Abstract
Ringtail is a pathologic condition of laboratory rodents characterized by annular constrictions of the tail. Traditionally, it is classified as an environmental disorder caused by low relative humidity, but other factors (temperature, dietary deficiencies, genetic susceptibility, and caging type) have also been proposed. Twenty litters of mice with ringtail lesions occurred from September 2010 to August 2013 in a facility located in the northern Italy. Mice were maintained under controlled environmental conditions and fed a standard diet. Retrospective analysis of environmental data (relative humidity, temperature) was carried out. Gross, histopathologic, scanning, and transmission electron microscopy examination of tails and limbs was performed. The incidence of ringtail was 0.075% (20/26 800) of all weaned litters over the 3-year period of examination. Temperature and relative humidity remained within accepted limits in all cases except one. We observed annular constrictions in tail, digits of pes, crus, and antebrachium in 116 (100.0%), 47 (40.5%), 11 (9.5%), and 2 (1.7%) of 116 affected mice, respectively. Histologic and ultrastructural examination revealed abnormal keratin desquamation and presence of a keratin ring encircling the tail, causing progressive strangulation of the growing tail with subsequent compression and ulceration of underlying soft tissues, resulting in circulatory changes (edema, hyperemia, thrombosis, hemorrhages), ischemic necrosis, and eventually auto-amputation distal to the constriction. On the basis of our findings, we suggest a disorder of cornification as the primary lesion of ringtail in mice. The cause of these cases, however, remained undetermined, even though traditional etiologic factors (relative humidity, temperature, diet, caging type) were reasonably excluded.
Ringtail is a pathologic condition of laboratory rodents characterized by annular constrictions affecting the tail and less frequently the hindlimbs, leading to distal necrosis and eventually spontaneous auto-amputation. 17,31 Ringtail has been mainly reported in laboratory rats (Rattus norvegicus), occurring either as a spontaneous disease in preweaning animals 3,4,16,29,31 or as an experimentally induced condition in adults. 1,2,7,9 Spontaneous cases of ringtail have been also described in the white-tailed hamster (Mystromys albicaudatus) 26 and the pouched mouse (Saccostomus campestris). 5 With regard to laboratory mice (Mus musculus), spontaneous cases of ringtail are sporadically observed and commonly reported in textbooks and reviews dealing with pathology of laboratory animals. 14,17 However, after a careful examination of the literature, we found only a brief report describing ringtail in a Swiss albino mice colony in India. 22 Lesions resembling ringtail were also reported in genetically engineered mice (GEM) with targeted mutations of genes involved in epidermal differentiation. 19,24,25,27
Although ringtail is well known, the cause and pathogenesis of this condition are not completely understood. Traditionally, it is considered an environmental disorder, and the main recognized cause is low relative humidity (<25%–40%). 17 Accordingly, ringtail is reported to be most frequent and most severe during winter and early spring, when the relative humidity level tends to be the lowest, and rare or absent in the summer. 29,31 Other factors, such as environmental temperature, dietary deficiencies (fatty acids, B vitamins, and zinc), genetic susceptibility, caging type, and degree of hydration, have been also proposed and may play a role in the incidence of the disease. 6,17,31 Even though ringtail is traditionally believed to be caused by the aforementioned factors, the etiology of most recent cases is elusive since nowadays laboratory animals are maintained under strict environmental conditions and fed standard and balanced diets. 3
Histopathologic examination of ringtail in rats identified epidermal hyperplasia and orthokeratotic/parakeratotic hyperkeratosis as the main features and likely the primary lesions of ringtail. In severe cases, vascular thrombosis and necrosis of underlying dermis were also described. 3,4 Previously, it was suggested that the low relative humidity causes cooling by evaporation of the naked newborn rat, leading to vasoconstriction in an attempt to reduce the heat loss. If vasoconstriction is prolonged for some time, necrosis of the tail may then result. 16,31
Several cases of ringtail in mice occurred in a facility over a 3-year period. The purpose of this study was to analyze the environmental data and characterize the gross, microscopic, and ultrastructural features of these cases of ringtail to shed light on the cause and pathogenesis of this still poorly understood condition.
Materials and Methods
Animals
We examined 20 litters with ringtail lesions, comprising a total of 116 affected mice. Ten unaffected littermates and 5 additional mice from 2 unaffected litters (2 female 21-day-old mice on C57BL/6 background and 3 male 21-day-old mice on FVB background strain) were used as controls. The strain details are as follows: FVB/NCrl (Charles River, Calco, Italy) or FVB/NHanHsd (Harlan Laboratories, San Pietro al Natisone, Italy); and C57BL/6J (Charles River, Calco, Italy) or C57BL/6JOlaHsd (Harlan Laboratories, San Pietro al Natisone, Italy). The cases occurred over 36 months (from September 2010 to August 2013) in 2 separate units (A and B) of a mouse facility located in the northern Italy. The facility housed approximately 350 different lines of GEM in different genetic backgrounds (predominantly C57BL/6 and FVB). Animals were maintained according to the guidelines set out in Commission Recommendation 2007/526/EC of 18 June 2007, for the accommodation and care of animals used for experimental and other scientific purposes, and were used in experimental procedures in accordance with the Italian laws (D.L.vo 116/92 and following additions), which enforces the Council Directive 86/609/EEC of 24 November 1986, on the approximation of laws, regulations, and administrative provisions of the member states regarding the protection of animals used for experimental and other scientific purposes.
Mice were housed in individually ventilated caging (IVC) systems (Sealsafe and Sealsafe Plus; Tecniplast, Buguggiate, Italy) equipped with polysulfone solid-bottom cages, provided with autoclaved sawdust bedding (Lignocel 3/4; Rettenmaier & Sohne, Ellwangen-Holzmühle, Germany) autoclaved diet (Teklad 2018 S Global 18% Protein Rodent Diet Sterilizable; Harlan Teklad Diets, Madison, WI), and autoclaved water ad libitum. A Mouse House (Tecniplast, Buguggiate, Italy) was added to each cage as the only environmental enrichment, and no nesting material was provided. Cages were checked every day by the animal care staff and changed every 2 weeks in a laminar flow changing station (CS5 Evo; Tecniplast, Buguggiate, Italy). The IVC systems consisted of 1 or 2 double-sided IVC racks connected to an air-handling unit, whose effluents were exhausted directly into the building’s heating, ventilation, and air condition (HVAC) system and set up in positive pressure at 75 air changes per hour. Animal holding rooms received 15 fresh air changes per hour. Temperature was controlled at room level at 22° ± 2°C, and relative humidity was controlled at unit level at 55% ± 10%. The 2 units were located in 2 separate buildings with independent HVAC systems and dedicated washing areas with independent steam sterilizers. Unit A was established in 2008 and housed approximately 6000 IVC cages. Unit B was established in 2004 and housed approximately 3000 IVC cages. From September 2010 to August 2013, the total number of weaned litters was approximately 12 700 in unit A and 14 100 in unit B.
