Abstract
o-Nitroanisole is an intermediate in the manufacture of azo dyes. In a National Toxicology Program stop-exposure study, o-nitroanisole induced hyperplasia, papillomas, and papillary carcinomas in the urinary bladder of Fischer 344/N rats. o-Nitroanisole was investigated since occupational or environmental exposure to aniline and azo dyes is a risk factor for urinary bladder cancer in humans. The current study describes the morphology of urinary bladder neoplasms seen in rats with respect to those observed in humans. This study also evaluated immunohistochemical expression of the cell cycle–related proteins cyclin D1 and p53 and the differentiation markers cytokeratin 20 and uroplakin III in hyperplastic (n = 11) and neoplastic (n = 6 papillomas, n = 11 carcinomas) lesions of the urinary bladder epithelium from rats treated with o-nitroanisole and in normal (n = 6) urinary bladders from untreated rats. The tumors observed were more similar to the papillary type rather than the muscle-invasive type of urinary bladder cancer in humans. The preneoplastic and neoplastic lesions observed suggest progression from hyperplasia to papilloma to papillary carcinoma. With neoplastic progression (hyperplasia to papilloma to carcinoma), cyclin D1 immunoreactivity progressively increased in intensity, percentage of cells staining, and distribution. Overexpression of p53 was not found. Cytokeratin 20 staining decreased in superficial cells, while uroplakin III staining increased in intermediate and basal cells with progression from hyperplasia to carcinoma. The results are consistent with increased cell cycle dysregulation or proliferation (cyclin D1), decreased differentiation (cytokeratin 20), and abnormal differentiation (uroplakin III) as lesions progress toward malignancy.
Keywords
Urinary bladder neoplasia is the sixth-most common cancer in humans in the United States, with urothelial carcinoma the most common subtype. 27 The most important risk factors for urinary bladder cancer in the Western world are cigarette smoking, followed by occupational or environmental exposure to aniline and azo dyes and aromatic amines. 25 o-Nitroanisole (2-nitroanisole, 2-methoxynitrobenzene) is a single-ring aromatic nitro compound used as an intermediate in the preparation of o-anisidine and the production of azo dyes. 22 o-Nitroanisole is reasonably anticipated to be a human carcinogen based on sufficient evidence of malignant tumor formation at multiple tissue sites in multiple species of experimental rodents, including the urinary bladder in rats of both sexes. 22 In the present study, we used immunohistochemistry to characterize the expression patterns of cell cycle–related markers (cyclin D1 and p53) and differentiation markers (cytokeratin 20 [CK20] and uroplakin III [UPIII]) in o-nitroanisole-induced urothelial (transitional cell) hyperplasia and neoplasia in rat urinary bladders to compare with findings reported for urinary bladder cancer in humans.
Bladder cancer in humans encompasses a heterogeneous group of tumors. There are 2 main phenotypes with different clinical behaviors. The majority (75%–85%) of patients present with papillary urinary bladder tumors, which are noninvasive or only superficially invasive. 5,6,15,25 These low-grade papillary tumors rarely metastasize but frequently (70%) recur after excision, and up to 25% progress to invasive phenotypes. 5,25 The second group of urothelial tumors are the muscle-invasive carcinomas, which are often flat lesions thought to arise from carcinoma in situ or, less frequently, from high-grade papillary tumors. 6,15,25 In rats, bladder cancer is typically of the papillary type. 7 Papillomas in rats have been shown to progress from urothelial hyperplasia to nodular or papillary hyperplasia through papilloma and eventually to higher-grade noninvasive and, ultimately, invasive papillary carcinomas. 6 One of the goals of the present study was to elucidate similarities and differences in immunoreactivity between rat bladder lesions along the progression from hyperplasia through papilloma to papillary carcinoma and the reported expression patterns of human papillary urothelial neoplasms.
Increased cellular proliferation is a hallmark of carcinogenesis. This study investigated expression of the proliferation-related protein cyclin D1, which regulates cell cycle progression from G1 to S phase via the Rb pathway, as well as that of the tumor suppressor gene p53. Cyclin D1 is believed to be an oncogene in some human cancers and is overexpressed in several human cancers, including urinary bladder cancer. 11,15 Papillary or nodular hyperplastic lesions with extensive cyclin D1 overexpression in rats may be more likely to progress to advanced lesions, such as papillomas or carcinomas. 16 Mutations in the tumor suppressor gene p53 have been proposed to be important in the development of some urothelial carcinomas in humans. 5 This study tested the hypotheses that cyclin D1 and p53 immunoreactivities progressively increase along the progression from hyperplasia through papilloma to carcinoma in rat urinary bladder carcinogenesis.
