Abstract
In October 2011, a fatal disease outbreak occurred in 3 civet species farmed for their use in the coffee industry in Thailand. The disease quickly killed 20 animals in a mixed population of Asian palm civets (Paradoxurus hermaphroditus; n = 18), a masked palm civet (Paguma larvata; n = 1), and small Indian civet (Viverricula indica; n = 1). Clinical signs consisted of severe lethargy, weakness, vomiting, and diarrhea with associated dehydration, dyspnea, nasal and footpad hyperkeratosis, and seizures. All civets were positive for canine morbillivirus using the commercial canine distemper virus (CDV) antigen test kit. Consistently observed necropsy findings consisted of severe pneumonia and hemorrhagic enteritis. Microscopic examination revealed severe gastroenteritis, bronchointerstitial pneumonia, lymphadenitis, necrotizing dermatitis, nonsuppurative polioencephalitis, and characteristic intranuclear/intracytoplasmic eosinophilic viral inclusions in multiple tissues. Immunohistochemical analysis revealed immunoreactivity of varying intensity, while virus isolation demonstrated typical cytopathic effects. To confirm CDV infection, reverse transcription–polymerase chain reaction against fusion (F), phosphoprotein (P), and hemagglutinin (H) genes showed bands of expected size using conjunctival swabs (9 civets, 1 dog [Canis lupus familiaris] living on the farm). Phylogenetic analyses and restriction fragment length polymorphism results indicated that the civets were infected by the Asia-1 strain of CDV commonly found in dogs in Thailand. The deduced amino acid sequences of the signaling lymphocyte activation molecule binding region of the CDV-H proteins revealed a Y549H mutation in both CDV-infected Asian palm civets (n = 4) and a co-located dog. We report a canine distemper outbreak in a civet colony with lineage classification and a Y549H mutation in noncanid species in Thailand.
Canine distemper virus (CDV) is a morbillivirus in the family Paramyxoviridae that occurs worldwide and most commonly affects members of the Canidae family. There are reports of susceptibility and infection in wildlife species as diverse as mink (Neovison vison), raccoons (Procyon lotor), civets (Paguma larvata), foxes (Vulpes vulpes), lions (Panthera leo), tigers (Panthera tigris), bears (Ursus americana), and lesser pandas (Ailurus fulgens). 1,6,15,31,33 Recently, CDV infection expanded its host range into species that are evolutionarily more distant to canids, including a nonhuman primate species (Japanese monkeys [Macaca fuscata]) and rodents (Asian marmots [Marmota caudata]). 19,23 CDV is considered one of the most lethal infectious agents in both susceptible free-living and captive carnivores, and there are frequent reports of disease in wildlife in many countries. 15,27,31 However in Thailand, where the disease is considered endemic in dogs, there are only limited studies of CDV infection of wildlife.
CDV has an unsegmented single-strand RNA genome encoding 6 structural proteins—the hemagglutinin (H), the fusion (F), the nucleocapsid (N), the phospho- (P), the large (L), and the matrix (M) protein—and 2 other nonstructural proteins, termed C and V protein. 12 The sequence analysis and genotypic characterization of the virus have been studied to describe the genetic relationship among CDV-susceptible species. Based on the alignment of the H gene, which has the highest variability within the genome, CDV has been classified into 10 lineages that include Asia-1, Asia-2, Asia-3, Europe, European wildlife, vaccine (America-1), America-2, South America, South Africa, and Arctic-like. 20,26,32,33 Recently, a novel genotype, Asia-4, was proposed, thereby suggesting that at least 3 CDV lineages circulate in the canine population in Thailand, consisting of the vaccine lineage, Asia-1, and the newly described Asia-4. 22
Clinical signs of CDV infection are relatively uniform in affected species. 6 Viral multiplication occurs in various lymphoid tissues, including the tonsils, spleen, gut-associated lymphoid tissue (GALT), and lymph nodes, and results in a systemic lymphopenia causing severe immunosuppression. 