Abstract
To investigate the molecular mechanisms of the oncogenic effects of avian leukosis virus subgroup J (ALV-J), we examined mutations in and the expression of p53 in the myelocytomas distributed in the liver, spleen, trachea, and bone marrow, as well as in fibrosarcomas in the abdominal cavity and hemangiomas in skin from chickens that were naturally or experimentally infected with ALV-J. Two types of mutations in the p53 gene were detected in myelocytomas of both the experimentally infected and the naturally infected chickens and included point mutations and deletions. Two of the point mutations have not been reported previously. Partial complementary DNA clones with a 122-bp deletion in the p53 gene ORF and a 15-bp deletion in the C-terminus were identified in the myelocytomas. In addition, moderate expression of the mutant p53 protein was detected in the myelocytomas that were distributed in the liver, trachea, spleen, and bone marrow. Mutant p53 protein was not detected in the subcutaneous hemangiomas or in the abdominal fibrosarcomas associated with natural and experimental ALV-J infection, respectively. These results identify mutations associated with abnormal expression of p53 in ALV-J–associated myelocytomas, suggesting a role in tumorigenesis.
Avian leukosis virus subgroup J (ALV-J) induces myelocytomas, myeloid leukosis (ML), nephromas, fibrosarcomas, and other types of malignancies in chickens. 6 Recent studies have shown that the malignant transformation of normal cells and the occurrence and development of malignant tumors result from the abnormal structure, function, expression, and regulation of oncogenes and tumor suppressor genes. The physiological roles of the p53 gene include inducing apoptosis and differentiation of abnormal cells, protecting the integrity of the genome, and inhibiting tumor cell growth. The p53 gene is closely associated with the onset and development of tumors. Mutations in the p53 gene and the expression of mutant p53 protein may be used as indicators of malignant tumors.
In the present study, we collected egg-type chickens that were suspected to be naturally infected with ALV-J and performed histopathologic analyses, isolated and identified the virus, and then experimentally infected specific pathogen-free (SPF) chickens with the isolated strain of the ALV-J virus. To determine whether p53 mutations were involved in the pathogenesis of chicken tumors resulting from exposure to ALV-J, we analyzed the molecular structure of the chicken p53 gene and the expression of the p53 protein. Here, we report that the tumors possessed mutations in the gene and demonstrate the expression of a mutant p53 protein.
Materials and Methods
Ethics Statement
All animal experiments were approved by the ethical review board of Harbin Veterinary Research Institute (HVRI) of the Chinese Academy of Agricultural Sciences (CAAS) and performed in accordance with approved animal care guidelines and protocols. The animal ethics committee approval number is Heilongjiang-SYXK-2006–032.
Clinical Sample Collection and Processing
Four affected commercial layers from the Henan Province of China were selected. Tissues, including skin with hemangiomas, as well as tumors from the liver, spleen, trachea, and bone marrow, were collected. One portion of each tissue sample was fixed in 10% formalin solution for 24 hours and then dehydrated, embedded in paraffin, and cut into 5-μm sections. Sections were stained with hematoxylin and eosin (HE) or indirect immunohistochemical staining to determine the expression of the p53 protein. Another portion of each sample was stored at −70°C for subsequent nucleic acid (genomic RNA and viral DNA) extraction, virus isolation, and p53 mutation analysis.
Detection of ALV-J
Ground suspensions of the above tissues were inoculated onto DF-1 cells, and then extracts of genomic DNA were tested by polymerase chain reaction (PCR) for ALV-J with H5/H7 primers and ALV-A and ALV-B with H5/AD1 primers by PCR testing. 8 ALV p27 antigen was detected in the cell suspensions, and indirect fluorescence antibody (IFA) testing was conducted with the ALV-J–specific monoclonal antibody (mAb) JE9 on the clinical samples. 7 Uninfected DF-1 monolayers were simultaneously used as a negative control. Specific primers and the genomic DNA templates were used to detect other suspected viruses, including Marek’s disease virus (MDV), 3 chicken infectious anemia virus (CIAV), 4 and reticuloendotheliosis virus (REV), 3 which may be associated with the disease.
