Abstract
In humans, cutaneous metastasis of transitional cell carcinoma (TCC) has been attributed to direct extension, lymphatic or hematogenous dissemination, or surgical implantation. The purpose of this study was to characterize the clinical and histologic features of cutaneous TCC metastasis, confirmed by uroplakin-III immunohistochemistry, in dogs. The 12 cases were 9 spayed female and 3 neutered male dogs, 6 to 14 years old (mean, 11 years). Four dogs had a history of urinary incontinence. Three had undergone abdominal surgery for TCC diagnosis or treatment. The primary neoplasms were 7 papillary infiltrating and 5 nonpapillary infiltrating TCC. Cutaneous lesions were detected at a mean of 123 days (median, 38 days) after diagnosis of the primary TCC and appeared as plaques, papules, or nodules in, with 1 exception, perineal, inguinal, or ventral abdominal dermis or subcutis. Of 8 dogs with dermal TCC, 5 had epidermal erosion or ulceration. In 10 dogs, TCC was detected in cutaneous lymphatic vessels, identified by endothelial immunoreactivity for Prox1. Metastases were also detected in lymph nodes in all dogs and at distant noncutaneous sites, usually the lungs, in 10 dogs. Mean survival after diagnosis was 162 days (median, 90 days). Despite medical treatment of 10 dogs after the development of cutaneous metastasis, remission was not achieved; 4 dogs had stable disease. Although TCC could have spread to skin by direct extension or lymphatic or vascular dissemination, the proximity of most cutaneous metastases to the vulva or prepuce raises the additional possibility of transepidermal spread through urine-scalded skin.
Keywords
In comparison to that of other domestic animals, neoplasia of the urinary bladder and urethra is relatively common in dogs. Most canine bladder and urethral tumors are epithelial and malignant; transitional cell carcinoma (TCC) is by far the most common diagnosis. 7 Metastatic disease is detected at the time of clinical diagnosis in 10% to 20% of dogs and in up to 50% by the time of necropsy. 12 Histologically, TCCs are classified as papillary and infiltrating, nonpapillary and infiltrating, papillary and noninfiltrating, and nonpapillary and noninfiltrating (carcinoma in situ). 8 The infiltrating varieties (papillary or nonpapillary) are more common than noninfiltrating TCC. The nonpapillary and infiltrating variety is considered the most likely to metastasize. 7,8 In anaplastic or metastatic neoplasms, immunohistochemistry for uroplakin III is useful in confirming urothelial origin. 13
TCC is also the most common cancer of the human urinary bladder or urethra and is a geriatric disease in both people and dogs, with increased risk after 60 years of age in humans and 9 to 11 years in dogs. 5,6,12 Although TCC is more common in men than in women, in dogs, the converse is true—it is more common in female than in male and more common in neutered than sexually intact dogs. 2,5
Common metastatic sites of human and canine TCC include lymph nodes, lung, and bone. 2,4,6,7,10 Cutaneous metastasis of TCC is rare—generally not reported in canine studies and accounting for < 1% of human bladder cancer cases 11 —and has been attributed to retrograde lymphatic dissemination, hematogenous spread, or surgical implantation. 3,14 The purpose of this study was to evaluate the frequency and clinical, histologic, and immunohistochemical features of cutaneous metastasis of canine TCC.
Materials and Methods
The archives of the Indiana Animal Disease Diagnostic Laboratory (ADDL) were searched from January 2001 through December 2011 for canine cases of TCC in which neoplastic cells were detected in the dermis or subcutis by histologic examination of biopsy or necropsy specimens. Cases in which the neoplastic cells were detected in the abdominal wall musculature (which could be the result of seeding from surgical procedures), rather than in overlying skin or subcutis, were excluded. Medical records of the cases were reviewed for pertinent history of urinary tract and integumentary disease and case management and outcome. The ADDL archives were also searched for the total number of canine necropsy accessions as well as necropsy accessions with a diagnosis of TCC during the same period.