The 2 units were included in a health monitoring program developed in accordance with Federation of European Laboratory Animal Science Associations (FELASA) guidelines 15 and tested negative for ecto- and endoparasites and for the following viruses and bacteria: mouse hepatitis virus, mouse rotavirus (EDIM), minute virus of mice, mouse parvovirus, pneumonia virus of mice, Sendai virus, Theiler’s murine encephalomyelitis virus, ectromelia virus, lymphocytic choriomeningitis virus, mouse adenovirus, mouse cytomegalovirus, reovirus type 3, Haantan virus, mouse K virus, mouse polyoma virus, mouse thymic virus, Citrobacter rodentium, Clostridium piliforme, Corynebacterium kutscheri, Mycoplasma spp, Salmonella spp, streptococci β-hemolytic (not group D), Streptococcus pneumoniae, Streptobacillus moniliformis, Bordetella bronchiseptica, and cilia-associated respiratory (CAR) bacillus. Both the units were positive for murine norovirus. Unit A housed colonies known to be positive for Helicobacter spp and Pasteurella spp, while unit B regularly tested negative also for these agents.
Environmental Data Monitoring and Analysis
Retrospective analysis of environmental data was carried out by careful examination of the temperature and relative humidity data recorded by both the Building Management System (BMS) (Sauter, Basel, Switzerland) and the IVC air handling units extending from 5 days before the putative date of birth until the day of clinical observation of ringtail of each affected litter. The BMS measured the temperature of each room of the unit and the relative humidity of the total air exhausted from all rooms of the whole unit (including animal holding rooms, IVC systems, corridors, cage-washing areas, etc), and the measurements were automatically recorded every 15 minutes. The IVC air-handling units measured the temperature and relative humidity of the air exhausted from the racks (1 or 2) serviced by each unit, and alarms were set for temperature below 20°C or above 25°C, as well as for relative humidity below 40% and above 70%. Temperature, relative humidity, and alarms displayed by each air-handling unit were manually recorded by the animal care staff on a daily basis.
For comparison, the relative humidity recorded by the BMS was analyzed in the 2 units in a control period when no ringtail lesions were reported. The interval considered was from May to November 2012 (6 months) in unit A and from December 2012 to July 2013 (7 months) in unit B.
Gross Examination
Affected litters were sacrificed as soon as possible after the clinical observation of ringtail lesions. Immediately after euthanasia, all affected and control mice underwent whole-body fixation (ie, thoracic and abdominal cavities were opened and the whole mouse was fixed in 10% neutral buffered formalin for at least 48 hours) until necropsy was performed. Macroscopically, affected tails and limbs were classified on the basis of the severity of the lesions as follows: grade 1, mild annular constrictions in otherwise normal tail/limb; grade 2, moderate annular constrictions with edema of the tail/limb; grade 3, severe annular constrictions with distal hemorrhage and/or necrosis; and grade 4, loss of distal portions (auto-amputation) (Fig. 1). In addition, the number and localization (proximal, middle, distal third) of annular constrictions in the tail and the localization of annular constriction in the digits of the pes were recorded for each affected mouse.

Macroscopic grading of ringtail in mice, classified on the basis of the severity of the lesions as follows: grade 0, no lesions; grade 1, mild annular constrictions in otherwise normal tail; grade 2, moderate annular constrictions with edema of the tail; grade 3, severe annular constrictions with distal hemorrhage and/or necrosis; and grade 4, loss of distal portions (auto-amputation).
Histopathology
Examined tissues included tail and limbs (after decalcification in 10% EDTA w/v) and main organs (dorsal haired skin, lungs, heart, thymus, liver, spleen, pancreas, gastrointestinal tract, kidneys, and adrenal glands) of case Nos. 1 to 12. All tissues were embedded in paraffin, sectioned at 4 μm, and stained with hematoxylin and eosin (HE) for standard histopathologic examination. To clarify the progression of ringtail, we examined tails and pedes according to their macroscopic grade of lesion.
Scanning Electron Microscopy Analysis
Samples of formalin-fixed tail and pes from 1 control and 3 affected mice with grade 1 and 2 lesions (case No. 13) were prepared for scanning electron microscopy (SEM) using established procedures. Briefly, samples were fixed with glutaraldehyde 1.2% in Na cacodylate buffer 0.1 M and postfixed with osmium tetroxide (OsO4) 1% in Na cacodylate buffer 0.1 M. After removing the OsO4 solution, samples were gradually dehydrated with an ethanol series and dried with hexamethyldisilazane. When specimens were dried, they were sputtered (Polaron E5100 Sputter Coater; Quorum Technologies Ltd, Lewes, United Kingdom) with gold and examined with a scanning electron microscope (Sigma field emission scanning electron microscope; Carl Zeiss Microscopy GmbH, Oberkochen, Germany) operated at 5 kV.
Transmission Electron Microscopy
For transmission electron microscopy (TEM) analysis, small sections of tail skin sampled from 2 control and 2 affected mice with grade 1 lesions at the level of the constriction ring (case Nos. 9, 17) were fixed with 2% glutaraldehyde and 2% paraformaldehyde in 0.15 M Na cacodylate buffer (pH 7.4) and processed for TEM observation. Briefly, samples were postfixed with osmium tetroxide (2% OsO4 in Na cacodylate buffer), rinsed, en bloc stained with 1% uranyl acetate in water, dehydrated, and embedded in epoxy resin (Epon 812; Electron Microscopy Science, Hatfield, PA) that was baked for 48 hours at 60°C. The regions of interest for the ultrastructural analyses were identified on semi-thin sections (0.5 mm) collected on glass slides, stained with 0.1% methylene blue and 0.1% toluidine blue in phosphate buffer, and observed with an optical microscope (DM 2500; Leica Microsystem, Vienna, Austria) with a 100× oil immersion objective. Thin sections were obtained with an ultramicrotome (EM UC6; Leica Microsystems, Vienna, Austria), stained with uranyl acetate and lead citrate, and examined with a Philips CM10 TEM (FEI, Eindhoven, Germany). Images were acquired with a Morada Olympus digital camera (Olympus Soft Imaging Solutions GmbH, Münster, Germany).
Results
Epidemiology
The incidence of ringtail was 0.075% (20/26 800) of the total weaned litters over the 3-year period of examination. Ringtail lesions were observed in 15 distinct lines of GEM, with prevalence ranging from 1.0% to 7.7% depending on the GEM line, and in 2 different background strains (FVB, C57BL/6). Percentage of affected mice per litter ranged between 20% and 100%. No marked sex bias was evident (M/F = 0.97). The mean age at clinical observation was 21 days (range, 8–42 days), whereas the mean age at sacrifice was 25 days (range, 12–49). In 9 of 20 litters (case Nos. 1, 2, 4, 6, 7, 9, 13, 15, 17), lesions were noticed before weaning during the routine daily checks because of their severity. The remaining cases were identified at weaning (3/20) or later (8/20). Cases were observed in both units of the facility and over the 4 seasons of the year, although most cases occurred in summer (8/20) and winter (6/20). Some litters shared common progenitors (Table 1).
