplemental Table S3).
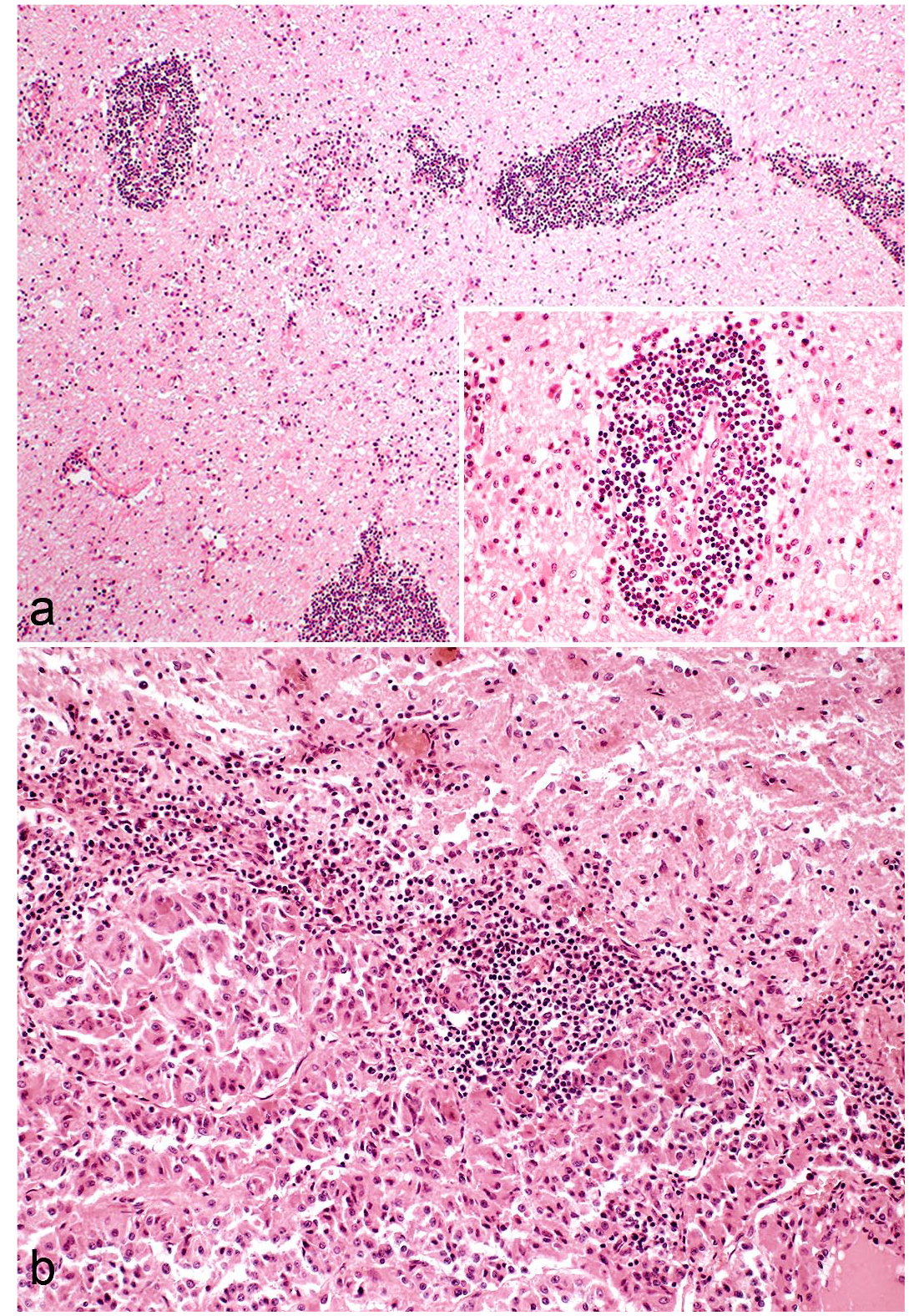
Severity of immune cell infiltration in nervous tissue of Borna disease (BD)-affected horses; brain, horse. Hematoxylin and eosin. (a) severe, multifocal, perivascular, predominant lymphocytic encephalitis was diagnosed in BD-affected horses (horse H1). Inset. Lymphocytes were the predominant inflammatory cell type, admixed with macrophages and few plasma cells. (b) A lymphocytic infiltration was evident at the transition of the Pars nervosa to the Pars intermedia of the pituitary gland (horse H7).
