Abstract
Bacteria from the genus Mycoplasma are common inhabitants of the respiratory, gastrointestinal, and genital tracts of mammals. The understanding of the pathological significance of mycoplasmas in seals is poor, as few studies have utilized the specific culture techniques required to isolate these bacteria. The current study surveyed for the Mycoplasma species present in Australian fur seals (Arctocephalus pusillus doriferus) and investigated the association between infection and pathology. Mycoplasmas were found in the nasal cavities of 55/80 (69%) of apparently healthy individuals. Isolates from 18 individuals were investigated through 16S ribosomal RNA sequencing, and 3 species were identified: M. zalophi, M. phocae, and Mycoplasma sp. (GenBank no. EU714238.1), all of which had previously been isolated from Northern Hemisphere pinnipeds. In addition, mycoplasmas were isolated from the lungs of 4 out of 16 juveniles and 1 out of 5 adults sampled at necropsy. Isolates obtained were M. zalophi, Mycoplasma sp. EU714238.1, and M. phocicerebrale, but infection was not associated with lung pathology in these age classes. Inflammatory disease processes of the heart and/or lungs were present in 12 out of 32 (38%) aborted fetuses on microscopic examination. Predominant findings were interstitial pneumonia, pericarditis, and myocarditis. Mycoplasma phocicerebrale was isolated from the thymus of an aborted fetus, and 3 out of 11 (27%) fetuses with inflammatory heart or lung lesions were PCR-positive for Mycoplasma. In conclusion, several species of Mycoplasma are part of the normal flora of the nasal cavity of Australian fur seals, and some mycoplasmas may be associated with abortion in this species of seal.
Introduction
Bacteria from the genus Mycoplasma are the smallest of all self-replicating organisms and are wildly distributed in the animal kingdom. 24 The customary habitats of mycoplasmas in their host species are the surfaces of mucous membranes, and, accordingly, they are commonly isolated from the respiratory, gastrointestinal, and genital tracts. 25 Although often found in apparently healthy animals, mycoplasmas are recognized as pathogens in a wide range of vertebrate species6,31,36 where they most commonly cause disease of the respiratory tract and joints.9,24 Other syndromes that are sometimes caused by Mycoplasma infection include genital tract inflammation, infertility, and spontaneous abortions.21,33
The ubiquitous nature of mycoplasmas means that the clinical relevance of their isolation from individuals without observable pathology is sometimes unclear. For some known primary pathogens, such as M. pneumoniae in human beings and M. mycoides subsp. mycoides in goats, isolation even from apparently healthy animals has significance, as infected animals may develop disease themselves or act as a source of infection for others. The pathological potential of many mycoplasmas found in wildlife species is poorly understood apart from some notable exceptions, such as the substantial mortalities of North American house finches (Carpodacus mexicanus) caused by M. gallisepticum. 31 For many mycoplasmas, the ability to cause disease is influenced by variations in host susceptibility and co-infections. A reduction in the immune competence of the host can increase susceptibility to disease caused by mycoplasmas. 4 Also, the role of mycoplasmas in polymicrobial infections, either as initiating or secondary agents, is well recognized.17,29 Therefore, in wildlife species, the absence of obvious pathology in the individual from which a Mycoplasma sp. is isolated cannot be used as the sole criterion to discount the clinical relevance of the isolation. Assessing the importance of an isolate must include detailed examination of epidemiological factors and specific diagnostic criteria as to the potential impacts of the Mycoplasma sp. in question.
Knowledge of the Mycoplasma spp. present in most wildlife species is poor, as the small size and fastidious nature of bacteria require specific culture techniques for reliable isolation. 4 In pinnipeds, for example, isolation has been largely limited to a few situations where pathologic lesions of the respiratory tract or joints suggested Mycoplasma infection, which was confirmed upon analysis. In 1980, more than 400 harbor seals (Phoca vitulina) on the New England coast of North America died of acute pneumonia caused by co-infection with an influenza virus and a novel Mycoplasma sp., M. phocae (originally described as M. phocidae).10,27 It was suggested that pathology in seals that were infected with both agents was much more severe than the pathology in individuals infected with the influenza virus alone. Two other novel Mycoplasma species, M. phocicerebrale (formerly M. phocacerebrale) and M. phocirhinis, were isolated from harbor seals that died during a 1988 Phocine distemper virus (PDV) epizootic in the North and the Baltic seas. 13 The detection of the Mycoplasma species in pathologically altered organs 13 and their observed cytotoxic capacity in vitro 30 suggested that they may have acted as secondary invaders in seals infected by PDV and exacerbated that disease. The role of mycoplasmas as either primary or significant secondary pathogens was unclear in both the 1980 influenza and 1988 PDV epidemics.
