Abstract
Pheochromocytoma-associated catecholamine-induced cardiomyopathy is a well-known entity in man, nonhuman primates, and mice but has not been described in dogs. In this retrospective study, 9 dogs were identified with pheochromocytomas and concurrent cardiovascular pathology observed histologically (n = 6), echocardiographically (n = 4), and/or electrocardiographically (n = 5). Cardiac lesions included multifocal cardiomyocyte necrosis with contraction bands, cardiomyocyte degeneration, myocardial hemorrhage, lymphohistiocytic myocarditis, and interstitial fibrosis. Clinical procedures, including electrocardiographic and echocardiographic examinations, Doppler blood pressure measurement, and auscultation, were available for 5 dogs and consistently revealed concentric or mixed (eccentric and concentric) ventricular hypertrophy. Additional changes observed included arrhythmias, systemic hypertension, and heart murmurs. The myocardial lesions observed in this series of dogs are similar to those observed in humans with pheochromocytoma-associated catecholamine-induced cardiomyopathy. Since the clinical manifestations of catecholamine-induced cardiac disease are amenable to medical treatment, recognition of this cardiomyopathy has the potential to reduce morbidity and mortality in dogs with pheochromocytoma.
Pheochromocytomas are the most common tumor of the adrenal medulla in the dog 21 and can be functionally active or inactive. Functionally active pheochromocytomas secrete catecholamines, most commonly norepinephrine, 21 that can lead to systemic effects. Significantly elevated catecholamine levels in dogs with pheochromocytomas have been demonstrated via urinary catecholamine to creatinine ratios. 17 Approximately half of canine pheochromocytomas are incidental findings at necropsy or surgery. 11 Systemic hypertension has been demonstrated in 43% of dogs with pheochromocytomas. 2 Functional pheochromocytomas have been reported to cause tachycardia, edema, and cardiac hypertrophy in dogs, 12,35 but cardiomyocyte necrosis with contraction bands has not been described. 36 The cardiotoxicity of endogenous catecholamines produced by pheochromocytomas has been well documented in humans, 15,23 nonhuman primates, 16,37 and rodents, 22 and both the clinical signs and progression of the resulting cardiomyopathy are frequently reported to be reversible with medical treatment in humans. 10,13,18,28,29,38 The purpose of this study is to characterize the pathologic cardiovascular abnormalities in dogs with pheochromocytomas using histopathologic, echocardiographic, and electrocardiographic methods.
Materials and Methods
Case Selection
A retrospective search of the archives at the Colorado State University (CSU) Veterinary Teaching Hospital was performed between 2002 and 2012, yielding 62 histologically confirmed cases of pheochromocytomas in dogs. Nine dogs with concurrent cardiovascular pathology were identified and database information for these cases included age, sex, breed, and presenting clinical signs (Supplemental Table S1).
Gross Pathology
Neoplasms were discovered and sampled at necropsy (case Nos. 1–6) or during surgery (case Nos. 7–9), and gross characteristics of the primary neoplasm were recorded in necropsy or surgery reports. Cardiovascular systems were grossly examined for case Nos. 1 to 6, and gross cardiac changes, or the lack thereof, were provided in each necropsy report. Dogs that did not receive necropsies had morphologic assessments of the heart performed via echocardiography (case Nos. 7–9).