The primary TCCs were classified histologically. 8 All histologic sections of skin and subcutis were evaluated for epidermal erosion or ulceration and for the presence of neoplastic epithelial cells in the dermis, subcutis, or lumen of vessels. Uroplakin III immunohistochemistry (mouse monoclonal antibody, Research Diagnostics, Flanders, New Jersey; diluted at 0.31 μg IgG/ml) was used with proteinase K antigen retrieval to confirm urothelial origin of neoplastic cells in cutaneous sections. 13 The number of labeled neoplastic cells was scored as 0–3 (none, < 25%, 25%–50%, > 50%, respectively). Prox1 immunoreactivity (rabbit polyclonal, AngioBio, Del Mar, California, with heat-induced epitope retrieval in citrate buffer) of endothelial cells was used to distinguish lymphatic vessels from blood vessels. 9,15 Immunoreactivity was detected with diaminobenzidine as chromogen.
Results
Case records for 12 dogs with histologically documented cutaneous or subcutaneous metastasis of TCC were identified. All case materials from dogs with cutaneous TCC were received during the 7 years from January 2005 through December 2011. During that 7-year period, 2430 canine necropsy accessions included 104 cases of TCC of the urinary bladder or urethra. No cases of cutaneous metastasis of TCC were diagnosed during the preceding 4 years from January 2001 through December 2004, when only 13 necropsy examinations were performed on dogs with TCC.
Case Histories
The signalment, history, clinical and necropsy TNM (tumor/node/metastasis) staging, 12 and pathologic findings are summarized in Table 1. The 12 dogs included 10 different breeds and 2 mongrels. The 3 castrated male and 9 spayed female dogs ranged in age from 6 to 14 years (mean, 11 years; median, 12 years) and in body weight from 6.6 to 28.2 kg (mean, 18.7 kg; median, 18.5 kg).