Animal Experiments
A total of eighty 5-day-old SPF chicken embryos were selected for the study and were divided into 2 groups. Group I (n = 50) was infected with an isolated ALV-J strain (4000 TCID50; the inoculum volume was 0.1 ml) via the yolk sac inoculation method, and group II (n = 30) received Dulbecco’s modified Eagle’s medium (DMEM) as a control. The 24 of 30 surviving chickens in group I and 20 of 26 surviving chickens in group II were placed in isolators and raised to the age of 35 weeks. The tissues collected from the experimental group were processed in the same manner as the clinical samples.
Immunohistochemical Analysis
Monoclonal mouse anti-p53 antibody and biotinylated goat anti–rabbit or goat anti–mouse IgG secondary antibodies were used, and conditions were optimized for indirect immunohistochemical staining. 11 Samples that were treated with phosphate-buffered saline (PBS) instead of the primary antibody were used as negative controls and produced negative results in the immunohistochemical analyses.
Amplification and Sequence Analysis of p53
Three pairs of primers that covered the full-length open reading frame of the p53 gene were designed based on the published complementary DNA (cDNA) sequence of the avian p53 gene (GenBank accession number X13057.1). Using gene-sequencing techniques, p53 mutations were assessed in genomic RNA extracted from the tumor nodules in the abdominal cavity; in the skin hemangiomas; in the tumors in the bone marrow, liver, spleen, and trachea; and in the normal tissue. These sequences were compared between the chickens that had been naturally infected with ALV-J, the SPF chickens that had been experimentally infected with ALV-J p53, and the SPF chickens that had been inoculated with DMEM (control group). The p53 sequence was analyzed to identify p53 mutations that arose during ALV-J infection.
Results
Clinical Signs and Gross Lesions
At the age of 17 weeks, the naturally affected chicken flock had mortality rates that ranged from 0.5% to 1% per week. The affected chickens were thin, anemic, and dehydrated. Subcutaneous hemangiomas ranging from 5 to 30 mm in diameter were observed on the claws, breasts, wings, and internal organs of a number of the chickens. Gross examination revealed visible yellow-white tumors in the liver, spleen, and trachea. Following experimental inoculation with the ALV-J virus isolated from clinically affected animals, experimentally infected chickens developed the same clinical signs and gross lesions in the liver, spleen, and trachea. Unlike the naturally affected chickens, 2 experimentally infected chickens developed nodular tumors in the abdominal cavity.
Histopathology and Detection of ALV-J Infection
Based on the PCR results, histopathologic examinations, and virus isolations, it was concluded that all tumor samples were infected with ALV-J and that none were infected with MDV, REV, or ALV-A and ALV-B.
Histopathologic examination of the tissues from the naturally infected chickens and the experimentally infected chickens revealed that the yellow-white tumors in the liver, spleen, and trachea primarily consisted of myeloid tumor cells that contained cytoplasmic eosinophilic granules (Fig. 1). The hemangiomas consisted of large subcutaneous blood-filled cavities that were lined by endothelial cells. The nodular tumors in the abdominal cavity of 2 experimentally infected chickens were formed by neoplastic spindle-shaped cells arranged in interlacing bundles and sheets consistent with fibrosarcomas (Fig. 2), not detected in naturally affected chickens. All 4 naturally affected chickens had both hemangiomas and myelocytomas. Of the and experimentally infected chickens, 2 developed hemangiomas, 2 developed myelocytomas, and 2 developed fibrosarcomas.