BoDV-1 Antigen Was Detected in the CNS, Peripheral Neurons, and Pituitary Glandular Cells of the Pars intermedia of BD-Affected Alpacas
Comprehensive immunohistochemical analysis identified BoDV-1 antigen in the CNS of all alpacas via antibodies directed against the viral N or P protein with virtually identical results (Supplemental Table S4, Fig. 3a, b). Viral antigen was primarily detected in the nucleus, cytoplasm, and cellular processes of neurons (Fig. 3a–e). In addition, virus antigen-positive astrocytes were also identified (Fig. 3c). In contrast, BoDV-1 was not found in microglia and CD3+ lymphocytes (Supplemental Figure S1). In the cerebellum, BoDV-1 antigen was predominantly expressed in Purkinje cells with their dendritic arbores (Fig. 3e).
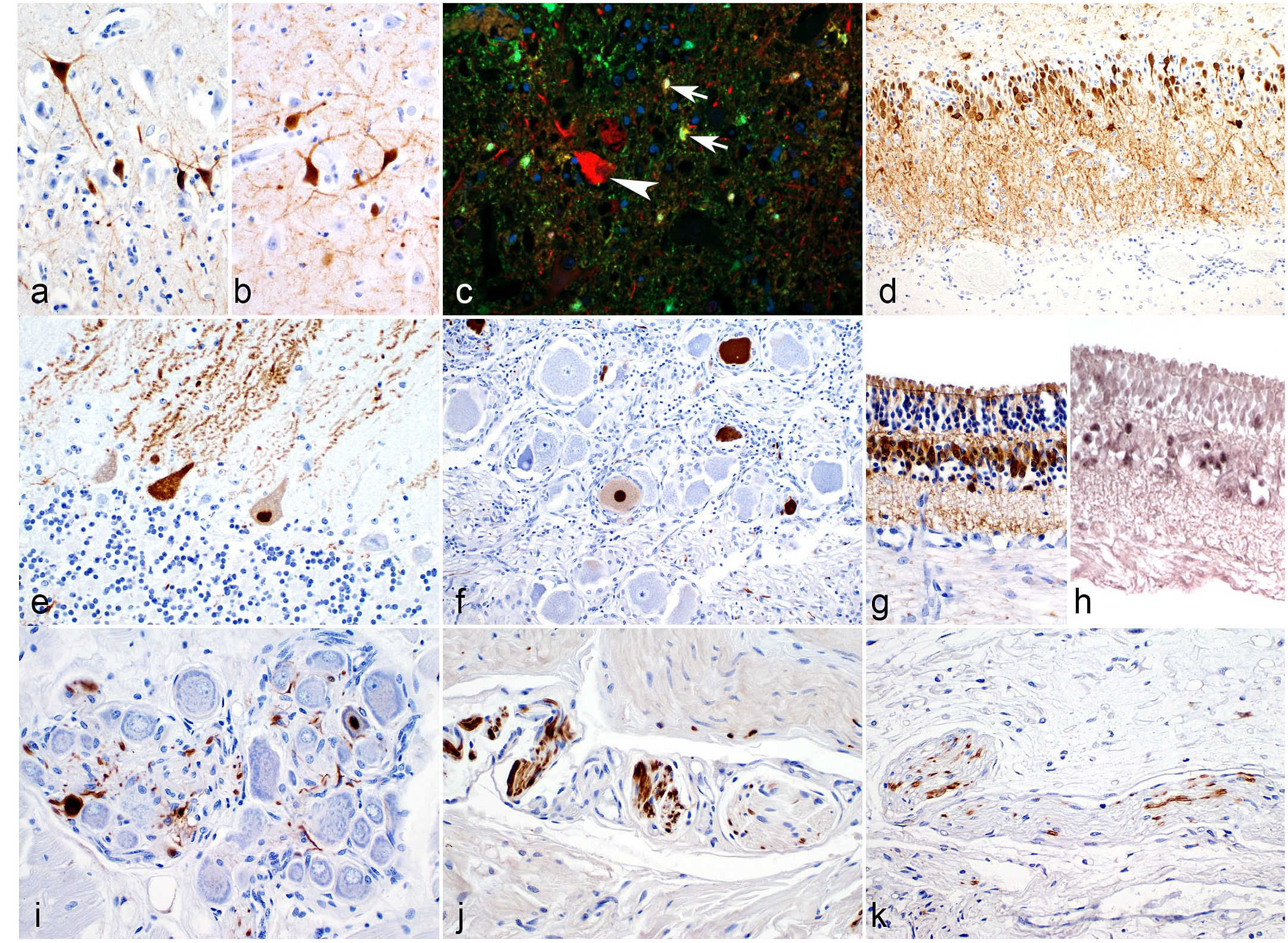
Borna disease virus-1 (BoDV-1) detection in nervous and retinal tissues of Borna disease-affected alpacas by immunohistochemistry or immunofluorescence (IF) and in situ hybridization (ISH). (a) BoDV-1 nucleoprotein was detected primarily in the nucleus and cytoplasm of neurons and in their cellular processes of the cerebrum (temporal cortex, alpaca A2). (b) BoDV-1 phosphoprotein was virtually identically localized (temporal cortex, alpaca A2). (c) BoDV-1 nucleoprotein (red) was identified by IF in neurons (arrowhead) and GFAP (green)-expressing cells (arrows, alpaca A1). (d) BoDV-1 nucleoprotein was present in the hippocampus (alpaca A6). (e) In the cerebellum, predominantly the Purkinje cells with their dendritic arbors expressed BoDV-1 nucleoprotein (alpaca A1). In addition to the brain, BoDV-1 nucleoprotein was expressed in (f) the trigeminal ganglion (alpaca A5) and (g) the retina (alpaca A5). (h) The cellular expression of BoDV-1 in the retina was confirmed by ISH. BoDV-1-antigen was also detected (i) the sinoatrial node of the heart (alpaca A5), and occasionally in diminutive nerve fibers of peripheral organs such as (j) the intestine (alpaca A5) and (k) the adrenal gland (alpaca A5).