14,24,29 Clinical signs include neurological signs such as seizures; myoclonus; repetitive, involuntary chewing movement; respiratory signs; gastrointestinal signs such as vomiting and diarrhea; and “hard pad” of the nasal planum and the footpads. The severity of these signs is dependent on the distribution of the infection within the individual animal. Postmortem findings typically parallel the clinical signs with necrotizing lesions described in the lungs, gastrointestinal tract, and skin. Microscopically, the respiratory lesions typically include a mucopurulent necrotizing rhinitis, bronchointerstitial pneumonia with formation of syncytia, and characteristic morbilliviral eosinophilic intranuclear and intracytoplasmic inclusion bodies. Gastrointestinal manifestations present as gastroenteritis associated with the depletion of Peyer’s patches. Infection of the cutaneous tissues can have quite variable features but typically include a pustular dermatitis often seen on the ventral abdomen as well as the stereotypical hyperkeratosis of the footpads and nasal planum. 2 Microscopic lesions are frequently exacerbated by secondary bacterial infections in both the respiratory and gastrointestinal systems and can contribute to increased mortality rates, especially in younger susceptible animals. 2,10,24
Molecular evolution analysis of the H gene, particularly signaling lymphocyte activation molecule (SLAM) receptor binding sites at amino acid residues 530 and 549, revealed that domestic dogs were infected with the 549Y CDV strain with the exception of a 549H-infected dog (Z47762/Dog/Canis lupus familiaris). Similarly, wild canids were more frequently infected by the 549Y than the 549H strain. Although noncanids are susceptible to both strains, they are associated more frequently with the 549H than the 549Y infection. 18,25 At position 530 of CDV-H gene, there are varied amino acid changes (G, D, N, E, R, V) regardless of host species. 16,18 Recently, Bieringer et al 3 revealed that substitution at residue 540 from D to G is sufficient to allow CDV to bind to human SLAM in vitro.
Wild-caught and captive-bred civets are maintained in Thailand for the production of a specialty coffee, which is a lucrative microindustry. Various species of viverridae, including civets and binturong, are used for the passaging of coffee beans through their gastrointestinal tract and are maintained in close quarters, frequently with exposure to dogs (C. lupus familiaris) living on the same premises.
In October 2011, a CDV outbreak occurred on a private farm used for the production of specialty coffee in the Kanchanaburi province of western Thailand. This epizootic killed 100% of the farmed civets on the property, resulting in significant economic losses to the coffee producer. There is no prior report of CDV infection in civet species in Thailand. The infection has been documented in wild masked palm civets (Paguma larvata) in Japan, at which time it was suggested that dogs represented a reservoir host for that particular CDV transmission based on phylogenetic analysis on the H gene. 27 The purpose of this study was to characterize the pathological and immunohistochemical features of CDV infection in civets and to investigate the genetic relationship of this and other CDV lineages in Thailand.
Materials and Methods
Animals and Clinical Signs
The study group consisted of a mixed population of 20 captive civets on a farm producing specialty coffee (18 Asian palm civets [Paradoxurus hermaphroditus], 1 masked palm civet [Paguma larvata], and 1 small Indian civet [Viverricula indica]). The wild-caught animals’ ages ranged from 4 months to 2 years, and there was no history of vaccination against CDV. The civets were individually housed in cages separated by a narrow path that was easily accessible to stray domestic dogs living in the area.
Significant clinical signs included severe lethargy and weakness, vomiting and diarrhea with associated dehydration, respiratory distress presenting as coughing and dyspnea with concurrent purulent oculonasal discharge, nasal and footpad hyperkeratosis, and seizures (Suppl. Table S1). A dog living on the same premises was observed with mild oculonasal discharge. All evaluated civets and a sampled dog were positive for CDV infection using a commercially available CDV antigen test kit (BioNote, Gyeonggi-do, Korea).
Pathology
Postmortem examination was performed in 4 animals immediately following death. These consisted of 3 Asian palm civets (Nos. 1–3) and 1 small Indian civet (No. 4). Fresh tissues were harvested from all 4 animals (nasal planum, footpad, brain, lung, heart, kidney, spleen, liver, stomach, small and large intestine, pancreas, tongue, lymph nodes, and salivary glands, as well as the uterus from civet No. 4) and were submitted for histopathology, immunohistochemistry, and viral isolation. For histopathologic study, tissues were fixed in 10% neutral buffered formalin, processed, and further embedded in paraffin. Samples were then cut at 4 μm thick and stained with hematoxylin and eosin (HE) using routine procedures prior to investigation under light microscopy.