List of Ringtail Cases and Summary of Environmental Data.
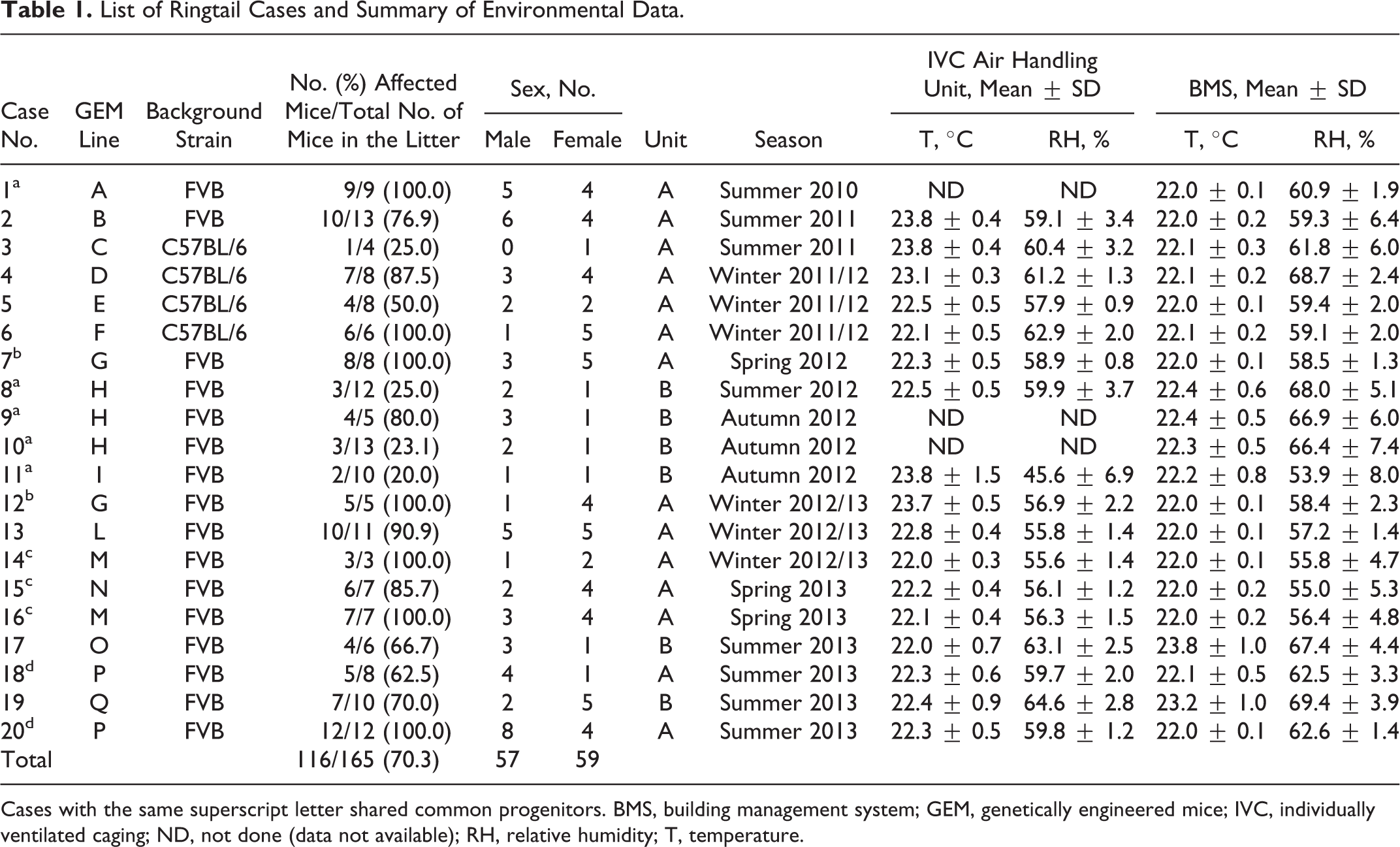
Cases with the same superscript letter shared common progenitors. BMS, building management system; GEM, genetically engineered mice; IVC, individually ventilated caging; ND, not done (data not available); RH, relative humidity; T, temperature.
Temperature and relative humidity remained within accepted limits during the examination periods with only 1 exception (case No. 11), where the relative humidity dropped below 40% the day before the reporting of the lesions. For each unit, we also examined the environmental data during a control period when no ringtail lesions occurred in the weaned litters. In unit A, only 2 short-term drops of humidity were recorded during the control period, while unit B experienced a malfunctioning of the humidifier of the air conditioning system, resulting in high fluctuations of relative humidity with frequent drops below 40%.
Gross Examination
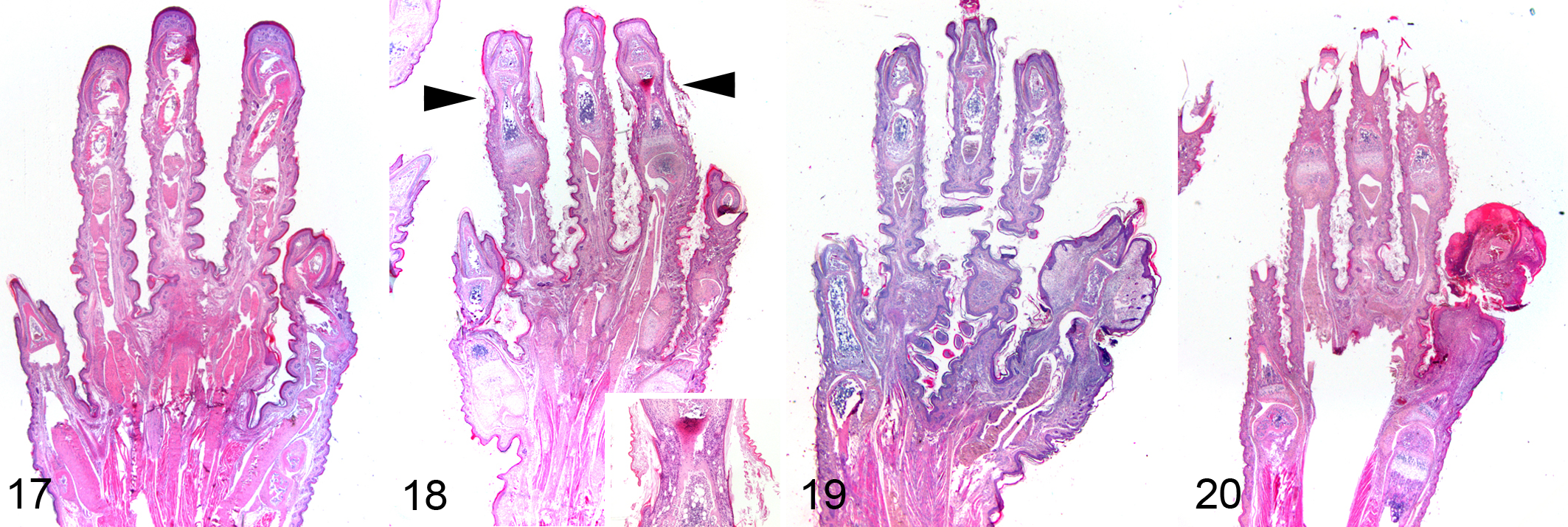
Results of the gross examination are summarized in Table 2. Annular constrictions of different grade of severity were found in tail, digits of the pes, crus, and antebrachium of 100%, 41%, 9.5%, and 2% of affected mice, respectively. Figure 2 shows an example of a litter in which all members were affected by ringtail, involving tail, digits, and crus. Tail lesions were found in all the affected mice, and lesions were most severe in the tail. Mean number of constrictions per tail was 3.2. The most affected portion of the tail was the distal third, with increasing involvement from the proximal to the distal third of the tail. Digits were affected in 11 of 20 litters, with overall moderate lesions, and the most frequently affected digit was the fifth, with increasing involvement from the first to the fifth digit. Crus and antebrachium were affected in 2 and 1 litters, respectively, and lesions were overall mild to moderate (Figs. 3, 4). Digital and limb constrictions were either monolateral or bilateral. Constrictions were restricted to the tail in 9 litters; the tail and digits in 9 litters; the tail, digits, and crus in 1 litter; and the tail, digits, crus, and antebrachium in 1 litter.