In alpacas A1 to A5, BoDV-1 antigen was found in different parts of cerebrum, brainstem, cerebellum, and spinal cord (Supplemental Table S4). In contrast, the distribution of the viral antigen was restricted to a few cerebral regions of animal A6, in which viral antigen was present mainly in the olfactory bulb and hippocampus (Supplemental Table S4).
In addition to the CNS, viral antigen was commonly localized to large peripheral nerves, the trigeminal ganglion (Fig. 3f), and the retina (Fig. 3g) in alpacas A3 to A5, but only sporadically in alpaca A6 (Supplemental Table S4). The detection of the virus in the retina in alpaca A6 was confirmed by ISH (Fig. 3h). In addition, the optic nerve and, occasionally, adjacent nerve fibers displayed positive signals.
Occasionally, viral antigen expression was also detectable in diminutive nerve fibers of peripheral organs and tissues (Supplemental Table S4), such as the nasal mucosa (A3), heart (A5, Fig. 3i), digestive tract (A5, Fig. 3j), and the adrenal (A3, A5, Fig. 3k) and lacrimal glands (A5, A6). However, the cells of the non-neural tissues were devoid of BoDV-1 antigen.
Despite the known neurotropism of BoDV-1 in spillover hosts, the virus seems to selectively target pituitary glandular cells of the Pars intermedia in alpacas. In accordance with signs of inflammation at the transition of the neural to the glandular part of the pituitary gland (Fig. 4a) and the viral expression in the neural pituitary compartment (Fig. 4b), viral antigen was found in cells of the pars intermedia in alpacas A2 to A5 with a patchy distribution pattern (Supplemental Table S4; Fig. 4c). Of note, the Pars distalis of the adenohypophysis lacked expression of BoDV-1 antigen (Fig. 4d). The pituitary gland of the control alpacas showed no positive BoDV-1 immunolabeling (Supplemental Figure S2).