Immunohistochemistry
To detect CDV antigen in a formalin-fixed, paraffin-embedded section, immunohistochemical staining was performed using the chain polymer-conjugated method. 11 After deparaffinization and rehydration of the sections, the antigen was retrieved by autoclaving at 121°C for 5 minutes in distilled water, followed by blocking endogenous peroxidase activity with 3% hydrogen peroxide in methanol for 5 minutes. Subsequent to triplicate washing with phosphate-buffered saline (PBS), samples were incubated with mouse monoclonal antibodies against CDV envelope (H and F) protein (1:200 dilution, Monotope; ViroStat, Portland, ME) used as a primary antibody at 37°C for 60 minutes and followed by the Dako REAL EnVision Detection System (Dako, Glostrup, Denmark) at 37°C for 60 minutes. After triplicate washings with PBS, a positive antigen-antibody reaction was observed by labeling with 3,3′-diaminobenzidine tetrahydrochloride (DAB) and counterstained with Mayer’s hematoxylin. Positive control consisted of brain tissue from a canine necropsy case previously diagnosed with CDV infection. 22
Viral Isolation
Tissues collected at necropsy were frozen and were further processed as described. 22 Briefly, tissues were homogenized with Dulbecco’s modified Eagle’s medium (DMEM) and centrifuged at 2500 rpm, 4°C for 10 minutes, and the supernatant was harvested. Subsequently, samples were absorbed on the monolayer Vero cells expressing canine signaling lymphocyte activation molecules (Vero-DST cells) at 37°C for 1 hour. The infected culture supplemented with DMEM, 10% fetal bovine serum, and Geneticin (G418, 0.4 mg/ml; Invitrogen, Carlsbad, CA) was maintained at 37°C in a 5% CO2 incubator for 4 to 5 days. When cytopathic effects affected more than 70% of the cell monolayer, the viral suspension was collected and kept at –80°C until it was used for polymerase chain reaction (PCR) assays.
Molecular Detection and Genetic Characterization
Conjunctival swabs taken from 9 civets (8 Asian palm civets [Nos. 5–7, 9–13], 1 masked palm civet [No. 8]) and a stray dog (No. 14), which was found living on the farm and had mild oculonasal discharge, were sampled and kept at –80°C until processing. Total RNA extraction and reverse transcription PCR (RT-PCR) protocols using the NucleoSpin Extract Viral RNA Kit (Macherey-Nagel, Düren, Germany) and the AccessQuick RT-PCR system kit (Promega, Madison, WI) were performed according to the manufacturer’s instructions. Oligonucleotide primers specific to the regions of F, P, and H genes and amplification cycles were optimized as described previously. 13,22 The positive bands were visualized at 1031, 390, and 1824 bp, respectively. Thereafter, the positive F gene PCR products were used for genotypic lineage classification by restriction fragment length polymorphism (RFLP) using the restriction enzyme Tag αI digestion (New England BioLabs, Ipswich, MA). The reaction was performed according to the manufacturer’s instructions. Different patterns of CDV-F–digested fragments were used to differentiate among Asia-1 or Asia-4 lineages or vaccine strains as described previously. 22
All amplicons were submitted for sequencing (1st BASE Pte Ltd., Singapore) and deposited in GenBank with the assigned accession numbers (Suppl. Table S1). The nucleotide sequences of partial F, P, and H genes were aligned with available published CDV strains using ClustalW program running under BIOEDIT 7.1.3.0 (Ibis Biosciences, Carlsbad, CA). 7 Maximum likelihood phylogenetic trees and molecular evolutionary analyses were conducted using MEGA software package version 5.2.1 (The Biodesign Institute, Tempe, AZ). 28 Bootstrap values were calculated on 1000 replicates of the alignment. Finally, the identity of the deduced amino acid at sites 530, 540, and 549 on the partial H gene was determined comparing with 75 CDV strains available in GenBank for which information on host species and location was available.
