Abstract
Clostridium perfringens type D causes enterotoxemia in sheep and goats. The disease is mediated by epsilon toxin (ETX), which affects the cerebrovascular endothelium, increasing vascular permeability and leading to cerebral edema. In the present study, we compared the distribution and severity of the cerebrovascular changes induced in lambs by C. perfringens type D strain CN1020, its isogenic etx null mutant, and the ETX-producing complemented mutant. We also applied histochemical and immunohistochemical markers to further characterize the brain lesions induced by ETX. Both ETX-producing strains induced extensive cerebrovascular damage that did not differ significantly between each other in nature, neuroanatomic distribution, or severity. By contrast, lambs inoculated with the etx mutant or sterile, nontoxic culture medium did not develop detectable brain lesions, confirming that the neuropathologic effects observed in these infections are dependent on ETX production. Lambs treated with the wild-type and complemented strains showed perivascular and mural vascular edema, as well as serum albumin extravasation, particularly severe in the cerebral white matter, midbrain, medulla oblongata, and cerebellum. Brains of animals inoculated with the ETX-producing strains showed decreased expression of glial fibrillary acidic protein and increased expression of aquaporin-4 in the end-feet processes of the astrocytes around blood vessels. Early axonal injury was demonstrated with anti–amyloid precursor protein immunohistochemistry. Perivascular accumulation of macrophages/microglia with intracytoplasmic albumin globules was also observed in these animals. This study demonstrates that ETX is responsible for the major cerebrovascular changes in C. perfringens type D–induced disease.
The virulence of Clostridium perfringens is mostly dependent on its ∼16 toxin armory. Toxin production varies from strain to strain, permitting classification of C. perfringens isolates into 5 toxinotypes (A, B, C, D, and E), based on the production of 4 typing toxins—namely, α (CPA), β, ∊ (ETX), and ι toxins. C. perfringens type D isolates produce both CPA and ETX. ETX, a 32-kDa pore-forming toxin, is the third most potent clostridial toxin after botulinum and tetanus toxins. Accordingly, ETX has been of interest as a potential bioterrorism agent, and it was included until recently on the US Centers for Disease Control and Prevention category B list of select toxins and in similar lists in other countries. Group B agents are considered moderately easy to disseminate and would result in significant morbidity if civilian populations were to be exposed. To assist in the development of vaccines against these agents, there needs to be a better understanding of the interaction between toxins such as ETX and their target tissues. This is particularly important for ETX because there is no information on the likely effect of this neurotoxin in humans, 12 although recently, ETX has been proposed as a possible environmental trigger of multiple sclerosis in humans. 20
Genetic and animal model studies have shown that ETX is necessary for C. perfringens type D isolates to produce disease in natural and experimental hosts. 7 ETX is produced as a relatively inactive prototoxin in the gastrointestinal tract of animals and becomes fully toxic by proteolytic removal of the C-terminal 14 amino acids. 14 Activated ETX is then absorbed through the intestinal mucosa and transported to several target organs, including, but not limited to, the brain, lungs, and kidneys. 24
C. perfringens type D causes enterotoxemia in sheep and goats and, more rarely, in cattle and perhaps other animal species. 13,24 The disease is mostly characterized by neurologic signs and lesions, which are believed to be the consequence of the action of ETX. The pathogenesis of the ETX-induced lesions in the brain of these natural hosts for type D disease has not been fully elucidated. ETX appears to bind to specific receptor sites on the luminal surface of cerebral vascular endothelial cells, which seem to be, at least initially, the principal target of toxin-induced brain injury. 3,23 While a direct and damaging effect of ETX on cerebral vascular endothelium is considered the fundamental basis of clinicopathologic changes in ETX-exposed sheep, the precise mechanism of this perturbation of the blood-brain barrier is incompletely understood. 19 We therefore used brains from lambs challenged with a wild-type strain of C. perfringens type D, its isogenic ETX null mutant, and an ETX-complemented strain, collected during a previous study, to investigate the pathogenesis of C. perfringens type D–mediated neurologic disease. 7
Materials and Methods
Bacterial Strains and Experimental Inoculations
Formalin-fixed brains from 24 Mulley lambs were available from a previous study. 7 Six animals each had been experimentally inoculated intraduodenally with 1 of 3 isogenic strains of C. perfringens type D: either the wild-type strain CN1020, its etx mutant JIR4981, or the etx mutant complemented in trans with the wild-type etx gene (JIR12604) or with sterile tryptone glucose yeast agar (TGY) medium (control). The construction of the mutant strains and the details of the inoculation procedures, as well as the clinical and pathologic data, tissue collection, and initial processing, were published elsewhere. 7,9 All procedures involving animals were reviewed and approved by the University of California–Davis Institutional Animal Care and Use Committee (permit 16383).
In the current study, the brains of all 24 lambs were sliced coronally at ∼0.5-cm intervals, and photographs of 5 selected slices (a–e from rostral to caudal) from each case were obtained. These images were then used to illustrate the topographic distribution of the microscopic lesions as follows (Fig. 1a–e). Slice a was obtained at the level of the forebrain, included the cerebral cortex and brainstem, and involved the following major structures: corona radiata, corpus callosum, corpus striatum (internal and external capsules, caudate nuclei, globus pallidus, and putamen), lateral ventricles, and cingulate gyri. Slice b also included the cerebral cortex and brainstem and involved the thalamus, hypothalamus, subthalamus, habenula, lateral ventricles, third ventricle, internal capsule, dentate gyrus, hippocampus, claustrum, and entorhinal cortex. Slice c included the brainstem only (midbrain), involving the cerebral aqueduct, periaqueductal gray matter, anterior and posterior colliculi, reticular formation, crus cerebri, cerebral peduncle, and oculomotor, red, and interpeduncular nuclei. Slice d included the medulla oblongata and cerebellum, involving the white matter of the cerebellar vermis and cerebellar hemisphere, anterior cerebellar peduncles, reticular formation, lingula, trapezoid body, and dorsal nuclei of the trapezoid body, cerebellar cortex, and cerebellar roof nuclei. Slice e included only the medulla oblongata at the level of the obex, which included several nuclei (external cuneate, hypoglossal nucleus, dorsal motor nucleus of vagus, nucleus and spinal tract of the trigeminal nerve, nucleus of the solitary tract, and ventral regions of the reticular formation).