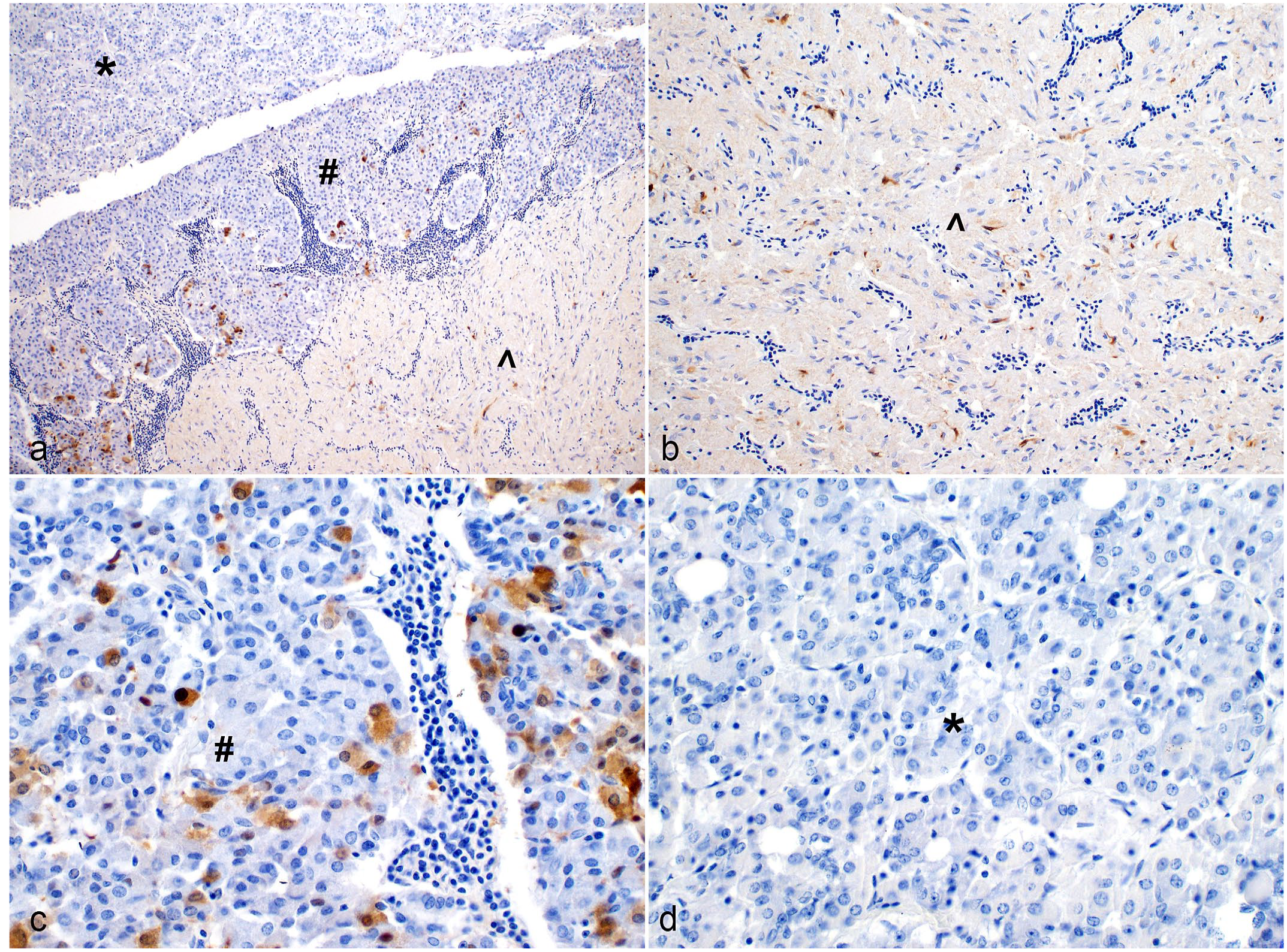
Borna disease virus-1 (BoDV-1) antigen expression in the Pars nervosa and intermedia of the pituitary gland of Borna disease-affected alpacas. Immunohistochemistry to detect BoDV-1 nucleoprotein. (a) Overview of the 3 parts of the pituitary gland: Pars nervosa (circumflex), Pars intermedia (hash), Pars distalis (asterisk). (b) BoDV-1 was consistently found in axonal projections of the Pars nervosa (circumflex) and (c) glandular cells of the Pars intermedia (hash) adjacent to the neurohypophysis. (d) The Pars distalis (asterisk) of the adenohypophysis was devoid of BoDV-1 antigen.
BoDV-1 Antigen Expression Was Restricted to the Nervous System and the Pituitary Pars intermedia in BD-Affected Horses
BoDV-1 antigen was found in the CNS (Supplemental Table S5, Fig. 5a), eye, and frequently, in large peripheral nerves of horses affected by BD. Similar to alpacas, BoDV-1 antigen was also identified in GFAP-expressing cells (Fig. 5b), but not in microglia or CD3+ lymphocytes (Supplemental Figure S1). In addition, pituitary glandular cells of the Pars intermedia were infected by BoDV-1 (Fig. 5c), whereas the Pars distalis of the adenohypophysis lacked BoDV-1 antigen. The pituitary gland of the control horse showed no BoDV-1 immunolabeling (Supplemental Figure S2). Viral antigen was also detectable in diminutive nerve fibers of the adrenal gland of horse H1 (Supplemental Table S5, Fig. 5d). No other tissue or cell types of the peripheral organs were immunolabeled for BoDV-1.
