Abstract
Funisitis, inflammation of the umbilical cord, is well recognized in human placentas. This report describes a case of funisitis associated with leptospiral infection in the placenta of a Thoroughbred foal born prematurely. The umbilical cord had diffuse superficial yellow discoloration along its entire length. Microscopic evaluation showed an exudate of neutrophils admixed with fibrin on the surface. Warthin-Starry staining showed spirochetes in the Wharton's jelly of the umbilical cord. A locally extensive, severe placentitis not involving the star and allantoic cystic hyperplasia were the other lesions observed in the allantochorion. Leptospira funisitis is similar to the funisitis of congenital syphilis in humans, although there are some major microscopic differences. in Leptospira funisitis, lesions were limited to the cord surface, whereas in lesions in human umbilical cords with Treponema pallidum infection, the changes are observed mostly around the vessels and in the Wharton's jelly.
Funisitis is a well-characterized lesion in human medical literature. 2 Reports are numerous of placentitis in the veterinary medical literature; however, there are no detailed reports of funisitis. Although funisitis was one of the significant findings in foals aborted in Kentucky during April and May 2001 and 2002 (mare reproductive loss syndrome), the specific cause of funisitis in those abortions has not yet been identified. 4
Several scientific studies report abortion, stillbirth, and premature live birth in horses as a result of leptospiral infection. 6, 10, 13 Although umbilical cord lesions are reported and leptospires are documented in the allantochorion, the umbilical cord has not been routinely examined for leptospires. 13 In utero infection by Leptospira spp. is well documented in the literature, but information on the pathological effects of Leptospira spp. on the umbilical cord is scant. This report describes a case of leptospirosis with funisitis and placentitis in a Thoroughbred mare that foaled 5 weeks prematurely.
The placenta from a Thoroughbred foal born 5 weeks prematurely was submitted to the University of Kentucky Livestock Disease Diagnostic Center in Lexington, KY. The foal was born live but weak. The foal was later euthanized because of progressive clinical deterioration. Grossly, the entire length of umbilical cord was yellow and thickened (Fig. 1). The chorionic surface of the allantochorion had several brownish yellow irregular raised areas on the pregnant horn. The allantoic surface in this area had multiple small microcysts ranging from 1 to 3 cm in diameter. The cervical star area had normal gross appearance. The amnion was markedly thickened, opaque, and light brown in color. Tissue samples of allantochorion from the cervical star area, body, pregnant horn and nonpregnant horn, amnion, and allantoic and amniotic segments of umbilical cord were fixed in 10% neutral buffered formalin, routinely processed, and embedded in paraffin. Five-micrometer sections were mounted on glass slides and stained with hematoxylin and eosin. Selected sections of placenta, amnion, and umbilical cord were stained by the Warthin–Starry (WS) method.
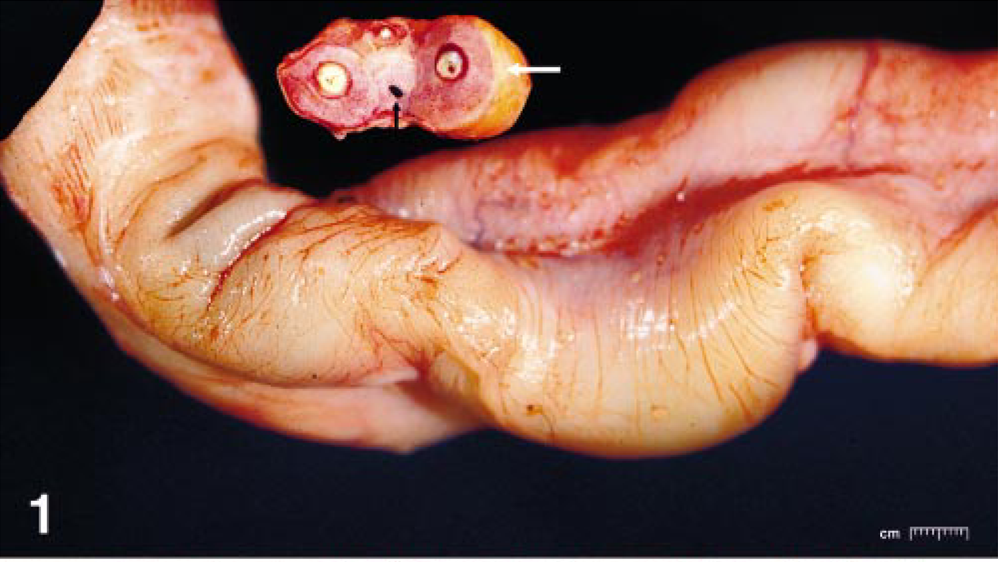
Umbilical cord; horse. Longitudinal view of cord with yellowish discoloration on surface. Cross section, circumferential yellowish discoloration on surface. White arrow shows the fibrin deposit on surface and black arrow shows urachus.