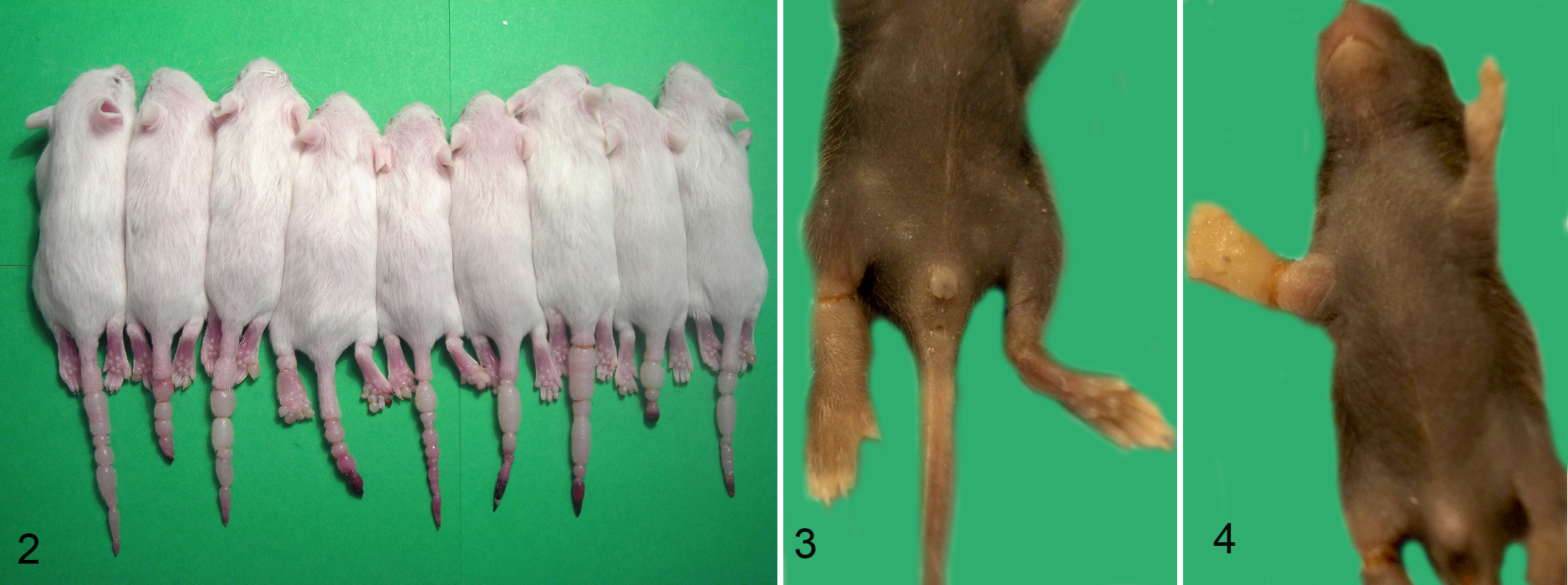
Litter of mice, 16 days old, FVB background (case No. 1). All members of this litter were affected by ringtail involving the tail, digits of the pes, and crus.
Summary of Gross Findings.
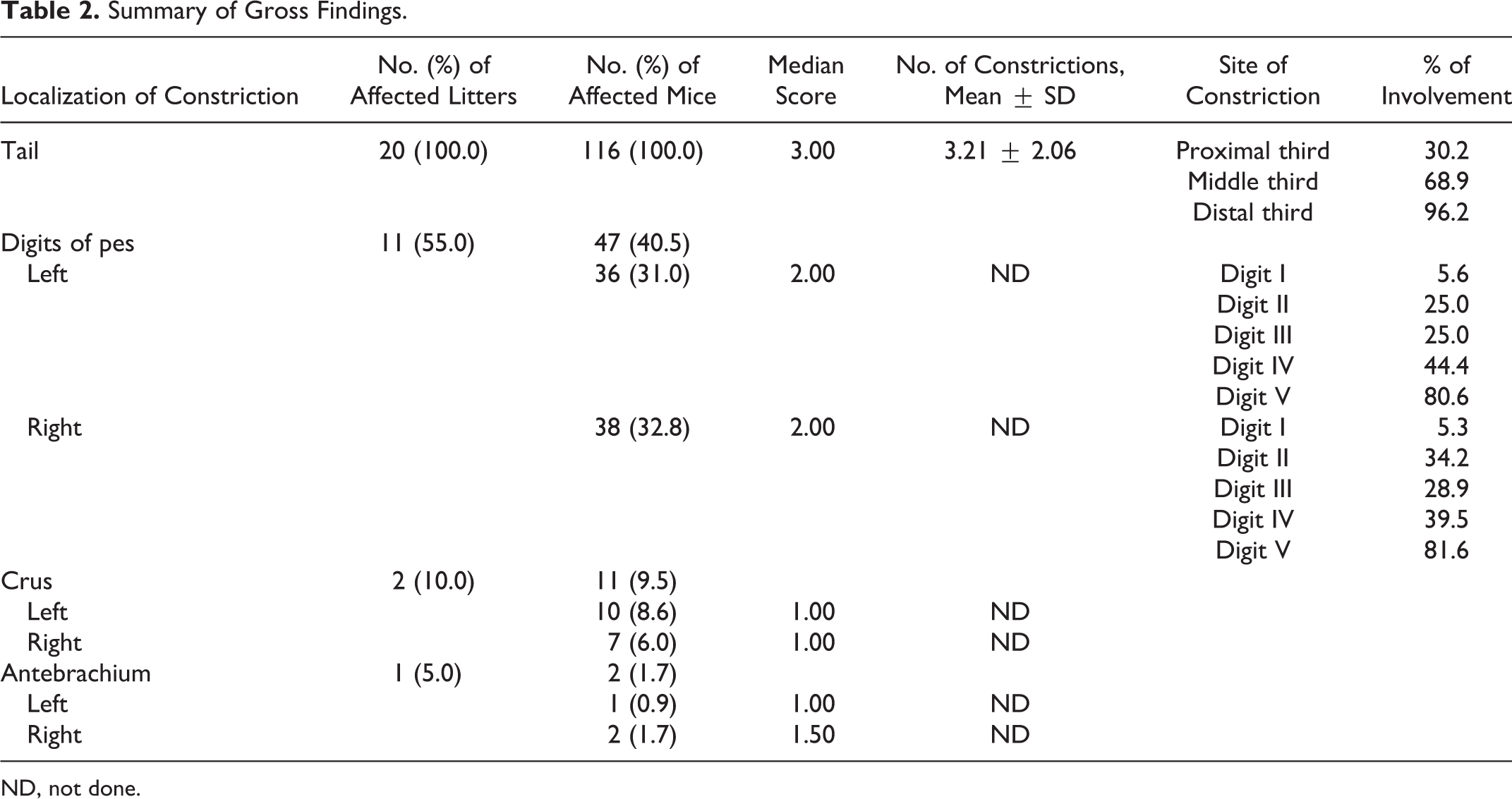
ND, not done.