In contrast to the potential co-infection role of mycoplasmas in harbor seals, M. zalophi, a novel species isolated from Californian sea lions (Zalophus californianus), appears capable of acting as a primary pathogen. 18 Infection with this organism was recognized as the cause of death at a rehabilitation facility of four Californian sea lions that were suffering from both severe pneumonia and pleuritis or septicemia that was secondary to septic polyarthritis. In other individuals in the same facility, M. zalophi was grown from subdermal and muscle abscesses, septic joints, and lymph nodes.
In addition to being potential pathogens of the pinnipeds themselves, mycoplasmas resident in pinnipeds may be passed to humans through bites or the handling of infected seal tissues. In particular, M. phocicerebrale is recognized as the likely cause of “seal finger” in human beings, a painful condition characterized by severe cellulitis with, in some cases, joint involvement.3,37
The Australian fur seal (Arctocephalus pusillus doriferus) is an Australian endemic otariid (fur seals and sea lions) species. Australian fur seals were heavily exploited by colonial-era commercial sealing operations, and the subsequent recovery of their population numbers has been slow compared to other, similarly exploited fur seal species.22,35 Low fecundity resulting from a high incidence of spontaneous abortions in the second half of gestation is a major contributory factor to the slow population growth in the Australian fur seal. 12 Currently, little is known regarding the pathogens present in Australian fur seals and their possible involvement in gestational failure. Moreover, no investigations of mycoplasmas have been conducted in any Southern Hemisphere pinnipeds. The aims of the current study, therefore, were to survey Australian fur seals for mycoplasmas, and, if present, investigate if infection is associated with disease.
Materials and methods
Animals
Nasal swabs were collected from Australian fur seals from 3 island-breeding colonies located in northern Bass Strait in southeastern Australia. In June 2007, 47 individuals (32 pups, 6 juveniles, and 9 adults) were sampled on Kanowna Island (39°10’S, 146°18’E). In October 2009, 12 individuals (10 juveniles and 2 adults) were sampled at Lady Julia Percy Island (38o42′S, 142o00′E). A further 15 individuals (13 juveniles and 2 adults) were sampled at this site in February 2010, and, in July 2010, 6 juvenile seals were sampled at Seal Rocks (38o30′S, 145o10′E). Sampling for this project was conducted concurrently with other research studies to limit disturbance to the colonies.
All animals were selected at random and captured with a large hoop net. a Swabbing of the nasal cavity was performed either under manual restraint or while under anesthesia. For the latter, a facemask was placed over the nose and mouth, and anesthesia was induced by 5% isoflurane b delivered in a closed circuit. c Swabs were immediately placed into Mycoplasma broth containing 20% porcine serum 19 and stored at ambient temperature (5–15°C) for up to 14 days before processing.
Juvenile and adult Australian fur seals found dead and dying along the Victorian coastline between 2007 and 2010 were collected for postmortem examination. Tissue samples were taken from the adrenal gland, brain, eye, heart, intestine, kidney, liver, lung, lymph nodes (axillary and mediastinal), spleen, and stomach and preserved in 10% neutral buffered formalin solution. In addition, swabs of lung tissue as well as pooled lymph node tissues from some animals were inoculated into Mycoplasma broths. Aborted Australian fur seal fetuses were collected between 2007 and 2010 during field operations at all 3 breeding colonies. If available, as seabirds had scavenged on some tissues prior to collection, the same set of tissues samples as for the older age classes along with tissues from the pharynx, placenta, and thymus were collected and stored in formalin fixative solution. Unfixed samples of body fat, liver, lung, placenta, and spleen were held frozen. Microbiological swabs from fresh, unfixed lung or thymus were collected into Mycoplasma broth from some individuals and held at ambient temperature (5–15°C) for up to 5 days prior to processing.