Histopathology, Immunohistochemistry, and Histochemical Staining
Neoplastic adrenal and cardiovascular tissues were preserved in 10% neutral-buffered formalin prior to being routinely processed at the CSU Veterinary Diagnostic Laboratory. Original hematoxylin and eosin (HE) slides containing pheochromocytoma were available for all cases. Slides were reviewed by a board-certified veterinary pathologist to confirm the original diagnosis of pheochromocytoma. For each of the diagnosed pheochromocytomas, paraffin-embedded tissues were available for immunohistochemical and histochemical evaluation. Antibodies to synaptophysin (RTU, PA0299, clone 27G12; Leica, Newcastle Upon Tyne, UK) and chromogranin A (1:1500, SP-1; Immunostar, Hudson, WI) were used to label primary tumors using a Leica Bond-Max autostainer and the Leica bond polymer refine red detection system (DS9390; Leica). Positive controls for immunohistochemistry were canine adrenal gland. For negative controls, primary antibodies were omitted; serial sections from each of the pheochromocytomas were used. Representative sections of the left ventricular free wall were available for case Nos. 1 to 6. Sections containing cardiac muscle were stained with phosphotungstic acid hematoxylin (PTAH) using standard protocols. 4
Cardiology Examination
All dogs diagnosed with pheochromocytoma in the study period were cross-referenced to the CSU Cardiology Service records to identify 5 patients who also received noninvasive cardiovascular evaluations. Cardiovascular examinations were conducted and interpreted by Cardiology diplomates of the American College of Veterinary Internal Medicine (ACVIM) and included auscultation, echocardiogram, electrocardiogram, and Doppler blood pressure measurement for 4 cases (Nos. 6–9). The cardiovascular examination for case No. 2 included electrocardiogram, auscultation, and Doppler blood pressure measurement interpreted by Cardiology diplomates of the ACVIM without an echocardiogram. The cardiovascular evaluations were performed prior to removal of the pheochromocytomas as part of a presurgical workup for case Nos. 7 to 9 and prior to euthanasia for case Nos. 2 and 6.
Results
Epidemiological and Clinical Findings
Of the 62 dogs diagnosed with pheochromocytomas during the study period, 9 dogs (14.5%) were identified with concurrent cardiovascular pathology either through histologic evaluation of tissue samples (case Nos. 1, 3–5), through clinical procedures including echocardiograms (case Nos. 7–9), or using a combination of clinical diagnostics followed by cardiac histopathology (case Nos. 2, 6). The average age at the time of diagnosis was 9.9 years (Suppl. Table S1). Of the 9 dogs, 6 were females (1 intact, 5 neutered), and 3 were neutered males. Dogs presenting for necropsies were euthanized (case Nos. 2, 4, 5–6) or died unexpectedly (case Nos. 1, 3). Of the 4 dogs with an echocardiogram, case No. 6 was euthanized due to poor prognosis and examined at necropsy the same day, case No. 9 died of surgical complications during adrenalectomy and a necropsy was not performed, and case Nos. 7 and 8 survived the adrenalectomy. Of the 2 dogs who survived the surgery, case No. 8 was lost to follow-up 6 months following the surgery, and case No. 7 was euthanized 4 years later for an unknown cause and no necropsy was performed. Potential causes for systemic hypertension, other than a productive pheochromocytoma, were explored and discovered in 3 dogs. Case Nos. 6 and 7 had evidence of proteinuria, and case No. 2 was receiving phenylpropanolamine for urinary incontinence.
Gross Pathology
Invasion of pheochromocytoma into the caudal vena cava was observed grossly in 7 of 9 dogs in this study (Supplemental Table S1); for case Nos. 1 and 3 to 6, invasion into the caudal vena cava was observed at necropsy, and in case Nos. 8 and 9, invasion was observed during surgery. There were no grossly visible myocardial lesions reported in postmortem examination of 4 of the 6 cases (Nos. 1, 2, 4, 5). For case No. 6, moderate biventricular hypertrophy was reported grossly. For case No. 3, multifocal petechial to ecchymotic myocardial hemorrhages in the left ventricle were described.