Another hallmark of carcinogenesis is decreased or altered cellular differentiation. CK20 is among several cytokeratins that are important in the immunohistochemical diagnosis of carcinomas derived from a limited number of sites, and it has been widely studied in human urothelial neoplasia. In normal urothelium, expression of CK20 is restricted to the superficial (umbrella) cell layer and is therefore a marker associated with terminal differentiation of urothelial cells. 19,20 In addition to CK20, another marker of urothelial differentiation is the integral membrane protein UPIII. UPIII is 1 of 4 membrane-associated uroplakins synthesized in terminally differentiated, superficial urothelial cells. Uroplakins—proteins conserved over a broad range of mammalian species—form a plaque-like complex and function in cell adhesion and maintenance of impermeability. 33 As the main differentiation-related membrane proteins of the urothelium, uroplakins would be expected to be downregulated during urothelial tumorigenesis. Previous experiments showed modification of uroplakin expression during chemically induced rat urinary bladder carcinogenesis. 23,34 Using the differentiation markers CK20 and UPIII, we tested the hypothesis that the ability of urothelium to terminally differentiate is progressively lost during tumor progression. As urinary bladder cancer is a common cancer in humans and one of the most expensive cancers to treat overall, 4 there is a need to identify similarities and differences between urinary bladder cancers in humans and those in rats, since rodents are important models in carcinogenicity testing.
Materials and Methods
Archived tissue blocks of urinary bladders from 6 control and 28 treated rats were obtained from a National Toxicology Program stop-exposure feed study of o-nitroanisole in Fischer 344/N (F344/N) rats. 22 Because of a high incidence of diffuse urothelial (transitional cell) hyperplasia of the urinary bladder and cases of urothelial papilloma and carcinoma in the highest dose groups in a prechronic (13 week) study, a stop-exposure study had been performed. 22 In the stop-exposure study, groups of 60 male and 60 female F344/N rats received diets containing 0-, 6000-, or 18 000–ppm o-nitroanisole for 27 weeks and were then maintained on control feed without further chemical exposure for 3, 6, 9, or 15 months. Urinary bladder samples were fixed in 10% neutral buffered formalin, routinely processed, and paraffin embedded (see National Toxicology Program 22 for details). The samples from the 6 control rats were free of proliferative lesions, and those from treated rats included 11 hyperplastic lesions, 6 papillomas, and 11 carcinomas. The 11 hyperplasias were from the 6000-ppm group (n = 8; 6- or 9-month scheduled sacrifice) and the 18 000–ppm group (n = 3; 3-month scheduled sacrifice). The 6 papillomas were from the 6000-ppm group (n = 5; 9- or 15-month scheduled sacrifice with 2 moribund sacrifices) and the 18 000–ppm group (n = 1; 15-month scheduled sacrifice). The 11 urothelial (transitional cell) carcinomas were from the 6000-ppm group (n = 3; all moribund sacrifices) and the 18 000–ppm group (n = 8; 15-month scheduled sacrifice with 2 natural deaths and 5 moribund sacrifices). The control urinary bladders were from the 3-month (n = 1) and 15-month (n = 5) scheduled sacrifices.
For immunohistochemistry, tissue sections (6 µm) were mounted on charged slides, deparaffinized in xylene, and rehydrated in graded ethanols. Endogenous peroxidase activity was quenched with 3% hydrogen peroxide for 15 minutes, followed by antigen retrieval via a Decloaking Chamber (Biocare Medical, Concord, CA) for 5 minutes at 120°C with citrate buffer at pH 6.0. Nonspecific binding was blocked with normal horse (cyclin D1, UPIII) or goat (CK20, p53) serum and an Avidin/Biotin blocking kit (Vector Laboratories, Burlingame, CA). Tissue sections were incubated with cyclin D1 mouse monoclonal antibody (Dako Corp, Carpinteria, CA) at a 1:250 dilution, UPIII mouse monoclonal antibody (American Research Products Inc, Belmont, MA) at a 1:100 dilution, CK20 rabbit polyclonal antibody (Abcam Inc, Cambridge, MA) at a 1:1000 dilution, or p53 rabbit polyclonal antibody (Vision Biosystems, Inc, Norwell, MA) at a 1:200 dilution for 1 hour at room temperature. For negative controls, normal mouse (cyclin D1, UPIII) or rabbit (CK20, p53) serum was used at the same dilution as the primary antibody. After rinsing with phosphate-buffered saline, tissues were incubated with biotinylated horse anti-mouse (cyclin D1, UPIII) or goat anti-rabbit (CK20, p53) secondary antibody for 30 minutes at room temperature. Avidin-biotin complex was used as the detection method and diaminobenzidine as the chromogen. Positive controls included rat gastrointestinal tract for cyclin D1 and CK20, mouse skin papilloma for p53, and rat urogenital tract for UPIII.