Formalin-fixed tissues were embedded in paraffin wax, sectioned at 5 µm, and stained with hematoxylin and eosin for histological examination. Frozen tissues from both fetuses and older age classes were routinely cultured on serum dextrose and horse blood agar plates and also underwent specific microbiological and molecular investigations for Brucella spp. 23
Mycoplasma cultivation and identification
Broths of nasal swabs were incubated at 37°C for 3 days then cultured onto Mycoplasma agar and incubated at 37°C in 5% carbon dioxide. Plates were examined weekly and resultant colonies examined under light microscopy. Individual colonies were selected for cloning and placed into Mycoplasma broth for incubation and subculture. The process was repeated 2 more times, and the final selected colony was incubated for a further 3 days in Mycoplasma broth prior to DNA extraction. The majority of inoculated broths collected from seals at postmortem were processed as for nasal swabs. A small number, however, were frozen prior to further processing after an initial incubation. The basic biochemical profile of the isolates was determined by applying tests for glucose fermentation, arginine and casein hydrolysis, and tetrazolium chloride reduction, phosphatase production, and formation of film and spots. 2
A sample of broth containing live mycoplasmas was spun at 14,000 RCF for 3 min to separate organisms from supernatant. The DNA was extracted from the organisms using a commercial kit, d as per the manufacturer’s method. Polymerase chain reaction (PCR) was used to amplify almost complete sequences of the 16S ribosomal RNA (rRNA) gene, as described, 7 and the resulting PCR products were purified using a commercial gel extraction kit. e Primers used in the PCR reaction were BSF8/20 and BSR1541/20. The nucleotides of the 16S rRNA gene were determined by direct sequencing of the PCR product with 6 primers, as described. 7 Sequencing was performed in an genetic analyzer f using a cycle sequencing kit. g Each of 6 sequences determined were analyzed and aligned using the SeqMan Pro module of the Lasergene ver. 8.0.2 software package. h The sequence was used as a query using the Basic Local Alignment Tool Search (BLAST) 1 to obtain a preliminary species identification. Frozen lung samples from fetuses were tested for the presence of mycoplasmas using a genus-specific PCR, 34 which amplifies a 280-bp fragment of the 16S rRNA gene using the primers GPO3 and MGSO. i
Results
Mycoplasma spp. were isolated from 55/80 (69%) nasal swabs collected from Australian fur seals (Table 1). Nine of the isolates from Kanowna Island (all pups), 5 from Lady Julia Percy Island (all juveniles), and 4 from Seal Rocks (all juveniles) were preliminarily identified by 16S sequencing and BLAST searching. Of these 18 isolates, 10 were most closely matched (≥98%) to M. zalophi, 5 to M. phocae (99% matched to BLAST sequence), and 3 to an unnamed Mycoplasma sp. (99% matched) previously isolated from Californian sea lions (16S rRNA nucleotide sequence GenBank ref. EU714238.1).
Mycoplasmas in Australian fur seals (Arctocephalus pusillus doriferus): numbers of isolates from the nasal cavity and seals sampled made between 2007 and 2010.*

The unnamed Mycoplasma sp. most closely matched Basic Local Alignment Search Tool (BLAST; GenBank no. EU714238.1). Numbers of isolates sequenced are in parentheses.
Isolation of mycoplasmas occurred from 5 out of 22 (23%) lung swabs collected from pups (0/1), juveniles (4/16) and adults (1/5) examined at necropsy. Four of the lung isolates were sequenced, and 3 were identified through 16S sequencing as the same unnamed Mycoplasma sp. (GenBank no. EU714238.1) that was found in nasal swabs. The remaining isolate collected from the lung of an adult female was identified as M. phocicerebrale (Table 2). Three juveniles and 1 adult had pooled axillary and mediastinal lymph node samples specifically cultured for Mycoplasma. All 3 juveniles were lung Mycoplasma-positive individuals, and all also had mycoplasmas isolated from their lymph nodes. In one of these seals, 2 Mycoplasma spp. were isolated from pooled lymph nodes, these being the unnamed Mycoplasma sp. (ref. EU714238.1) and M. zalophi. One of the remaining 2 juveniles that were both lung and lymph node Mycoplasma-positive had 16S sequencing performed on the lymph node isolate. The isolate was identified as M. phocicerebrale. The biochemical profiles test results for all Mycoplasma spp. isolated in the current study are shown in Table 3.