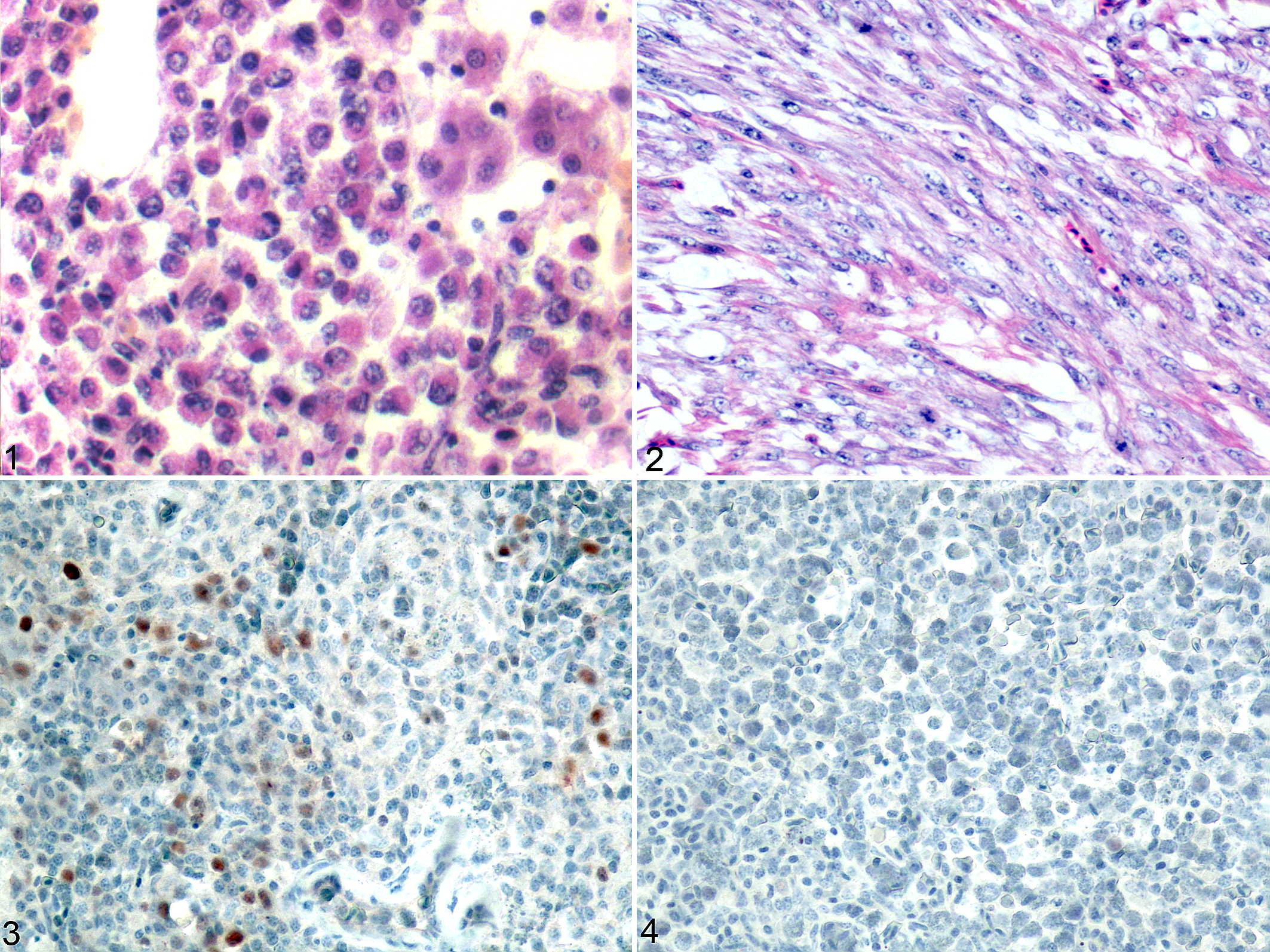
Tumors associated with avian leukosis virus subgroup J (ALV-J) infection; chickens.
Analysis of the Expression of the p53 Protein
In the spleens of the experimentally infected chickens and the clinically collected sick chickens, the nuclei of the myeloma cells that underwent multifocal proliferation were stained brown by immunohistochemistry (Fig. 3). Positive staining was also detected in the tumor cells in the liver and trachea tissues but not in the hemangiomas or fibroblastomas. Neither positively stained cells nor positively stained proteins were detected in the samples without tumor cells or the samples obtained from the control group (Fig. 4).