Microscopically, the surface of the umbilical cord was diffusely coated by a dense exudate of mostly nondegenerate neutrophils that were admixed with fibrin (Fig. 2). These neutrophils infiltrated from the cord surface minimally into the Wharton's jelly. Lesions were not identified in cord vessels. The sections of the umbilical cord that were stained with WS demonstrated numerous spirochetes in the Wharton's jelly (Fig. 3). The organisms were also observed in the stroma of the allantochorion, in which gross lesions were observed, but not in the cervical star area or the amnion. The allantoamnion had marked diffuse infiltration of neutrophils and lesser numbers of lymphocytes with necrosis. Multifocally, the stroma underlying the chorionic epithelium in the sections from the pregnant horn had an infiltration of neutrophils, with lesser numbers of lymphocytes and plasma cells. These inflammatory cells were mostly observed in the perivascular areas. The epithelium of chorionic villi in these areas was necrotic. A few blood vessels in the chorionic stroma contained thrombi. The allantoic epithelium exhibited mild hyperplasia with a few microcysts. Direct fluorescent antibody test with the use of multivalent Leptospira fluorescent antibody conjugates (containing antibodies against serogroups canicola, grippotyphosa, hardjo, copenhageni, pomona, and tarassovi) was conducted on the allantochorion and was positive for Leptospira spp. The specific serogroup of Leptospira was not identified.
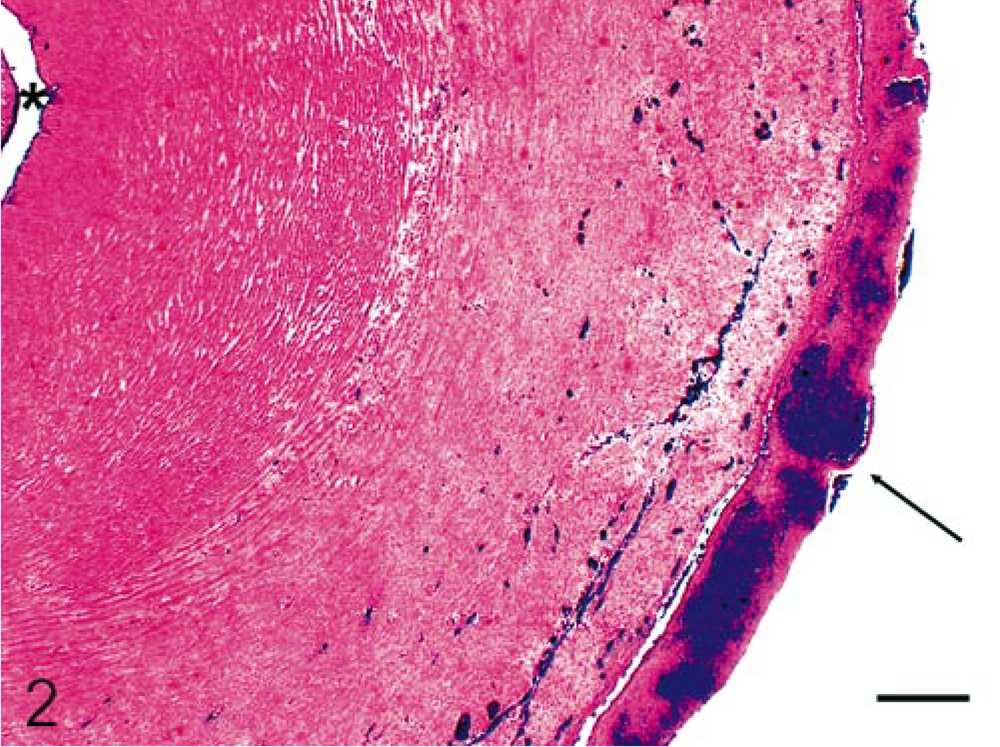
Umbilical cord; horse. Histological appearance of the yellowish material on the surface of the umbilical cord shown in Fig. 1. Surface of the umbilical cord with inflammatory exudate (arrow). Lumen of the umbilical artery (∗). HE. Bar = 250 μm Inset. Degenerate neutrophils (40×) on the surface of the umbilical cord; also, congested vessels are visible.