Mycoplasmas in Australian fur seals (Arctocephalus pusillus doriferus): numbers of tissues sampled and isolates made between 2007 and 2010.*
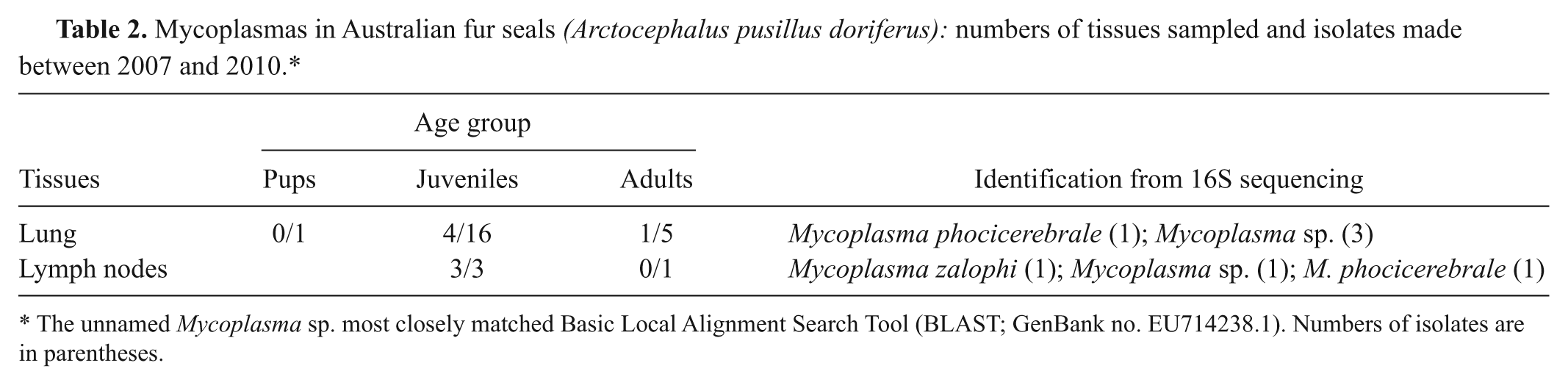
The unnamed Mycoplasma sp. most closely matched Basic Local Alignment Search Tool (BLAST; GenBank no. EU714238.1). Numbers of isolates are in parentheses.
Mycoplasmas in Australian fur seals (Arctocephalus pusillus doriferus): biochemical profiles for Mycoplasma spp. isolated between 2007 and 2010.*

Reference strains obtained from the Department of Comparative Pathobiology, Purdue University, IN.
Microscopically, none of the lung sections from Mycoplasma-positive juveniles and adults had evidence of pneumonia. Tracheal tissue was examined from 12 seals (8 juveniles and 4 adults). Lymphocytic and plasmacytic tracheitis was detected in 4 (3 juveniles and 1 adult) of these individuals. Three of the 4 seals with this lesion were positive for Mycoplasma on lung culture.
Microscopic findings on aborted fetuses examined in the current study have been reported. 23 Significant findings were noted in 14 out of 36 (39%) fetuses. For 32 of these fetuses, the heart and lungs were available for examination, and 12 out of 32 (38%) had pneumonia and/or either myocarditis or pericarditis. Five fetuses had both cardiac and pulmonary inflammatory disease processes, 4 only lung lesions and 3 only cardiac. Pneumonic lesions, observed in 6 out of 32 (19%) fetuses, were characterized by perivascular collections of lymphocytes and plasma cells and interstitial edema. Suppurative bronchiolitis with accumulation of neutrophils and macrophages within the terminal airways was also observed and this varied in its severity between affected fetuses (Figs. 1, 2). An additional 2 fetuses had inflammatory foci within the lung; one of these individuals had peribronchial infiltrates of lymphocytes and the other necrotic foci (neutrophils and macrophages) of the epithelial surfaces of the bronchioles. Myocarditis or pericarditis was noted in 8 out of 32 (25%) of fetuses and 3 of these also displayed pneumonia. Three of the 8 fetuses with cardiac lesions exhibited pericarditis, characterized by infiltrates of lymphocytes and neutrophils. In the remaining 5 fetuses, there was perivascular neutrophilic myocarditis/epicarditis (Fig. 3). One fetus with severe pneumonia and significant myocarditis also had lymphoplasmacytic phlebitis of hepatic vessels and mild, diffuse, meningitis characterized by lymphocyte accumulation (Fig. 4). No inclusion bodies were noted in any of the tissue sections.