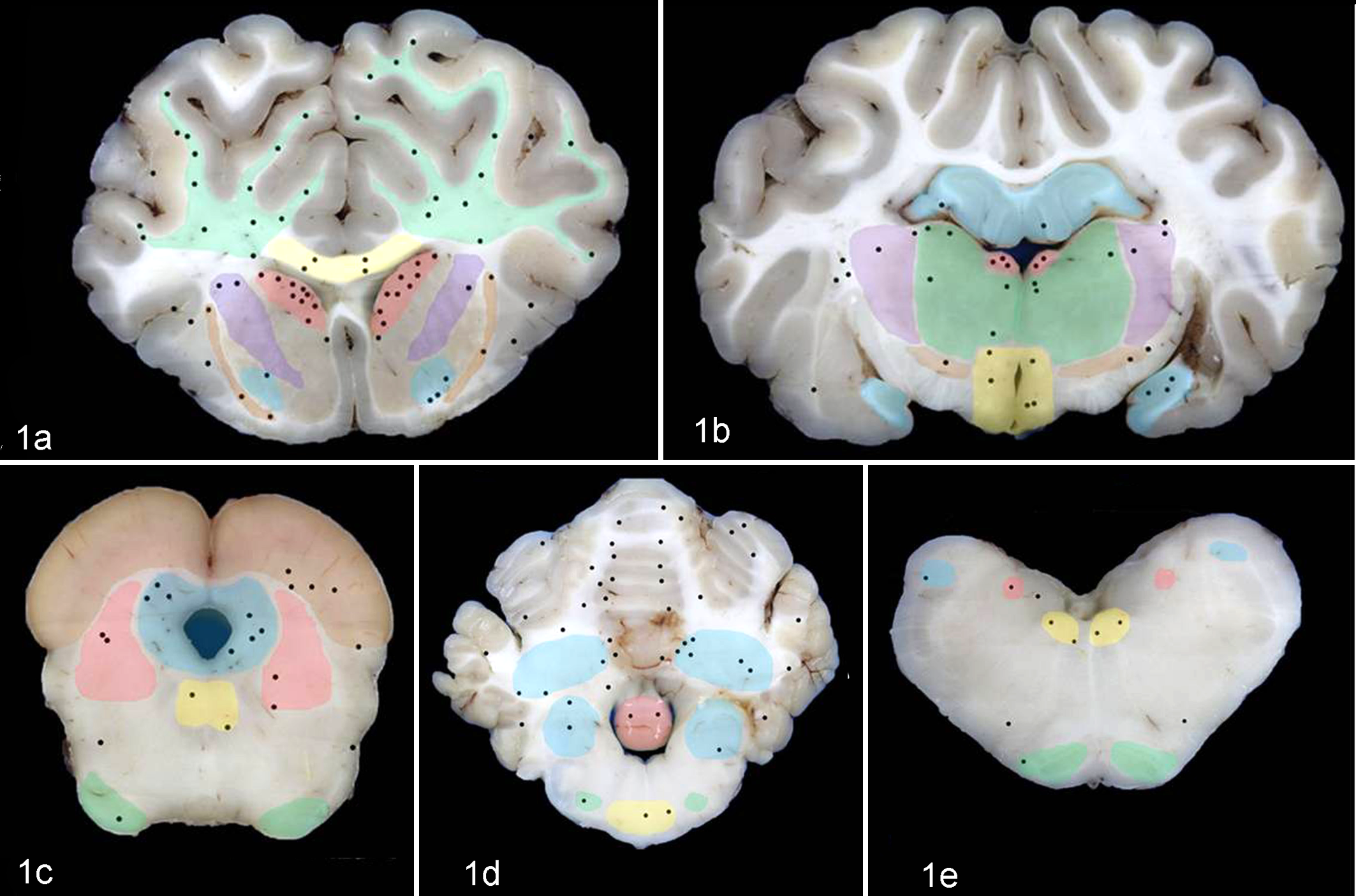
Brain; sheep. Schematic representation of the topographic distribution of the perivascular edema (black dots) in the brain of lambs inoculated intraduodenally with the wild-type Clostridium perfringens type D strain CN1020. Coronal sections include cerebral cortex (a, b); corpus striatum (a) and thalamus (b); midbrain (c); cerebellum, cerebellar peduncles, and pons (d); and medulla oblongata (e). The most significantly affected structures are color-coded as follows: (a) white matter of the cerebrum and corona radiata (green), corpus callosum (yellow), internal capsule (violet), external capsule (brown), caudate nucleus (salmon), and globus pallidus (light blue); (b) hippocampus (blue), thalamus (green), internal capsule (violet), habenula (salmon), hypothalamus (yellow), and subthalamus (brown); (c) rostral colliculus (brown), periaqueductal gray matter (blue), oculomotor nucleus (yellow), reticular formation (salmon), and crus cerebri (green); (d) white and gray matter of the cerebellum (uncolored), vestibular nuclei (blue), lingula (salmon), trapezoid body (yellow), and dorsal nuclei of the trapezoid body (green); and (e) spinal tract of the trigeminal nerve (blue), nucleus of the solitary tract (salmon), hypoglossal nucleus (yellow), ventral regions of the reticular formation (uncolored), and pyramidal tract (green).