Results
Pathologic Findings
Of 20 affected civets, 4 were submitted for complete necropsy. The carcasses were in poor body condition with evidence of dehydration and fecal staining. The integument of the nose, cranial muzzle, and footpads showed significant degrees of diffuse hyperkeratosis (Fig. 1). The most consistent gross lesion observed was a severe interstitial pneumonia with pulmonary edema and congestion (Fig. 2), followed by cerebral congestion. There were numerous multifocal to coalescing necrotic foci in all lobes of the liver in civet No. 4 (Fig. 3). Gastrointestinal findings included mild thickening of the intestinal mucosa (civet No. 1), catarrhal gastroenteritis (civet No. 2), gas accumulation in the stomach and intestine (civet No. 3), and multifocal gastric ulceration with concurrent mucohemorrhagic enteritis (civet No. 4; Fig. 4).
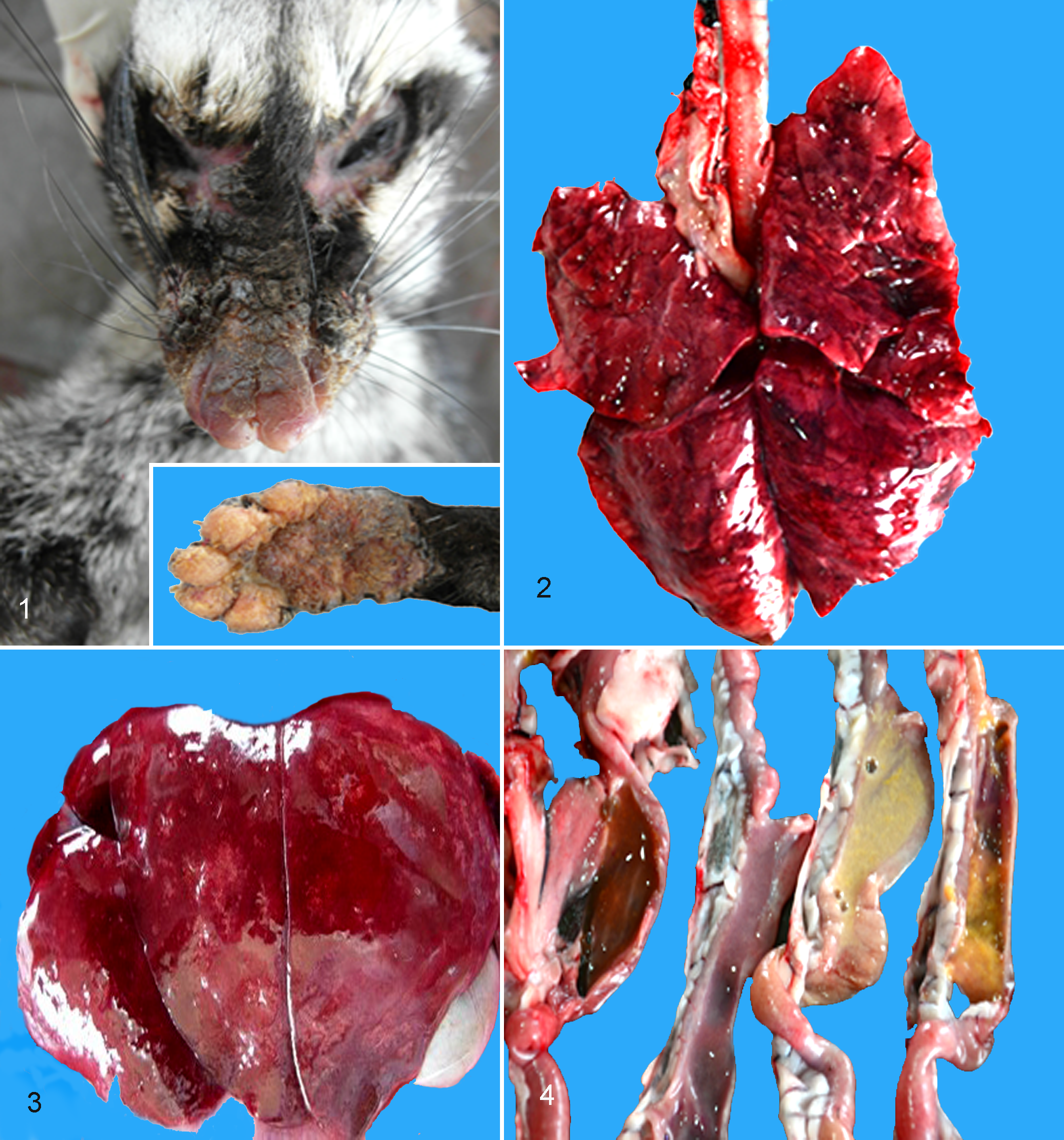
Canine distemper infection.