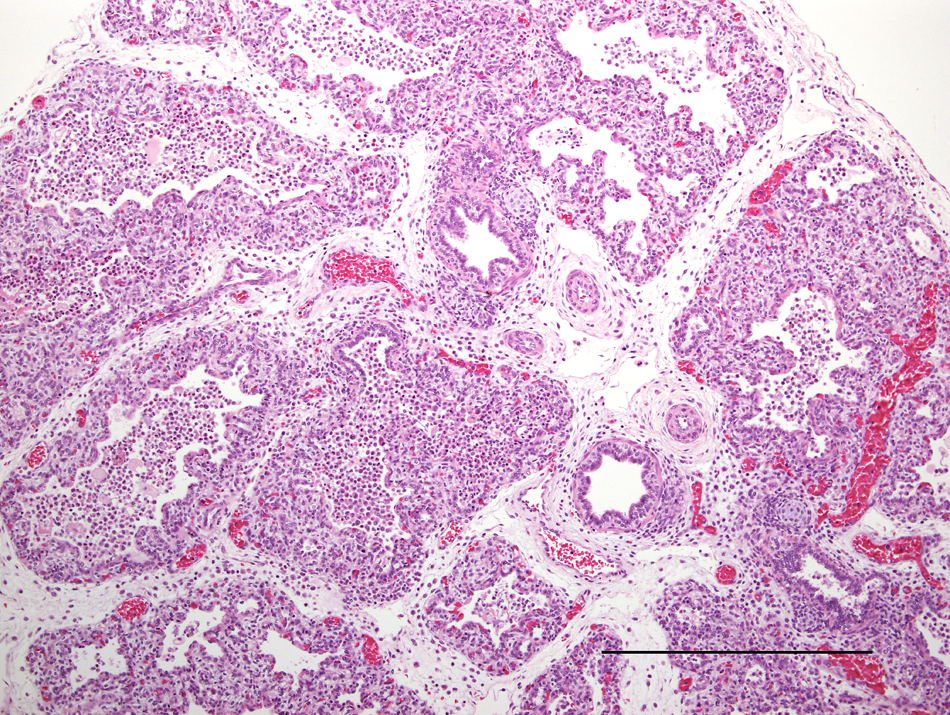
Lung; Australian fur seal fetus. Large numbers of neutrophils and macrophages obscure the bronchioles and extend into the adjacent alveolar sacs. The connective tissues within the interlobular septae are loosely arranged and expanded. Occasional plasma cells and lymphocytes expand the perivascular connective tissues, the lymphatic vessels are mildly ectatic, and the blood vessels are engorged. Hematoxylin and eosin. Bar = 500 μm.
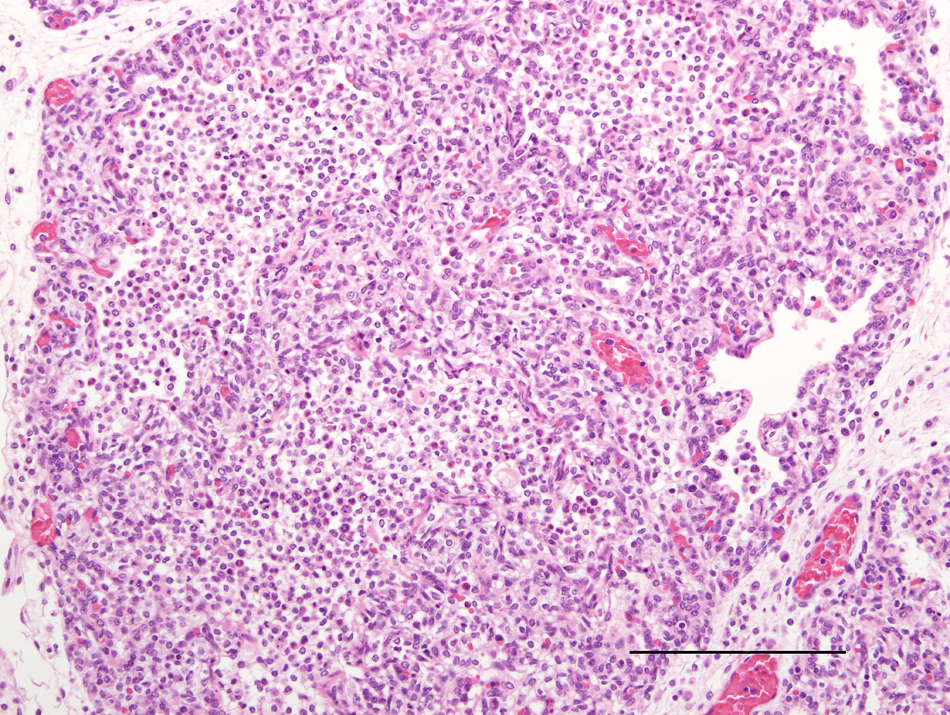
Lung; Australian fur seal fetus. Higher magnification of Figure 1 above. Large numbers of neutrophils and macrophages severely obscure the bronchioles and adjacent alveolar sacs. Hematoxylin and eosin. Bar = 200 μm.
