Abstract
Porcine periweaning failure-to-thrive syndrome (PFTS) is a clinical syndrome characterized by anorexia and progressive debilitation of newly weaned pigs. The objectives of the current case-control study were to describe the histopathologic features of PFTS in North America and test for selected pathogens in case and control pigs on 8 farms allegedly fulfilling the clinical definition of PFTS. Based on observations during farm visits, 5 farms fully met the case definition (PFTS farms), whereas 3 farms only partially fulfilled the definition (NON-PFTS farms). Necropsy and histopathologic examination were performed on case (n = 8 or 9) and control (n = 4) pigs from each farm. Superficial gastritis, which was mainly localized in the fundus and characterized by attenuation of superficial foveolar cells, was significantly more frequent in case pigs from PFTS farms compared with all the other pigs (odds ratio [OR], 16.7). The same was found for thymic atrophy (OR, 30.1) and small intestinal (SI) villous atrophy in the duodenum (OR, 28.7), jejunum (OR, 67.4), and ileum (OR, 56.3). All pigs with PFTS had at least 2 of these 3 lesions: gastritis, thymic atrophy, and SI villous atrophy. PFTS was not associated with any relevant porcine pathogen tested. We propose the diagnosis of PFTS be based on the fulfillment of the clinical case definition, the presence of the above lesions, and exclusion of other common swine diseases and pathogens. However, PFTS can be ruled out if debilitated pigs do not have at least 2 of the above 3 lesions.
Keywords
Porcine periweaning failure-to-thrive syndrome is a clinical syndrome of anorexia, lethargy, and progressive debilitation affecting nursery pigs within 2 to 3 weeks of weaning. 10 Although sporadic “postweaning starve-outs” have existed in the swine industry for many years, it was not until 2008 that outbreaks of starve-outs accompanied by substantial increases in nursery mortality were first recognized as a distinct clinical syndrome. 3,6 Early reports of the syndrome used various terms such as postweaning catabolic syndrome, postweaning wasting-catabolic syndrome, failure-to-thrive syndrome, and postweaning fading pig–anorexia syndrome 3,8,10 until the consensus name PFTS was adopted in 2010. 10 Many nutritional, environmental, and medical interventions have been attempted to reduce the impact of PFTS, but none has proven effective. 3,6,10 A recent survey conducted in North America estimated the prevalence of PFTS to be about 4.3% of nursery flows, where a nursery flow was a farm-level parameter defined as consecutive groups of pigs sourced from a single sow operation that supplied 1 or more nurseries, or consecutive groups of pigs sourced from multiple sow operations that supplied 1 or more nurseries. Moreover, the 4 most commonly reported clinical signs were anorexia, loss of body condition, prolonged standing, and repetitive oral behavior. 14 The current status of PFTS on other continents is unknown, but a recent report suggests it may be present in Spain. 17
At present, diagnosis of PFTS is based on fulfillment of the proposed case definition and exclusion of other common swine diseases. As the etiology is not known, the herd-level case definition is mainly based on clinical presentation and is paraphrased as follows: PFTS is defined by the presence in a farm of progressive debilitation of newly weaned pigs within 2 to 3 weeks of weaning, resulting in variable morbidity but high case fatality, with repetitive oral behaviors such as chomping and chewing observed in at least some affected pigs in all affected farms, but without residual illness from the suckling phase or discernible and detrimental infectious, nutritional, managemental, and environmental factors that can explain the clinical signs. 10
A number of diagnostic investigations in clinically affected farms have been completed to date by our group 9 and others. 16,17 These investigations have identified a number of gross and histopathologic lesions that may be associated with PFTS, including thymic atrophy, superficial gastritis, small intestinal villous atrophy, superficial colitis, nonsuppurative meningoencephalitis, neutrophilic lymphoplasmacytic rhinitis, and bronchopneumonia. In addition, the involvement of common swine pathogens, including porcine reproductive and respiratory syndrome virus (PRRSV), porcine circovirus type 2 (PCV2), influenza A virus (FLUAV), transmissible gastroenteritis virus, (TGEV), Mycoplasma hyopneumoniae, Brachyspira hyodysenteriae, and Brachyspira pilosicoli, was ruled out. However, a number of swine pathogens, such as Suid herpesvirus 2 (SuHV2), porcine enteric calicivirus (PECV), swine torque teno virus 2 (TTSuV2), pathogenic Escherichia coli (defined as possessing any of the following factors: LT, STa, STb, Stx2e, and Eae), rotavirus (RV-A), hemagglutinating encephalomyelitis virus (HEV), and coccidia, were detected in affected pigs, age-matched unaffected pigs, or both depending on the investigation. 9,16 Given that the investigations completed to date were limited to a few farms, were conducted by different investigators, and had insufficient experimental power and scope to be conclusive, the objectives of this study were to characterize the pathology and pathogens associated with PFTS more conclusively using a larger scale case-control investigation involving North American farms. Our specific goals were to determine if debilitated pigs from different PFTS farms have pathological findings that are consistent among farms and to determine if 1 or more common swine pathogens are consistently associated with lesions across farms. This experiment was purposefully not designed to determine whether any such lesions were specific to or pathognomonic for PFTS.