Cyclin D1 and CK20 immunoreactivities were scored semiquantitatively, and any differences were resolved by consensus of 2 pathologists. Samples were divided into 5 categories based on percentage of immunoreactive cells in all layers of the urothelium for cyclin D1 or in the luminal epithelium for CK20 (category 0, 0%–4%; 1+, 5%–10%; 2+, 11%–30%; 3+, 31%–50%; and 4+, >50%). Overexpression of p53 was considered positive if >5% of cells in the urothelium exhibited nuclear labeling. 31 UPIII was evaluated by the percentage of cases with >5% of cells positive within the 3 urothelial cell layers (superficial/luminal, intermediate, or basal).
Cyclin D1 and CK20 categories were compared among the 4 morphologic groups (normal, hyperplasia, papilloma, carcinoma) using the Jonckheere-Terpstra (J-T) test. 14 The J-T test tests associations between the semiquantitative immunoreactivity scores and neoplastic progression through the morphologic groups (normal, hyperplasia, papilloma, carcinoma). In addition, where significant associations were found, followup J-T tests compared each pair of morphologic groups. The J-T test was also used to test the association between extent of UPIII immunoreactivity and neoplastic progression. P values were 1-sided and considered statistically significant if <.05.
Results
The urothelium from vehicle control rats was histologically normal with 3 to 4 layers of thickness, composed of a single row of basal cells and ≥1 rows of intermediate cells covered by a single row of flattened luminal/superficial, terminally differentiated (umbrella) cells (Fig. 1). Hyperplastic urothelium from treated rats was thickened and often consisted of >30 cell layers (Fig. 2). The hyperplasia was generally diffuse, and the urothelium often formed papillary folds that protruded into the lumen or nodules that extended into the underlying stroma (Fig. 2). Papillomas were exophytic, pedunculated masses within the bladder lumen (Fig. 3). They were composed of a complex, branching, fibrovascular core covered by a thickened urothelial layer. The thickness of the urothelium on papillomas varied from 3 to >30 cell layers, and the cells histologically resembled those of hyperplastic urothelium. Carcinomas were also exophytic masses, but the neoplastic cells grew in several patterns, including sheets, cords, and nests or clusters with some occasional glandular differentiation (Figs. 4, 5). There was