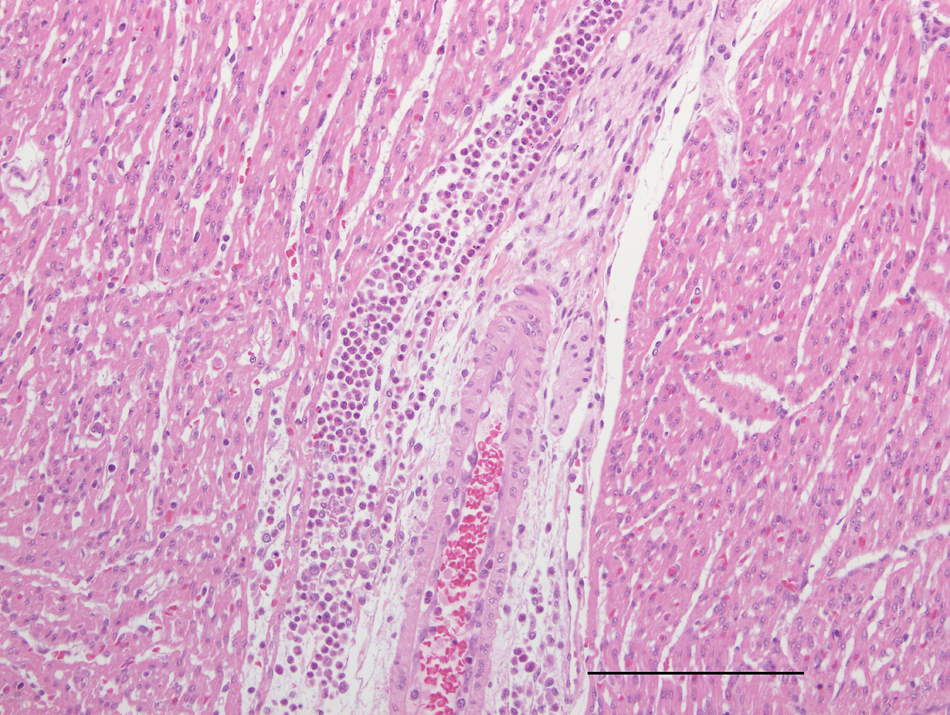
Heart; Australian fur seal fetus. Myocardial, perivascular infiltrate consisting predominantly of neutrophils and mononuclear cells. Hematoxylin and eosin. Bar = 200 μm.
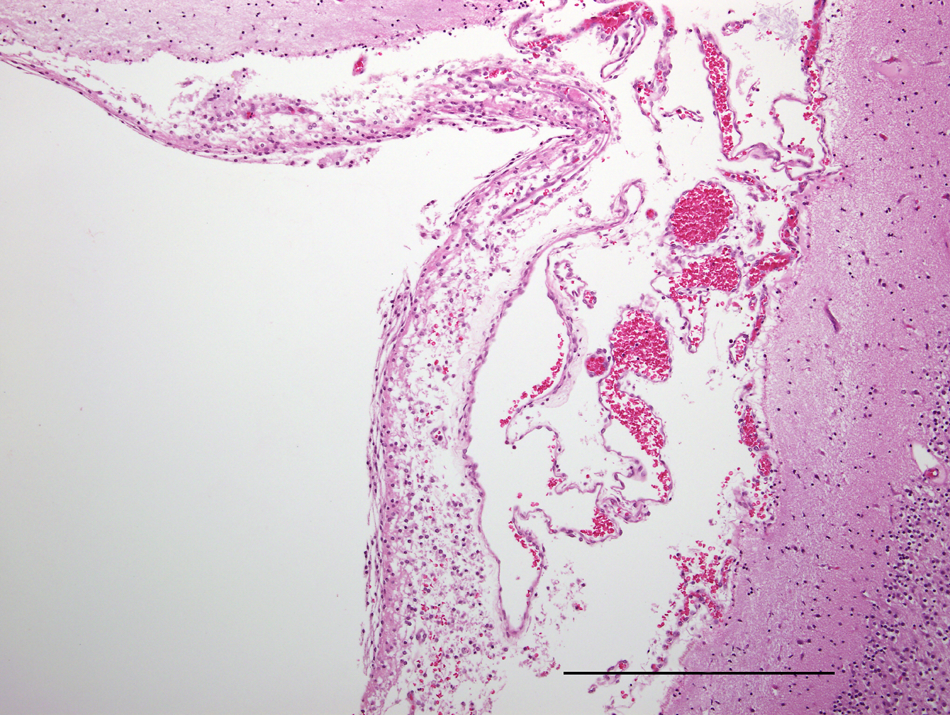
Brain; Australian fur seal fetus. Mild, diffuse meningitis characterized by lymphocyte accumulation. Hematoxylin and eosin. Bar = 500 μm.