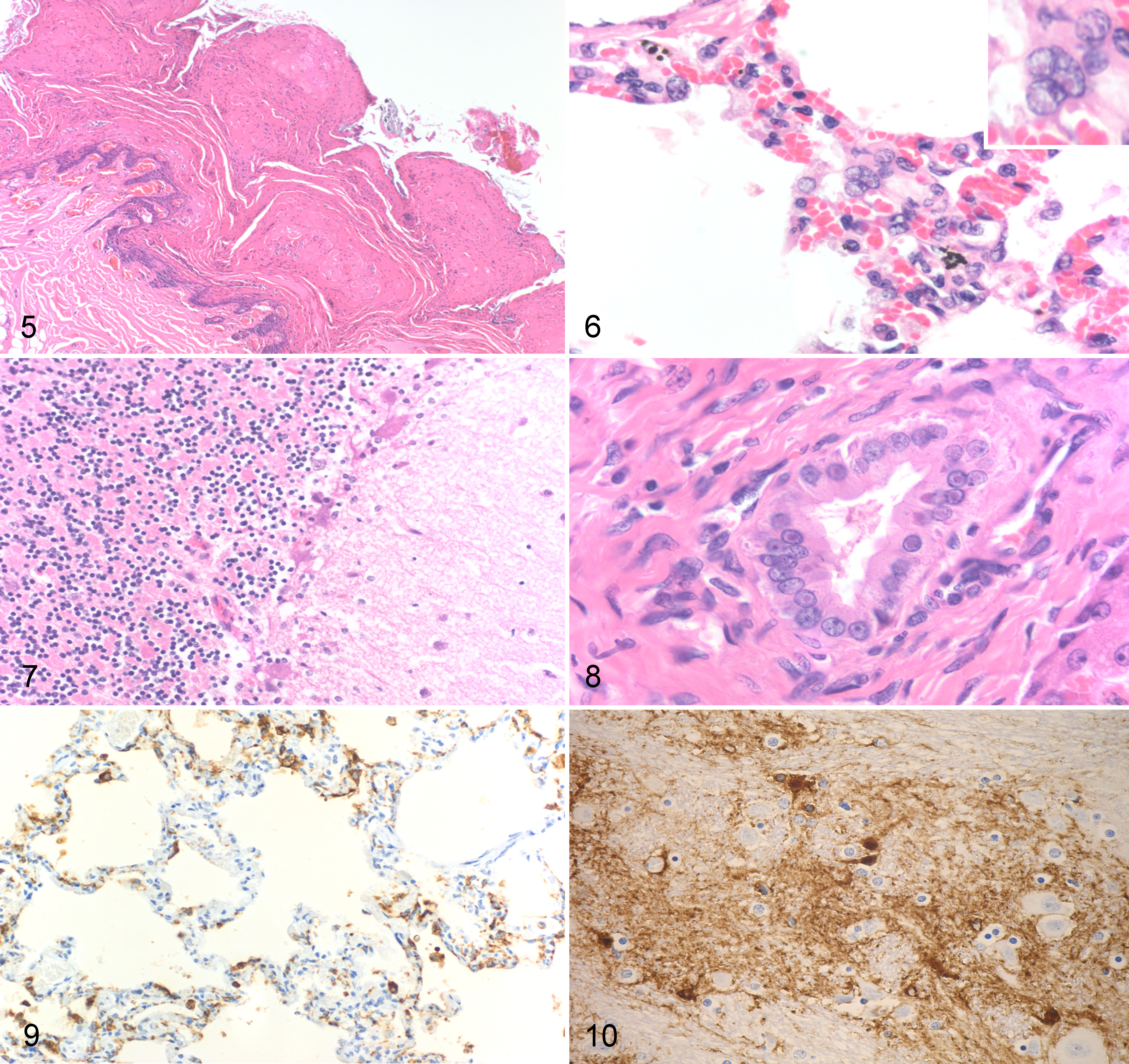
Histopathologic examination revealed lesions of canine distemper in most tissues examined. The most severe lesions were in the epithelium of the nasal planum and footpad, as well as the bronchiolar and bronchial epithelium. Less severe findings were observed in the liver, central nervous system, lymph nodes, gastrointestinal tract, pancreas, and kidney, with the heart and uterus having no significant findings. The nasal planum and the footpads contained variably severe lesions of acanthosis, parakeratotic hyperkeratosis, superficial cellular crusts, and rare vesicle formation (Fig. 5). Within all layers of the epithelium, but concentrated in the stratum spinosum, were foci of spongiosis and frequent intranuclear and intracytoplasmic inclusion bodies. In civet No. 3, there was a significant necrotizing folliculitis with lytic necrosis of the hair follicles and viral inclusions in the germinal epithelium.