Methods
Farm Visits and Sample Collection
This work was approved by the University of Saskatchewan’s Animal Research Ethics Board and adhered to the Canadian Council on Animal Care guidelines for humane animal use. Between July 2011 and April 2013, the authors were contacted by swine veterinarians in Canada and the United States who reported client farms with elevated nursery mortality due to the presence of pigs with clinical signs typical of PFTS. Relevant aspects of the history, clinical signs, and health status were obtained, and farms appearing to fulfill the proposed clinical definition of PFTS (elevated mortality, anorexia, weight loss within 2–3 weeks of weaning) 10 were investigated. Recent diagnostic reports were reviewed if available, and farms that had undertaken no recent diagnostic testing were asked to submit 2 to 3 characteristic pigs for necropsy examination to their local diagnostic laboratory to verify that no known diseases or pathogens were responsible. Eight farms were included (Suppl. Table S1), all having PCV2 vaccination programs in place. Pigs that were convincingly thin to emaciated and that demonstrated repetitive oral behavior but no other signs of disease were assigned as cases (CASE). Pigs from the same batch (in the same airspace where possible) that were robust, were active, and had a full abdomen (indicative of normal appetite) were selected conveniently as controls (CTRL). No attempt was made to match pigs by sex or litter. All pigs had been weaned less than 15 days at the time of selection. From each farm, 8 or 9 CASE and 4 CTRL pigs blocked by number of days since weaning were identified, transported to the local diagnostic laboratory, and held overnight with feed and water. The following day, animals were humanely euthanized using intravenous barbiturate, and a comprehensive necropsy examination was performed. The authors performed all farm visits, made clinical observations, and performed all necropsy examinations.
Formalin-fixed and fresh tissues were collected for routine histologic examination and further diagnostic tests. Tissues collected included cerebrum, cerebellum, brainstem, trigeminal ganglion, salivary gland, lung, heart, thoracic thymus, stomach (fundus and pylorus), duodenum, jejunum (2 sections), ileum, spiral colon, cecum, pancreas, liver, kidney, adrenal gland, second rib, tonsil, bronchial lymph node, mesenteric lymph node, superficial inguinal lymph node, ventral nasal conchae, and spleen. Fresh tissues were stored at –80°C until further use. Histologic examinations were carried out by the author (Y.H.) while blinded to the identities of the animals. In addition, the lengths and depths of 10 randomly selected intact villi and crypts in the ileum of each pig were measured using a 10× objective and micrometer.
Categorization of Farms Based on Clinical Presentation
During the visits to the 8 selected farms, it was determined that 5 (farms 1–5) completely fulfilled the proposed clinical definition of PFTS (PFTS farms). 10 However, the nursery pigs in farms 6 and 7 did not demonstrate progressive debilitation (emaciation and wasting) during the farm visits, despite the fact that increased nursery mortality was reported by the herd veterinarian prior to the visit. CASE pigs from these 2 farms were selected on the basis of demonstrating a flat or hollow abdomen, lack of interest in the available feed, and repetitive oral behavior (chomping). They were not in advanced stages of debilitation. In farm 8, necropsy examination revealed bronchopneumonia of various severities in all CASE but no CTRL pigs. As these latter 3 farms did not fully meet the proposed PFTS case definition—specifically, the lack of progressive debilitation in CASE pigs of farms 6 and 7 and the presence of bronchopneumonia in all CASE pigs from farm 8—they were designated “NON-PFTS” farms. As a result, pigs used in this study were further divided into 4 subgroups: affected pigs from PFTS farms (PFTS-CASE), nonaffected pigs from PFTS farms (PFTS-CTRL), affected pigs from NON-PFTS farms (NON-PFTS-CASE), and nonaffected pigs from NON-PFTS farms (NON-PFTS-CTRL). The NON-PFTS farms were included in the analyses because they potentially represented a control group with pigs appearing to be affected by other diseases with clinical signs similar to PFTS or without the debilitation. That being said, these farms may have had less severe PFTS or were in a period of waning clinical signs at the time of the visit. Regardless, they did not fulfill the case definition at the time of the visit and provided a useful comparison to the PFTS farms in this study.
