Abstract
The placenta from an embryo transfer–recipient mare and live foal was examined. The placenta was effaced by multifocal masses, which ranged from less than 1 cm to 14 cm in diameter. The foal represented at 52 days for lethargy, ataxia, and urine dribbling; due to a poor prognosis, the foal was euthanized. At necropsy, the liver was effaced by multifocal, pale, irregular nodules. The lumbar vertebrae and other skeletal sites had multifocal lytic lesions. The placenta had 4 populations of neoplastic cells, including a spindle cell population, tall columnar and transitional epithelial cell populations, and an undifferentiated polygonal cell population. The foal’s liver had similar populations and patterns of cells as those in the placenta. The lesion in the placenta and the masses in the foal were diagnosed as a mixed germ cell tumor and metastatic mixed germ cell tumor, respectively.
The placenta of a 4-hour-old, 54-kg (118-lb) quarter horse filly born to an embryo transfer–recipient mare was presented to Iowa State University. The filly and mare had no significant clinical signs. Routine postpartum examination of the mare’s reproductive tract was unremarkable. The foal was reported to be healthy until 52 days of age, at which time she presented for a 5-day history of hind limb weakness, prolonged periods of recumbency, and difficulty rising. Both the placenta and the foal were examined by the necropsy service at Iowa State University.
The fetal surface of the chorioallantois and allantoamnion were covered by numerous, nodular, friable, firm to gritty, red to yellow, variably sized masses (Fig. 1). These masses did not appear (grossly) to extend into the chorionic surface of the placenta. The largest mass on the chorioallantois was approximately 14 cm long and 5 cm thick and located at the umbilical cord and chorioallantois junction 11 ; it was surrounded by numerous similar but smaller nodules (Fig. 1). These masses did not appear to extend to portions of the placenta occupying the distal pregnant or nonpregnant horns. Another large cylindrical shaped mass (12 × 5 cm) was present on the chorioallantois. These masses tended to localize around the umbilical cord and smaller vessels. On cut surface, the masses were white to yellow and had a mineralized core.
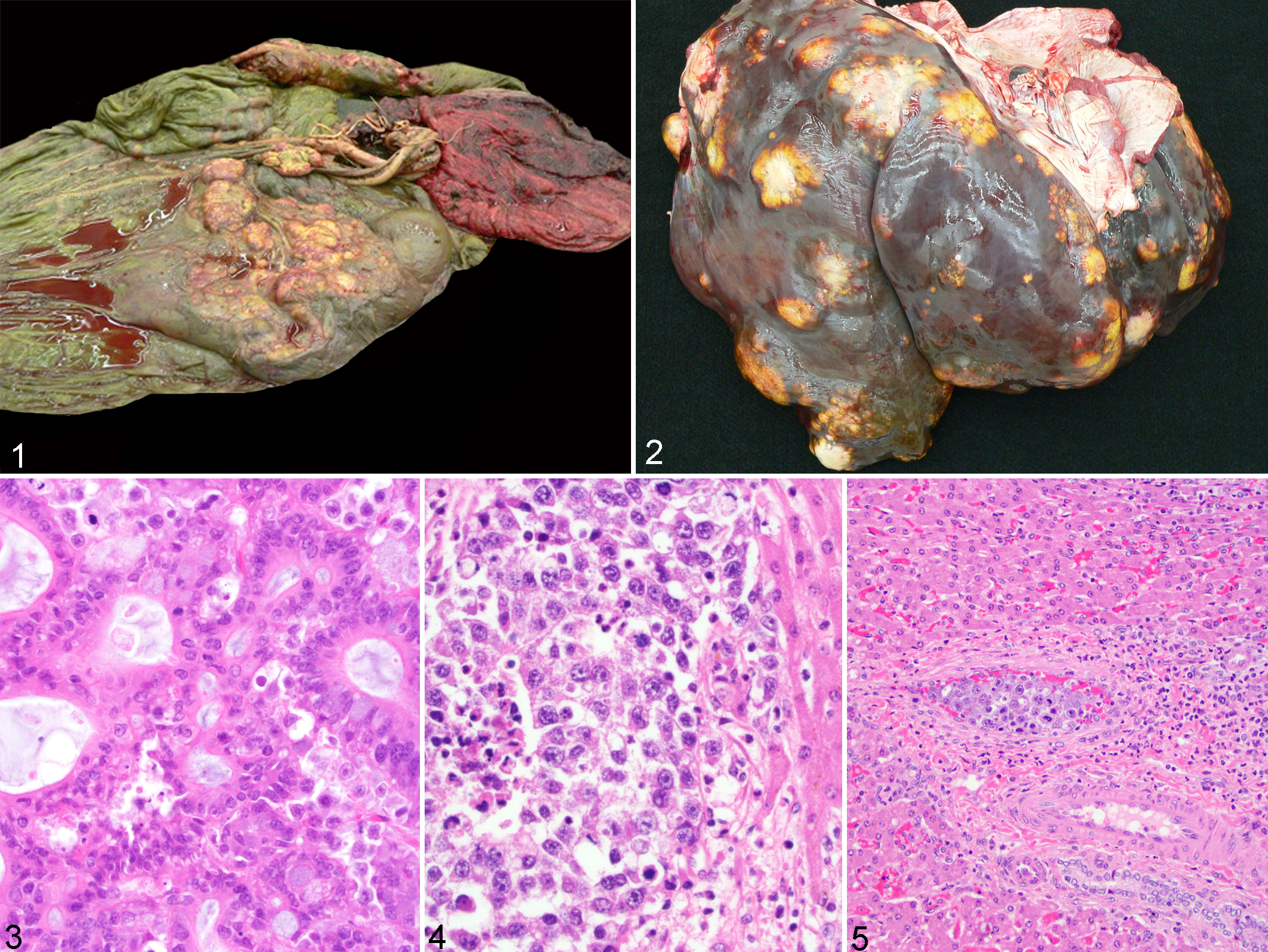