Canine distemper infection.
The lungs of all animals had varying degrees of bronchointerstitial pneumonia characterized by mononuclear inflammation, frequent eosinophilic intranuclear and intracytoplasmic inclusion bodies within inflammatory cells and bronchiolar epithelium, and syncytial cells in the bronchiolar epithelium (Fig. 6). Civet No. 2 had copious necrotic debris, edema, fibrin, and sloughed cells occluding the larger airways.
The midbrain sporadically had reactive gliosis bounding hippocampal and cortical neurons (satellitosis) with rare eosinophilic intranuclear inclusion bodies in glial cells. Within these areas, there was also edema and rarefaction of the neuropil. Multifocally, few gemistocytic astrocytes were observed, as was rare perivascular hemosiderosis. Within the cerebellum, there was a multifocal loss of Purkinje cells characterized by empty baskets as well as both cellular swelling (degeneration) and occasional hypereosinophilia, angulation, and both somal and nuclear fragmentation (necrosis). Sporadically within the Purkinje cell layer, there were increased numbers of reactive glial cells (Fig. 7).
In the liver, the biliary epithelium displayed similar inclusions and syncytia as noted in the lung. In civet No. 4, there was a marked necrotizing hepatitis with a periportal to midzonal bridging pattern accompanied by marked biliary hyperplasia. Within areas of necrotic debris, adjacent biliary epithelium, Kupffer cells, and rare hepatocytes contained both intranuclear and intracytoplasmic inclusion bodies (Fig. 8). Other tissues that displayed viral lesions included the spleen, lymph nodes, gastrointestinal tract, tongue, urinary bladder, pancreatic ductal tissue, and, to a lesser degree, the kidney.
Immunohistochemical analysis revealed CDV antigen was observed in epithelium of the airways, epidermis and adnexa, stomach, intestines, pancreas, bile ducts, urinary bladder, endometrium, and tongue, as well as in mononuclear cells of the lymph nodes and spleen, inflammatory cells in the lungs, and rare glia in the brain (Figs. 9, 10; Suppl. Table S2).
Virologic Findings
Viral isolation was done using Vero-DST cells expressing dog SLAM receptors, which were infected with minced pooled tissues from CDV-infected civets. The cytopathic effect (CPE) demonstrated syncytial formation compatible with CDV infection.
All samples (fresh tissues and infected cultures) from civets Nos. 1 to 4, as well as conjunctival swabs from civet Nos. 5 to 13 and a feral dog found on the farm (No. 14), were positive for CDV infection using an RT-PCR assay. Selected amplicons of 3 target genes were sequenced and aligned with the homologous portion of other reference strains (Suppl. Table S1; GenBank accession numbers KC415037-KC415045 [F gene], KC415046-KC415049 [H gene], and KC415050-KC415062 [P gene] from CDV-infected civets; JX876873 [F gene], JX876874 [H gene], and JX886803 [P gene] from dog No. 14).
The F gene RT-PCR products were further analyzed using RFLP. All samples revealed uniformly digested patterns of the Asia-1 lineage with 2 fragments of 393 and 638 bp in length on 2% agarose gel electrophoresis (Fig. 11).