mild anisokaryosis and moderate pleomorphism with stellate, fusiform, and round to polygonal forms and frequent mitotic figures (Fig. 6). Regions of central necrosis were common. There was evidence of early invasion into the submucosa in 1 carcinoma from a male rat, but no metastases were reported. Foci of squamous metaplasia were present in all neoplasms and in many of the hyperplastic lesions (Fig. 2). Urinary bladder calculi were not reported.
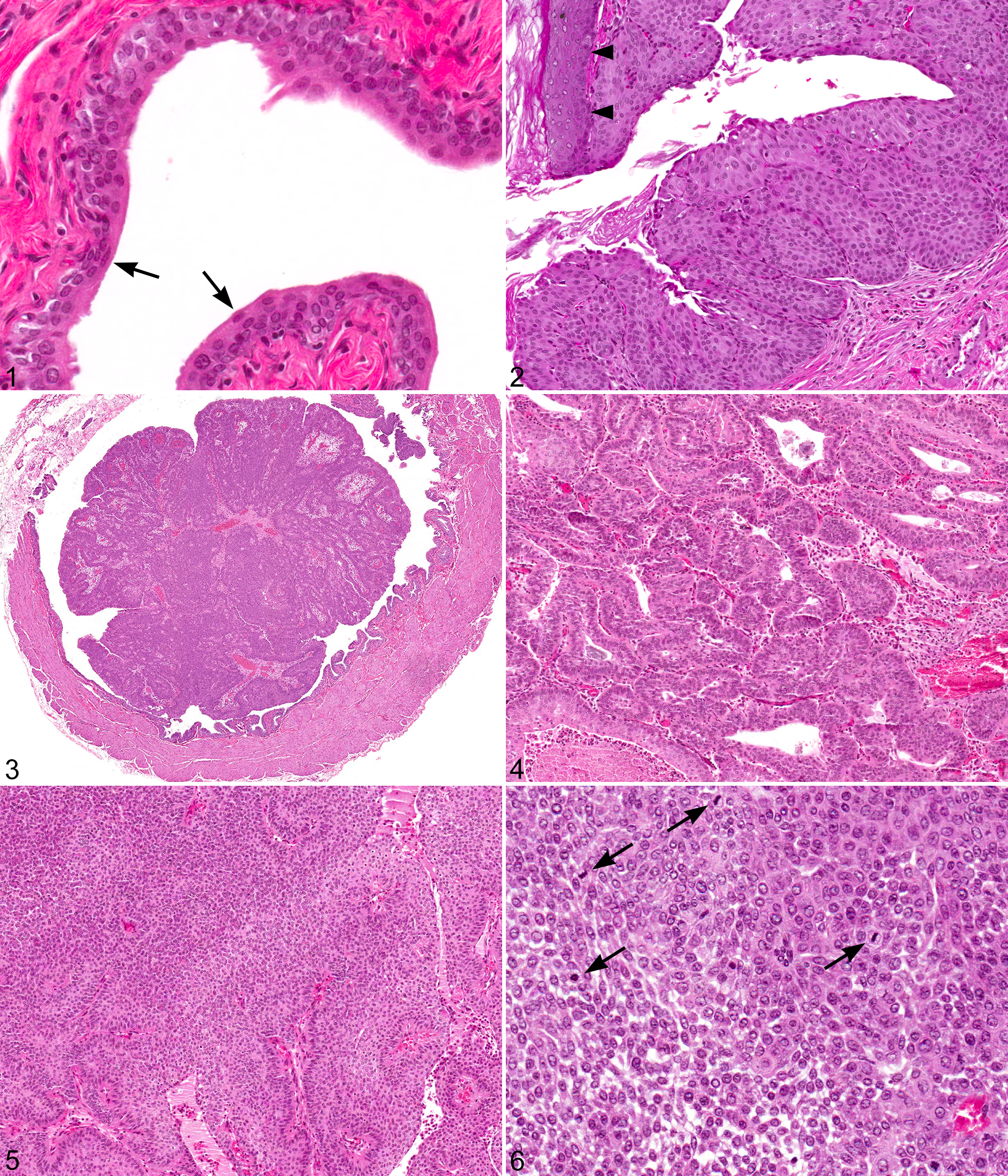
Urinary bladder, male F344/N rat. Hematoxylin and eosin (HE).
With progression from hyperplasia to papilloma to carcinoma, cyclin D1 immunoreactivity occurred in an increased number of cells, within and beyond the basal layer, and with increased intensity (P < .001 by the J-T test; Table 1; Figs. 7a, 8a, 9a, 10a). Cyclin D1 acts in the nucleus; however, cyclin D1 can also be found in the cytoplasm, where it undergoes ubiquitin-dependent degradation. 10 Thus, the antibody can also stain cytoplasm in tissues such as the urothelium of the urinary tract, as observed in the present study. Comparisons were based, however, on only nuclear staining, not cytoplasmic staining. Cyclin D1 immunoreactivity in normal bladder urothelium was restricted to basal cells, generally reacting in <5% of basal cells and with relatively weak intensity (Fig. 7a). In hyperplasias, cyclin D1 expression generally was noted in 5% to 30% of cells (Table 1), with extension beyond the basal cell layer into intermediate cells, and with greater intensity than that of normal urothelium (Fig. 8a). Papillomas showed distributions and intensities between hyperplasias and carcinomas, with staining typically occurring in basal cells and some intermediate cells but not in superficial cells (Fig. 9a). In carcinomas, cyclin D1 immunoreactivity was patchy, and the majority of cases were positive in 31% to 50% of cells; 2 had >50% reactivity throughout the urothelium (Table 1). Greater cyclin D1 staining was seen in carcinomas than in normal urinary bladders as well as hyperplasias and papillomas (Table 1; Figs. 7a, 8a, 9a, 10a). In 7 of 11 carcinomas, all 3 cell layers reacted to cyclin D1, albeit with less intensity in superficial cells than in intermediate or basal cells. Immunoreactivity of another cell cycle–related protein, p53, was negative in normal, preneoplastic, and neoplastic (papilloma, carcinoma) urinary bladder samples (data not shown).