Histopathology
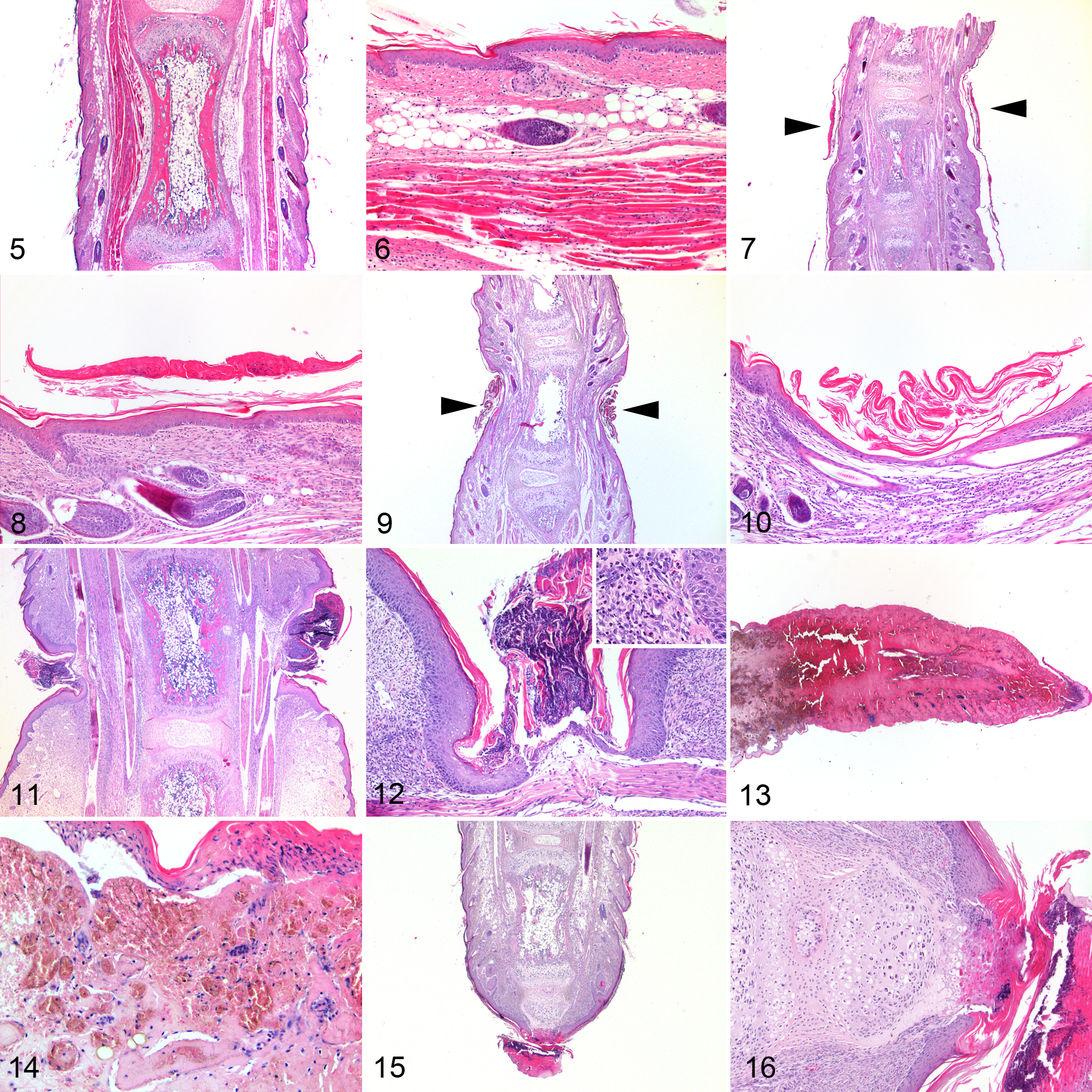
No histologic lesions were observed in the tails from control mice (Figs. 5, 6). Early grade 1 lesions had none to minimal focal constrictions associated with laminated or compact orthokeratotic hyperkeratosis overlying an epidermis of normal thickness (Figs. 7, 8). In more advanced cases of grade 1 lesions, folded keratin lamellae were found within a groove overlying a thinned epidermis (Figs. 9, 10). Grade 2 lesions had moderate to severe constrictions, and epidermis could be either thinned or ulcerated, usually moderately hyperplastic at the edges. Folded keratin lamellae were frequently found in the grooves, associated with serocellular crusts and neutrophilic inflammation of underlying soft tissues. Marked edema and lymphangectasia were evident distal to the constrictions (Figs. 11, 12). In grade 3 lesions, in addition to severe tail constriction, severe circulatory changes (hyperemia, thrombosis, hemorrhages) and ischemic coagulative necrosis of distal portions were found (Figs. 13, 14). Grade 4 lesions were characterized by discontinuity of vertebral bodies surrounded by necrosis and hemorrhages, followed by healing with progressive reepithelialization of the amputated portion (Figs. 15, 16). Similar lesions were observed in the digits of the pes (Figs. 17–20), crura, and antebrachia. No relevant pathologic changes were found in the other examined organs.
Normal tail; mouse. Note the regular profile of the tail.
Normal pes; mouse.