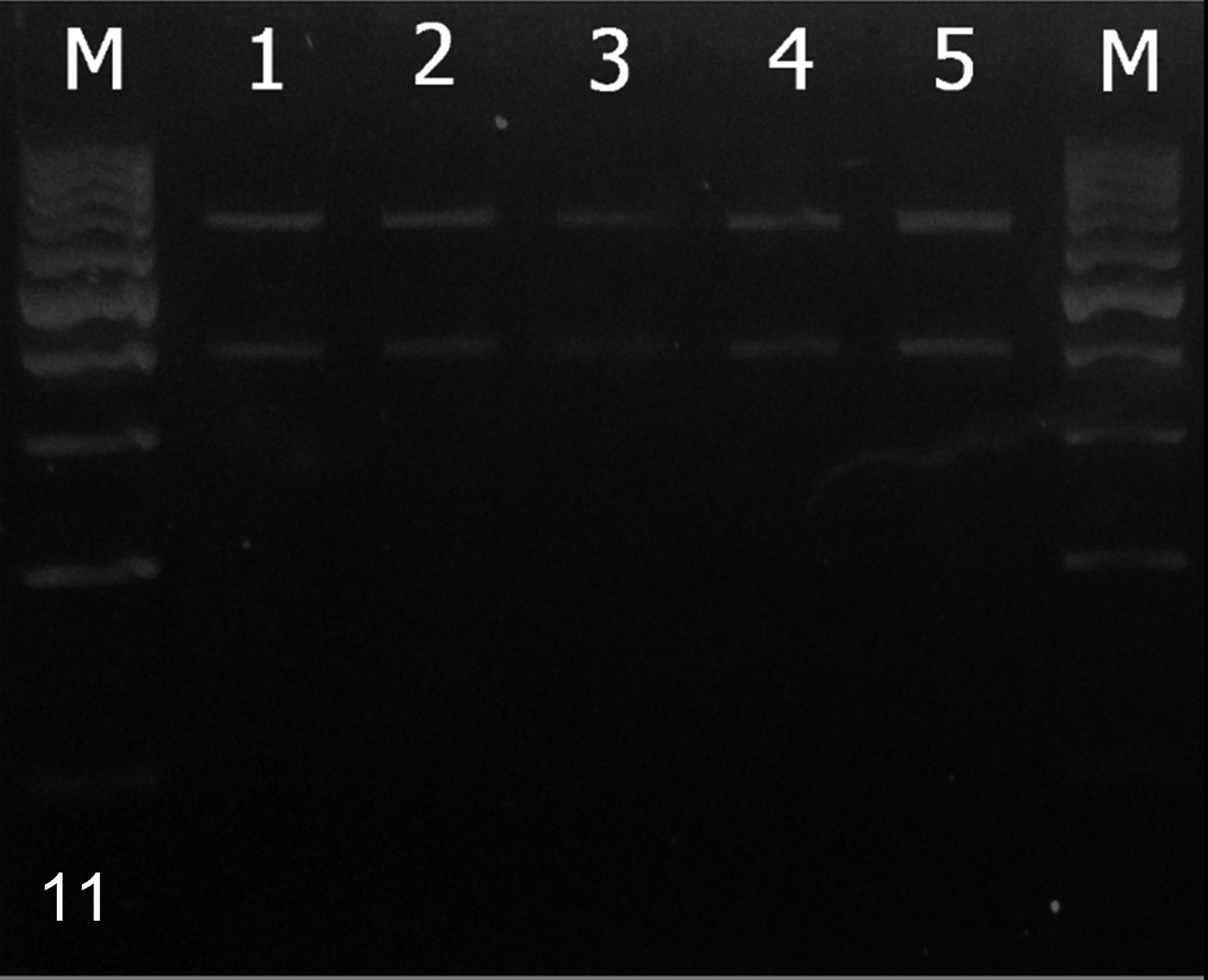
Restriction fragment length polymorphism analysis on F gene fragments for canine distemper virus lineage identification. M: 100-bp DNA ladder. Lanes 1–5: civets No. 1, 2, 3, and 4 and dog No. 14. The specific fragments at 393 and 638 bp indicate Asia-1 lineage.