Analyses of p53 Gene Mutations
cDNA samples that were obtained from tissues of the chickens infected naturally with ALV-J (clinical samples), the SPF chickens experimentally infected with the ALV-J strain hn10py01, and the SPF chickens given DMEM instead of virus were PCR amplified using the primer pairs p53F58/p53R522, p53F346/p53R1182, and p53F496/p53R1182 (Suppl. Table S1). DNA fragments with sizes of approximately 460 bp, 680 bp, 560 bp, and 840 bp were successfully amplified from the 3 group. The nucleotide sequences of these amplified DNA fragments of p53 cDNA corresponded to positions 58 to 1182 in the p53 open reading frame for all samples (Fig. 5). The tumor samples and normal tissue were compared with the wild-type chicken p53 cDNA sequence that has been reported previously. The results revealed that mutations in the p53 gene were detected in tumor samples from both the experimentally infected group and the naturally affected group, and these mutations were not detected in tissues from the control group. Two types of mutations, deletions and point mutations, were detected in the p53 gene. The point mutations included single-point mutations and multiple-point mutations, and their locations are presented in Supplemental Table S2. No mutations were identified in the hemangiomas or fibrosarcomas. The point mutations were not identical across the same tumor type from different chickens. Deletions mutations were located between 1011–1025 bp and between 741–862 bp compared to the reference sequence (GenBank accession number: X13057.1; Fig. 5).
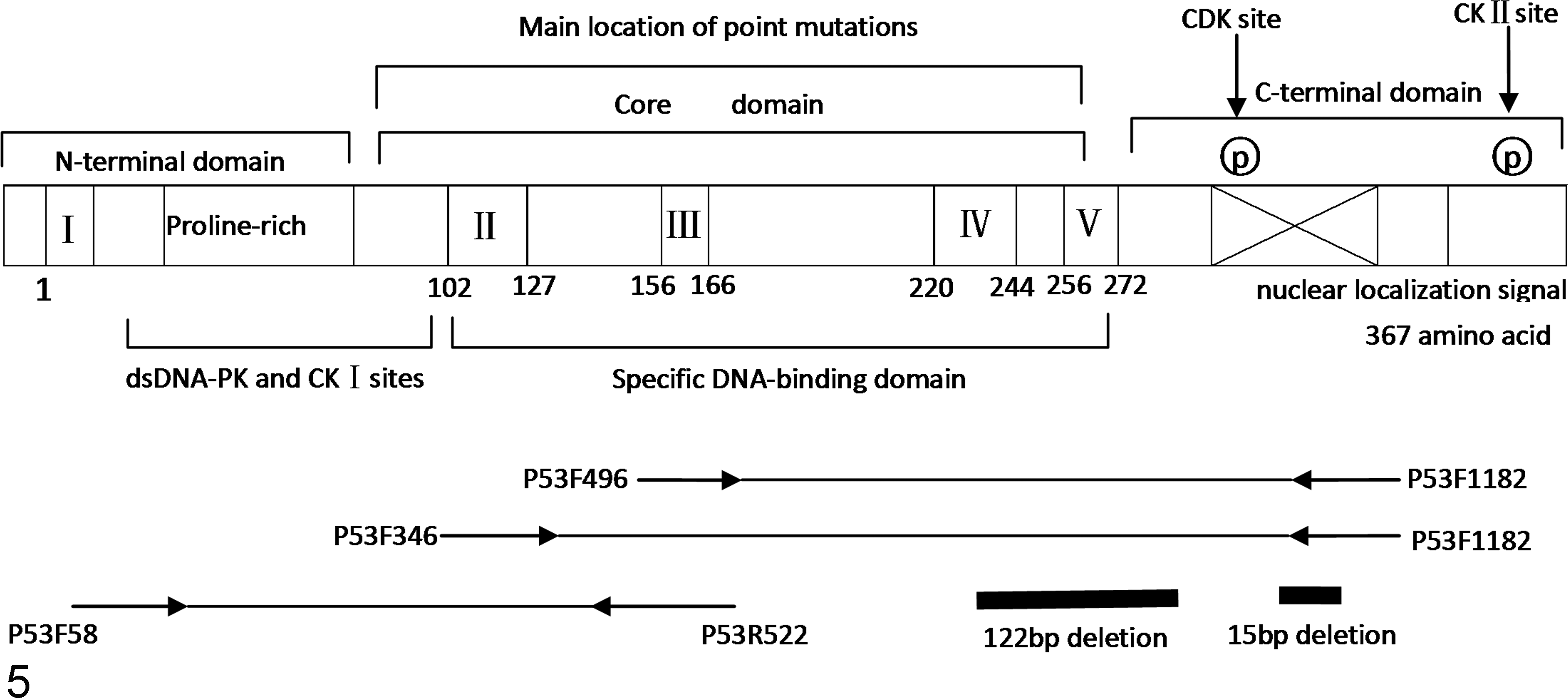
Schematic diagram of the structure of the p53 gene and the locations of the deletion mutations.