Necropsy of the foal revealed a grossly enlarged and irregular liver, along with multifocal bone lesions. The liver weighed 6.8 kg (15 lb) and had multifocal, irregular, raised nodules that were tan to yellow and often had central cavitations (Fig. 2). Nodules ranged from 0.5 to 3.0 cm in diameter and were located throughout the liver, with multiple visible masses present on the diaphragmatic surface and a large 10-cm-diameter white mass present on the ventral surface. A fracture and osteolysis of the first sacral vertebral body was present, along with brown exudate within the spinal canal at the lumbosacral junction. Bone lesions were also present on the 8th right rib, 11th left rib, and distal right third metacarpal bone, which had hyperostosis of the periosteum.
The chorioallantoic and allantoamnionic masses were unencapsulated, poorly demarcated, and widely expansile masses of similar histomorphology. The masses were located primarily within the allantois, although they extended into the chorion. They were most numerous near the umbilical vasculature and often surrounded and infiltrated allantoic vessels. The neoplastic cells extended into the chorionic villi but did not penetrate beyond the trophoblasts. The masses were composed of 4 distinct populations of neoplastic cells arranged as densely cellular sheets and tubules. The most abundant neoplastic cells (composing approximately 80% of the neoplasm) were round to polygonal with distinct cell borders and moderate amounts of eosinophilic cytoplasm and large, round, vesicular nuclei. There were 2 or 3 prominent nucleoli with marked anisocytosis and anisokaryosis and occasional bizarre mitotic figures. These cells had a mitotic index of 14 per 10 fields (400×). A second population of neoplastic cells consisted of tall columnar epithelium arranged in tubules with a brush border and variably spaced goblet cells. An additional neoplastic population consisted of a stratified cuboidal epithelium, also arranged in tubules, resembling urinary transitional cell epithelium. A fourth neoplastic population consisted of trabeculae formed by spindle-shaped cells with elongate nuclei consistent with smooth muscle. Approximately 40% of the masses were necrotic. Approximately 10% of the masses were mineralized within areas of central necrosis; portions of the allantoic stroma were mineralized. Last, there was a focal area containing thin hair shafts surrounded by a stratified squamous epithelium. Neoplastic cells surrounded allantois arteries and were within the umbilical artery effacing the tunica adventitia and impinging on the tunica muscularis (Fig. 3).