Nucleic Acid Extraction and Reverse Transcription of RNA
DNA and RNA from tonsil, brain, lung, ileum, and stomach were extracted using the Qiagen AllPrep DNA/RNA Mini Kit (Toronto, ON, Canada) according to the manufacturer’s instructions. Reverse transcription of the extracted RNA was performed using the Bio-Rad iScript cDNA Synthesis Kit (Mississauga, ON, Canada) according to the manufacturer’s instructions. DNA, RNA, and complementary DNA (cDNA) were stored at –80°C before testing for different pathogens by polymerase chain reaction (PCR).
PCR Detection of Common and Relevant Swine Pathogens
Detection of nucleic acid of HEV in the tonsil; 9,18 porcine enteroviruses CPE group 1, 2, and 3 (PEV-1, 2, and 3) in the brain; 23 PECV in the ileum; 9,22 TGEV in the ileum; 21 PCV2 in the tonsil; 5 M. hyopneumoniae in the lung; 12 and Helicobacter pylori–like and Helicobacter heilmannii–like organisms in the stomach 2 were performed according to previously published assays using the Qiagen HotStarTaq Plus DNA Polymerase Kit. PCR detection of FLUAV in lung tissue was performed as previously described 9,19 by using the Qiagen QuantiFast Probe PCR Kit according to the manufacturer’s instructions. Detection of PRRSV RNA from lung was performed using the Tetracore EZ-PRRSV Kit (Rockville, MD, USA) according to the manufacturer’s instructions. PCR detection of RV-A, B, and C was performed by the Veterinary Diagnostic Services Laboratory of the Government of Manitoba (Winnipeg, MB, Canada) using a protocol developed by the Minnesota State Veterinary Diagnostic Laboratory. 11
Statistical Analyses
Generalized estimating equations (GEEs) adjusted for clustering effects of farm were employed to build logistic regression models, in which the outcomes were the presence or absence of specific lesions and pathogens. The predictors for all models were farm type (PFTS or NON-PFTS farms), pig type (CASE or CTRL), and the interaction of these 2 factors. Post hoc pairwise comparisons of the estimated means of each combination of farm type and pig type were performed when the interaction term was statistically significant or when both farm type and pig type were significant (Table 1 and Suppl. Table S2). When the interaction of the initial GEE was significant (ie, superficial gastritis, duodenal villous atrophy, jejunal villous atrophy, and ileal villous atrophy) or the prevalence of the lesion was obviously higher in PFTS-CASE pigs than in the others (ie, thymic atrophy and colitis), further analyses were completed comparing the odds of lesions in PFTS-CASE vs all other groups (Table 1 and Suppl. Table S2). When the prevalence of lesions was obviously higher in pigs from PFTS farms than in those from NON-PFTS farms (ie, encephalitis and rhinitis), further analyses were pursued to compare the odds of lesions between PFTS farms and NON-PFTS farms (Table 1 and Suppl. Table S2). Linear regressions were built by GEEs to predict ileal villous lengths, crypt depths, and villi-to-crypt (V/C) ratios, with the predictors being farm type, pig type, and the interaction term. The raw residues of the linear regression models were checked for normality and homogeneity of the variances (Suppl. Table S3). All statistical analyses were performed using SPSS version 21 (SPSS, Inc, an IBM Company, Chicago, IL). P values less than .05 were regarded as statistically significant. Ninety-five percent confidence intervals (CIs) of sensitivities and specificities were calculated as described. 20
Prevalence and odds ratios indicating significance of histopathological lesions observed in PFTS case and control pigs.