Discussion
The loss of normal p53 function is primarily caused by gene mutations. 1 Previous studies have shown that p53 gene mutations, including point mutations and deletions, occur in a variety of neoplastic cells. Of the mutations in the p53 gene, 95% are located in regions that encode the sequence-specific DNA-binding domain of the p53 protein (amino acid residues 102–292), which contains the highly conserved functional domains II to V. The locations and types of mutations in the p53 gene vary across different tumors. Mutations in the p53 gene primarily occur in exons 5 to 8 (amino acids 129–146, 171–179, 234–260, 270–287), which are called “hotspots.” The mutations are predominantly point mutations, followed by base deletions and insertions; both of the latter mutations typically result in missense mutations. 2 Of the 393 codons in the human p53 gene, mutations have been found in 280.
In neoplastic diseases of poultry, the mutational hotspots in the p53 gene in MD cell lines and clinical samples of ALV infection are at codons 146, 188, 202, 253, and 356. Among these hotspots, mutations occur most frequently at codon 146. 5 In the present study, multiple types of p53 gene mutations, mainly point mutations, were detected in tumors from chickens that were experimentally or naturally infected with ALV-J. In addition to point mutations 236, 243, and 277, we detected other point mutations that were not localized to the “hotspots.”
The mutations occurred most frequently at codons 277 and 289. However, mutations at other locations, such as codons 218 and 263, were also common. The present study also demonstrated the coexistence of mutations at 2 or more locations in the same sample. These results suggest that the mutation spectrum of the p53 gene in the experimental group and in the clinical samples was rather broad.
Interestingly, 2 short forms of the p53 transcript were detected in the bone marrow obtained from the experimental group that was infected with ALV-J and from the naturally affected group. The deleted sequences were located in the regions that encode the DNA-binding domain and the C-terminal basic region of the p53 protein, which corresponds to bps 741 to 862 and 1011 to 1025, respectively, of the open reading frame in the reference sequence. The deletion of bps 741 to 862 are predicted to result in a frameshift with formation of missense mutations that would lead to the premature termination of translation at amino acid 269. The deletion of bps 1011 to 1025 would also cause a frameshift, resulting in the alteration of all amino acids downstream of residue 336. This is the first report to detect the deletion of bps 1011 to 1025 in chicken tumors. The DNA-binding domain and the C-terminal basic region of the p53 protein contain the nuclear localization signals, the p53 oligomerization domain, and a domain that mediates the non-sequence-specific interaction of p53 with DNA and RNA. 9,10 The present study revealed that, in the ALV-J infected chickens, deletions and point mutations occurred in p53 in the region that encodes the p53 C-terminal domain and the DNA-binding domain. In addition, nonsense mutations caused by 122-bp and 15-bp deletions in the regions that encode the DNA-binding domain and the C-terminal domain, respectively, resulted in the complete loss of the p53 C-terminal domain. The loss of the C-terminal domain may affect the formation of the tetramer and prevent the nuclear localization of the p53 protein and thereby result in the loss of normal p53 function.
In the present study, abnormal expression of mutant p53 was detected by immunohistochemistry in the tumor cells of the bone marrow, liver, spleen, and trachea. However, our results do not provide sufficient evidence to determine the specific mutation loci that influenced the expression of the p53 protein.
Our results indicate that mutation of the p53 gene occurs in tumors resulting from ALV-J infection and suggest a role in tumorigenesis of these mutations in and overexpression of p53. In the chickens that were naturally infected with ALV-J and the chickens that were experimentally infected with the same viral strain, some parts of the mutations in the p53 gene were identical (eg, the deletions of bps 1011–1025), and others were different (eg, the point mutations and deletions in other regions).
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is funded by the National Natural Science Foundation of China (31172296), the Modern Agro-industry Technology Research System (no. nycytx-42-G3-01) and the State Key Laboratory of Veterinary Biotechnology Foundation (SKLVBF201005).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