Within the liver were large, unencapsulated, densely cellular, and poorly demarcated multilobular masses that consisted primarily of round to polygonal cells similar to the primary population within the placental mass. These cells had round, vesiculate nuclei, coarsely stippled chromatin, and 1 to 5 prominent nucleoli. There were 1 to 5 mitotic figures per 400× field (Fig. 4). Neoplastic emboli were present within portal veins, lymphatics, and sinusoids that compressed hepatic cords (Fig. 5). In contrast to the placental tumors, the hepatic masses were composed primarily of the polygonal cell population with few tubules or acini. Throughout the liver were large areas of necrosis with associated mineralization affecting up to 70% of the neoplasm in some sections. Similar neoplastic cells were within the vertebral body, ribs, and distal right metacarpal bone. These cells replaced bone marrow. Nearby trabecular bone was necrotic, and new woven bone was present.
Immunohistochemistry was performed on the placental and hepatic masses with a variety of cell markers to try to determine the tissue origin (Table 1). All neoplastic tissues were negative for all markers except pancytokeratin and oct3/4. Neoplastic cells within the placenta, including the round to polygonal cell population, were weakly immunoreactive, and the columnar cells forming tubules were strongly immunoreactive for pancytokeratin and slightly immunoreactive for c-kit (CD117). Neoplastic cells within the placenta and liver were positive for oct3/4. Normal hepatocytes were immunoreactive for hepatocyte marker 1 and c-kit (CD117); subpopulations of cells, particularly the large multinucleated cells and cells of the interstitial stroma, were immunoreactive for vimentin.
Immunoreactivity Produced by Various Primary Antibodies on Formalin Fixed Placental or Hepatic Masses.a
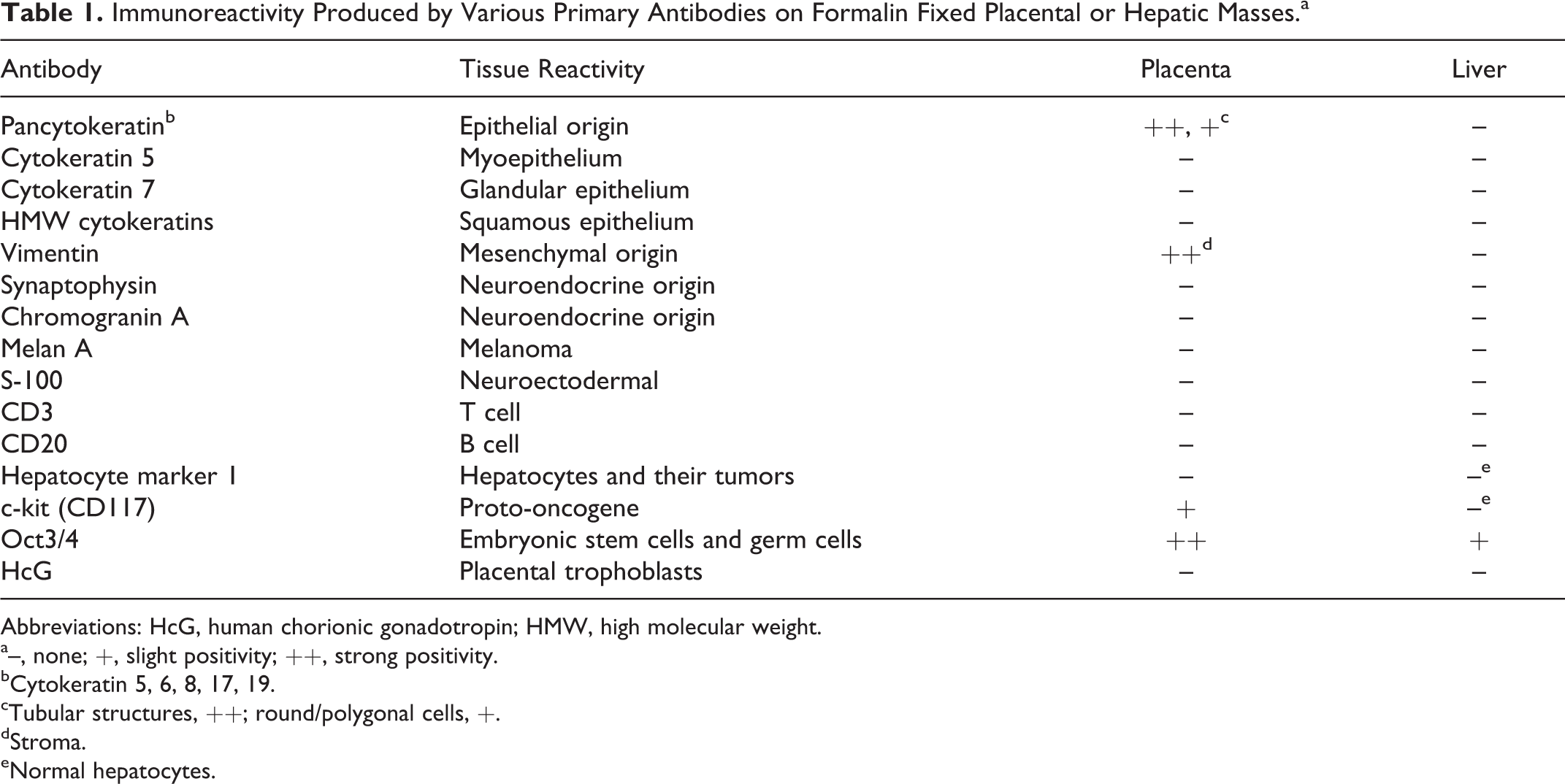
Abbreviations: HcG, human chorionic gonadotropin; HMW, high molecular weight.
a–, none; +, slight positivity; ++, strong positivity.
bCytokeratin 5, 6, 8, 17, 19.
cTubular structures, ++; round/polygonal cells, +.
dStroma.
eNormal hepatocytes.
Oct3/4 is a marker for embryonic stem cells and germ cells and is often used to support a diagnosis of human seminomas. The neoplastic cells within the placenta and liver were both immunoreactive for oct3/4, supporting a diagnosis of a mixed germ cell tumor. The neoplastic cells in both organs were negative for hepatocyte marker 1, a marker of hepatocytes and their tumors. These results rule out hepatoblastoma or other hepatocyte origin neoplasms.