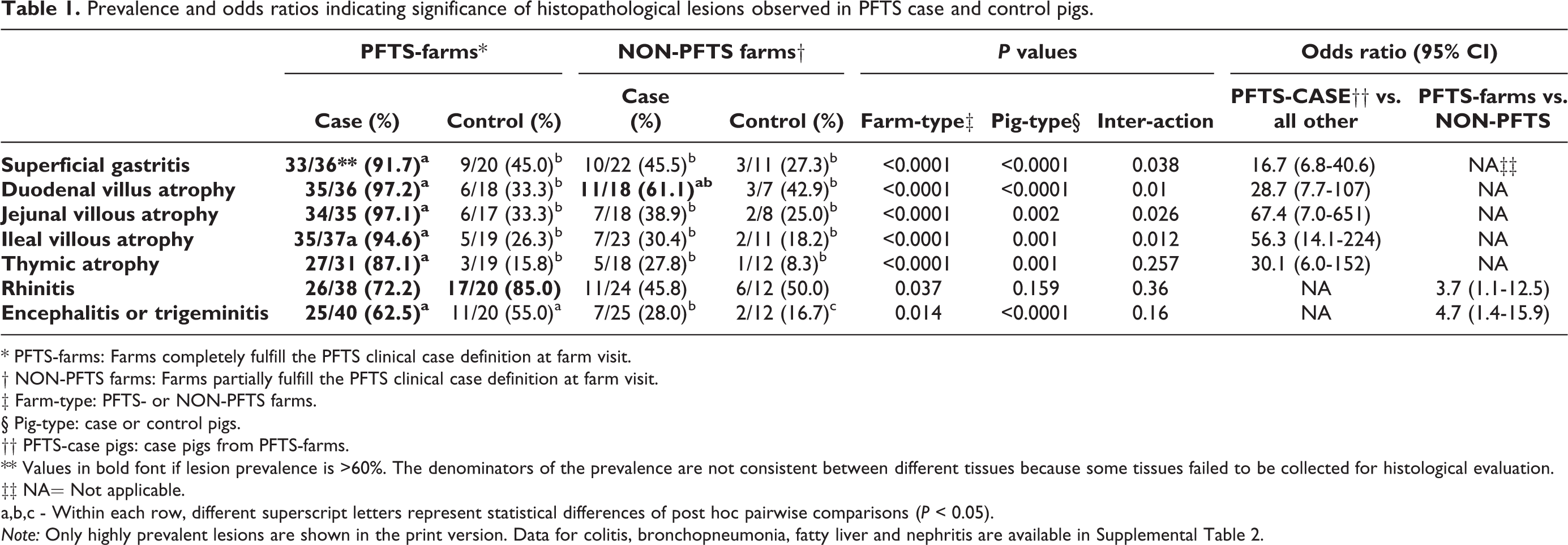
* PFTS-farms: Farms completely fulfill the PFTS clinical case definition at farm visit.
† NON-PFTS farms: Farms partially fulfill the PFTS clinical case definition at farm visit.
‡ Farm-type: PFTS- or NON-PFTS farms.
§ Pig-type: case or control pigs.
†† PFTS-case pigs: case pigs from PFTS-farms.
** Values in bold font if lesion prevalence is >60%. The denominators of the prevalence are not consistent between different tissues because some tissues failed to be collected for histological evaluation.
‡‡ NA= Not applicable.
a,b,c - Within each row, different superscript letters represent statistical differences of post hoc pairwise comparisons (P < 0.05).
Note: Only highly prevalent lesions are shown in the print version. Data for colitis, bronchopneumonia, fatty liver and nephritis are available in Supplemental Table 2.
Results
The most notable gross lesions in PFTS-CASE pigs were thymic atrophy and empty gastrointestinal tracts. Frequencies of different histologic lesions in each farm are presented in Supplemental Table S4. Histologic lesions that were more than 60% prevalent in PFTS-CASE were superficial gastritis; small intestinal villous atrophy of the duodenum, jejunum, and ileum; thymic atrophy; encephalitis or trigeminal ganglioneuritis; and suppurative or lymphoplasmacytic rhinitis (Table 1). The gastritis was mainly observed in the fundic stomach. Compared with normal (Fig. 1), the superficial foveolar cells of the affected fundus lacked or had reduced cytoplasmic mucus and were attenuated, low cuboidal, squamous, or multifocally absent (Fig. 2). The same epithelial changes occasionally extended deeper, affecting the mucous neck cells and upper glands. Increased numbers of lymphocytes were observed in the lamina propria of some pigs, without formation of lymphoid follicles. However, at times, these changes were subtle and difficult to distinguish confidently from normal.