Umbilical cord; horse. Silver stain demonstrating numerous spirochetes in Wharton's jelly. Warthin–Starry staining. Bar = 25 μm.
Funisitis in human medicine has been subclassified as necrotizing funisitis, acute funisitis, and subacute necrotizing funisitis. It is almost always associated with inflammation of the chorioamnion and ascending infection. 5, 12 Although several infectious agents have also been associated with necrotizing funisitis, the most common disease in human beings that causes necrotizing funisitis is congenital syphilis. 5, 8, 11, 15 Congenital syphilis results from the transplacental passage of Treponema pallidum subsp. pallidum, in a manner presumed to be similar to leptospirosis in horses. The diagnosis of congenital syphilis is often based on serological tests and clinical presentation of the mother and the infant. The demonstration of spirochetes in the chorioamnion or umbilical cord is usually interpreted for clinical and therapeutic purposes as a definitive indicator of fetal infection. 15 In the study of human umbilical cords from mothers with congenital syphilis, the most frequent pathological finding is necrotizing funisitis (36%), and spirochetes were detected in 89% of the umbilical cords by silver and immunofluorescent staining. 15 In a recent study testing umbilical cords for funisitis from T. pallidum infection, 11% of the umbilical cords tested from infants whose mothers were positive for T. pallidum infection had necrotizing funisitis with spirochetes, and 48% of the umbilical cords had varying degrees of funisitis, which underscores the importance of histopathological examination of umbilical cord for infectious agents. 8 In the same study, 9% of the umbilical cords of infants from mothers negative for T. pallidum infection also showed mild funisitis, indicating that all cases of funisitis are not caused by congenital syphilis. Necrotizing funisitis in human is a deep-seated inflammatory lesion within the umbilical cord, which may be accompanied by phlebitis and thrombosis. 5 Several infectious agents have been associated with necrotizing funisitis, but in many cases, the specific cause is not identified. Although T. pallidum is the most important infectious agent causing necrotizing funisitis, herpes simplex virus and Candida albican are also commonly associated with necrotizing funisitis. 9 Herpes simplex funisitis consists of necrotizing funisitis with mild plasma cell infiltrates. 9 Candida albicans funisitis is characterized by grossly appreciable small focal necrotizing lesions on the surface of the cord. 3
In a study conducted to identify the causes of equine abortion and stillbirth among 3,527 horses, a cause was identified in 84% of cases. 6 Among these cases, 18% were due to bacterial infection, and Leptospira spp. accounted for 12.5% of the bacterial abortions (2.2% of 3,527 cases), indicating that leptospirosis is an important cause of infectious abortion and stillbirth. 6 The gross allantochorion lesions associated with equine leptospirosis in the earlier studies consisted of nodular cystic allantoic masses, edema, necrotic areas of chorion, and necrotic mucoid exudate coating the chorion. The microscopic lesions in allantochorion were thrombosis, vasculitis, mixed inflammatory cell infiltration of the stroma and villi, cystic adenomatous hyperplasia of the allantoic epithelium, villi necrosis, and calcification. 13 The gross and microscopic lesions of the umbilical cord included mild to severe edema, focal to multiple sacculations filled with fluid, and coating of the surface with a fibrinous exudate similar to the case of this study, but umbilical cord was not examined for leptospires. Microscopic lesions were observed in 52% of the umbilical cords. 13 In earlier studies, the tissues of choice for demonstration of leptospiral organisms in aborted fetuses were kidney and allantochorion. 13 Because umbilical cords were not always examined in the earlier studies, finding organisms in umbilical cord might increase detection, especially if kidney is not available for testing. The Leptospira serovar/serogroup was not identified in this study because the test was conducted with a multivalent Leptospira fluorescent antibody conjugate. The most common serogroup/serovar isolated from equine leptospirosis-associated abortions/placentitis is Leptospira interrogans serogroup pomona serovar kennewicki. 13