Histopathology and Histochemistry
In all animals, multiple samples for histology were obtained bilaterally from slices a to e (Fig. 1a–e), so each slice was fully examined histologically. All samples were processed routinely, embedded in paraffin, and sectioned to obtain 4-μm-thick sections, which were then stained with hematoxylin and eosin (HE). Detailed microscopic examination of all sections was performed and the microscopic lesions detected in HE-stained sections were localized topographically using the macroscopic images of the brains obtained during trimming (Fig. 1a–e). The degree of severity of perivascular edema, defined as perivascular accumulation of eosinophilic proteinaceous material (Fig. 2), present at each neuroanatomical location was scored using a subjective scale from 0 (no edema) to 5 (severe edema) (Table 1). For this, 2 pathologists examined independently each section in a blinded fashion and came up with a subjective numerical score. The average of these 2 values was used as a final score.
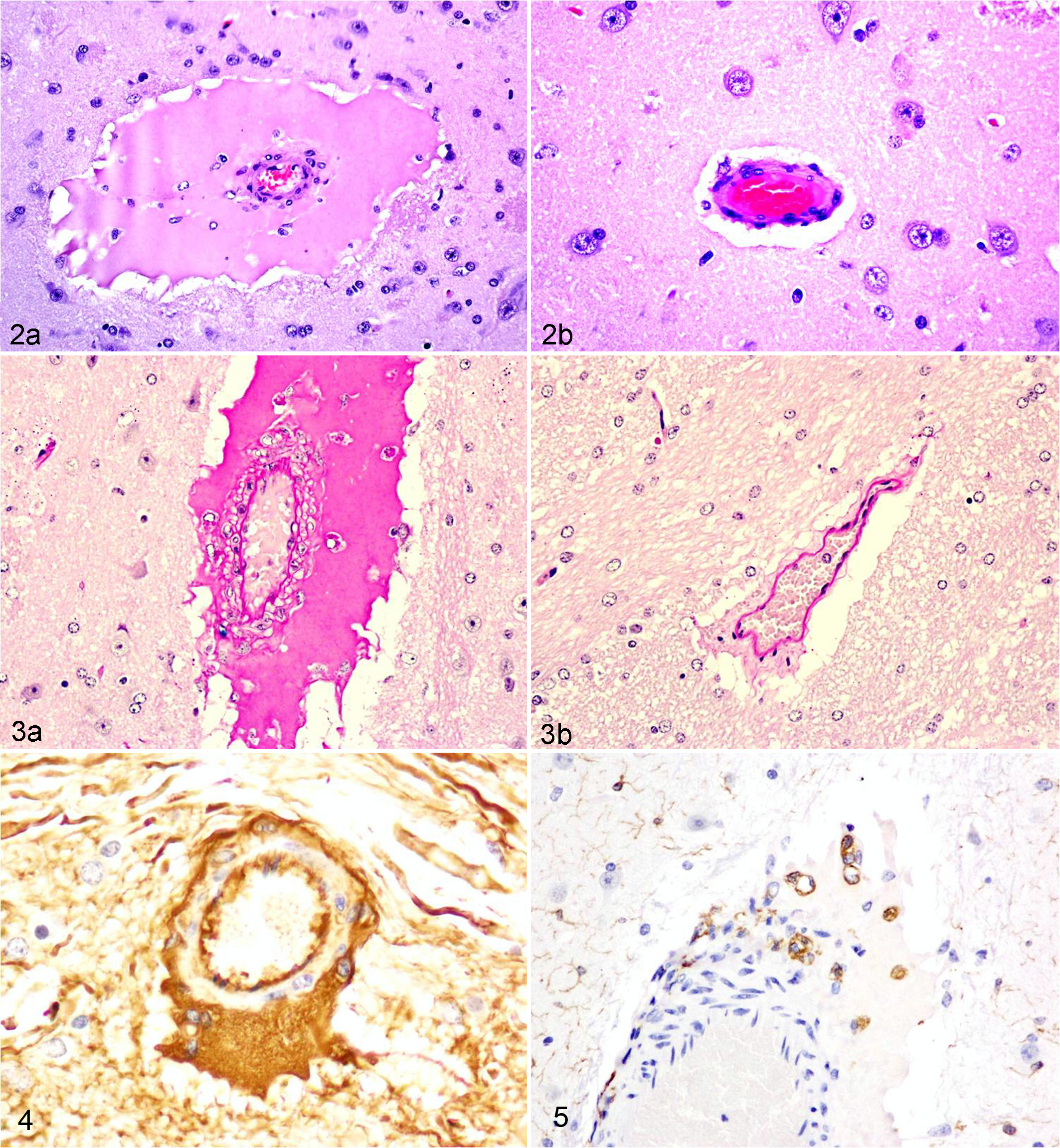
Brain; sheep. (a) Perivascular edema in the globus pallidus of a lamb inoculated with Clostridium perfringens type D wild-type strain CN1020. The perivascular space of a small arteriole in the gray matter is markedly and circumferentially expanded by deeply eosinophilic homogeneous proteinaceous fluid (edema) that displaces and compresses the adjacent neuroparenchyma. Note mild spongiosis of the neuropil immediately adjacent to the edematous perivascular space. Floating freely in the proteinaceous fluid are scattered nuclei of polygonal cells consistent with macrophages/microglial cells (see Figs. 3, 5, and 7 and text for further detail). (b) A normal similar-caliber arteriole in the same anatomic location of a control lamb is surrounded by a thin clear halo, which represents artifactual separation/retraction of the neuropil/vessel during histologic processing. Hematoxylin and eosin (HE).
Degree of Microscopic Perivascular Edema Observed in the Brain of 6 Lambs Inoculated With the Wild-Type Clostridium perfringens Type D Strain CN1020.a

aThe degree was scored using a scale of 0 to 5, with 0 indicating no edema and values between 1 and 5 indicating increasingly severe edema.
bOverall score = average ± standard deviation.