Signalment, Clinical, and Pathologic Findings in 12 Dogs With Cutaneous Metastasis of Transitional Cell Carcinoma
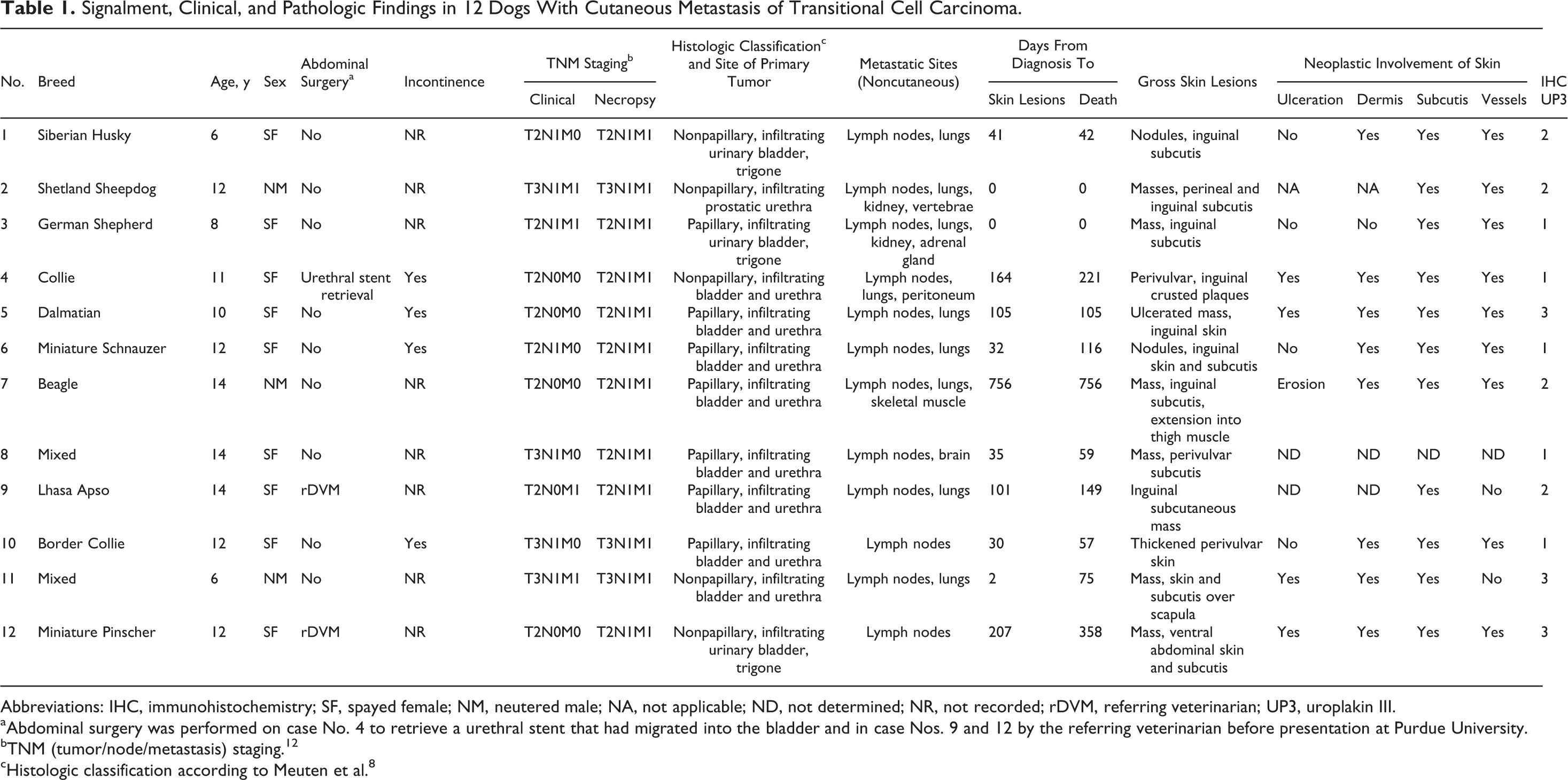
Abbreviations: IHC, immunohistochemistry; SF, spayed female; NM, neutered male; NA, not applicable; ND, not determined; NR, not recorded; rDVM, referring veterinarian; UP3, uroplakin III.
aAbdominal surgery was performed on case No. 4 to retrieve a urethral stent that had migrated into the bladder and in case Nos. 9 and 12 by the referring veterinarian before presentation at Purdue University.
bTNM (tumor/node/metastasis) staging. 12
cHistologic classification according to Meuten et al. 8
At diagnosis, 7 dogs (Nos. 1–3, 6, 8, 10, 11) had nodal metastases, and 4 dogs (Nos. 2, 3, 9, 11) had distant metastases. Cutaneous lesions were grossly apparent from 0 to 756 days (mean, 123; median, 38) after diagnosis of the primary TCC. Cutaneous or subcutaneous metastasis of TCC was documented antemortem by histologic examination of a biopsy specimen in 4 dogs (Nos. 4, 10–12).
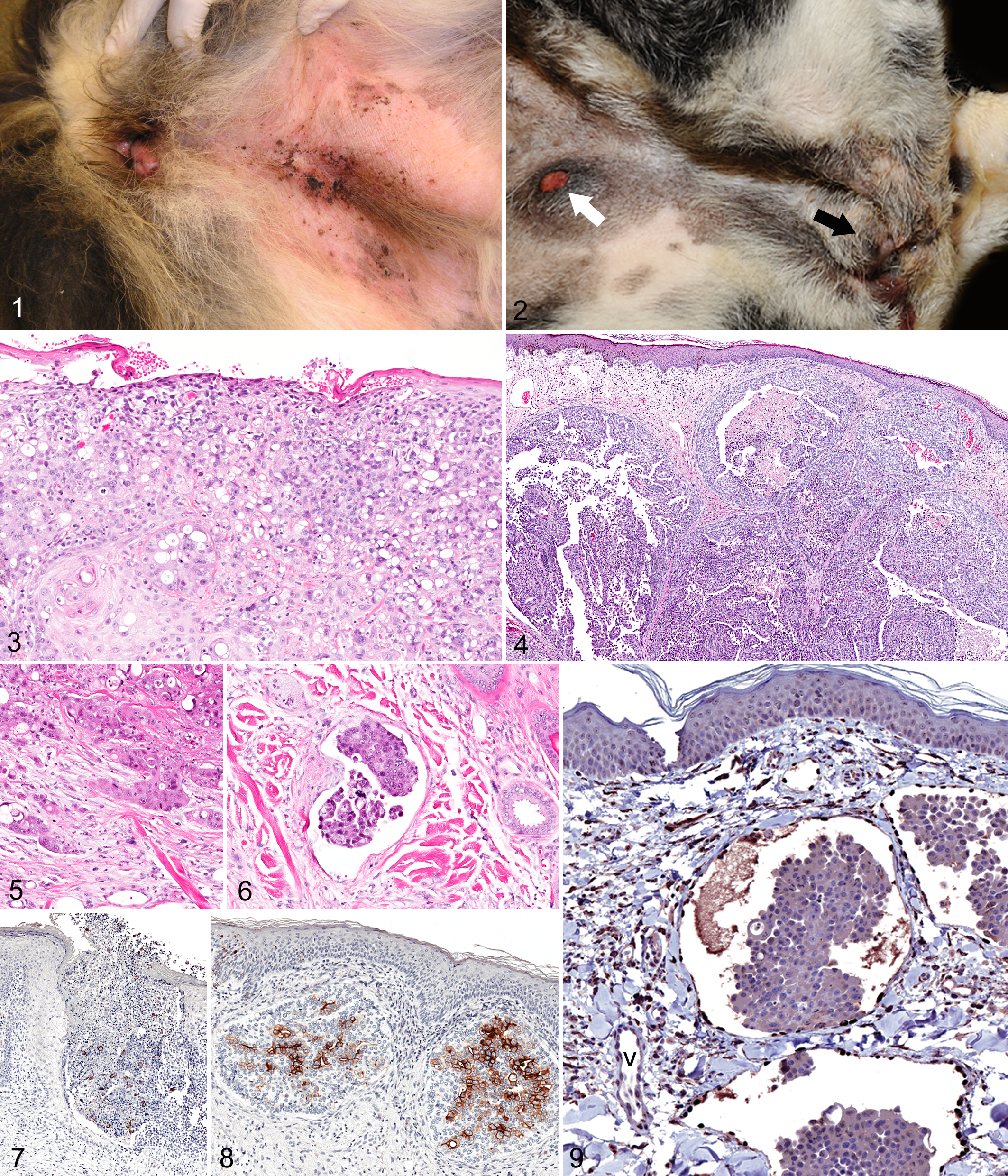
The initial clinical presentation in 9 dogs with lesions in perineal, inguinal, or abdominal skin was dermatitis or a cutaneous/subcutaneous mass or thickening. Some lesions, interpreted clinically as dermatitis or pyoderma, were erythematous and eroded with formation of crusted plaques and papules (Fig. 1). A major clinical finding in 1 dog (No. 3) was bloody urine on fur around the vulva. According to the owner, another dog (No. 10) frequently licked the vulva and surrounding area.