Histopathology, Histochemical, and Immunohistochemical Staining
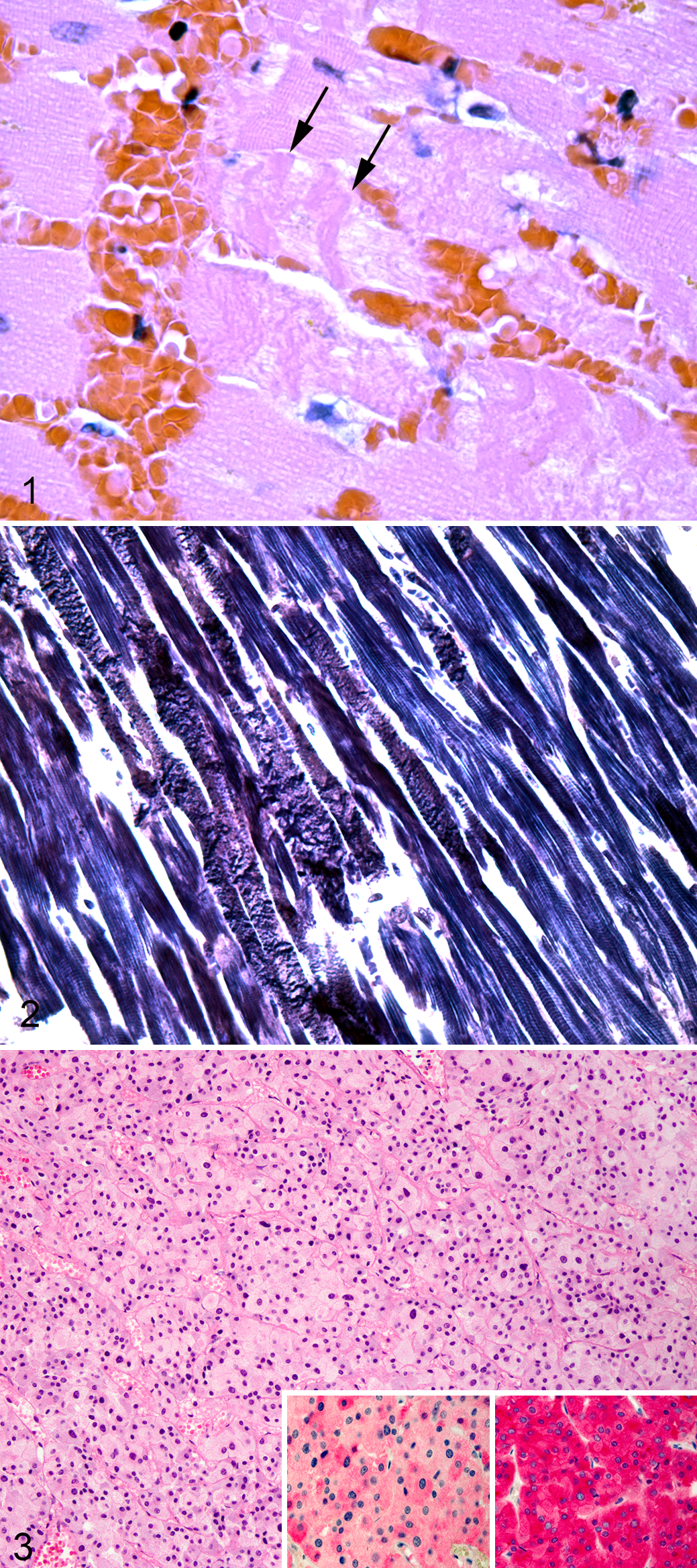
Cases Nos. 1 to 6 contained randomly scattered, polyphasic cardiomyocyte necrosis with contraction bands, cardiomyocyte degeneration, fibrosis, and lymphohistiocytic myocarditis (Suppl. Table S1). Cardiomyocyte necrosis and degeneration most often affected single cardiomyocytes or groups of 2 to 10 cardiomyocytes randomly distributed throughout the left ventricle (case Nos. 1–6) and affected approximately 5% to 25% of the ventricular muscle examined. Individualized necrotic cardiomyocytes were hypereosinophilic and shrunken with fragmented sarcoplasm and pyknotic, karyorrhectic, or absent nuclei. Hypereosinophilic sarcoplasm often contained multiple, amorphous, deeply eosinophilic bands (contraction bands) that measured approximately 2 to 4 μm in width and transversed the necrotic cardiomyocyte (Fig. 1). Contraction bands were accentuated using PTAH staining, demonstrating loss of myofibrillar organization and fragmentation and coalescence of the sarcoplasm (Fig. 2). Case Nos. 1, 3, and 5 contained mineralization of necrotic cardiomyocyte sarcoplasm characterized by deeply basophilic, granular material arranged in globules 1 to 3 μm in diameter replacing sarcomeric myofibrils. In addition to individualized cardiomyocyte necrosis and degeneration, case Nos. 3 and 6 contained foci of necrosis and hemorrhage up to 2 mm in diameter. Scattered randomly throughout the myocardium in all cases were empty myofiber spaces containing histiocytes and small lymphocytes. Along with inflammatory cells, areas of myocardial loss contained increased numbers of fibroblasts amid loosely arranged collagenous, eosinophilic stroma (fibrosis). Mild to moderate arteriosclerosis was observed in medium-caliber arteries within the myocardium in case Nos. 1 to 5 along with hypertrophy of arteriolar smooth muscle cells within the tunica media. Case No. 6 contained regions in which cardiomyocytes displayed moderate variation in diameter and enlarged nuclei (hypertrophy). The diagnosis of pheochromocytoma was confirmed for each case using immunohistochemistry for chromogranin A and synaptophysin (Fig. 3). Immunopositivity was intracytoplasmic and strong for synaptophysin and intracytoplasmic and moderate for chromogranin A for all cases.