Sections of the corpus striatum of most animals in the 4 groups were stained with periodic acid–Schiff stain (PAS) to evaluate the integrity of the basement membranes of the blood vessels affected by edema and to further characterize the perivascular edema. In addition, to assess the integrity of the tunica adventitia (collagen type III) of the blood vessels, Gomori’s reticulin stain was performed on sections of the corpus striatum from most animals in all 4 groups.
Immunohistochemistry
Selected sections from all areas of the brain from most animals in all groups were also processed for immunohistochemical detection of injured axons (amyloid precursor protein [APP]), astrocytic reaction (glial fibrillary acidic protein [GFAP]), the water channel protein, aquaporin-4 (AQP4), serum albumin, and a microglial/macrophage marker (ionized calcium-binding adaptor molecule 1 [Iba1]). The following primary antibodies were used: a mouse monoclonal antibody anti-APP (clone 22C11, Chemicon; Millipore, Billerica, MA), a rabbit polyclonal antibody (Pab) anti-GFAP (Dako, Carpinteria, CA), a rabbit Pab against AQP4 (Millipore, Burlingame, CA), a goat Pab against rat albumin (which cross-reacts with sheep albumin; Cappel, West Chester, PA), and a rabbit Pab against Iba1 (Wako Pure Chemical Industries, Osaka, Japan). Rabbit, mouse, or goat IgGs were used as primary antibodies in negative control sections. The Dako EnVision+ horseradish peroxidase (HRP) kit (Dako) was used for APP, according to the instructions of the manufacturer and standard operating procedures of the California Animal Health and Food Safety Laboratory, UC Davis. For GFAP, an avidin-biotin-peroxidase method (Vectastain Elite Kit; Vector Laboratories, Burlingame, CA) was used according to the manufacturer’s instructions and standard operating procedures of the Veterinary Medical Teaching Hospital, UC Davis. Serum albumin, AQP4, and Iba1 were incubated with a streptavidin-conjugated peroxidase tertiary (Pierce, Pasadena, CA), according to the instructions of the manufacturer. For APP and GFAP, immunoreactivity was visualized using the chromogen 3-amino-9-ethylcarbazole (AEC K4001; Dako). Albumin, AQP4, and Iba1 were visualized with 3,3′-diaminobenzidine tetrahydrochloride (DAB). After chromogen incubation, all sections were counterstained with hematoxylin, cleared, and mounted. Positive and negative control tissues were included in these protocols.
Results
Comparative analysis of infected sheep revealed that animals inoculated with the wild-type or the etx-complemented strains showed almost identical microscopic and immunohistochemical alterations in the brain, while no significant abnormalities were observed in animals inoculated with the etx mutant or sterile TGY medium (controls). For those animals inoculated with the wild type, the topographic distribution of these microscopic lesions is depicted in Fig. 1a–e, and the degree of severity of the perivascular edema scored for each neuroanatomical location is shown in Table 1.
The topographical distribution and severity of lesions in lambs inoculated with the complemented strain (data not shown) did not differ significantly from those described in lambs inoculated with the wild type. In animals inoculated with the wild-type or complemented strains, the areas of the brain most frequently affected within the brain sections obtained at the level of the corpus striatum were found in the white matter of the cerebral cortex, corona radiata, corpus callosum, internal capsule, external capsule, caudate nucleus, and globus pallidus (Fig. 1a). In the sections obtained at the level of the thalamus, areas of microangiopathy were found in the hippocampus, thalamus, hypothalamus, subthalamus, habenula, and internal capsule (Fig. 1b). In the midbrain, the main affected regions were the anterior colliculus, periaqueductal gray matter, oculomotor and red nuclei, reticular formation, and crus cerebri (Fig. 1c). Within the brainstem and cerebellum, perivascular edema was found within the white matter of the cerebellar vermis and cerebellar hemisphere, lingula, cerebellar nuclei, trapezoid body, and dorsal nuclei of the trapezoid body (Fig. 1d). In the sections of the medulla oblongata at the level of the obex, the most severe lesions were found in the spinal tract of the trigeminal nerve, nucleus of the solitary tract, hypoglossal nucleus, ventral regions of the reticular formation, and pyramidal tract (Fig. 1e).
In HE-stained brain sections of animals inoculated with the wild-type or etx-complemented strains, the perivascular spaces of the microvasculature (postcapillary venules and capillaries) and midsize blood vessels (arterioles and venules) were often markedly distended with lakes of extravasated eosinophilic, homogeneous, proteinaceous edema fluid (Fig. 2a). This change was not seen in the brains of any of the control animals or in those animals inoculated with the etx mutant (Fig. 2b).
The extravasated fluid observed in the brains of animals inoculated with the wild-type or complemented strains also stained strongly positive with PAS (Fig. 3a) and was strongly immunoreactive with antibody to albumin (Fig. 4). This perivascular edema sometimes extended to infiltrate the neuroparenchyma surrounding affected blood vessels. The albumin immunolabeling intensity was stronger in the perivascular spaces and progressively became weaker with increasing distance from injured vessels. Occasionally, a large number of hyaline droplets rather than lakes of fluid distended the perivascular space. Large polygonal mononuclear cells with round to reniform nuclei and abundant, acidophilic, vacuolated cytoplasm were frequently observed immersed in the perivascular edema fluid. These cells were immunoreactive with anti-Iba1 (Fig. 5). When evaluated with PAS stain and immunohistochemistry for albumin, these macrophages contained abundant large intracytoplasmic PAS-positive (Fig. 3a) and albumin-immunoreactive globules. Uptake of leaked albumin by nearby neurons was also sometimes evident with immunohistochemistry for albumin. In brains of animals inoculated with the etx mutant strain or in control brains, extravasation of serum albumin was not detected.