Ten dogs received treatment for TCC at or following the emergence of the skin lesions. Treatments included vinblastine in 4 dogs (Nos. 6, 8, 9, 10), mitoxantrone and cyclooxygenase inhibitor in 3 dogs (Nos. 6, 9, 11), leukeran in 4 dogs (Nos. 4, 6, 7, 12), and piroxicam in 2 dogs (Nos. 7, 11). One dog each was treated with cisplatin (No. 1), carboplatin (No. 5), firocoxib (No. 4), and deracoxib (No. 4). No dogs had remission. Stable disease (< 50% change in tumor volume and no new tumor lesions) was noted in 2 (Nos. 6, 8) of 4 dogs receiving vinblastine, in 1 (No. 11) of 3 dogs receiving mitoxantrone and cyclooxygenase inhibitor, and in 1 (No. 11) of 2 dogs receiving piroxicam. The remaining dogs had progressive disease (≥ 50% increase in tumor volume or the development of new cancer lesions). The dogs survived 0 to 756 days (mean, 161; median, 90) after diagnosis. Ten dogs were euthanized, and 2 dogs (Nos. 8 and 12) died naturally because of various complications of TCC.
Pathologic Findings
Necropsy was performed at the ADDL in 10 cases and by the referring veterinarian for 1 dog (case No. 10). The owner of dog No. 12 did not allow a necropsy.
Sites and histologic classification of the primary TCC are in Table 1. In dog Nos. 1, 3, and 12, the TCC was localized to the trigone of the urinary bladder; in dog No. 2, it was localized to the urethra. The other 8 dogs had TCC in the bladder and urethra. All primary neoplasms were classified as infiltrating (at least focally) and papillary in 7 dogs and nonpapillary in 5 dogs.
Cutaneous or subcutaneous metastatic TCC involved the perineal, inguinal, or ventral abdominal skin in all but 1 case (dog No. 11, with a cutaneous and subcutaneous mass over the scapula). In 3 dogs (Nos. 1, 7, 8), skin lesions were not recognized until necropsy. Gross integumentary lesions included epidermal erosion or ulceration, subcutaneous edema, and dermal or subcutaneous plaques, nodules, or masses. Some dermal nodules had an ulcerated surface (Fig. 2).