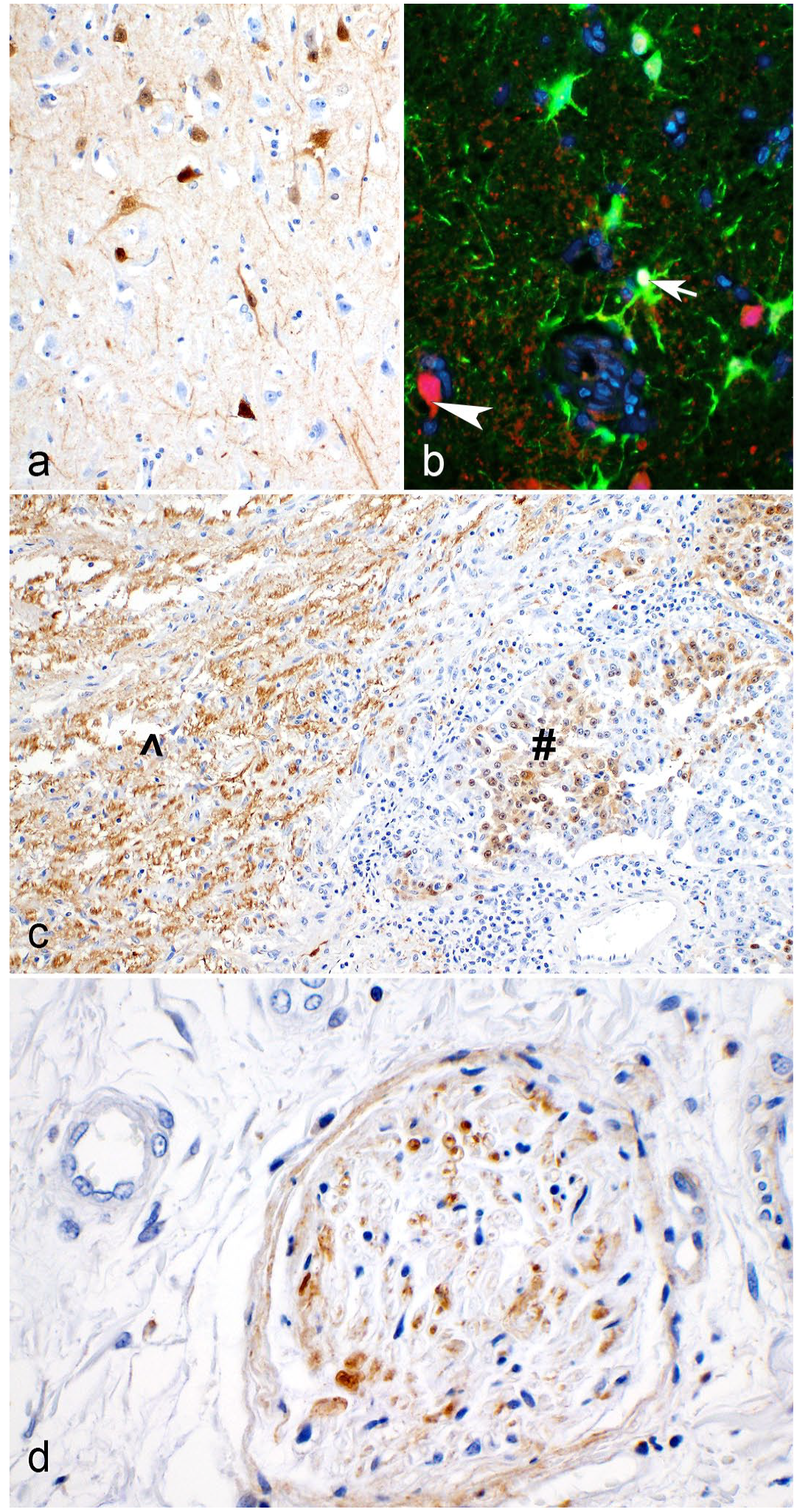
Neural Borna disease virus-1 (BoDV-1) antigen detection of Borna disease-affected horses. Immunohistochemistry (IHC) and immunofluorescence (IF) to detect BoDV-1 nucleoprotein. BoDV-1 nucleoprotein was immunohistochemically detected in (a) neurons of the cerebrum. (b) In addition, BoDV-1 (red, arrowhead) was found in GFAP (green)-expressing cells (arrow). (c) Glandular cells of the Pars intermedia (hash) and axonal projections of the Pars nervosa (circumflex) of the pituitary as well as in (d) diminutive nerve fibers of the adrenal gland were also positive for BoDV-1. Horse H1, IHC, or IF to detect BoDV-1 nucleoprotein.
Discussion
New World camelids have only sporadically been described as spillover hosts of BoDV-1 to date1,26,27,34,53; however, alpacas appear to be particularly susceptible to develop BD and may even serve as sentinels for BoDV-1 infection.34,52 Here, we analyzed the topography of histopathological lesions and the BoDV-1 cell and tissue tropism in 6 BD-affected alpacas in comparison to naturally infected horses.