PAS staining was used to evaluate the integrity of the basement membranes of the blood vessels affected by edema in the brain of animals inoculated with the wild-type or complemented strains. The basement membrane in affected vessels did not lose the affinity reaction for this stain, and it was evident as a continuous deeply PAS-positive laminar layer subjacent to the endothelium (Fig. 3a,b) in brain vessels of all groups studied.
Gomori’s reticulin stain was performed to assess the integrity of the tunica adventitia of blood vessels. In those blood vessels affected by perivascular edema in lambs inoculated with the wild-type or complemented strains, the fluid dissected through and partially disrupted the reticulin meshwork of the tunica adventitia, but it was often still contained within this layer rather than being perivascular. The reticulin fibers containing the edema within the vessel walls (Fig. 6a) allowed for the differentiation between the mural edema and the clear perivascular area of artifactual separation due to vascular/parenchymal retraction (Fig. 6b).
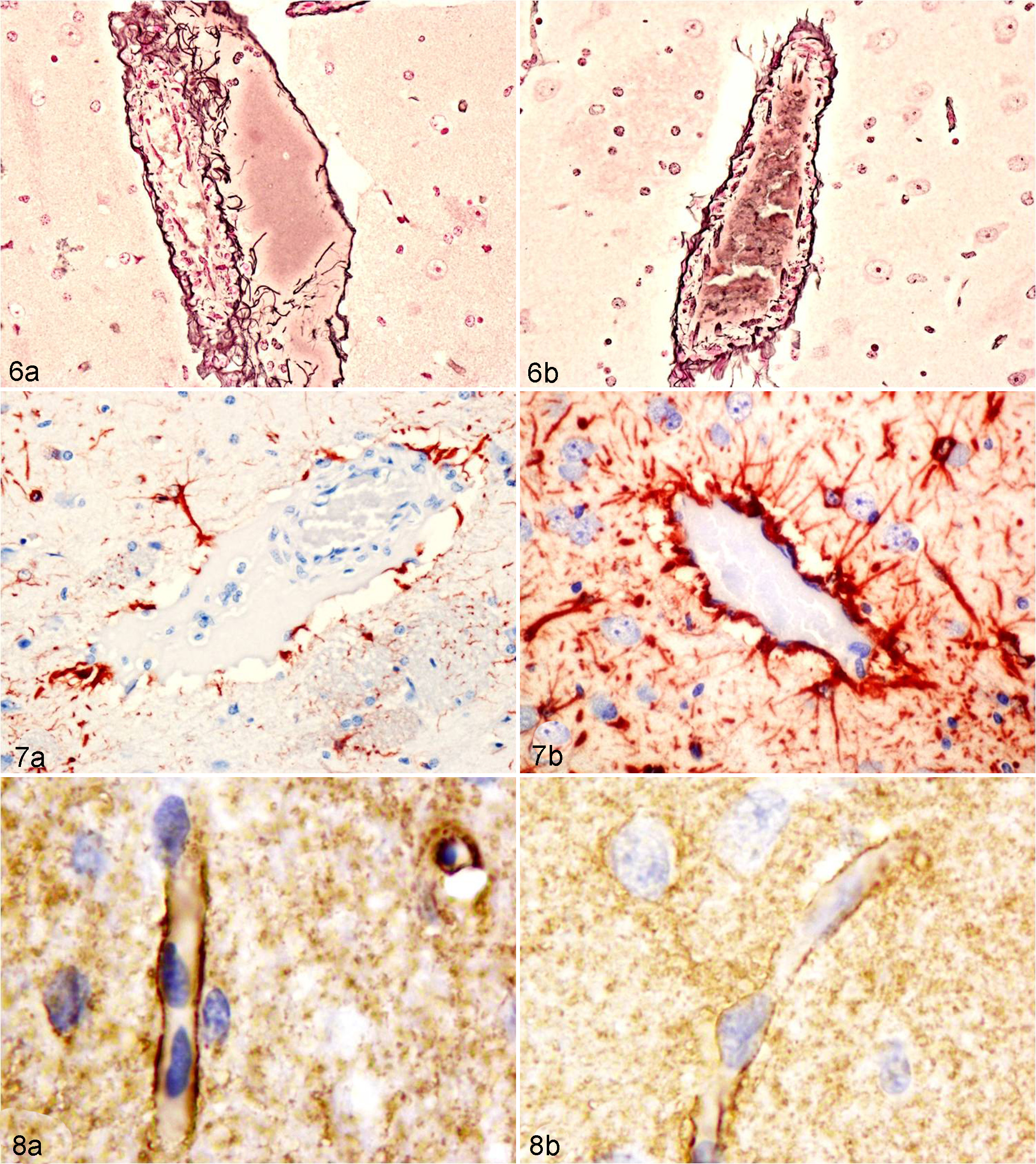
Sheep; brain. Gomori’s stain of affected and nonaffected blood vessels. (a) Caudate nucleus of a lamb inoculated with the wild-type strain. Edema fluid dissects through and disrupts the reticulin meshwork of the arteriolar adventitia, causing pathologic separation from the tunica muscularis. The edema fluid is still contained within the adventitia, indicating mural rather than perivascular edema. (b) In a control lamb, the reticulin fiber meshwork of the tunica adventitia of a normal arteriole (not affected by mural edema) is closely apposed to the tunica muscularis and externally surrounded by a clear zone that artifactually separates the tunica adventitia and the surrounding neuropil. Gomori.
In a few blood vessels of lambs from the wild-type or complemented-inoculated groups, the reticulin meshwork was completely disrupted and permitted the leakage of the edema fluid directly into the Virchow-Robin space (perivascular edema). In the control animals and in the lambs inoculated with the etx mutant, the tunica adventitia of all blood vessels, regardless of their size and location, was characterized by a continuous meshwork of interwoven reticulin fibers that completely surrounded and delineated the most external aspect of the vessels (Fig. 6b). The tunica adventitia was in close apposition to the tunica media in arterioles or the tunica intima in the capillaries and venules.