Histologically, metastatic TCC appeared as superficial dermal plaques or deeper coalescing nodules of neoplastic epithelial cells. The plaque-like TCC abutted the eroded or ulcerated epidermal surface (Fig. 3), whereas discrete nodules of metastatic TCC were found beneath ulcerated or intact epidermis (Fig. 4). The borders of nests of neoplastic cells were indistinct, and neoplastic cells invaded adjacent dermis or subcutis (Fig. 5). Neoplastic cells were also found in the lumen of dilated lymphatic vessels (Fig. 6).
The cytologic features of the cutaneous or subcutaneous metastases of TCC closely resembled those of the primary tumors. The neoplastic cells were polyhedral or rounded with a round to oval hyperchromatic nucleus, variable nuclear diameter, prominent or multiple nucleoli, 2 to 4 mitotic figures in most 400× fields, and ample to abundant eosinophilic cytoplasm with distinct cell borders. Many cells had large (≥ 25 μm in diameter) unstained cytoplasmic vacuoles. Glandular or squamous differentiation was seldom observed; however, focal proximity of neoplastic epithelial cells to the epidermis or apocrine sweat glands required distinction from squamous cell carcinoma or apocrine adenocarcinoma.
The histologic preparation from 1 dog could not be oriented for lack of overlying skin in the section. Skin sections in the other 11 cases were evaluated for neoplastic cells in the dermis, subcutis, and vasculature. Cutaneous erosion (1 case) or ulceration was documented histologically in 5 of 9 cases withepidermis in the plane of section. All 8 dogs with dermal TCC also had subcutaneous TCC. In addition, subcutaneous TCC was documented in 3 dogs that did not have histologically detected dermal TCC.
The number of neoplastic cells that were immunoreactive for uroplakin 3 in the cutaneous or subcutaneous metastatic lesions varied among cases (Figs. 7, 8). Only 3 dogs received a score of 3 with over 50% of neoplastic cells immunoreactive for uroplakin; 5 dogs received a score of 1 (< 25% of neoplastic cells immunoreactive); and 4 received a score of 2 (25%–50% of cells immunoreactive). Intravascular neoplastic cells were detected in skin or subcutis in all but 2 dogs. Embolized vessels, in all dogs in which they were found, were identified as lymphatic vessels by the strong immunoreactivity of the endothelial cells for Prox1 (Fig. 9).
All 12 dogs with cutaneous metastasis of TCC also had metastasis to other organs (Table 1). Regional or distant lymph nodes (12 cases) and lungs (9 cases) were the most common metastatic sites. Less commonly, metastatic TCC was found in the kidney (2 cases) and, in 1 case each, vertebrae, adrenal gland, peritoneum, skeletal muscle, or brain.
During the 7-year period (2005–2011) in which the 12 canine TCC cases with cutaneous metastasis were accessioned, there were 104 necropsies performed on dogs with TCC. The 104 cases of TCC comprised 4.3% of 2430 canine necropsy cases in that period. These 104 cases included 10 of the 12 TCC cases with cutaneous metastasis and 94 cases of TCC in which cutaneous metastasis was not detected. The 94 dogs included 37 males (24 [65%] with metastatic disease) and 57 females (35 [61%] with metastatic disease); only 1 female and 1 male dog were sexually intact. In 46 (49%) of the 94 dogs, the primary tumor involved both the bladder and the urethra; in 35 (37%), it was localized to the bladder; in 9 (10%), to the urethra only. Thirty-two of 46 (70%) of the tumors that involved both the bladder and the urethra metastasized; 20 of 35 (57%) bladder-confined tumors metastasized; 7 of 9 (78%) tumors localized to the urethra metastasized. The TCC in the remaining 4 dogs involved various portions of the urinary tract from the renal pelvis to the urethra; all 4 cases had metastasized. The most commonly recorded sites of metastasis were lungs (52 cases), lymph nodes (37 cases), and bones (12 cases). The histologic classification of the 94 cases without cutaneous metastasis included 50 papillary and infiltrating (32 [64%] metastasized), 41 nonpapillary and infiltrating (31 [76%] metastasized), and 3 papillary and noninfiltrating (none metastasized) TCCs. The distribution of the 3 different histologic types of TCC was not obviously related to the site of the primary tumor.