Scanning Electron Microscopy
In the tail and digits of the pes from the control mouse, the hairs were uniformly distributed over the skin, and the interfollicular epidermis was covered by flat overlapping layers of corneocytes (Figs. 21, 22). In the tail and digits with grade 1 lesions, a ring of keratin encircling the tail and digits was evident at the level of the constriction. The tail and digits were normal in size, but hair density was reduced and hairs were thinner and shorter compared with control. Corneocytes formed irregular scales lifting over the surface and partially entrapping the base of hair shafts (hyperkeratosis) (Figs. 23, 24). In the tail and digits of grade 2 lesions, single or multiple keratin rings at the level of constrictions appeared more tight and associated with marked enlargement of the distal portions (edema). Hair density and length were markedly reduced. Scales of corneocytes were irregular, lifting over the surface and diffusely attached to the hairs (hyperkeratosis) (Figs. 25, 26).
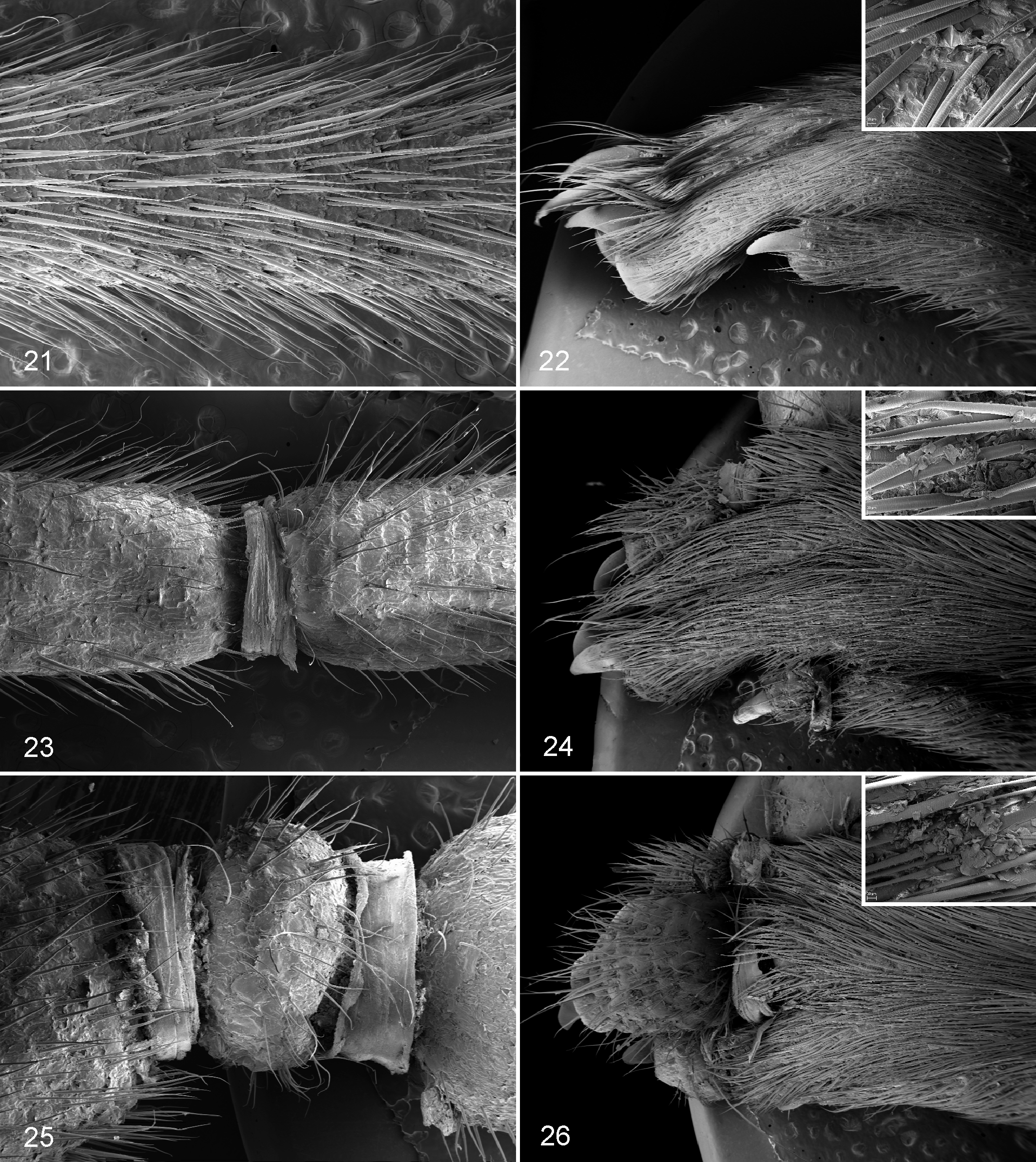
Normal tail; mouse. Tail has a regular profile, and hairs are uniformly distributed over the skin. Scanning electron microscopy (SEM).
Transmission Electron Microscopy
Ultrastructural changes were restricted to the upper stratum corneum of the tail epidermis, located at the level of the annular constrictions of both mice affected by grade 1 ringtail lesions, while underlying epidermal layers appeared normal. In the control mice, the stratum corneum was composed of flat corneocytes that progressively separated in the uppermost portions, which is consistent with normal skin desquamation. Corneocytes were characterized by a fibrillar to homogeneous electron-dense cytoplasm, devoid of recognizable nucleus and organelles (Fig. 27). Mice with ringtail had increased cohesiveness of the upper cornified layers compared with the control mouse, with retention of a thin layer of intercellular lipid matrix. In addition, corneocytes had intracytoplasmic oval to irregularly shaped, homogeneous to granular, variably electron-dense inclusions (lipid inclusions) (Fig. 28).