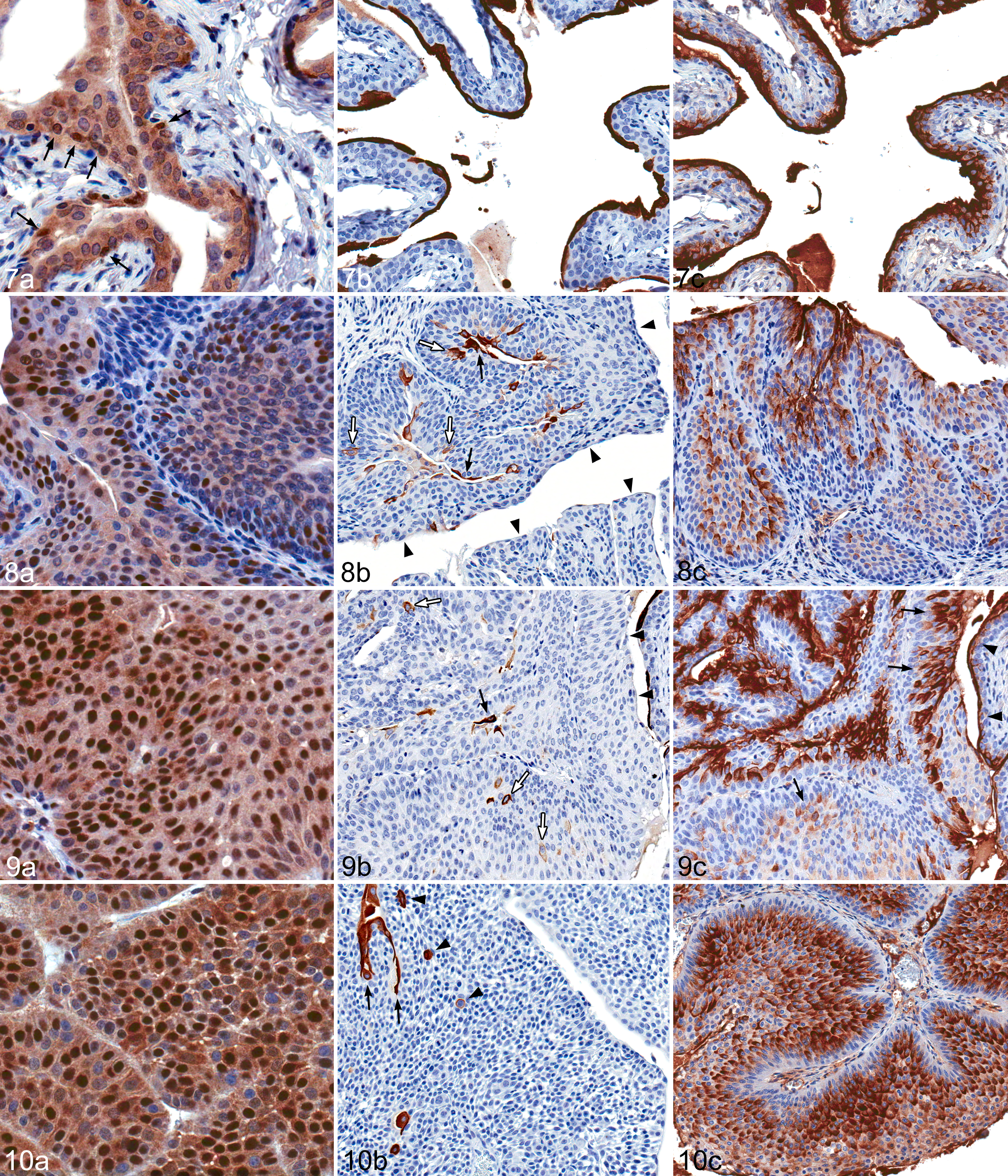
Urinary bladder; male F344/N rat. Immunohistochemistry.
Nuclear Cyclin D1 Immunoreactivity by Morphologic Category Based on Prevalence of Staining Throughout the Urothelium.a

aCyclin D1 immunoreactivity increased with neoplastic progression from hyperplasia through papilloma to carcinoma (P < .001 by the Jonckheere-Terpstra test). Hyperplasias, papillomas, and carcinomas had significantly more staining than controls (P < .001). Carcinomas were significantly more reactive than papillomas (P < .05) and hyperplasias (P < .001).
CK20 immunoreactivity was present in the cytoplasm diffusely within the superficial cell layer in normal bladders, and the percentage of superficial cells expressing CK20 generally decreased with tumor progression from hyperplasia to papilloma to carcinoma (P < .001 by the J-T test; Table 2; Figs. 7b, 8b, 9b, 10b). In all 6 normal bladders, CK20 expression was limited to superficial cells (Fig. 7b), with >80% of the superficial cell layer immunoreactive for CK20 (Table 2). In hyperplasias, most superficial cells were positive for CK20 as seen in control animals, but 4 of 11 cases had <50% immunoreactivity in luminal/superficial cells (Table 2). CK20 immunostaining in papillomas was variable but generally less than that of hyperplastic lesions (Table 2). In 4 of 11 carcinomas, <11% of superficial cells reacted with CK20, but there was reactivity in some intermediate cells, especially in the few areas of squamous metaplasia present (Table 2; Fig. 10b).