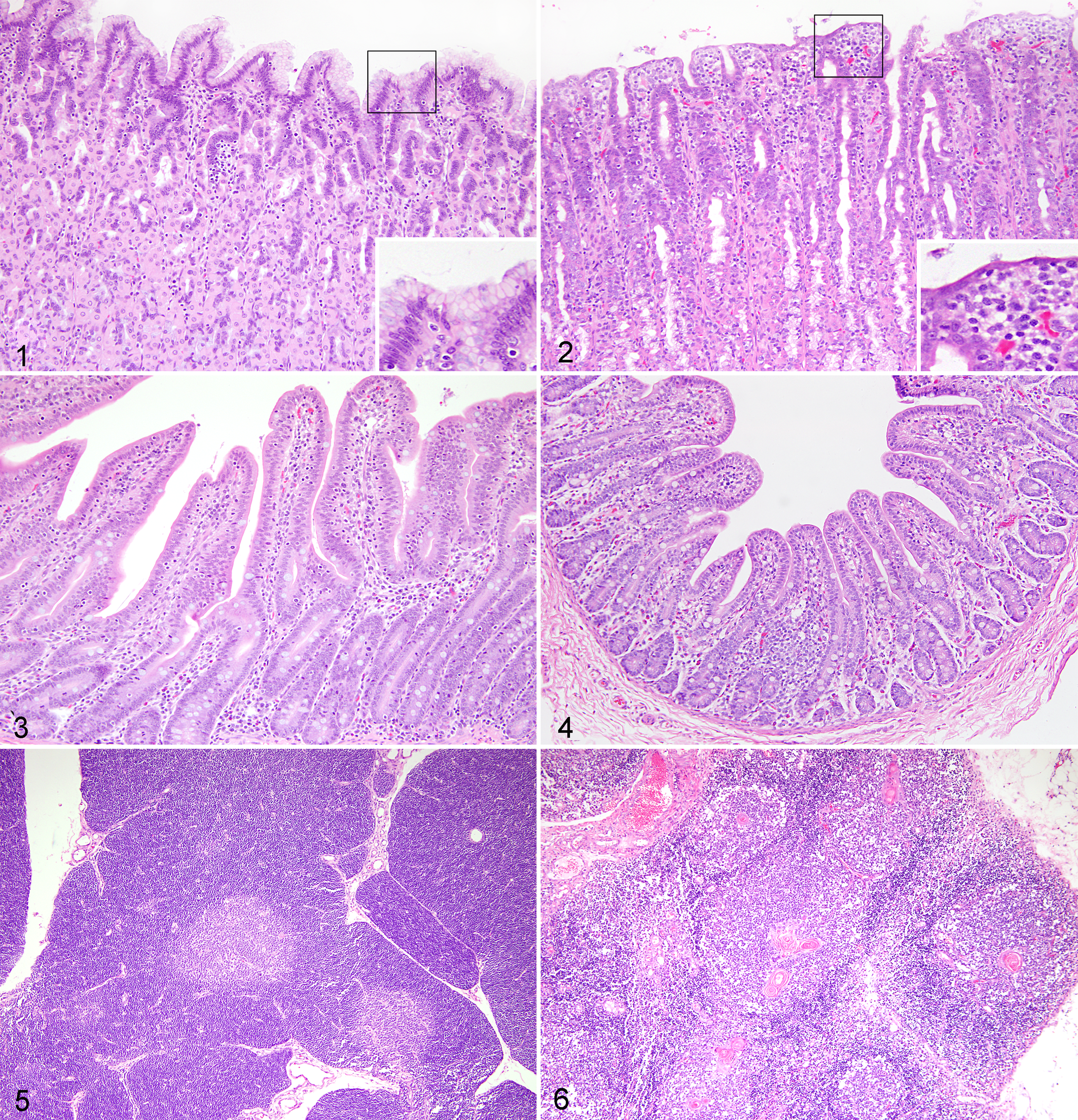
Normal. Gastric fundus; porcine. Non–periweaning failure-to-thrive syndrome control (NON-PFTS-CTRL) pig. Abundant cytoplasmic mucus in foveolar cells (insert). Hematoxylin and eosin (HE).