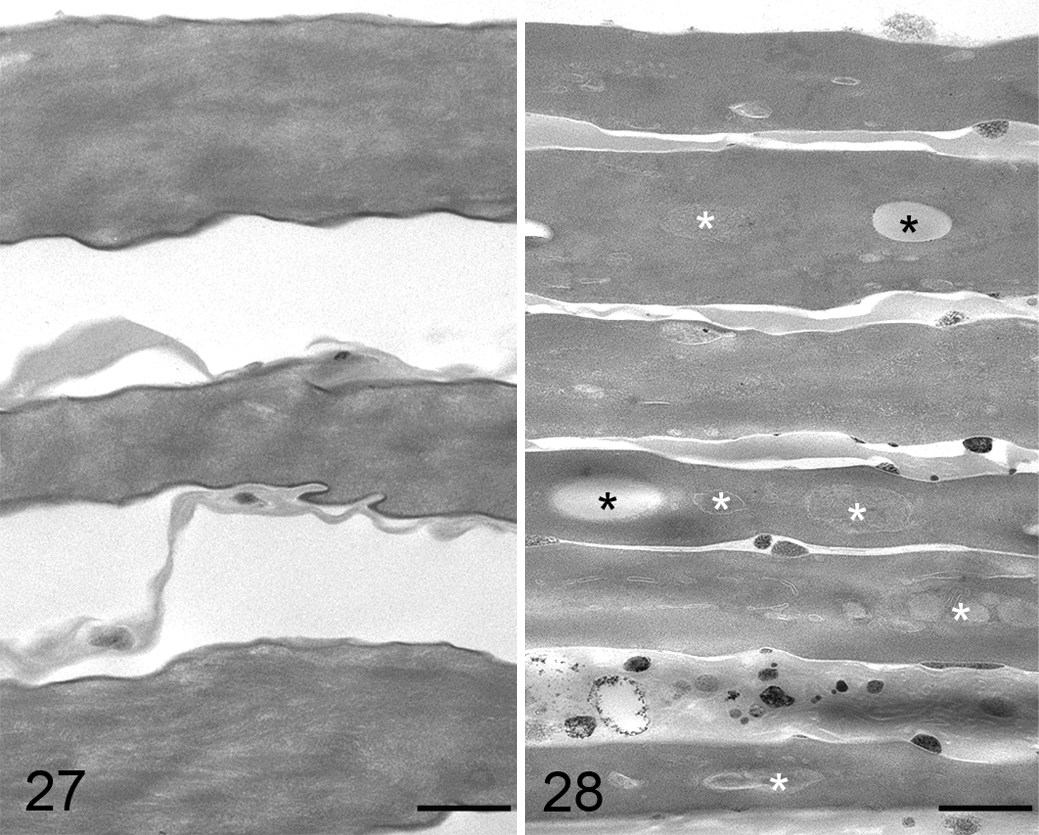
Tail, epidermis, control mouse. Upper portion of the stratum corneum composed of regular layers of flat corneocytes undergoing progressive detachment. Corneocytes have a homogeneous electron-dense cytoplasm devoid of nucleus and cytoplasmic organelles. Transmission electron microscopy (TEM). Bar = 500 nm.
Discussion
Cases of ringtail in mice were observed in a facility located in the northern Italy from September 2010 to August 2013. No cases were recorded before this period, even though facility B was established in 2004 and over the years experienced some failures in environmental humidity control. The overall incidence of ringtail was very low, reaching 0.075% of all weaned litters over this 3-year period of examination. The mean age at observation was 21 days, but lesions were most likely already present at an early age, as reported for rats in which ringtail is commonly observed in preweaning animals and can be observed as early as 2 days after birth. 3,4,16,29,31
Detailed gross evaluation of affected litters revealed that the tail was the most frequently affected site, followed by the digits of the pes. We observed, for the first time, constrictions also at the level of the crus and antebrachium in 2 and 1 of 20 affected litters, respectively. Constrictions were restricted to the appendages, and there was an increasing involvement from the proximal to the distal portion of the tail and from the first to the fifth digits of the pes. Since the distal portion of the tail and the lateral digits of the pes are more exposed and likely more prone to suffer repeated mechanical injuries than proximal tail and medial digits, a potential role of trauma as a cofactor in the development of ringtail is suspected, although its contribution is not clear.
Histologically, the mildest lesion we found was compact or laminated orthokeratotic hyperkeratosis overlying an epidermis of normal thickness. Successively, folded keratin lamellae were observed within a groove associated with thinning or ulceration of the epidermis, progressive compression of underlying tissues, and worsening of circulatory changes, with edema, stasis, thrombosis, and eventually ischemic coagulative necrosis of surrounding tissues. Epidermal hyperplasia, previously suggested to be a primary lesion of ringtail in rats, 3 was found only at the periphery of moderate to severe constrictions, which were usually ulcerated and thus considered associated with a healing response (reepithelialization) of the wounded skin. Histologic differences regarding the presence and role of epidermal hyperplasia between mice and rats could be related to the different trimming procedures applied for histologic examination of the tail lesions (longitudinal vs cross sections), which might have led in the previous study to the examination of cross sections of the peripheral areas of the constrictions, resulting in the identification of hyperplasia and misinterpretation of this finding.
The histologic presence of folded keratin lamellae within a groove in association with compression and ulceration of underlying soft tissues prompted us to formulate the hypothesis that ringtail lesions were associated with the presence of a constricting ring of keratin around the tail. To verify this hypothesis, we analyzed a few samples of tail and pes by SEM that confirmed the presence of a keratin ring encircling the tail and digits at the level of the constrictions. Other causes of appendage constriction that can eventually lead to the sloughing of extremities, such as cotton nesting material, 17 hair fibers, or remnants of birthing material wrapping around limbs or digits, were ruled out since nesting material was not added in breeding cages and given the histologic absence of any foreign body material.
TEM analysis of the affected tails at the level of constrictions revealed the presence of ultrastructural abnormalities localized in the uppermost stratum corneum (increased cohesiveness of the upper cornified layers and intracytoplasmic inclusions within corneocytes), suggesting an impaired desquamation process, although no conclusions can be drawn about the cause of the disorder and whether these findings are the cause or consequence of the keratin ring formation.
The histopathologic and ultrastructural examination of the different macroscopic grades of ringtail allowed us to identify a keratin ring at the level of the constrictions and thus clarify at least in part the pathogenesis of ringtail lesions, although it should be noted that the grading is not directly correlated to the age of mice or the progression over time of the lesions, because the affected mice were sacrificed shortly after the clinical observation of the lesions for animal welfare reasons, and we had no opportunity to perform a follow-up study.
On the basis of our histologic (presence of compact or laminated orthokeratotic hyperkeratosis without associated epidermal hyperplasia) and ultrastructural (increased cohesiveness of corneocytes in the uppermost layers of the stratum coneum) findings, we suggest that the primary lesion of ringtail is a disorder of cornification characterized by retention hyperkeratosis and abnormal desquamation, resulting in the formation of a nondeformable and persistent keratin ring encircling the tail and causing the progressive strangulation of the rapidly growing tail of young mice. As the tail grows, the constriction becomes progressively tighter (with respect to the diameter of the tail) and causes distal blood flow alterations, eventually resulting in ischemic necrosis and auto-amputation of the distal portions.
In humans, there are lesions, called ainhum and pseudoainhum, strikingly similar to ringtail for digital localization and presentation, characterized by constricting bands of digits of hands and feet that can progress to auto-amputation of the involved body part. Ainhum (also known as dactylolysis spontanea) is a very specific spontaneous condition affecting only the fifth toe of black people whose cause is currently unknown. 8,10 Primary pseudoainhum has no racial or digit preference, can affect both hands and feet, and occurs as a result of an identifiable or associated disease process. Pseudoainhum has been described in many cases of hereditary or acquired dermatoses, usually involving defects of epidermal differentiation, including pachyonychia congenita, Vohwinkel syndrome, loricrin keratoderma, lamellar ichthyosis, mal de Meleda, fungal infection, leprosy, psoriasis, and trauma. 18,21 These human conditions share many similarities with mouse ringtail, such as the peculiar localization and clinical progression of the constrictions, the genetic predisposition (suspected in ainhum and identified in some forms of pseudoainhum), and the obscure etiopathogenesis.