Cytoplasmic Cytokeratin 20 Immunoreactivity by Morphologic Category Based on Prevalence of Staining in Luminal Urothelial Cells.a
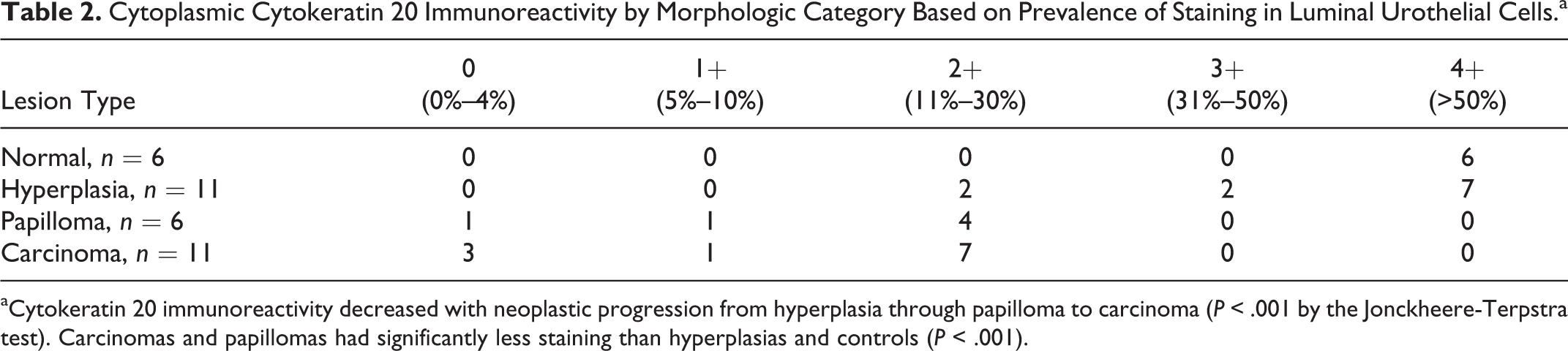
aCytokeratin 20 immunoreactivity decreased with neoplastic progression from hyperplasia through papilloma to carcinoma (P < .001 by the Jonckheere-Terpstra test). Carcinomas and papillomas had significantly less staining than hyperplasias and controls (P < .001).
Normal rat urinary bladders demonstrated intense and uniform UPIII immunoreactivity on the luminal surface membrane of the terminally differentiated superficial umbrella cells (Fig. 7c). In hyperplasias, papillomas, and carcinomas, UPIII expression generally persisted in superficial cells; however, UPIII immunoreactivity tended to also appear in greater numbers of intermediate cells with progression from preneoplastic to malignant lesions from 0 of 6 in normal urothelium to 3 of 11 hyperplasias, 3 of 6 papillomas, and 10 of 11 carcinomas (P < .001 by the J-T test; Table 3; Figs. 7c, 8c, 9c, 10c). In 6 of 11 carcinomas, UPIII immunoreactivity also appeared in basal cells (Table 3). UPIII immunoreactivity became discontinuous in the superficial layer and more cytoplasmic rather than restricted to the luminal membrane with tumor progression from hyperplasia to papilloma to carcinoma. In carcinomas, staining was multifocal, with areas of positively staining cells adjacent to areas of negatively staining cells. UPIII reactivity was usually greater toward the cell surface away from the basement membrane (ie, at the luminal surface); however in a few regions of some carcinomas, UPIII reactivity was observed throughout the cytoplasm (Fig. 10c).
Uroplakin III Immunoreactivity by Morphologic Category Based on Location of Staining in the Urothelium: Superficial Layer, Intermediate Cells, and Basal Layer.a

aUroplakin III immunoreactivity in the intermediate and basal layers of the urothelium increased with neoplastic progression (P < .001 by the Jonckheere-Terpstra test). Immunoreactivity in the intermediate and basal layers of carcinomas was significantly greater than that in controls and hyperplasias (P < .001) and papillomas (P < .05).