Cytokeratins 5 and 7 and high molecular weight cytokeratins are markers for myoepithelium, glandular epithelium, and squamous epithelium, respectively. Pancytokeratin is a broad-spectrum antikeratin reagent that reacts with intermediate and low molecular weight keratins and cytokeratins 5, 6, 8, 17, and 19. The neoplastic cells within the placenta reacted only with the pancytokeratin marker and not with the other cytokeratin markers. The neoplastic cells within the liver did not react with any epithelial markers. The neoplastic cells resembling epithelium lacked staining for epithelial markers, which was likely due to the cells being poorly differentiated.
Since many of the neoplastic cells had morphology similar to round cells, immunohistochemistry was performed for melan A (melanocyte marker), CD3 (T-cell marker), and CD20 (B-cell marker). The neoplastic cells did not react with these markers. These results rule out several round cell tumors. Neuroendocrine tumors were ruled out by negative immunohistochemistry results for S-100, chromogranin A, and synaptophysin. Human chorionic gonadotropin is a marker that is immunoreactive for trophoblasts, and negative results helped rule out a syncytiotrophoblastic origin of the mass.
On the basis of the morphology and immunohistochemistry results, a diagnosis of a germ cell tumor was made. Differential diagnoses include teratoma, dysgerminoma, choriocarcinoma, and embryonal carcinoma. Although less likely, a yolk sac tumor was considered due to the location of the placental mass at the allantoic portion of the umbilical cord, near the area of the yolk sac remnant. However, the morphology was not consistent with a yolk sac tumor, as there was a lack of yolk sac endoderm and papillary structures composed of a fibrovascular core lined by columnar cells. The possibility of a teratomatous twin was also considered. However, this was considered less likely due to the lack of supportive morphology, such as more mature skeletal structures and organization around a vertebral axis. Also, at the time of the assessment of the placental lesions, there were no known lesions in the foal. Finally, allantoic adenomatous hyperplasia was also considered. This was ruled out by the presence of a neoplastic round cell population rather than cystic or glandular structures lined by a columnar epithelium.