Discussion
In this study, 12 dogs with TCC of the urinary bladder or urethra had uroplakin-positive neoplastic urothelial cells in the dermis or subcutis. In all cases, this cutaneous metastasis was accompanied by metastasis to lymph nodes and other distant sites. Uroplakin immunohistochemistry was useful to distinguish TCC from other metastatic carcinomas, but the variation in number of labeled cells—in 5 of 12 cases, < 25% of neoplastic cells were labeled—emphasizes the importance of a careful search for positive cells.
The treatment response during and following the emergence of cutaneous metastasis was poor. None of 10 dogs treated at that point had remission of TCC; 4 dogs had stable disease. Standard chemotherapy for canine TCC typically results in a 35% remission rate and 40% stable disease rate. 1,5
At 4% of canine necropsy cases, TCC may be more commonly encountered at Purdue University than at other diagnostic laboratories because of case referrals to the Purdue Comparative Oncology Program, which has conducted multiple clinical trials in dogs with TCC. The female predominance, the most common metastatic sites, the histologic classification, and the correlation of metastatic disease with the histologic classification parallel what has been reported for canine TCC. No obvious correlation between sex, site of the primary tumor, or histologic classification and the presence of cutaneous metastasis was apparent. There was also no indication that the proportion of TCC cases with cutaneous metastasis has increased over the past decade.
The integument is a rare site of TCC metastasis, accounting for less than 1% of human bladder cancer cases, 11 and generally has not been recognized as a site of metastasis in canine TCC. The cases of cutaneous metastasis of TCC in this study were accessioned during a 7-year period in which cutaneous metastasis was documented in 9.6% of 104 canine TCC necropsy accessions. These findings suggest that cutaneous metastasis of TCC may be more common in dogs than previously believed and may be more common than it is in people. With the exception of 1 dog, cutaneous metastatic TCC was detected in proximity to the vulva or prepuce. When evident clinically, the cutaneous lesion was variably interpreted as dermatitis or as a mass or nodule. These clinical appearances are also typical in cutaneous metastasis of human TCC. 11,14
Neoplastic cells of TCC are believed to invade the skin by direct extension from the primary neoplasm, by retrograde passage through blood or lymphatic vessels, or by iatrogenic implantation during surgery or cystocentesis. 11 Only 3 dogs in this study had a history of abdominal surgery, and these dogs did not have TCC in abdominal wall musculature as would have been expected with surgical seeding of the cancer. These findings suggest that routes of cutaneous metastasis other than surgical seeding were in effect in the majority of the dogs. Neoplastic cells were found in lymphatic vessels (identified by Prox 1 immunoreactivity) in most of the cutaneous metastases, and this could suggest lymphatic spread. However, it is not possible to know if this represents retrograde metastasis from the primary TCC or invasion of afferent lymphatic vessels from the dermal or subcutaneous metastases. Although neoplastic cells were not detected in arteries or veins in the cutaneous sections, this does not rule out vascular spread. Because of the proximity of cutaneous metastatic lesions to the urinary tract (and, hence, the primary neoplasm) in most cases, direct extension from the primary TCC is possible even though this was not appreciated at necropsy. Another noteworthy aspect of TCC is that dogs with advanced disease commonly have urinary incontinence, and some of these dogs also have limited mobility and are prone to develop urine scalding of the skin. Neoplastic urothelial cells shed in the urine could be seeded into the erosions and ulcers of urine-scalded skin. The proximity of the metastatic lesions in most dogs in this study to the external urethral orifice would also be consistent with a transepidermal route for cutaneous metastasis.
In summary, the skin is a less common site than lymph nodes, lung, or bone for metastasis of TCC; however, cutaneous metastasis was detected in almost 10% of canine TCC necropsy accessions. Thus, cutaneous TCC may be more common in dogs than previously recognized and more common than in humans. 11 Possible routes of migration of neoplastic urothelial cells to the skin include direct extension, lymphatic or hematogenous spread, surgical implantation, and transepidermal seeding in urine-scalded skin. The identification of cutaneous metastasis may predict a poor response to chemotherapy, although further study is required. Importantly, cutaneous metastasis of TCC may resemble an inflammatory dermatosis, so metastatic neoplasia should be included in the differential diagnosis for any dog with TCC that develops plaques, papules, or nodular masses, especially in perineal, inguinal or ventral abdominal skin.
Footnotes
Acknowledgements
We thank Patty Bonney for technical assistance and help with data retrieval, as well as the staff of the ADDL Histopathology Laboratory and Carol Bain of the Purdue Histology and Phenotyping Laboratory for histologic and immunohistochemical preparations.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
