Abstract
Macrophages are an important leukocyte component of the microenvironment of neoplasms. Macrophages have classically been subdivided into M1 and M2, depending on their roles in immune response, wound healing, and promotion or inhibition of tumor growth. In human breast cancer, increased presence of M2 macrophages has been associated with poor prognosis. The authors hypothesized that rat mammary carcinomas have increased macrophage influx compared to benign mammary proliferative lesions and normal mammary glands as well. In humans, both M1 and M2 macrophages express CD68, while CD163 is expressed primarily by M2 macrophages. Based on a single immunolabeling protocol with anti-CD68 and anti-CD163 antibodies, the extent of macrophage influx was investigated by morphometry to quantitate the immunopositive cells in normal rat mammary glands, benign mammary proliferative lesions, and mammary carcinomas. In mammary carcinomas, there was significantly higher percentage of CD68+ cells compared to benign mammary proliferative lesions and normal mammary glands. There was also higher percentage of CD163+ cells in mammary carcinomas compared to benign mammary proliferative lesions. Thus, increase in CD68+ and CD163+ macrophages corresponded to increased malignancy of rat mammary tumors in this study.
One determinant of tumor growth is the ability of neoplastic cells to proliferate within the tumor microenvironment, which provides a supportive and interactive setting for neoplastic cells. The tumor microenvironment comprises stroma and, commonly, inflammatory cell infiltrates. In mammary tumors, influx of macrophages is an important component of the tumor microenvironment, and their presence has been associated with poor prognosis. 2,19 Earlier studies in mice found that tumor-associated macrophages are present in greater proportion in metastatic compared to nonmetastatic mammary tumors, suggesting that macrophages may facilitate tumor cell escape from immune surveillance and subsequent metastasis. 12 Infiltrating macrophages have been associated with necrotic and hypoxic areas of tumor tissue, and the resultant upregulation of proangiogenic genes by these cells may be associated with tumor malignancy. 11 More recent studies showed that the presence of macrophages in neoplasms is generally, although not always, correlated with increased rate of metastasis and, hence, poor prognosis, while reduced macrophage infiltration has been associated with inhibition of tumor growth and reduced metastasis. 19
M1 and M2 macrophages have been described in inflammatory and healing lesions. M1 or classical macrophages are induced by interferon gamma, tumor necrosis factor α, and lipopolysaccharide and are typically regarded to have microbiocidal and tumoricidal functions. 17,21 M2 or alternative macrophages are induced by interleukins 4, 10, and 13 and glucocorticoids, as well as immune complexes. 17,21 M2 macrophages exhibit scavenging and angiogenic and antiparasitic activity and promote tissue repair and tumor progression. 18,19
In humans, glycoprotein CD68 is the predominant marker used for detection of macrophages in tumors, labeling both M1 and M2 macrophages. 7 CD163 is highly expressed in M2 macrophages and can be used to discriminate between M1 and M2 macrophages. 7 Based on human breast cancer studies using CD68 as a marker for macrophages, large numbers of intratumoral CD68+ macrophages are associated with decreased survival and poor prognosis. 3 In laboratory rodents, CD68 is similarly used as a macrophage marker, although this glycoprotein is also expressed in phagosomes and lysosomes of other leukocytes and melanocytes. 15 In contrast, the expression of CD163 in laboratory rodents is mostly confined to monocytes and macrophages. 15
The histologic architecture, developmental growth pattern, and hormonal receptors of mammary glands in the rat mimic those of the human mammary glands. 1,9 The rat is therefore a relevant and useful comparative model for the study of human estrogen receptor–positive and progesterone receptor–positive breast cancers, which are the most common type of human breast cancers.
In this study, we hypothesized that rat mammary carcinomas have increased macrophage influx compared to benign proliferative mammary lesions and normal rat mammary glands. Both CD68 and CD163 markers were used in this study to determine if CD68+ and CD163+ cells are present in different proportions within the 3 histologic groups and to compare a more specific macrophage marker (CD163) to one that also labels other phagocytic cells (CD68). Quantitative morphometry was performed to analyze the immunolabeling using these 2 markers.