There are few reports of equine placental neoplasia. Those reported include a teratoma and a teratocarcinoma. 1,5 Differential diagnoses to consider for placental nodules in all species include partial and complete hydatidiform moles, choriocarcinoma, metastatic tumors, granulomas, yolk sac tumor, and allantoic adenomatous hyperplasia. 5 Metastasis of another tumor to the placenta was considered unlikely due to the apparent health of the mare.
Based on immunohistochemistry results and tumor morphology, the neoplasm was classified as a mixed nonseminomatous germ cell tumor (NSGCT). 7,10 These neoplasms are composed of pleomorphic cells that have components of a teratoma, embryonal carcinoma, choriocarcinoma, and/or a yolk sac carcinoma and can occur separately or simultaneously. 10 Germ cell tumors can be further classified as either seminomatous or nonseminomatous. Seminomatous tumors occur in the ovaries or testes, whereas nonseminomatous tumors occur elsewhere. 10 NSGCTs are further divided, depending on the basic morphologic pattern recognized, into embryonal carcinomas, teratomas, choriocarcinomas, and yolk sac tumors. Tumors that have features of both embryonal carcinoma and teratoma are referred to as teratocarcinoma. 10 Some tumors exhibit a combination of these patterns and are referred to as mixed NSGCTs. Mixed germ cell tumors are primarily reported within the ovaries or testes, whereas the placenta is rarely affected. 7
Equine placental and hepatic neoplasms are rare. Hepatic and skeletal metastasis from placenta neoplasia, as reported herein, is extremely rare. Primary hepatic neoplasia in horses is uncommon. Hepatoblastoma is the most frequently reported liver tumor in horses and has been reported in animals ranging from late-term aborted fetuses to 3 years of age. 2,4 Reports of liver tumors in foals include hepatocellular carcinoma, 6,8 mixed hamartoma, 9 and mesenchymal hamartoma. 2,9
This case is unique as it describes a placental germ cell tumor that metastasized to the liver and skeletal structures of a foal. Although rare, metastasis from the placenta to the fetus can occur either from a primary placental mass or from a metastatic placental mass. Regardless of the origin of the neoplastic cells (maternal or placental), if those cells reach the fetal circulation via the umbilical vein, the fetal liver is the first organ encountered. The opportunity of neoplastic cells to metastasize outside the fetal liver is possible, especially considering the presence of the ductus venosus, which shunts a portion of the umbilical blood to the caudal vena cava. The most likely explanation for the lesions in this case is the development of a placental germ cell tumor, which spread to the fetal liver hematogenously via the umbilical vein during gestation. This was followed by dissemination to the skeletal system. Choriocarcinomas are one of the few reported placental neoplasias in people with metastatic potential to fetal tissues. 3 When choriocarcinomas metastasize, they most frequently (73%) spread to the liver. 3
A case of placental metastasis has been reported in a 2.5-month-old Arabian foal, although the diagnosis was teratocarcinoma. 1 In that report, a 5-year-old Arabian mare delivered a healthy, full-term colt, and a mass was identified within the placenta. The placental mass was composed of poorly organized islands of bone, cartilage, epithelial tissues, and nests of cells that were reminiscent endocrine tissue. 1 The colt presented at 2.5 months for colic and died. Necropsy of the colt revealed disseminated, multilobulated masses up to 15 cm wide within the liver, with similar smaller masses in the omentum, splenic ligament, sublumbar lymph nodes, and splenic lymph nodes. 1 Immunohistochemical stains were not useful in identifying the origin of the undifferentiated components of the placenta and hepatic masses. Both foals were clinically normal until at least 2 months of age, at which time neoplasm-related lesions resulted in rapid clinical decline.
The case presented herein describes a unique tumor with metastases that is rare in both humans and horses. Mixed germ cell tumors are uncommon in veterinary species. Such tumors with metastasis from the placenta to the fetus are even less frequent. While these neoplasms are rare, their occurrence presents a unique opportunity to further document metastatic neonatal neoplasms.
Footnotes
Acknowledgements
We thank Deborah Moore (Iowa State University) and the personnel of the Purdue Animal Disease Diagnostic Laboratory for immunohistochemical staining, as well as Drs Ron Myers and Michael Yaeger for their help with revisions and edits. We also thank Major Chris Schellhase of the Joint Pathology Center (Silver Spring, MD) for his expert consultation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