The degree, character, and distribution of the lesions as well as the topographic and cellular distribution of BoDV-1 is conspicuously similar between alpacas and horses with comparable disease duration, which suggests that there are common pathomechanisms in these 2 spillover species (Tables 2, 3). In this study, a predominantly lymphocytic meningoencephalitis was diagnosed in all alpacas and viral antigen was consistently detected in particular regions of the CNS, such as the olfactory bulb, temporal cortex, thalamus, Nucleus caudatus, and hippocampus. The virus had a predominant cellular tropism to neurons and was additionally detected in GFAP-expressing cells of alpacas, similar to horses. IBA-1+ microglia and CD3+ lymphocytes seem not to be targeted by BoDV-1, which had also been reported in humans suffering from BD. 33
Summary of the distribution and severity of the immune cell infiltration in the brain and pituitary gland of Borna disease-affected alpacas and horses.

: No perivascular immune cell infiltration (PVI) or immune cell infiltration at the transition to the glandular part of the pituitary gland
: Few vessels with <2 layers of PVI/< 2 layers of immune cell infiltration
: Moderate number of vessels with mostly 2 to 3 layers of PVI/mostly 2 to 3 layers of immune cell infiltration
: Many vessels with >3 layers of PVI/> 3 layers of immune cell infiltration.
Abbreviation: n.a., not analyzed.
Summary of the spatial tissue distribution of Borna disease virus-1 (BoDV-1) antigen in Borna disease-affected alpacas and horses.
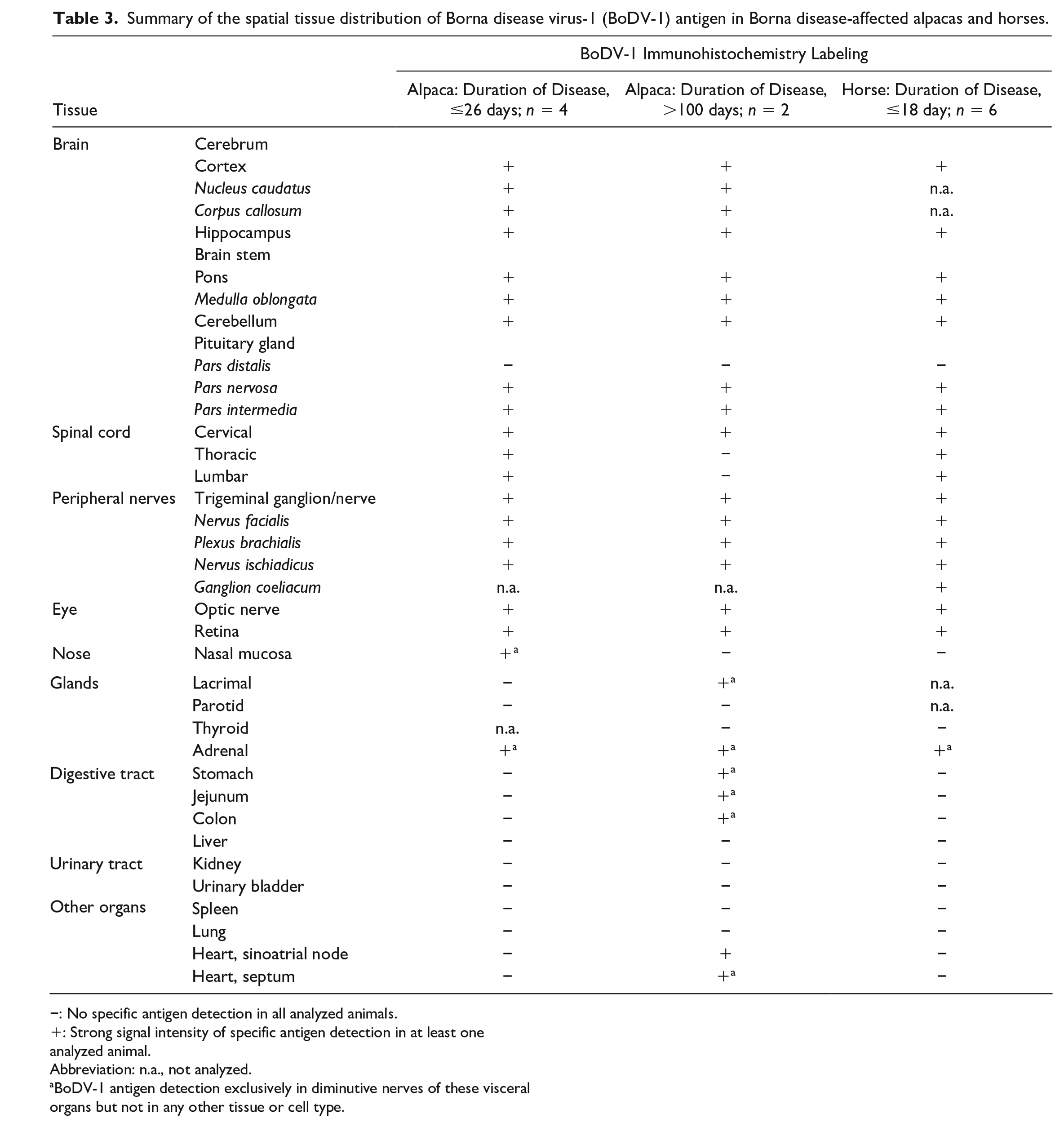
−: No specific antigen detection in all analyzed animals.