Fresh tissue from 4 fetuses was cultured for mycoplasmas; 3 had the lung sampled and 1 the thymus. No mycoplasmas were isolated from the lung swabs, and none of these individuals displayed significant microscopic findings. Mycoplasma phocicerebrale was isolated from the thymus of the remaining individual. No significant microscopic findings were noted in this individual’s thymus, pharynx, eye, and brain; no other tissues were available for examination. Frozen lung samples from 22 fetuses (11 with and 11 without lesions) were tested for the presence of mycoplasmas by the genus-specific PCR. These 22 fetuses were in addition to the 4 fetuses that had fresh tissues cultured for mycoplasmas. Four animals were positive for Mycoplasma and 3 of these individuals had inflammatory lesions. Of the 18 PCR-negative animals, 8 had inflammatory lesions. Sequences obtained from the fetal lung PCR products were used as queries in a BLAST search. The sequence from the fetus that showed no significant histological lesions had a 98% match with an unnamed Mycoplasma sp. previously isolated from the conjunctiva of a California sea lion (ref. EU859984.1). One fetus with inflammatory lesions had a sequence 99% matched to M. phocae, and the remaining two individuals had products with only a 94% match to an unnamed Mycoplasma sp. (no. FJ876266.1) isolated from the reproductive tract of a harbor porpoise (Phocoena phocoena).
Discussion
The present study reports on the isolation of mycoplasmas from a Southern Hemisphere pinniped species and from aborted marine mammal fetuses. The 3 Mycoplasma spp. isolated from nasal swabs of apparently healthy Australian fur seals (M. phocae, M. zalophi, and Mycoplasma sp. EU714238.1) have all been previously found in either harbor seals or Californian sea lions.18,27 A fourth species, M. phocicerebrale, found in the thymus of an aborted pup and in older aged seals, has also been isolated from harbor seals and previously reported as the cause of the zoonotic disease, “seal finger.”3,13 The sequencing and biochemical test results from isolates made from Australian fur seals suggest that they represent the same Mycoplasma spp. found in Northern Hemisphere pinnipeds.
The current study extensively surveyed apparently healthy seals for mycoplasmas. The isolation of these bacteria from the nasal cavities of 72% of seals indicates that mycoplasmas are common inhabitants of the upper respiratory tract of Australian fur seals. In domestic cats and dogs, mycoplasmas are part of the normal flora of the pharynx, larynx, oral, and nasal cavities. 9 Their presence in the lower airways (trachea and lungs) is usually considered pathologically significant though, as Mycoplasma-positive animals have a higher rate of pulmonary disease compared to negative individuals.8,9 Whether the isolation of mycoplasmas from the lungs of juvenile and adult Australian fur seals at postmortem was of pathological significance remains unclear; however, as it did not appear to be associated with pneumonic lesions. Tracheitis was present in some individuals, but it is unknown if this was associated with mycoplasmas, as this site was not directly swabbed in the current study. Further investigations are required to establish if mycoplasmas are associated with pulmonary diseases in Australian fur seals, either acting as primary pathogens or co-infective agents.
Mycoplasma infections could be associated with abortion in Australian fur seals. Infections with mycoplasmas and the closely related ureaplasmas can result in abortion in several other mammals, either through spreading to the fetus from genital tract infections or by transplacental transfer following circulation of the organism through the mother’s blood stream.32,33 In human beings, the risk of late-term abortion is increased in women with cervicovaginal M. genitalium or M. hominis infections.5,20 Mycoplasmas can cause abortion in domestic cattle following transplacental transfer. Microscopic findings in aborted bovine fetuses, are characterized by suppurative bronchopneumonia and myocarditis with mixed collections of neutrophils and macrophages and perivascular infiltrates of plasma cells and lymphocytes.21,32 Histological lesions in aborted caprine fetuses resulting from infection with M. mycoides subsp. mycoides were confined to the lung. 26 They consisted of patchy to diffuse pneumonia with the most prominent lesions in the bronchoalveolar areas. Alveolar macrophages, scattered neutrophils, and lymphocytes were observed in the terminal airways and perivascular lymphocytic infiltrates were present. Histological lesions observed in the aborted Australian fur seal fetuses in the current study were consistent with those reported for cattle and goats infected with mycoplasmas.