Small intestinal villous atrophy was evaluated subjectively and objectively. By blinded subjective evaluation, CTRL nursery pigs generally had small intestinal V/C ratios greater than 1 (Fig. 3), whereas the ratios in PFTS-CASE intestines were usually less than 1 (Fig. 4). Second, an objective measurement of villous lengths and crypt depths in ileum demonstrated that PFTS-CASE pigs had shorter villi and lower V/C ratios than all other pigs (Suppl. Table S3). The average villous length of PFTS-CASE pigs was 163 μm compared with greater than 320 μm for all other groups (P < .0001; Suppl. Table S3). The average V/C ratio of PFTS-CASE pigs was 0.9 compared with greater than 1.5 for all other groups (P < .0001; Suppl. Table S3). Crypt depths of PFTS-CASE pigs were not different from other groups (Suppl. Table S3). The subjective evaluation of villous atrophy in the ileum was shown to be highly sensitive (100%) and specific (89%) in estimating the quantitative measure of the V/C ratio (Suppl. Table S5). Epithelial cells on the villous tips were often cuboidal and occasionally squamous. Coccidia, one potential pathogen associated with villous atrophy, were inconsistently observed in various life cycle stages in the jejunal or ileal epithelium from 3 PFTS farms and 1 NON-PFTS farm, and its presence was not associated with CASE or CTRL (P > .05).
Compared with normal thymus (Fig. 5), the thymic cortex of PFTS-CASE pigs demonstrated markedly reduced numbers of lymphocytes (Fig. 6). As a result, there was moderate to severe reduction in the thickness of the thymic cortex in PFTS-CASE pigs, but the medulla was relatively unaffected (Fig. 6).
In the ventral nasal conchae of the pigs affected by rhinitis, large numbers of neutrophils were mixed with mucus without observable bacteria. Moderate to large numbers of lymphocytes and plasma cells infiltrated the lamina propria of the nasal turbinate and either diffusely or multifocally surrounded the nasal glands. Intranuclear inclusion bodies typical of SuHV2 were observed in 5 of 8 CASE pigs from 1 PFTS farm and 2 of 12 CASE and 1 of 4 CTRL pigs from 2 NON-PFTS farms. Less frequently observed lesions included superficial colitis, bronchopneumonia, hepatic cellular fatty change, and multifocal lymphoplasmacytic nephritis, which varied in prevalence among herds but were unrelated to pig type (Suppl. Table S2).
Lesions of >20% prevalence in PFTS-CASE pigs were statistically analyzed. Compared with all other groups, PFTS-CASE pigs had significantly higher odds of showing superficial gastritis (odds ratio [OR], 16.7; 95% CI, 6.8–40.6); small intestinal villous atrophy of the duodenum (OR, 28.7; 95% CI, 7.7–107), jejunum (OR, 67.4; 95% CI, 7.0–651), and ileum (OR, 56.3; 95% CI, 14.1–224); thymic atrophy (OR, 30.1; 95% CI, 6.0–152); and superficial colitis (OR, 14.6; 95% CI, 2.0–107) (Table 1 and Suppl. Table S2).
Stomach, small intestine, and thymus were examined histologically in 29 PFTS-CASE pigs and 43 other pigs (PFTS-CTRL, NON-PFTS-CASE, NON-PFTS-CTRL). Based on a case definition requiring the presence of at least 2 of superficial gastritis, small intestinal villous atrophy, and thymic atrophy, the sensitivity and specificity of correctly identifying PFTS-CASE were 100% and 67%, respectively (Suppl. Table S6). Based on a case definition requiring the presence of all 3 of the above lesions, sensitivity decreased to 86% while the specificity increased to 85% (Suppl. Table S6).
For rhinitis and encephalitis or trigeminal ganglioneuritis, the pig type × farm type interactions were not significant in the initial GEE, and statistical analysis was used to compare PFTS with NON-PFTS farms. All pigs from PFTS farms had significantly higher odds of having suppurative or lymphoplasmacytic rhinitis and nonsuppurative encephalitis or trigeminal ganglioneuritis than all pigs from NON-PFTS farms. The odds of pneumonia and liver and renal lesions did not differ by farm type or pig type (Table 1 and Suppl. Table S2).
RV-A, B, and C; HEV; and PEV-1, 2, and 3 were detected variably by PCR in both pig types and farm types (Suppl. Table S7). The odds of detecting these viruses did not differ by farm type or pig type (P > .05). The likelihood of detecting RV-C was greater in pigs from PFTS farms compared with those from NON-PFTS farms (P = .023; OR, 3.3; 95% CI, 1.2–9.3). One PFTS-CASE (from farm 1) tested positive for PCV2, but PCV2-associated disease was ruled out because this pig did not have histiocytic inflammation in lymphoid organs or kidney. All pigs in this study tested negative for PRRSV, FLUAV, PECV, TGEV, M. hyopneumonia, and H. pylori–like and H. heilmannii–like organisms. The detection of HEV and PEVs was not associated with the presence of nonsuppurative meningoencephalitis or trigeminal ganglioneuritis (P > .05 for all).