: Strong signal intensity of specific antigen detection in at least one analyzed animal.
Abbreviation: n.a., not analyzed.
BoDV-1 antigen detection exclusively in diminutive nerves of these visceral organs but not in any other tissue or cell type.
However, interindividual differences regarding tissue distribution, degree of inflammatory infiltrates, and existence of viral antigen were identified, which appeared to correlate with disease duration. Alpacas that died after a shorter disease course of up to 25 days had more prominent inflammation as compared to those that had suffered from chronic disease for several months. The 2 alpacas with a disease duration of more than 100 days that were euthanized due to severe apathy had mild CNS pathology. It had been reported that the clinical signs of BoDV-infected rats transitioned from severe neurological signs, such as convulsion and tremor, to a passive phase characterized by apathy. 54 Concurrently, the cytokine expression profile changed and it has been speculated that the reduction of inflammation and amelioration of clinical signs could be due to the production of certain cytokines. 54 It remains speculative if the viral infection and changes in the cytokine expression profile causes persistent neuronal biochemical disorders without corresponding histopathology which could explain the clinically severe apathy.
Similar to the alpacas with a shorter duration of disease, severe inflammation was also observed in the horses analyzed in this study that showed a peracute to subacute course of disease (Table 2). An impact of the duration of the disease had also been demonstrated in experimentally infected rats, in which the intensity of inflammation, and partly also viral loads, subsided after 40 to 50 days postinfection.11,13,22,38 However, numerous other factors, such as infectious dose, route of entry, individual susceptibility, constitution, and immune status of the host, may contribute to this variation.4,11,20,32,37,38,39 Hence, the cause of these substantial inter-individual differences remains speculative to date.
Pathognomonic viral inclusion bodies were only observed in one alpaca with a short and one alpaca with a longer duration of disease. Viral inclusions of BoDV-1 are membrane-less spherical structures in the nucleus containing the viral genome, antigenome, and viral proteins. 35 The complex viral and cellular factors which may initiate the formation of inclusions bodies have not been elucidated to date. The interindividual finding of viral inclusion bodies may also be a stochastic phenomenon.
Viral antigen and RNA were also commonly found in the optic nerve and retina in the alpacas investigated in this study, as reported for other susceptible species.3,47 However, no inflammatory or degenerative lesions were diagnosed in the ocular tissues in the present cases. Contradictory results have been reported on BoDV-1-associated ocular lesions in the past. One study did not observe inflammatory lesions in the retina of naturally infected horses despite clinical signs of blindness, which were attributed to central lesions in the visual pathway. 2 In contrast, ocular lesions were reported in another study on naturally infected horses 14 as well as in experimentally BoDV-1-infected rabbits, rats, and rhesus monkeys.30,31,39,55 Thus, it remains elusive to date which factors determine a pathological response to ocular viral infection.
Furthermore, viral antigen was sporadically detected in peripheral nerves and nervous tissues of peripheral organs, including the nasal mucosa, heart, intestinal tract, and lacrimal and adrenal glands in the alpacas and horses in this study. However, these tissues were devoid of histologically conspicuous inflammatory reactions. Infection of peripheral nerves had also been described in other naturally infected accidental hosts, such as humans,33,51 in experimentally infected species,32,37 and in the reservoir host.17,43,61 In general, viral neuropathies are rarely present in BoDV-1-infected spillover hosts and tend to appear late in the course of a virus infection. 7 This assumption is supported by our study in which alpaca A5, which had a prolonged disease duration, exhibited the broadest BoDV-1 antigen distribution in the nervous tissue of peripheral organs. However, the viral distribution in alpaca A6, affected by a presumably comparable or longer disease duration, was restricted to fewer regions of the peripheral nerve system (PNS). Hence, additional factors other than disease duration must be considered to influence PNS viral distribution.
In humans affected by BD, infection of the PNS may cause clinical symptoms resembling Guillain-Barré syndrome. 12 However, clinical signs compatible with peripheral neuropathy have never been reported in other accidental BoDV-1 hosts, including alpacas.