In Australian fur seals, the isolation of M. phocicerebrale from a fetal thymus, the demonstration of mycoplasmas in fetal lung by PCR, and the presence of inflammatory histological lesions consistent with Mycoplasma infection suggest that these bacteria can potentially cause gestational failure in pinnipeds. Normal fetal tissues are a sterile environment so the isolation of bacteria indicates either infection or postmortem invasion. Postmortem invasion, however, is not associated with the formation of histologic lesions, whereas significant pathology characterized by interstitial pneumonia and inflammatory lesions in the heart was present in the Australian fur seal fetuses. In addition Mycoplasma spp. are highly unlikely to be postmortem invaders due to their fastidious growth requirements. 4 Thus, in utero infection with Mycoplasma appears to be associated with the mortality of at least some Australian fur seal fetuses.
Australian fur seals exhibit a low fecundity compared to other fur seals, probably largely due to their relatively high rates of abortion (up to 37%) in the second half of gestation. 12 Rates of gestational failure have been estimated in only a few pinniped species. Annually, up to 20% of Californian sea lion pups on San Miguel Island, California, are either aborted or born prematurely and die soon after. 14 Multiple etiologies have been suggested for this scenario, including interrelationships between disease agents and environmental contaminants, 14 ingestion of toxic algae and bacterial infections.15,28 In the Cape fur seal (A. p. pusillus), a gestational failure rate of 21% was measured between 1988 and 1991 but the relative contributions of failure of embryonic implantation and late-term spontaneous abortions to this figure were not established. 16 In both Cape and Australian fur seals, nutritional factors probably influence annual variations in abortion rates as fewer pups are produced in years when females have a lower mean body condition index.11,16 Although the present study suggests that Mycoplasma spp. are involved in some instances of abortion in Australian fur seals, the contribution of additional potential predisposing factors is unknown. Whether co-infections or the pregnant female’s age, nutritional status, toxin load, and immune competency are involved in Mycoplasma-induced abortion is yet to be established. The incidence of Mycoplasma-induced abortion may also vary temporally. As evidence for this, most fetuses (9/14 cases) showing histological lesions were collected on a single visit to Seal Rocks in September 2009.
Future studies could be directed at whether mycoplasmas can be recovered from the Australian fur seal female’s genital tract and if this is a risk factor for gestational failure. In addition, the attempted isolation of mycoplasmas from multiple sites within fetuses and increasing the number of individuals examined would assist in clarifying the importance of this pathogen to Australian fur seals. The possibility that the rate of Mycoplasma-induced abortion varies between years also requires further investigation.
In conclusion, the current study presented evidence that Mycoplasma spp. are common inhabitants of the nasal cavity of Australian fur seals and also can be recovered from the lower respiratory tract of juveniles and adults. Their pathological significance in these age classes is unclear and warrants further investigation. The isolation of the causative agent for “seal finger” from Australian fur seals should be noted for its zoonotic potential and appropriate care taken when handling seals or their tissues. The study also concluded that mycoplasmas could potentially cause abortion in Australian fur seals and their role in the substantial rate of gestational failure observed in this species should be further explored.
Footnotes
Acknowledgements
Research was conducted under the Deakin University Animal Welfare Committee Approval A12/2007 and Department of Sustainability and Environment (Victoria) Wildlife Research Permits 10003586 and 10004947. The authors gratefully acknowledge the work of Adrian Howard in coordinating Melbourne Zoo’s stranded seal response, which led to collection of many of the animals for necropsy. Special thanks to pathology staff from the Department of Primary Industries, Attwood, in particular Malcolm Lancaster (deceased), Jemma Bergfeld, and Mark Hawes for their substantial inputs into the research program.
a.
Research Nets Inc., WA, and Fuhrman Diversified Inc., TX.
b.
100% vol/vol Forthane®, Abbott Australia Pty Ltd., New South Wales, Australia.
c.
Stinger, Advanced Anaesthesia Specialists, North Ryde, Australia.
d.
QIAamp DNA Mini Kit (no. 51304), Qiagen, Australia.
e
QIAquick Gel Extraction Kit (no. 28704), Qiagen, Australia.
f.
ABI 3130XL, Applied Biosystems, Carlsbad, CA.
g.
BigDye Terminator v3.1, Applied Biosystems, Carlsbad, CA.
h.
DNASTAR Inc., Madison, WI.
i.
Super Taq polymerase, HT Biotechnology, Cambridge, England.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Phillip Island Nature Parks, Melbourne Zoo, Department of Agriculture, Fisheries and Forestry (Canberra), and the Winifred Violet Scott Trust provided the funding of the study.