GFAP immunohistochemistry was performed to evaluate the reaction of astrocytic end feet that envelop cerebral blood vessels. Regardless of the size of the blood vessel, in control animals, etx mutant-inoculated animals, and nonaffected brain regions of the animals inoculated with either the wild-type or complemented strains, GFAP revealed strong immunoreactivity that identified astrocyte foot processes around the adventitia of the vast majority of brain blood vessels. In those vessels affected by edema in brains of wild-type and complemented-inoculated animals, the edema fluid expanding the adventitia and Virchow-Robin space affected the astrocyte end-feet processes of the blood-brain barrier, resulting in partial or total loss of immunoreactivity around the expanded adventitia (Fig. 7a). In those nonaffected vessels of the brain from the 4 groups of animals, in which there was artifactual separation between the vascular adventitia and the adjacent brain parenchyma, the astrocyte end-feet processes remained attached to the vascular adventitia and sometimes the astrocytic cytoplasmic processes could be visualized crossing through the zone of artifactual separation (Fig. 7b). The free-floating polygonal mononuclear cells that were seen in the edema fluid on HE-stained sections did not stain with anti-GFAP, indicating that these cells were not astrocytes.
In the brain of animals inoculated with the wild-type or complemented strains, astrocytes presented numerous coarse AQP4-immunopositive granules, particularly in foot processes surrounding microvessels (Fig. 8a) and the inner and outer glia limitans. In the brains of control animals and those inoculated with the etx mutant strain, AQP4 immunopositivity was expressed as a fine granularity in astrocytic processes, with slightly increased expression where end feet were in contact with capillaries and postcapillary venules (Fig. 8b) and the inner (ependymal) and outer (pia mater) surfaces of the brain (glia limitans). Iba1 immunohistochemistry was helpful to identify macrophages/microglial cells in the perivascular spaces of vessels affected by edema in the animals inoculated with the wild-type or complemented strains (Fig. 5). In these sheep, numerous APP-immunoreactive axons (injured axons) were found in the neuropil and white matter adjacent to vessels showing perivascular edema (Fig. 9a,b) in the cerebral cortex, corpus striatum, thalamus, anterior colliculus, cerebellum, and rarely in the medulla oblongata. Uninjured axons did not stain with this immunohistochemical technique in brains from any of the 4 treatment groups (Fig. 9c).
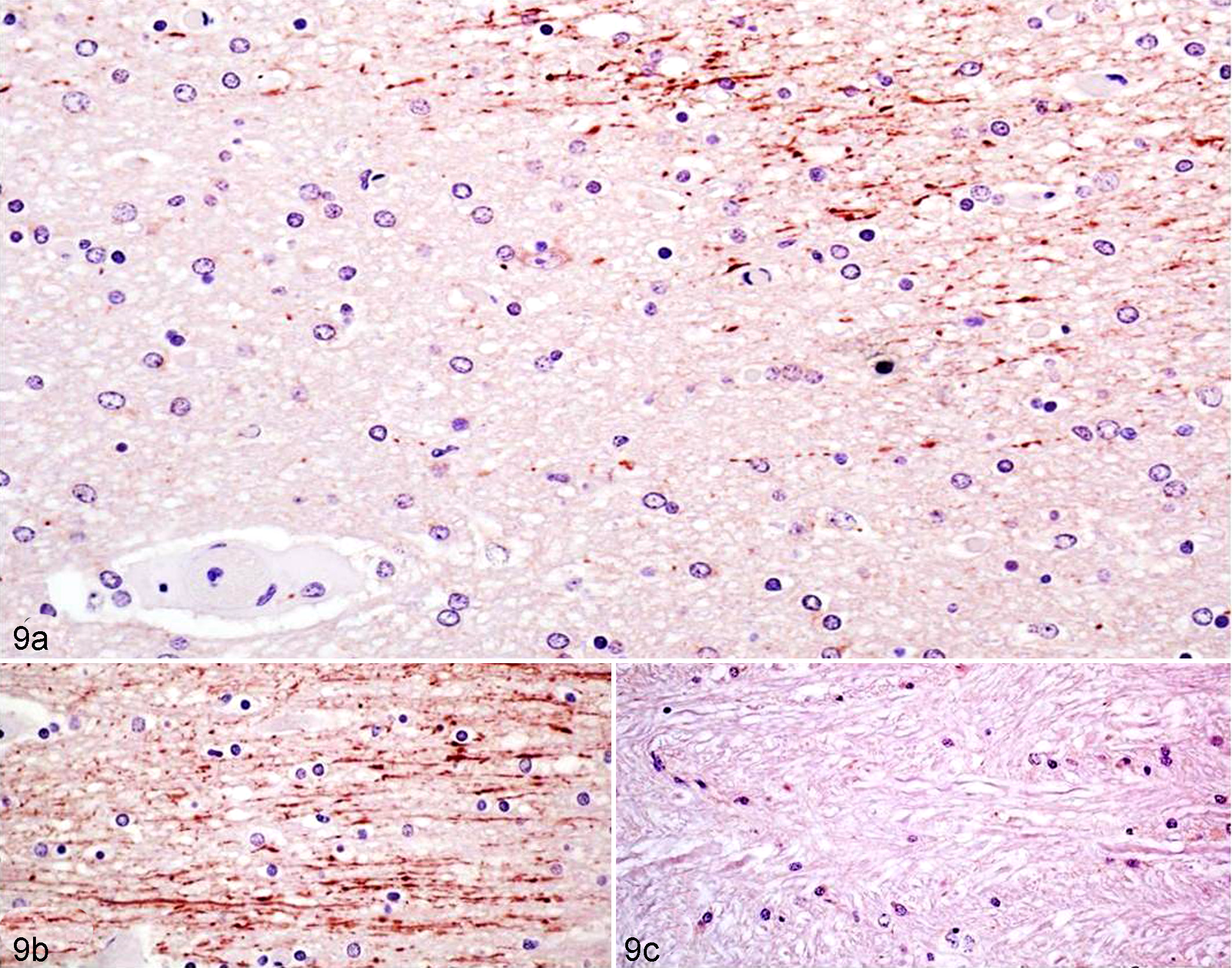
Sheep; brain. (a) Increased amyloid precursor protein (APP) immunoreactivity in white matter tract near a vessel affected by perivascular edema in a lamb inoculated with the wild type. (b) White matter of the cerebral cortex of the same inoculated lamb showing strong linear granular APP immunoreactivity confirming the presence of early axonal injury. (c) White matter of the cerebral cortex in a control lamb, showing total lack of axonal labeling. Anti-APP immunohistochemistry.