Of note, viral antigen was not detected in non-neuronal cells of peripheral organs in any of the alpacas or horses investigated in this study. This strict neurotropism of BoDV-1 observed in alpacas and horses coincides with previous work in naturally infected humans33,41 and other non-reservoir hosts,3,19,24,56 substantiating their status as dead-end hosts. This suggests that the virus is not shed by these hosts, which is supported by the absence of BoDV-1 RNA in nasal, conjunctival, and oral swabs or feces from the same alpacas in a previous study. 52 Identically to other spillover hosts, alpacas can be classified as dead-end hosts for BoDV-1 infection and may not pose a risk for infection for humans or other species.
The mechanisms restricting the tropism of BoDV-1 in spillover hosts to the CNS, predominantly to the neurons and also to astrocytes, remains speculative to date. In addition to factors such as the host’s immune response or the route of entry, the expression of receptors for cellular entry might contribute to this selective tropism. It has been speculated that the virus requires cellular receptors; however, their nature still remains to be identified. 15
Recently, BoDV-1 antigen was detected in the renal epithelial cells of a single human patient who had been infected by receiving an infected kidney transplant. 51 To our knowledge, this is the only report of BoDV-1 infection of non-neuronal cells in extraneural tissues in a naturally infected nonreservoir host. However, it is important to note that this patient had received immunosuppressive therapy which may be responsible for viral spread to non-neuronal tissues. Similarly, infection of non-neuronal cells has been described after experimental infection of immunocompromised rats56 –58 and occurs regularly in the reservoir species.6,42,61
A further factor in overcoming the strict neurotropism of BoDV-1 in nonreservoir hosts may be close contact between nerves and other cell types. In contrast to peripheral organs, viral antigen was detectable in glandular cells of the adenohypophyseal intermediate lobe of alpacas and horses in this study. The pituitary gland of 2 alpacas had been available for analysis by RT-qPCR, and BoDV-1 RNA was detected with a Ct value of 24.9 in one of these animals. 52 However, the targeted cell type was unknown. The glandular part of the intermediate lobe is in close contact with the Pars nervosa of the pituitary gland, where BoDV-1 was located in nerve fibers. It is known that corticotrophic basophil cells of the intermediate lobe may migrate into the Pars nervosa, which is termed basophil invasion 44 and may facilitate viral transition from neurons to glandular cells. The hypothesis of viral switchover by close contact of neurons to other cell types is supported by the observation that BoDV-1 antigen was not detectable in the glandular cells of the larger part of the adenohypophysis, which is anatomically detached from the nervous part of the organ. In humans, BoDV-1 RNA and antigen had been detected in the neurohypophysis but not in the adenohypophysis. 33 Of note, in contrast to other species, including alpacas, the intermediate lobe of the adenohypophysis can be very small in size or entirely absent in adult humans. Therefore, species-specific anatomy may affect the viral distribution and must be considered also for BoDV-1 infection.
In summary, the BD-affected alpacas in this study developed a predominant lymphocytic meningoencephalitis with striking interindividual differences regarding the distribution and severity of the inflammation. BoDV-1 antigen was predominantly detected in the CNS and only rarely in extraneural organs, where it was restricted exclusively to supplying nerves. The viral distribution showed conspicuous parallels to other spillover hosts (Supplemental Table S6) and underlines that alpacas, like other nonreservoir hosts, likely represent dead-end hosts that do not shed and transmit BoDV-1. Of note, BoDV-1 was able to infect non-neuronal glandular cells of the adenohypophyseal intermediate lobe in alpacas and horses, which had not been described previously.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231185107 – Supplemental material for Borna disease virus 1 infection in alpacas: Comparison of pathological lesions and viral distribution to other dead-end hosts
Supplemental material, sj-pdf-1-vet-10.1177_03009858231185107 for Borna disease virus 1 infection in alpacas: Comparison of pathological lesions and viral distribution to other dead-end hosts by Jenny Fürstenau, Madita T. Richter, Nancy A. Erickson, Reinhard Große, Kerstin E. Müller, Daniel Nobach, Christiane Herden, Dennis Rubbenstroth and Lars Mundhenk in Veterinary Pathology
Footnotes
Acknowledgements
We thank Nicole Huth, Kathrin Steffen, and Arnt Ebinger for technical support.
Authors’ Note
The manuscript is prepared in the Uniform Requirements format.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was in part funded by the Federal Ministry of Education and Research within the Research Network Zoonotic Infections to the “Zoonotic Bornavirus Consortium” (ZooBoCo; grant no. 01KI2005A to DR, no. 01KI1722E/01KI2005E to CH).
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