Discussion
The results of the current study provide new knowledge on the pathology and microbiology of PFTS. Since a detailed discussion of the significance of the lesions associated with PFTS was made previously, 9 our goal in this article is to expand understanding rather than reiterate a similar discussion.
While it was our intention to visit only farms fulfilling the PFTS clinical case definition, 3 farms failed to do so despite careful previsit screening. This was unexpected, and it remains uncertain whether the NON-PFTS farms had a condition similar to PFTS but did not meet the case definition on the day of the visit, were affected by other diseases, or both. Nonetheless, these 3 NON-PFTS farms may provide a serendipitous control group for comparison to the PFTS farms in this study and contribute to the understanding of the biological significance of lesions in PFTS-CASE pigs. For example, the healthy pigs from the NON-PFTS farms (NON-PFTS-CTRL) were the most likely group in this study to be truly healthy pigs (the PFTS-CTRL pigs might have been subclinical animals). On the other hand, in a diagnostic setting, pathologists may receive submissions of unthrifty or anorexic pigs that demonstrate repetitive oral behavior and be asked to rule in or rule out PFTS. The NON-PFTS-CASE pigs represent such pigs, as they were in poor body condition, demonstrated repetitive oral behavior, but were not from PFTS farms. The results of this study are illuminating, in that some NON-PFTS-CASE pigs had the same lesions as did the PFTS-CASE pigs but at lower frequency (Table 1 and Suppl. Table S6). Accordingly, one can rule out but cannot rule in PFTS based on pathology (Suppl. Table S6). This emphasizes that PFTS is a clinical syndrome and does not have pathognomonic pathology. Furthermore, to diagnose PFTS, the herd-level clinical case definition must be fulfilled.
Superficial colitis was highly prevalent in our previously reported investigation in both PFTS and nonaffected groups and suggested that colitis was a background lesion in the farm investigated. 9 In the current study, the overall prevalence of superficial colitis in PFTS-CASE pigs was lower (31%), but the prevalence varied widely by farm (0%–67%). This further supports the observation that colitis is not a consistent finding of PFTS and does not likely contribute significantly to the condition.
In our previous diagnostic investigation, 9 we also reported that rhinitis and nonsuppurative meningoencephalitis were lesions possibly associated with PFTS. In the present study, rhinitis and nonsuppurative meningoencephalitis or trigeminal ganglioneuritis were not related to pig type (CASE or CTRL), but these lesions were at higher odds of being present in pigs from PFTS farms than in those from NON-PFTS farms. Whether they are risk factors for farms developing PFTS is unknown and needs to be verified in a larger study. That being said, the high prevalence of encephalitis or trigeminal ganglioneuritis in the PFTS-CASE pigs (62.5%; Table 1) is of great interest, even though it did not differ significantly from the prevalence in PFTS-CTRL pigs (55%; Table 1). If the NON-PFTS farms were not included in the analyses, one may incorrectly conclude that encephalitis or trigeminal ganglioneuritis is a common lesion in pigs. Importantly, these neural lesions were significantly less prevalent in the pigs from NON-PFTS farms (<30%; Table 1).
The high prevalence of superficial gastritis, small intestinal villous atrophy, and thymic atrophy of PFTS-CASE pigs is largely consistent with those we have previously reported. 9 While these lesions are not pathognomic for PFTS, they are consistent across farms that fulfill the case definition. This is very relevant, because if the pathology varied among farms, one may conclude that PFTS was a heterogeneous condition. The results of this study indicate that the pigs affected by PFTS had similarities in clinical and pathologic features.