Although similarities are present between equine Leptospira funisitis and human T. pallidum funisitis, major differences do exist. In leptospiral funisitis, lesions were limited to the cord surface and the Wharton's jelly of the umbilical cord, with no visible involvement of the three primary blood vessels, in contrast to the lesion in T. palladium–infected human umbilical cords, in which the changes are observed primarily around the vessels and in the deeper Wharton's jelly. A possible explanation for this could be that T. pallidum infection is an immune-mediated event through lipoproteins; hence, it might be oriented more toward the blood vessels. 14, 16 It is apparent that T. pallidum human infection differs considerably from the equine leptospira infection. The human T. pallidum infection is the consequence of the host's immune response and is characterized by a mononuclear cell infiltrate. The lesion is segmental and often looks like a “barber pole,” and organisms are found in the media and adventitia of vessels only; calcification occurs with predominantly vasculitis. The human and equine umbilical cord also have anatomical differences. The human umbilical cord is devoid of lymphatics and small blood vessels, which prevent the rapid removal of inflammatory and necrotic debris, leading to perivascular infiltrates. 12 A possible explanation for this pattern of inflammatory cell distribution could be the numerous small blood vessels in the Wharton's jelly of the equine umbilical cord, which are absent in human umbilical cord. Another explanation could be leukoattractants in the amniotic fluid, which stimulate leukocytes to diffuse through the Wharton's jelly from the umbilical vessels to the amniotic fluid. 7, 17 In some cases, although leukoattractants are present in the amnion, no lesions are observed, which remains unexplained. 7, 17
Human and equine placental development are markedly varied, and the role of placentation in the different inflammatory responses in human and equine placentas is unknown. Placentation in equines is complex, with six layers (epitheliochorial) separating fetal and maternal circulations, whereas human placentation has only three layers. At 35 days of gestation, the trophoblast of the equine conceptus separates into invasive and noninvasive components. The invasive component invades the maternal endometrium to form the unique endometrial cups. These secrete a chorionic gonadotrophin that synergizes with pituitary follicle-stimulating hormone to induce secondary luteal development in the maternal ovaries. The noninvasive trophoblast of the allantochorion establishes a stable microvillous contact with the endometrial epithelium around day 40. Over the next 100 days, it develops into a complex multibranched interdigitation with the endometrium to form microcotyledonary hemotrophic exchange units that cover the entire surface of the diffuse epitheliochorial placenta. 1 The involvement of placentation in the different inflammatory responses of human and equine placentas is unknown.
Despite the comparative differences between equine leptopsiral funisitis and human syphilitic funisitis, there are important similarities. In both leptospirosis and syphilis, the organism enters the placenta causing fetal infection with placentitis and funisitis. Both newborn foals and human infants can be alive, but premature or weak and unhealthy at term. It is important to examine placentas and umbilical cords from these foals if leptospirosis is suspected or to be considered. The demonstration of leptospirosis in the allantochorion or umbilical cord by fluorescent antibody tests or WS staining could aid in rapid diagnosis and have important clinical and therapeutic indications. This case report and previous publications on leptospirosis, in which the umbilical cord is one of the primary components of the placenta affected, suggests that the umbilical cord could be a useful tissue for identifying Leptospira spp. as a cause of equine abortion and stillbirth.
Footnotes
Acknowledgements
This work is published as University of Kentucky College of Agriculture publication 03-14-159.