Clinical Cardiology Examination
Ventricular hypertrophy was the single echocardiographic abnormality present in all cases examined by this modality (case Nos. 6–9) (Suppl. Table S2). Case No. 6 had marked concentric left and right ventricular hypertrophy noted echocardiographically and confirmed grossly and histologically. Case Nos. 7 and 8 each had a moderate, concentric, and eccentric (mixed) left ventricular hypertrophy pattern. Case No. 9 had moderate, concentric left ventricular hypertrophy. Hypertension was observed in case Nos. 2 and 7 to 9, and blood pressures at the upper limit of normal were observed in case No. 6. In addition, case Nos. 6 and 7 had documented murmurs on auscultation. Arrhythmias were present in case Nos. 2, 6, and 8. Case No. 2 had complete (third-degree) atrioventricular (AV) block. Case No. 6 had premature atrial contractions and idioventricular rhythm. Cardiac arrhythmias in case No. 8 were characterized by premature ventricular contractions of left ventricular origin.
Discussion
In this retrospective case series, we describe myocardial degeneration associated with pheochromocytomas in dogs and suggest that the cardiomyocyte changes observed are secondary to excessive release of catecholamines by neoplastic cells. Although the myocardial lesions described here are not specific for catecholamine cardiotoxicity, the dogs in this series had no evidence of alternative causes of myocardial necrosis. Other causes of myocardial necrosis in the dog include primary cardiomyopathy, thiamine deficiency, neurogenic cardiomyopathy, myocardial ischemia, various toxicants including plants, infectious etiologies such as toxoplasmosis or leishmaniasis, and doxorubicin therapy.
Experimentally induced catecholamine cardiotoxicity has been reported in a variety of animals, including dogs, and the resulting cardiac lesions are well characterized. 1,9,19,33,34 The lesions of cardiomyocyte necrosis with contraction bands, myocarditis, myocardial hypertrophy, and fibrosis observed in this study are similar to those observed in experimental studies using exogenous catecholamines in dogs, 33,34 which supports the hypothesis that the cardiac pathology in these cases was a result of excess catecholamines produced by the pheochromocytomas. While catecholamines have been shown to directly cause cardiomyocyte necrosis, it cannot be ruled out that some of the lesions observed in our study were the result of catecholamine-induced hypertension rather than a direct toxic effect of catecholamines on the myocardium.
Due to the retrospective nature of this descriptive report and the small number of cases available for evaluation, limitations are present. The overlap between complete functional cardiac examinations and necropsy results are limited and, because of this, correlations between clinical diagnostic abnormalities and postmortem changes are limited. In addition, neither blood nor urine was available for the cases to assess catecholamine levels directly.