Thymic atrophy and small intestinal villous atrophy can be caused by lack of feed intake 7,16 or by infection that causes apoptosis or necrosis of thymic lymphocytes and intestinal villi. There are few published data describing the lesions of anorexia or starvation in pigs. In mice and rats, food deprivation results in gastric erosion and ulceration; 1,13,15 however, lesions have developed in the fundus of mice 15 but in the nonsecretory regions of rats. 1,13 In pigs, gastric ulcers associated with feed disruption almost always have developed in the nonsecretory pars esophagea, 4 but the lesions observed in PFTS-CASE pigs were almost exclusively in the fundus. Furthermore, the erosions described in mice and rats have been grossly visible, 1,13,15 but those in PFTS pigs were microscopic and ulceration was not present. Although the attenuation of foveolar cells in PFTS pigs might represent disuse atrophy associated with anorexia, the increased lymphoplasmacytic infiltrate cannot be easily explained by anorexia alone. Thus, the relationship between superficial gastritis and anorexia in PFTS-CASE pigs is uncertain. It should be noted that in another PFTS investigation using a small number of pigs, superficial gastritis was observed in healthy pigs on the day of weaning, in pigs fasted for 4 days, and in pigs affected by PFTS. 16 This suggests that superficial gastritis is present in healthy pigs around the time of weaning. It is, however, not known if the apparently healthy pigs at weaning with superficial gastritis experienced any degree of anorexia or reduced feed intake before sampling, nor was it possible to demonstrate prospectively that these pigs would not develop PFTS following weaning.
Based on the present results, the microscopic examination of the stomach, small intestine, and thymus potentially provides relevant information in the diagnostic workup of debilitated pigs. A highly sensitive pig-level diagnostic criterion involves a pig having characteristic lesions present in at least 2 of these 3 organs (ie, having at least 2 or 3 of superficial gastritis, small intestinal villous atrophy, or thymic atrophy). This criterion has a high sensitivity (100%) but moderate specificity (67.4%). Alternatively, a moderately sensitive diagnostic criterion involves a pig having characteristic lesions present in all 3 organs. With this criterion, the false-positive rate will decrease considerably, but one may misdiagnose some PFTS pigs as not having the syndrome. We suggest this latter criterion be used when larger groups of animals are examined for herd-level diagnosis.
The results of the current study do not support a causal role for any of the pathogens investigated, which is consistent with previous reports. 9,16 The evidence to date indicates that PFTS is not caused by PCV2, PRRSV, FLUAV, RV-A and B, PECV, TGEV, M. hyopneumonia, or H. pylori–like or H. heilmannii–like organisms. PFTS is unlikely to be caused by HEV and PEVs, since these viruses are detectable in both PFTS- and NON-PFTS farms and in CASE and CTRL pigs with no difference in the odds ratio. RV-C was associated with PFTS at the farm level, but the prevalence was high in PFTS and NON-PFTS farms. The presence of HEV or PEVs was not associated with lesions in the brain or trigeminal ganglion. Thus, although nonsuppurative meningoencephalitis and trigeminal ganglioneuritis were present in pigs from farms with PFTS, the etiology is not known. Furthermore, cytomegaloviral inclusion bodies were not consistently detected in PFTS farms. Thus, although HEV, PEV, and SuHV2 may contribute to the clinical signs of some PFTS-CASE pigs, there is no clear causal relationship with PFTS.
In conclusion, PFTS-CASE pigs have significantly higher odds of exhibiting superficial gastritis, small intestinal villous atrophy, and thymic atrophy compared with NON-PFTS pigs. Moreover, PFTS can be ruled out upon gross and histologic examination alone if pigs do not have at least 2 of these 3 lesions. By contrast, PFTS should be considered if debilitated pigs have all 3 histologic lesions, although approximately 15% false-positive and false-negative rates would be expected. Fulfillment of the clinical case definition, the presence of the above lesions, and the absence of underlying pathology and pathogens need to be considered together to diagnose PFTS at the farm level. The etiology of PFTS is not yet determined, and the current evidence does not support a causal role for any of the swine pathogens investigated in this study. Finally, nonsuppurative meningoencephalitis, neutrophilic lymphoplasmacytic rhinitis, and RV-C infection appear to be associated with PFTS at the farm level, but their potential roles in pathogenesis need further study.
Footnotes
Acknowledgements
We greatly appreciate the swine practitioners’ (Drs Henry, Dubois, Megan, Magrath, Kaminski, O’Sullivan, and Van Assen) and diagnostic laboratories’ (Prairie Diagnostic Services, Kansas State Veterinary Diagnostic Laboratory, Animal Health Laboratory, University of Guelph, Veterinary Diagnostic Services of Manitoba Agriculture, Food and Rural Initiatives) participation and assistance in this project. We are grateful for the technical assistance of Crissie Auckland and Dakota Wightman. We also appreciate Dr Brendan O’Connor for his comments on the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research was funded by Canadian Swine Health Board (grant number 2011-C5390), Saskatchewan Ministry of Agricultural (Agriculture Development Fund, grant number 2009-268), and Saskatchewan Pork Development Board (grant number 2009-01).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