Sequence analyses of 3 partial target genes revealed that all isolates were identical. Phylogenetic analysis of partial H gene sequences revealed that the CDV strain described from this study belonged to the Asia-1 lineage. Our CDV-Civet strain had 99.7% identity to the strain isolated from the dog (No. 14) in this study (JX886774/Dog/C. lupus familiaris) but lower identity (82.9%) to strain AB016776/Raccoon dog/Nyctereutes procyonoides. The sequences had lower identity to Asia-2 lineages (AB252717/Dog/C. lupus familiaris, 78.3% identity; AB025270/Dog/C. lupus familiaris, 78.2% identity) and Asia-4 lineage (JX886788/Dog/C. lupus familiaris, 93.4% identity; Suppl. Fig. S1). This finding was in agreement with the partial F gene sequence, which contained a thymine (T) to cytosine (C) transition at the 393th base of the fragment (corresponding to the 1152nd base of the complete F gene), resulting in a restriction site of Tag α I (5′ … T˅CGA … 3′). The 5 CDV strains isolated from 4 Asian palm civets and one co-located dog in this study showed identical deduced amino acid residues 530G, 540D, and 549H (Suppl. Fig. S1). CDV strains isolated from domestic dogs globally, with the exception of a single CDV-infected dog (Z47762, America-2 lineage), have been reported to contain the 549Y residue, but in this study, results from the feral CDV-infected dog on the farm instead had the 549H residue.
Discussion
Previous reports have demonstrated that CDV is a lethal infectious agent to susceptible free-living and captive Viverridae, including civets. 8,15,27,33 In this outbreak, the infected civets presented with severe acute clinical signs suggestive of respiratory, gastrointestinal, and neurologic disease and demonstrated characteristic lesions of CDV infections. 6,9,17 Recently, several investigations have reported CDV infection in a variety of wildlife in East Asia (Korea, Japan, and China) associated with the Asia-1 lineage identified by phylogeny. 1,24,33 Those investigations and the findings of the present study suggest an expanding geographic distribution of this CDV genotype, rather than a change in susceptibility of host species. 6,22,32
Devastation of natural habitat forces wild animals to expand their territory into urban environments, with increased probability for transmission of viral pathogens such as CDV. In Japan, wild-caught masked palm civets were naturally infected with CDV Asia-1 lineage and suspected to have interacted with domestic dogs, which are believed to have acted as the viral reservoir of CDV in that instance. 8,27 All 4 CD sequences from civets showed high identity to the sequence from the co-located dog sampled during this study, although the evidence remains inconclusive that the outbreak originated from that particular dog. However, the 549H strain described in this study was highly virulent to civets but relatively mild for the 1 dog that was observed to be affected. Although another dog-adapted 549H strain of America-2 lineage (Z47762/Dog/C. lupus familiaris) has been identified since 1989 (Suppl. Fig. S1), 4,5 these findings of the present study suggest that this Asia-1 lineage CDV strain adapted to and was spread by wild-caught civets rather than from the local dog population.
The lucrative nature of the civet-coffee industry in Thailand makes it likely that civets will continue to be removed from their native environment and placed in crowed breeding farms. Heterogeneous immune backgrounds and virus- and stress-induced immunosuppression likely increase the susceptibility of civets to CDV infection and enhance viral virulence in these species. Possible routes of viral transmission to the captive wild animals could be through direct contact with infected dogs or by indirect transmission via human handlers and contaminated equipment. 21,30
Footnotes
Acknowledgements
We would like to thank Professor Dr Ryoji Yamaguchi, University of Miyazaki, Japan, for providing Vero-DST cells.
Author Contributions
S. Techangamsuwan contributed to conception and design; contributed to acquisition, analysis, and interpretation of data; drafted manuscript; critically revised manuscript; and gave final approval. W. Banlunara contributed to conception and design, contributed to analysis and interpretation of data, drafted manuscript and gave final approval. A. Radtanakatikanon contributed to acquisition of data, drafted manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. A. Sommanustweechai contributed to analysis of data, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. B. Siriaroonrat contributed to interpretation of data, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. E. D. Lombardini contributed to analysis and interpretation of data, drafted manuscript, critically revised manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. A. Rungsipipat contributed to analysis and interpretation of data, drafted manuscript, critically revised manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Authors’ Note
E. D. Lombardini is a Lieutenant Colonel in the US Army. The opinions or assertions herein are those of the authors and do not necessarily reflect the view of the Department of the Army or The Department of Defense.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was co-funded by the Thailand Research Fund (MRG5480116) and Grants for the Development of New Faculty Staff (second year, GDNS 55-011-31-003), Chulalongkorn University.