Materials and Methods
The present study used archive material from a previous study by one of the authors (A.K.) 10 of 17 cases of malignant mammary neoplasms (2 cribriform adenocarcinomas, 6 acinar adenocarcinomas, 5 solid adenocarcinomas, 4 papillary adenocarcinomas), 13 cases of benign proliferative mammary lesions (3 papillary adenomas, 9 fibroadenomas, 1 simple adenoma), and 17 normal mammary glands (as control) from Sprague-Dawley rats with DMBA-induced mammary tumors (ie, 7, 12-dimethylbenz[α]anthracene). All tissues had been fixed in 10% buffered formalin and paraffin embedded. Five-micrometer sections from each block were cut and stained with hematoxylin and eosin, and tumors were evaluated by a board-certified veterinary pathologist (I.M.L) following previously published criteria. 4 Serial sections of each case were processed for immunohistochemistry.
Immunolabeling was performed with a Leica Bond-Max autostainer (Leica Microsystems, Buffalo Grove, IL). Antigen retrieval was performed by incubating the sections with citrate buffer antigen retrieval solution (S1968, Dako, Carpinteria, CA). Nonspecific antibody binding was blocked with protein-blocking agent (X0909, Dako). Sections were then incubated with 1 of the following 2 primary antibodies: mouse monoclonal anti-rat CD68 (ED1, MCA341R, AbD Serotec, Raleigh, NC) and mouse monoclonal anti-rat CD163 (ED2, MCA342GA, AbD Serotec), both at a dilution of 1:100. A streptavidin-biotin labeling system (LSAB2-K0675, Dako) was used for immunolabeling, and reactions were visualized with 3,3'-diaminobenzidine (K3467, Dako). All slides were counterstained with Gill’s III hematoxylin (Surgipath, Richmond, IL). Sections of rat liver with parasitic granulomas were used as positive controls. Nonspecific labeling was excluded by using a mouse IgG1 negative control antibody (MCA1209, AbD Serotech) at the appropriate concentration.
Slides representing each case were scanned and digitalized using the virtual slide system VS110 (Olympus, Hicksville, NY) with a 20× objective. An automated random subsampling of 50 images was performed on the digitalized slides with the NewCast software (Visiopharm, Hoersholm, Denmark) at the same magnification. Evaluation of the percentage of immunopositive cell labeling per mammary parenchyma (consisting of mammary glandular neoplastic and, if present, nonneoplastic cells, surrounding adipose tissue, fibrous stroma, and areas of necrosis) was carried out with the STEPanizer software (http://www.stepanizer.com). 20 A fine-point grid containing 20 × 20 points (area/point = 222.89 μm2) and a coarse grid of 5 × 5 points (area/point = 3566.28 μm2) were superimposed over the images. The fine-point grid was used to estimate the area of immunopositive cell labeling (for CD68 or CD163), and the coarse grid was used to estimate the reference area (ie, the entire mammary parenchymal tissue) per image by a count of the number of points hitting the immunopositive cells and the mammary parenchyma, respectively. The total area of immunopositive cell labeling within the mammary parenchyma was then estimated by multiplying the number of points with the area/point per section. The percentage of CD68 or CD163 immunolabeling per section was calculated by dividing the area of macrophage labeling by the reference area.
Percentages of CD68 and CD163 immunolabeling were analyzed with analysis of variance, and pairwise multiple comparisons (Student-Newman-Keuls method) were used to compare the percentages of CD68 and CD163 immunohistochemical labeling, within and among the groups: normal mammary glands, benign mammary proliferative lesions, and mammary carcinomas. Significance level was set at P < .05 for each test.
Results
Histologically, the stroma and the tumor microenvironment of rat mammary carcinomas were markedly infiltrated by CD68+ and CD163+ cells, whereas fewer positively labeled cells were present in benign mammary proliferative lesions and normal mammary glands (Figs. 1–9). Rat mammary carcinomas had abundant fibrous stroma (desmoplasia), as well as multifocal areas of necrosis that were variably infiltrated by macrophages. Macrophages formed the predominant leukocyte population within mammary gland tissues in this study. There were also fewer neutrophils and rare mast cells. No melanocytes were observed. CD68+ and CD163+ cells were present in both the stroma and the neoplastic cell aggregates. The percentages of CD68 and CD163 immunolabeling per mammary tissue area, morphometrically quantitated, are shown in Figure 10.