Discussion
Vaccination with crude ETX toxoids has proven successful in reducing the incidence of ETX-induced neurologic disease in sheep and goats. Moreover, administration of epsilon prototoxin to laboratory rodents appears to block neural receptor sites and prevent or ameliorate the effects of experimentally inoculated ETX. 11,17 However, no prophylactic methods for ETX intoxication are currently available for use in humans. In these cases, new or developing pharmacological interventions are necessary. However, the careful targeting of these treatments will be predicated upon a better understanding of the interaction between ETX and neural structures. The brain is the most nonhomogeneous tissue in the body, and neural insults do not affect the brain uniformly. Accordingly, we sought to better define the cerebrovascular changes produced by this potent neurotoxin, as well as their neuroanatomic distribution.
In this study, we examined in detail the topographical distribution of vascular lesions in the brain of sheep infected with a toxigenic wild-type strain of C. perfringens type D and compared them with the effects of an isogenic etx mutant and its complemented derivative or sterile TGY medium. Significant differences were observed between animals infected with the wild-type or the complemented strains versus sheep challenged with the etx mutant or sterile TGY medium, which confirms that ETX is necessary to produce the pathologic changes characteristic of type D enterotoxemia in the brain of sheep. Since the fundamental lesion in the neurologic disorder produced by ETX is vascular injury, we also used immunohistochemistry and histochemical techniques to further study the mural changes produced by this toxin in affected blood vessels, as well as the perivascular cell response.
ETX produces 2 principal clinicopathologic syndromes in sheep, the expression of its neurotoxicity probably depending on the amount of circulating toxin that reaches the brain and the duration of the clinical course of disease. 5 With high circulating toxin levels, injured microvascular endothelial cells swell, lose their cytoplasmic organelles, and form blebs on their luminal surface. The cytoplasm is eventually reduced to an attenuated, electron-dense band with nuclear pyknosis. 4,5,8,15 This breakdown in the blood-brain barrier then results in a rapid extravasation of fluid and plasma proteins into the extracellular space, producing a severe, generalized vasogenic cerebral edema. As a consequence of these lesions, affected sheep develop severe convulsions, become comatose, and soon die. This severe acute form of the disease is not associated with grossly visible necrotizing lesions in the brain that are typical of the subacute to chronic form but is characterized by microscopic perivascular aggregates of proteinaceous fluid and more generalized edema. Herniation of the cerebellar vermis through the foramen magnum (cerebellar coning), presumably as a result of generalized brain edema and rapid increase of the intracranial pressure, has been described at necropsy, albeit rarely, in this acute form of ETX-induced enterotoxemia. 22 In our previous experiments, from which the brains were obtained for the current study, we successfully reproduced acute clinical type D disease in sheep. 7 However, none of the clinically affected animals in that study developed grossly visible lesions of cerebellar vermis herniation (although some had severe widespread microscopic cerebral edema), supporting the fact that this is an uncommon pathologic manifestation, at least in animals that succumb spontaneously or are euthanized within 24 hours postinoculation.
When the ovine brain is exposed to lower ETX levels (or in partially immune sheep), the clinical course is more prolonged, and at necropsy, macroscopic bilaterally symmetrical foci of necrosis (termed focal symmetrical encephalomalacia), with or without hemorrhage, are found in selectively vulnerable brain regions, particularly the corpus striatum, internal capsule, thalamus, midbrain, and cerebellar peduncles. 2,25
Experimental studies have shown that the development of neural damage occurs in a seemingly dose- and time-dependent manner. 5 High doses of ETX tend to widen the distribution of brain lesions, although still favoring certain neuroanatomical regions, while lower toxin doses produce smaller, well-circumscribed necrotic foci that are confined to fewer vulnerable sites. 5 Moreover, as the time from toxin administration to death increases, so does the distribution of brain lesions. 5 In the present study, the topographical pattern of extravasated plasma protein after acute ETX exposure was not restricted to these susceptible neuroanatomical regions but was more widely distributed, also involving the white matter of the cerebral cortex and cerebellum, as well as the medulla oblongata. In all neuroanatomical sites examined, perivascular edema was more commonly found in the white rather than the gray matter, which is a characteristic distribution of vasogenic edema. This white matter preference is believed to be consequence of the sparse cellularity and larger extracellular spaces in white matter, which permits greater and more rapid accumulation and spread of edema fluid. 10
In this study, perivascular albumin extravasation was widely distributed in the brains of lambs inoculated with the wild-type and etx-complemented strain and was often found as lakes of pooled, leaked plasma albumin, as determined by immunohistochemistry, although intramural (adventitial) vascular edema was also a feature of ETX-induced brain damage. Albumin-rich edema fluid leakage from capillaries and small- to medium-size blood vessels also correlated with widely upregulated AQP4 immunopositivity in edematous toxin-exposed brains, especially in astrocytic processes abutting damaged capillaries and postcapillary venules, but also in subpial and subependymal sites. Aquaporins (AQP) are a family of 13-membrane channel proteins that facilitate the movement of water in the brain. Of these, AQP4 is the most abundant in the brain, and it is expressed in astrocytes at fluid-parenchymal interfaces in the blood-brain and subependymal/subpial-cerebrospinal fluid barriers. Cells expressing AQP4 on plasma membranes have up to 50-fold higher osmotic water permeability than membranes that are devoid of these proteins. 26 AQP4 is found in high concentration in astrocytic perivascular end feet, which, together with the dense astrocytic network under the pia and ependyma, suggests that this protein is involved in regulation of water movement at the blood-brain and cerebrospinal fluid–brain barriers, respectively. 18,27 The functions of AQP4 in the pathogenesis of vasogenic and cytotoxic brain edema seem to be opposed. In vasogenic edema, extra water enters the extracellular space independently of AQP4. However, this excess water is removed from the brain through AQP4 channels in the astrocytic foot-processes. By contrast, the AQP4 channels in the astrocytic foot-processes have an active role in the formation of cytotoxic edema. 16 In the only other study of AQP4 expression in ETX-treated brains, Finnie et al 6 found that the diffuse cerebral edema produced in rats given ETX was correlated with widespread upregulation of AQP4 in astrocytes. This result suggested that AQP4 plays a role in clearance of edema fluid from the brain by astrocytic uptake, moving water from the extracellular space into these glial cells and reducing osmotic stress on surrounding neurons. 6 The increased expression of AQP4 in these rat brains was concordant with the findings in sheep in the present study.