Discussion
In this study, o-nitroanisole induced hyperplasia, papillomas, and papillary carcinomas in the rat urinary bladder (Figs. 1–6). Foci of squamous cell metaplasia were observed in all neoplasms and in many hyperplastic lesions (Fig. 2). This is consistent with previous findings that rat urinary bladder carcinomas frequently have a squamous cell component admixed with the urothelial component. 7 Our data support previous findings that chemically induced urinary bladder neoplasia in rats usually arises as simple hyperplasia, progresses to nodular and/or papillary hyperplasia, then to papilloma, and ultimately to papillary carcinoma. 6,7 Of the 2 main types of urinary bladder cancer in humans, tumors in rats most closely resemble the papillary form. 6,7 However, in humans, the papillary neoplasms tend to not be invasive but frequently recur after treatment, whereas the papillary neoplasms in rats eventually become invasive, as noted in 1 rat in the present study. 7
Cyclin D1 expression increased with progression from normal to preneoplastic to malignant lesions of the urinary bladder in rats (Table 1; Figs. 7a, 8a, 9a, 10a). Amplification of the cyclin D1 gene (CCND1) has been reported in 10% to 20% of human bladder tumors, and overexpression of cyclin D1 has been reported in significant numbers of lower-grade human urothelial carcinomas. 15,16 Another study of chemically induced rat bladder carcinogenesis demonstrated a positive correlation between cyclin D1 expression and tumor progression with 0% of cells in normal urothelium, 17% of cells in papillary or nodular hyperplasias, 66% of cells in papillomas, and 69% of cells in carcinomas expressing cyclin D1. 17 We cannot exclude the possibility in this study that increased cyclin D1 expression may simply reflect increased cell proliferation. However, others have shown that cyclin D1 overexpression is not dependent on proliferation status. For example, cyclin D1 immunoreactivity was heterogeneous and did not correspond to PCNA immunoreactivity in preneoplastic and neoplastic lesions induced by N-butyl-N-(4-hydroxybutyl)nitrosamine in rat urinary bladders. 17 Our findings of cyclin D1 immunoreactivity in more cells, with greater intensity, and beyond the basal cell layer into intermediate cells of the urothelium suggest increased cell proliferation, cell cycle dysregulation, or possible cyclin D1 transforming activity as lesions progress toward malignancy (Table 1; Figs. 7a, 8a, 9a, 10a).
Others found that p53 mutations are rare in rat urinary bladder tumors. 2 In accordance, we did not find increased immunoreactivity of p53 (which would result from mutation) in hyperplastic or neoplastic lesions in rat urinary bladders (data not shown). Our data in rats are consistent with the superficial, papillary, low-grade types of neoplasms in humans that tend to recur but have a good prognosis and typically do not exhibit p53 mutations. 5
Our results support the findings of other studies that CK20 expression in normal urothelium occurs exclusively in the terminally differentiated superficial cells (Fig. 7b). 20 A normal CK20 expression pattern (limited to superficial cells) in noninvasive urothelial neoplasms is predictive of a lower risk of tumor recurrence in humans. 3,29 With tumor progression, CK20 immunoreactivity was gradually lost from the superficial cell layer (Table 2). A similar decline in superficial urothelial cell immunoreactivity in urothelial neoplasms has been shown in several human and rat studies. For example, others have found that normal rat control urothelium displays CK20 immunoreactivity in all superficial cells with none in intermediate and basal cells. 34 In rats administered the urinary bladder carcinogen N-butyl-N-(4-hydroxybutyl)nitrosamine, hyperplasias and low-grade papillary carcinomas had decreased superficial cell immunoreactivity, and high-grade papillary carcinomas had none. 34 In the current study, hyperplasias, papillomas, and carcinomas induced by o-nitroanisole had rare cells in the intermediate layer positive for CK20 expression (Figs. 8b, 9b, 10b). The abnormal distribution of staining in intermediate cells appears to be a clinically relevant feature in humans. The abnormal expression of CK20, defined as presence in >10% of intermediate cells, in human urothelial tumors is associated with higher tumor grade and increased risk of recurrence and progression. 3,12,24 We propose that decreased CK20 expression in superficial urothelial cells in urothelial hyperplasia and neoplasia in the present study is consistent with a progressive reduction in terminal differentiation of the superficial cells during carcinogenesis.