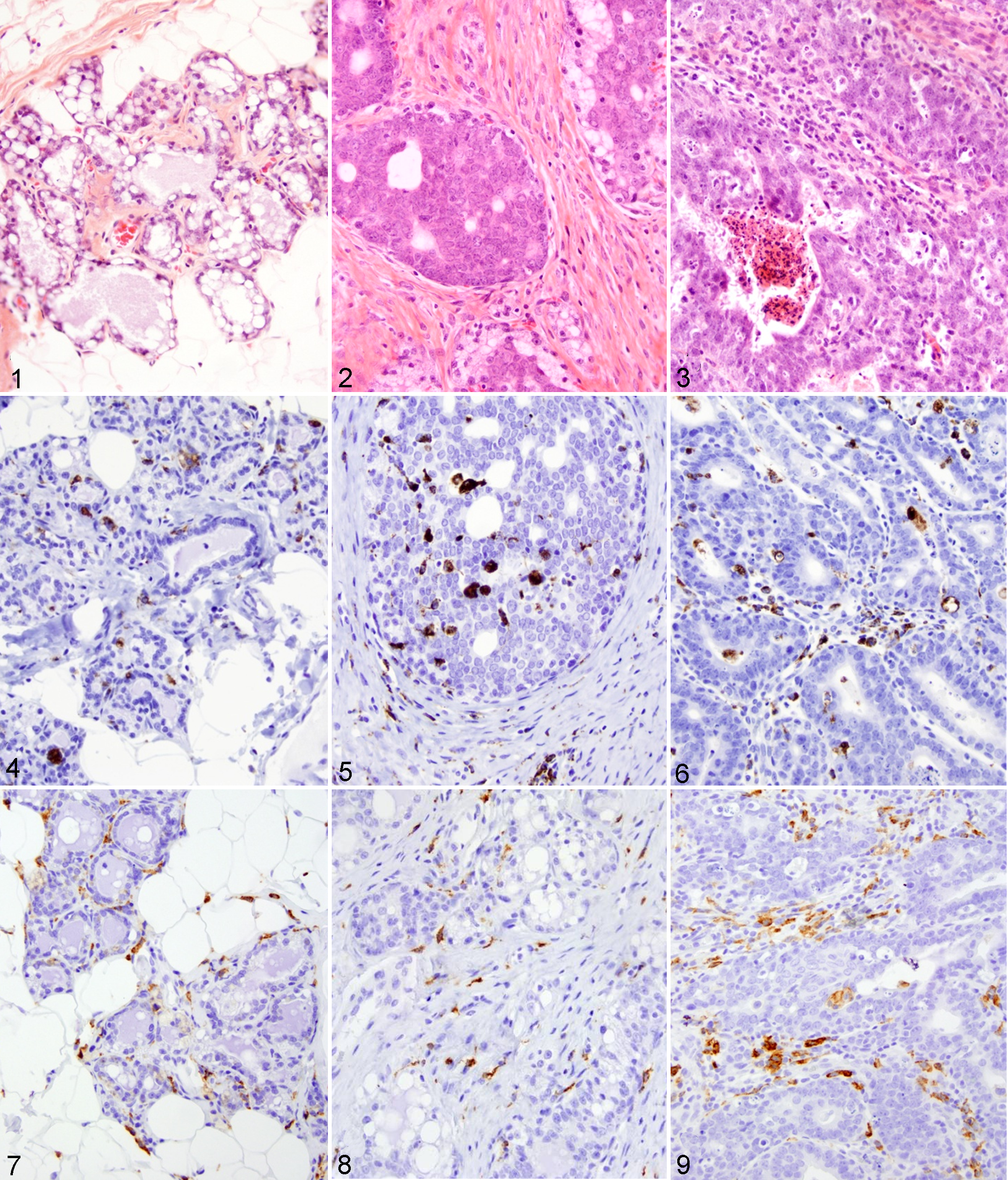
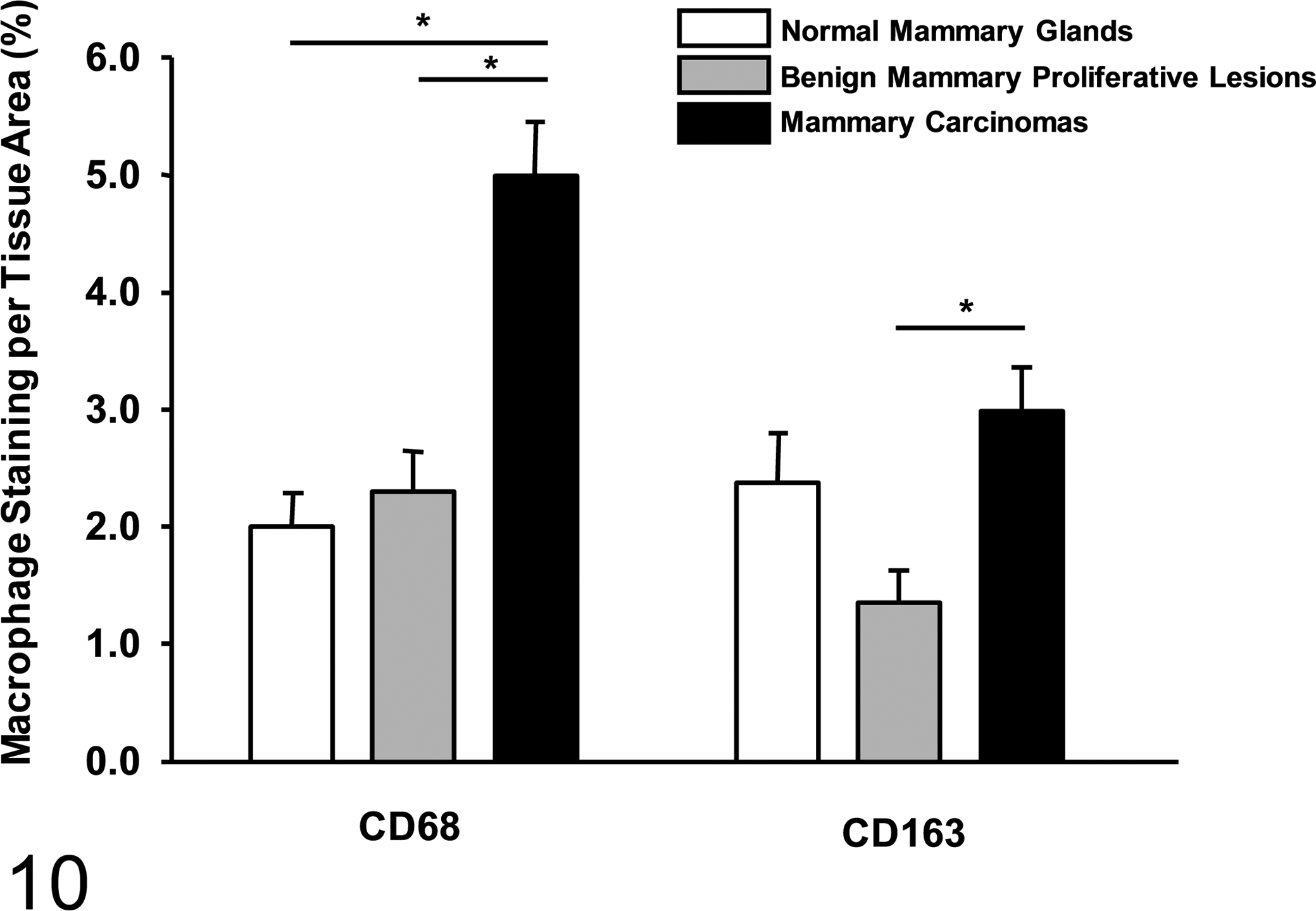
Morphometric quantification of percentage CD68 and CD163 tissue stain. Extent of macrophage infiltration in DMBA-induced rat mammary carcinomas, benign mammary proliferative lesions, and normal rat mammary glands, represented by percentage of CD68 and CD163 immunolabeling per tissue area. Error bars represent standard error of means, and asterisks represent statistically significant differences (P < .05).
Based on pairwise comparison, the percentage of CD68 immunolabeling was significantly higher in the mammary carcinomas group compared to that in the benign proliferative lesions group (P < .001) and that in the normal mammary glands group (P < .001). This supported the hypothesis that increased CD68+ macrophage infiltration is associated with malignancy of mammary tumors in the rat. As for CD163 macrophage marker, there was a significantly higher percentage of tissue immunolabeling in the mammary carcinomas group compared to that of the benign mammary proliferative lesions group (P = .033) but not to that of the normal rat mammary glands group (P = .245). In the rat mammary carcinomas group, the percentage of CD68 immunolabeling was significantly higher than that of CD163 immunolabeling (P < .001), indicating that more CD68+ macrophages were present than CD163+ macrophages. In the benign mammary proliferative lesions and normal mammary gland groups, the percentages of CD68 and CD163 immunolabeling were not significantly different (P = .246 and P = .754, respectively).