GFAP, the principal intermediate filament cytoskeletal protein in mature astrocytes, is widely used as an immunohistochemical marker of these glial cells. 21 In the present study, GFAP expression in perivascularly disposed astrocytic end feet was markedly reduced in those blood vessels showing edema compared with that in unaffected vessels. It is possible that the resulting intracellular edema in perivascular astrocytes mediated by AQP4 structurally alters the GFAP filaments, reducing their detection by immunohistochemistry. The results of GFAP immunohistochemistry also suggest that the perivascular edema detaches the astrocytic end feet from the affected blood vessels, rather than infiltrating the parenchyma surrounding these processes.
In addition, Iba1-immunoreactive macrophages/microglial cells accumulated within the edema fluid in perivascular spaces and these cells contained abundant intracytoplasmic globules that had an affinity for PAS, which was comparable to that of the extravascular edema fluid. Furthermore, these intracytoplasmic globules were immunoreactive with anti–albumin antibody. These findings suggest that, after ETX-induced vascular damage, macrophages/microglial cells incorporate extravasated albumin by endocytosis in an attempt to resolve the edema. This represents the first time intracytoplasmic uptake of albumin has been demonstrated in perivascular macrophages in ETX-damaged vessels.
To evaluate the integrity of the vascular basement membranes in the brain, we used PAS staining on brains from all groups. PAS stain intensity of basement membranes did not differ between these groups, and no changes in the morphology of this component of the blood-brain barrier could be detected with this stain.
In the present study, APP immunohistochemistry was used to detect early axonal injury. APP is normally transported along the axon in an anterograde direction by fast axoplasmic transport as a membrane-bound vesicular protein that accumulates rapidly proximal to the site of injury. Only injured axons are immunopositive, while normal, uninjured axons are unlabeled. APP is the most sensitive, early immunohistochemical marker of early axonal injury, with immunopositive injury being detected as early as 1.75 to 3 hours after injury, the number and size of the axonal swellings (spheroids) increasing over the next 24 hours. 1 Although no axonal damage was detected during routine examination of HE-stained brain sections in any of the brains from the 4 groups of sheep, numerous APP-immunoreactive injured axons were found in some edematous areas near injured blood vessels in brains of sheep inoculated with the wild-type or etx-complemented strains. This axonal injury could have been the result of hypoxia-ischemia from an edema-associated reduction in cerebral perfusion or a direct cytotoxic effect of extravasated ETX. However, since APP axonal immunopositivity cannot distinguish axons injured by ischemia-hypoxia from other etiologies, 1 the precise cause of the observed axonal injury is uncertain.
Conclusion
In this study, we characterized the distribution and severity of cerebral edema caused by a wild-type ETX-producing strain of C. perfringens type D. This pathology was not observed in an isogenic etx mutant but was restored upon complementation of that mutant with the wild-type etx gene. These data provide clear evidence that the observed pathology resulted from the effect of ETX. In addition, we have demonstrated that the acute vascular injury and attendant perivascular extravasation of proteinaceous fluid induced by ETX was widely distributed in ovine brains, involving the white matter of the cerebral cortex, midbrain, medulla oblongata, and cerebellum. Moreover, the pattern of proteinaceous fluid extravasation differed in topographical distribution from, and was substantially more widespread than, the multifocal necrosis produced by subacute or chronic intoxication. We have also demonstrated that, at least in initial stages of the pathogenesis, there is not only perivascular edema but also intramural (adventitial) vascular edema in the brain of affected sheep and that this edema expands the perivascular spaces, disrupting the astrocytic component of the blood-brain barrier, as demonstrated with GFAP immunohistochemistry. In addition, our results suggest that overexpression of AQP4 in the astrocyte end foot of the blood-brain barrier and perivascular macrophage uptake of extravasated serum albumin are 2 putative mechanisms involved in attempting to resolve the vasogenic edema in this condition. Finally, axonal injury as demonstrated with APP immunohistochemistry has been described herein for the first time in ETX-exposed sheep brains.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was generously supported by grant R01 AI056177 from the National Institute of Allergy and Infectious Diseases. Research at Monash University was also supported by funding provided by the Australian Research Council to the Australian Research Council Centre of Excellence in Structural and Functional Microbial Genomics.