Changes in UPIII expression during bladder carcinogenesis, although somewhat different from the patterns observed for CK20, also represent alterations in differentiation. As with CK20, the expression of uroplakins characterizes normal terminal differentiation of the superficial urothelium. 24,26 The transmembrane uroplakin proteins are normally expressed on the luminal aspect of superficial umbrella cells (Fig. 7c). UPIII expression is highly specific for a carcinoma of urothelial origin, so it can be useful if the site of origin is in question, especially when used with other urothelial-limited markers, such as CK20. 33 Uroplakins, as products of terminal differentiation, could be expected to exhibit decreased expression in urothelial tumors. Some degree of UPIII expression loss in the superficial cells, noted sporadically as discontinuity in papillomas and carcinomas in the present study, is associated with increasing tumor development, grade, or stage in human, canine, bovine, and chemically induced mouse and rat urothelial tumors. 1,9,18,23,28 The loss of UPIII immunoreactivity was minimal in the present study, and UPIII loss in chemically induced urothelial neoplasms has been found to be greater in mice than rats. 23 In a large study in humans, negative UPIII expression was significantly associated with risk of recurrence and cancer-specific mortality in cases of muscle-invasive urothelial carcinomas but not in noninvasive carcinomas. 13 In general, about half of human muscle-invasive carcinomas retain UPIII expression. The general retention, albeit with some discontinuity, of UPIII expression in superficial cells seen here in urothelial neoplasms in rats and in other species suggests that urothelial neoplastic transformation does not always equate with significant downregulation of terminal differentiation products, even in advanced cases.
Although downregulation is not a consistent feature, UPIII expression appears to be substantially perturbed, as aberrant or even increased expression appears to be common in urothelial carcinomas. In the present study, there was an increase in cases immunoreactive for UPIII beyond the normal location of the superficial layer into intermediate cell layers with progression from hyperplasia to papilloma to carcinoma, with extension into the basal layer in some carcinomas (Table 3; Figs. 7c, 8c, 9c, 10c). The increase in immunoreactivity in intermediate cells and in a patchy pattern has also been observed in chemically induced rat bladder neoplasia in other studies, as well as in some human, bovine, and canine urothelial carcinomas. 1,13,23,26,28 There are several possible reasons for the remarkable increase in the number of intermediate cells with strong membranous or cytoplasmic UPIII immunoreactivity in hyperplastic and neoplastic lesions. First, immunoreactive intermediate cells may have elevated, rather than decreased, uroplakin synthesis. Second, uroplakin-containing vesicles may be accumulating in the cytoplasm because their incorporation into the apical urothelial surface, as part of normal differentiation, may be hindered in preneoplastic and neoplastic lesions. Third, neoplasia may be associated with abnormal migration of differentiated cells toward the lumen. It may be true that in neoplasms, luminal cells do not become fully differentiated “umbrella” cells and thus do not have uroplakins assembled into urothelial plaques or asymmetric membrane units. 23 We did observe some evidence of cells with less distinct polarity and more staining on all sides of the cytoplasmic membrane or diffuse cytoplasmic staining, as has been observed in bovine urothelial carcinomas (Fig. 10c). 9 Overall, in many urothelial tumors, uroplakins may be redistributed rather than downregulated. 26 Immunoreactivity of UPIII in carcinomas is an example of neoplastic expression of a highly specialized, tissue-specific differentiation marker that may occur even in advanced tumors. 21
Chemicals may induce urinary bladder carcinogenesis via genotoxic or nongenotoxic mechanisms, with nongenotoxic compounds leading to increased cell proliferation, or by a combination of genotoxicity and cell proliferation. 6,8,32 o-Nitroanisole was mutagenic in the Ames test with the Salmonella typhimurium T100 strain, increased chromosomal aberrations in Chinese hamster ovary cells, and induced sister chromatid exchanges. 22 o-Nitroanisole has also been shown to bind covalently to DNA in vitro and form DNA adducts in the urinary bladder in male Wistar rats in vivo. 30 The cumulative data are consistent with o-nitroanisole contributing to urinary bladder carcinogenesis at least in part via genotoxic mechanisms.
In summary, rat urinary bladder neoplasms induced by the genotoxic dye intermediate o-nitroanisole were consistent with progression from hyperplasia to papilloma to carcinoma. Rat urinary bladder neoplasms were most similar to the papillary type observed in humans. Immunohistochemical expression generally changed along the progression from hyperplasia to papilloma to carcinoma, with increased expression of the cell cycle regulatory protein cyclin D1, decreased expression of the differentiation marker CK20, and altered distribution of the differentiation marker UPIII.
Footnotes
Acknowledgements
We thank the personnel of the Histology Core Laboratory, Cellular and Molecular Pathology Branch, National Institute of Environmental Health Sciences, for preparation of histology slides. We also thank Natasha Clayton, Tiwanda Marsh, Yvette Rebolloso, Geoffrey Hurlburt, and David Olson of the Immunohistochemistry Core, National Institute of Environmental Health Sciences, for technical assistance with immunohistochemistry. Dr Dave Malarkey offered useful comments on the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Institutes of Health, National Institute of Environmental Health Sciences.