The hypothesis that ringtail could be the result of a disorder of cornification is further supported by the fact that ringtail-like lesions have been described in GEM with mutations of genes involved in epidermal differentiation or integrity, such as the profilaggrin, loricrin, neutral cysteine protease bleomycin hydrolase (Blmh), and connexin26, 19,24,25,27 and that some of these GEM are models of inherited disorders that can be associated with pseudoainhum. 18,21 However, at present, we have not sufficient data to support a primary disorder of cornification, and a secondary defect of cornification due to interaction of several factors should be considered probable as well.
We can further speculate that this disorder of cornification (in terms of a defective desquamation process) is already present at birth (or develops immediately afterward) and results in the early formation of a cornified envelope around the tail (or digits), which then cracks into several rings during the rapid growth of the tail in the preweaning mice. Probably, most of these rings break up during growth, while others remain, causing the progressive constriction of the tail. In case constricting rings resolve spontaneously, no additional lesions usually develop in affected mice, suggesting that this disorder is transitory. Some similarities may be found with an uncommon human congenital condition named “collodion baby” (babies that are born encased in a tight and shiny hyperkeratotic membrane that completely sheds after a few weeks). 28,33 Collodion baby is a phenotype resulting from different congenital disorders of keratinization, and it is usually the first expression of some forms of ichthyosis (nonbullous congenital ichthyosiform erythroderma, lamellar ichthyosis, and ichthyosis vulgaris) 28, 33 or, less frequently, other genodermatoses, such as loricrin keratoderma. 12,34 Occasionally, collodion babies can resolve spontaneously (self-healing collodion baby) 13,20,30,32 and, although rarely, may develop constricting hyperkeratotic bands and eventually auto-amputation of digits (pseudoainhum). 12,23
The topical application of lanolin, an emollient used in dry skin conditions of humans, has been reported to be beneficial in the treatment of this condition in rats, although it is controversial if the effectiveness of such treatment was attributed to the moisturizing properties of lanolin or to the mechanical effect of applying the ointment to the tail, resulting in the removal of constricting rings. 29 In light of the identification of the persistent keratin ring encircling the tail, now both hypotheses appear conceivable and might work in concert (ie, mechanical removal of a previously softened keratin ring). Hence, although not tested yet, treatment by removal of constricting rings will likely result in the healing of ringtail, particularly in cases with grade 1 and 2 lesions.
In this study, the availability of BMS and IVC air-handling unit data of temperature and relative humidity allowed us to retrospectively evaluate the environmental conditions at unit, room, and rack levels. Based on this retrospective analysis, relative humidity and temperature values remained within accepted limits in all cases but one. Even if these parameters were not measured at the single cage level, our results indicate that the traditional environmental factors (low relative humidity, temperature) are probably not the primary cause of ringtail, as traditionally suggested. 17 The refutation of temperature and humidity as the traditionally accepted cause of ringtail is further supported by the absence of ringtail cases in unit B during a period of improper functioning of the humidifier of the air conditioning system, resulting in frequent periods of low environmental humidity (<40%). Diet and caging type were the same for all mice housed in the facility, and hence it is also reasonable to exclude a direct causative role of these factors in ringtail. Although cases of ringtail were observed in several GEM lines, originating from 2 distinct background strains (FVB, C57BL/6), some affected litters had common progenitors, and some GEM lines were more affected than others; thus, we cannot completely exclude that genetic factors can play a role in the development of ringtail. To our knowledge, the affected GEM lines have targeted mutations unrelated to epidermal integrity and differentiation and have no correlation with GEMs with ringtail-like lesions. 19,24,25,27 In this facility, most of the breeding cages housed a trio (1 male and 2 females), and in the case of genetic predisposition, this may account for the high variability of the percentage of affected mice per litter (ranging between 20% and 100%), since pups present in the cage could have been born to a single or both mothers.
Although it is difficult to speculate that a genetic mutation might result in a transient lesion, the skin phenotype and also the ringtail-like lesions that occurred in some GEM models were in fact transient, usually resolving by weaning or even before. 11,19,24,25,27 The study of these GEMs suggested that the transitory nature of their skin lesions might be the result of the existence a compensatory mechanism that is induced in an attempt to restore the epidermal barrier function. 11
Although this condition is traditionally considered an environmental disorder mainly caused by low relative humidity, other factors, such as environmental temperature, dietary deficiencies (fatty acids, B vitamins, and zinc), genetic susceptibility, caging type, and degree of hydration, also have been demonstrated or proposed as causative agents. 6,17 All of these etiologic factors can act alone or in combination, and usually their combination increases the incidence and/or the severity of the lesions, as demonstrated by the fact that the lowering of the relative humidity can enhance skin lesions of mice lacking Blmh 25 or that rats housed in cages with wire mesh bottoms are more prone than rats housed in solid-bottom cages to develop ringtail at low relative humidity. 6,31 Thus, all of these etiologic factors suggest that this syndrome might be the clinical expression of more than one causative agent or that more than one causative agent may be necessary to induce ringtail.
In conclusion, ringtail in laboratory mice is a rare spontaneous condition affecting the tail, digits, and less frequently the limbs of preweaning animals. The cause of the present cases of ringtail remains undetermined, even though traditional etiological factors (relative humidity, temperature, diet, caging type) were reasonably excluded, as already suggested in previous studies in laboratory rats. 3 Most likely, ringtail is a multifactorial disease, but at the moment, no conclusions can be drawn about the inherited, congenital, or acquired nature of these lesions. Although the etiology remains elusive, a disorder of cornification with abnormal desquamation is proposed as the primary lesion of ringtail, resulting in the formation of a persistent keratin ring encircling and progressively strangulating the rapidly growing tail of young mice with subsequent compression and ulceration of underlying soft tissues, resulting in circulatory changes (edema, hyperemia, thrombosis, hemorrhages), ischemic necrosis, and eventually auto-amputation distal to the constriction. Additional studies, such as analysis of proteins and genes involved in epidermal differentiation (eg, loricrin, filaggrin), and genetic studies (to assess if there is a way of transmitting this character) are necessary to further elucidate the pathogenesis of this well-known but poorly understood condition of laboratory rodents, even though the extremely low incidence of ringtail and its unpredictable onset make future studies challenging.
Footnotes
Acknowledgements
We thank Davide Marchesi for the excellent technical support for SEM analysis. A special thanks to Charles B. Clifford for his support and advice that accompanied the study.
Author Contribution
Conception or design: ES. Data acquisition, analysis, or interpretation: CR, SMB, LB, FB, MC, AG. Drafting the manuscript: CR, SMB, LB, FB, MC, AG. All authors participated in critically revising the manuscript, gave final approval, and agree to be accountable for all aspects of work to ensure integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