Discussion
CD68 is present in phagolysosome membranes in rats. Its expression can therefore be correlated with the phagocytic activity of macrophages. 5 Actively phagocytic macrophages could, at least in part, account for the higher percentage of CD68 immunolabeling in rat mammary carcinomas than that in benign mammary proliferative lesions and normal mammary glands in this study. Rat mammary carcinomas, especially of the cribriform type, often have foci of necrosis characterized by accumulation of cellular debris accompanied by macrophage influx. 13 CD68+ and CD163+ macrophages have also been suggested to induce fibrogenesis through production of galectin 3 and transforming growth factor β1. 8 In the present study, the desmoplastic response within rat mammary carcinomas may have been stimulated by CD68+ and CD163+ macrophages due to the higher percentages of CD68 and CD163 immunolabeling in this group. CD163 is a member of the scavenger receptor cysteine-rich group B family and has been shown to function as a scavenger receptor for hemoglobin–haptoglobin complexes. 14 It is normally expressed by macrophages in mature tissues, such as splenic red pulp macrophages, thymic cortical macrophages, hepatic Kupffer cells, and bone marrow macrophages. 14 CD163+ macrophages produce nitric oxide, tumor necrosis factor α, and interleukins 1β and 6. 14 The higher percentage of CD163 immunolabeling seen in the mammary carcinomas in the present study therefore indicates a proinflammatory microenvironment. Inflammation plays a role in initiation and promotion of cancer; it can also contribute to tumor rejection. 6 In addition, inflammation can contribute to changes in the tumor microenvironment. 6 Inflammatory cells can induce factors that limit cell death, secrete enzymes that alter the extracellular matrix and facilitate tumor invasiveness and metastasis, increase angiogenesis, and activate epithelial-to-mesenchymal transition of the neoplastic cells. 6
In a previous study by Yamashiro et al that evaluated the distribution pattern of macrophage subpopulations in transplanted rat mammary carcinomas, 22 both CD68+ and CD163+ cells were noted extratumorally and intratumorally as in our cases. According to that study, there were slightly more CD163+ cells than CD68+ cells within the tumor and nearly the same number of both cell types in the extratumoral space. Such a proportion of positive cells is in contrast to our study, in which there were more CD68+ cells compared to CD163+ cells in the mammary carcinomas group as well as in the benign proliferative lesions. In contrast to the study by Yamashiro et al, the current study investigated macrophage population in mammary tumors induced by DMBA exposure, while that by Yamashiro et al involved transplanted tumor cells. The disparity in results between the 2 studies could be related to the different methods of generating a rat tumor model, as well as the different analytical techniques.
Our results are more similar to a quantitative study of CD68+ and CD163+ cells in rat liver tissue by Santos et al, in which the authors found more CD68+ cells than CD163+ cells. 16 The authors of this study considered all CD163+ cells to be CD68+, whereas 16% of CD68+ cells were immunonegative for CD163. 16 Considering that in humans CD68 is expressed by both M1 and M2 macrophages and CD163 is expressed predominantly by M2 macrophages, 7 it is tempting to interpret the current immunolabeling results as representing varying populations of M1 and M2 macrophages in rat mammary glands and mammary tumors. However, in rats, there is no conclusive evidence of a correlation of M1 and M2 macrophages and the expression of CD68 and CD163. Alternatively, because CD68 can also be expressed in leukocytes other than macrophages, it is possible that some CD68+ CD163– nonhistiocytic leukocytes were present among the inflammatory infiltrate of mammary gland tissues in our study. It is undetermined why there was a relatively high number of CD163+ macrophages in normal mammary glandular tissue. In the absence of inflammation, they are most likely resident or tissue-fixed macrophages. 22 Another possible explanation is that resident macrophages were diminished in neoplastic lesions, in which case malignant lesions could have had similar numbers as normal glands due to possible compensation of lost resident macrophages by tumor-associated macrophages and inflammatory cells.
In summary, in this immunohistochemical study, increased numbers of CD68+ cells were present in DMBA-induced rat mammary carcinomas compared to benign rat mammary proliferative lesions and normal rat mammary glands. More CD163+ cells were also present in rat mammary carcinomas than in benign mammary proliferative lesions. The difference in percentages of tissue CD68 and CD163 immunolabeling indicates that rat mammary carcinomas are associated with increased infiltration of CD68+ cells and CD163+ cells compared to benign proliferative lesions.
Footnotes
Acknowledgements
We thank Tom Wood and the histology section of the Diagnostic Center for Population and Animal Health for slide processing, Dr Jack Harkema of the Department of Pathobiology and Diagnostic Investigation at Michigan State University for use of the laboratory’s slide scanner, and Sarah Hession at the Michigan State University Center for Statistical Training and Consultation for statistical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