The mechanism by which excessive catecholamines lead to cardiomyocyte necrosis is multifactorial—a combination of relative hypoxia, oxidative damage, microcirculatory effects, and sarcolemmal permeability—and can produce a variety of clinical manifestations and gross changes. 8,30,31,32 Molecular mechanisms of catecholamine-induced cardiomyocyte pathology have been thoroughly investigated. Excessive adrenergic stimulation leads to vasoconstriction and vasospasms in the coronary artery, resulting in relative myocardial hypoxia or infarction and irreversible cardiomyocyte damage. 30 Abnormally high catecholamine concentrations have also been shown to cause increased permeability of the sarcolemma with secondary increases in intracellular calcium within cardiomyocytes, which has a direct toxic effect on cardiomyocytes. 3 In addition, the oxidation of excess catecholamines produces highly reactive quinine compounds, which have been implicated in cardiomyocyte damage. 8,31 It has also been demonstrated that pheochromocytomas can induce a greater degree of cardiomyopathy than equivalent doses of norepinephrine alone, 22 suggesting that pheochromocytomas may secrete additional factors that act synergistically with catecholamines. 22 Excess catecholamines cause systemic hypertension, and the resultant pressure overload induces hypertrophy of cardiomyocytes and secondary hypertrophic cardiomyopathy. 14 In our canine cases, ventricular hypertrophy was a consistent finding. In addition to hypertrophic cardiomyopathy, which is the most commonly observed pattern in catecholamine-induced cardiomyopathy, 22 dilated 7 and tachycardia-related 27 cardiomyopathies have also been described in humans with catecholamine excess due to pheochromocytomas.
A wide variety of arrhythmias occur in humans with catecholamine-induced cardiomyopathy. 5,6 Three of the dogs in this study were diagnosed with arrhythmias, including idioventricular rhythms, premature atrial and ventricular contractions, and complete (third-degree) AV block. Third-degree AV block has been described in a case series of 2 dogs with pheochromocytoma 20 and is rarely described in human literature. 24 Specific electrocardiographic abnormalities associated with pheochromocytomas in humans include prolonged QT interval (QTc) and abnormalities of the ST segment and T wave 6 consistent with impaired ventricular repolarization. Prolongation of the QT interval and ST and T wave abnormalities were not present in any of the dogs in this series that were evaluated by electrocardiogram (ECG).
Arteriosclerosis and smooth muscle hypertrophy were observed in cardiac sections from a subset of dogs in this study. This has been described previously in 7 dogs with pheochromocytomas and was attributed to paroxysmal hypertension in these cases. 12
Early identification and treatment of catecholamine-induced cardiomyopathy has been shown to minimize cardiovascular pathology. Multiple descriptions of cardiomyopathy reversal have been reported in humans following treatment. 10,13,18,28,29,36 Medical treatments employed in the treatment of functional pheochromocytomas in human patients include α-adrenergic blocking agents, β-adrenergic blocking agents, captopril, and calcium channel blockers. 15 Captopril is an angiotensin-converting enzyme inhibitor and may also act to scavenge free radicals, which have been shown to contribute to the pathology of catecholamine-induced cardiomyopathy. 26,31 Calcium channel blockers are useful in the treatment of catecholamine-induced cardiomyopathy in that they minimize coronary vasospasm and myocarditis. 25
Because early detection and treatment of catecholamine-induced cardiovascular disease is likely to prevent or reverse progression of clinical signs and myocardial necrosis, all dogs with an antemortem diagnosis of functional pheochromocytoma should have a complete, noninvasive cardiovascular examination, including arterial blood pressure measurement, standard electrocardiogram, and Doppler echocardiographic examination. Furthermore, in those with normal blood pressure or ECG, repeated pressure measurements and 24-hour ambulatory ECG recording (Holter monitoring) may be needed to document the presence of intermittent abnormalities of cardiac rhythm and arterial pressure. The cardiac lesions detected in this study occurred in only a subset of dogs with pheochromocytomas (approximately 15%), demonstrating that not all dogs with pheochromocytomas develop secondary cardiomyopathies.
Conclusion
Pheochromocytoma in dogs can be accompanied by a cardiovascular disease similar to the catecholamine-induced condition observed in other species. This entity is characterized by contraction band necrosis of cardiomyocytes, arrhythmias, systemic hypertension, and varying patterns of ventricular hypertrophy. Based on human data, early clinical identification and treatment could potentially reverse or prevent the progression of catecholamine-induced cardiomyopathy and hypertension in dogs with pheochromocytomas.
Footnotes
Acknowledgements
We gratefully acknowledge the Colorado State University Veterinary Diagnostic Laboratory histopathology technicians for their assistance with histochemical and immunohistochemical staining.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
