Abstract
In humans, horses, and rodents, an association between pulmonary fibrotic disorders and gammaherpesvirus infection has been suggested. In dogs, canine idiopathic pulmonary fibrosis (CIPF), a progressive fibrotic lung disease of unknown origin and poorly understood pathophysiology, has been reported to occur in West Highland white terriers (WHWTs). The present study investigated the potential association between CIPF and herpesvirus infection. A PCR assay, using a mixture of degenerate and deoxyinosine-substituted primers targeting highly conserved regions of the DNA polymerase gene (DPOL) of herpesviruses, was applied on both lung and blood samples from WHWTs affected with CIPF and controls. Herpesvirus DPOL sequence could not be amplified from any of 46 lung samples (28 affected WHWTs and 18 control dogs of various breeds) and 38 blood samples (19 CIPF WHWTs and 19 control age-matched WHWTs) included. An association between CIPF and herpesvirus infection is therefore unlikely. Investigation of other causes of the disease is warranted.
Canine idiopathic pulmonary fibrosis (CIPF) is a progressive fibrotic lung disease which is most commonly reported in aged dogs of the West Highland white terrier (WHWT) breed. 6 Clinical signs in WHWTs affected with CIPF include progressive dyspnea, exercise intolerance and cough. 6 In addition, inspiratory Velcro-like crackles are commonly noticed on lung auscultation. 6 The cause and pathogenesis of CIPF are currently unknown, but a genetic basis is strongly suspected due to the breed predisposition. 6 Not all dogs from the WHWT breed develop CIPF at an advanced age, which suggests the involvement of triggering events in the development of the disease. CIPF shares several clinical features with human IPF, although histopathological differences have been described. 11 In human IPF, repetitive alveolar epithelial cell injuries and subsequent disordered tissue repair are considered to be the main pathological process involved in this fibro-proliferative disease. 3 Several studies have implicated viral infections, particularly Epstein Barr virus (EBV) infection, as a cause of epithelial injury and therefore as an important factor in the pathogenesis of IPF. 10 Indeed, EBV has been frequently detected in the lungs of IPF patients compared with control patients across several studies. 10 In horses, a fibrotic pulmonary disease called equine multinodular pulmonary fibrosis was proven to be associated with gammaherpesvirus infection. Equine herpesvirus 5 has been consistently isolated from lung tissue of affected horses, while none of the control horses were found positive. 13 Compelling evidence for gammaherpesvirus involvement has also been observed in experimental murine models of pulmonary fibrosis. 8,9 Based on these results obtained in humans, horses and rodents, and in view of the fact that a causative agent for CIPF has not yet been identified, we investigated the potential association between herpesvirus infection and CIPF in WHWTs.
For this purpose, a generic PCR assay 5 targeting highly conserved regions of the DNA polymerase gene (DPOL) of alpha-, beta- and gammaherpesviruses was applied to lung and blood samples collected from WHWTs affected with CIPF and from control unaffected dogs. Lung tissue samples collected from 15 male and 13 female WHWTs affected with CIPF (age: 9.1 to 16.3 years, median 13.6 years), and 18 control dogs (6 male and 12 female) of various breeds (5 WHWTs, 6 Jack Russell terriers, 2 Mixed breed, 1 Beagle, 1 Bulldog, 1 Newfoundland, 1 Maltese, 1 American Staffordshire) (age: 3.7 to 15.0 years, median 8.6 years) were included in the study. From 19 of the above mentioned WHWTs with CIPF, EDTA blood samples were also collected and included in the study. EDTA blood samples from 11 male and 8 female unaffected WHWTs, aged from 5.7 to 15.0 years (median 10.4 years) were included for control. The CIPF diagnosis was achieved according to a previously published approach and was confirmed by lung histopathology in all dogs. 6 Health status of the control WHWTs was assessed by taking a complete history and by performing a physical examination, hematology, serum biochemistry, echocardiography and thoracic high-resolution computed tomography, which did not reveal any abnormalities. Lung tissues used as controls were obtained from dogs euthanized for reasons unrelated to the study. These dogs had no history or clinical signs of lower respiratory disease at the time of euthanasia and no abnormalities were identified on necropsy and histopathological examination of their lungs. Lung biopsies were obtained within 30 minutes after euthanasia, snap-frozen in liquid nitrogen and stored at –80°C until further processing. Blood was collected from the jugular vein into EDTA tubes, aliquoted in 1.5 mL plastic cryotubes and stored at –80°C until analysis. DNA was extracted from both lung and blood samples with a commercially available kit following manufacturer instructions (DNeasy Blood & Tissue kit, QIAGEN GmbH, Hilden, Germany), with an overnight incubation in proteinase K at 56°C for lung samples. DNA samples were stored at –80°C until analysis. The panherpesvirus generic DPOL PCR was performed with a mixture of degenerate and deoxyinosine-substituted primers in a nested format as described previously. 5
PCR analysis of the 46 lung and 38 blood samples from WHWTs affected with CIPF and control dogs did not result in any amplification of a herpesvirus DPOL sequence (expected product size: approximately 210 bp–230 bp). However, identical bands of approximately 280 bp and 150 bp length appeared in all lanes (Figs. 1 and 2); these were of host origin as revealed by exemplary sequence analysis. In contrast, samples (spleen, lymph node, blood and other matrices) of human and animal origin (including carnivorans) that had previously tested positive for members of the herpesvirus subfamilies Alpha-, Beta-, and Gammaherpesvirinae, and were analyzed here for control, gave rise to the expected DPOL amplification products (Fig. 3). Although the generic PCR employed here may not detect all vertebrate herpesviruses with equal sensitivity, this molecular technique has previously been shown to successfully identify a plethora of novel alpha-, beta- and gammaherpesviruses in a wide range of host species including carnivorans. 4,5 We are therefore confident that the negative results obtained here were due to the absence of herpesvirus DNA in the dog samples (at least in clinically relevant numbers) rather than to a limitation of the method. However, based on the present results, a herpesvirus contribution to the pathogenesis of CIPF cannot completely be excluded, as “hit and run” mechanism 1 may be at work or a very low load of latent or persistent herpesvirus may exist that can be missed with the methodology employed here. So far, few reports have suggested the existence of an EBV-like virus in dogs, associated with lymphoma and oro-nasal tumor based on serological and molecular data. 2,7 However, contrasting negative results were reported in a very recent study using degenerate PCR assays on 112 canine tissue samples. 12 Consequently, these conflicting published results together with the results presented here do not sufficiently support the existence of gammaherpesviruses in dogs and encourage further research in this field.
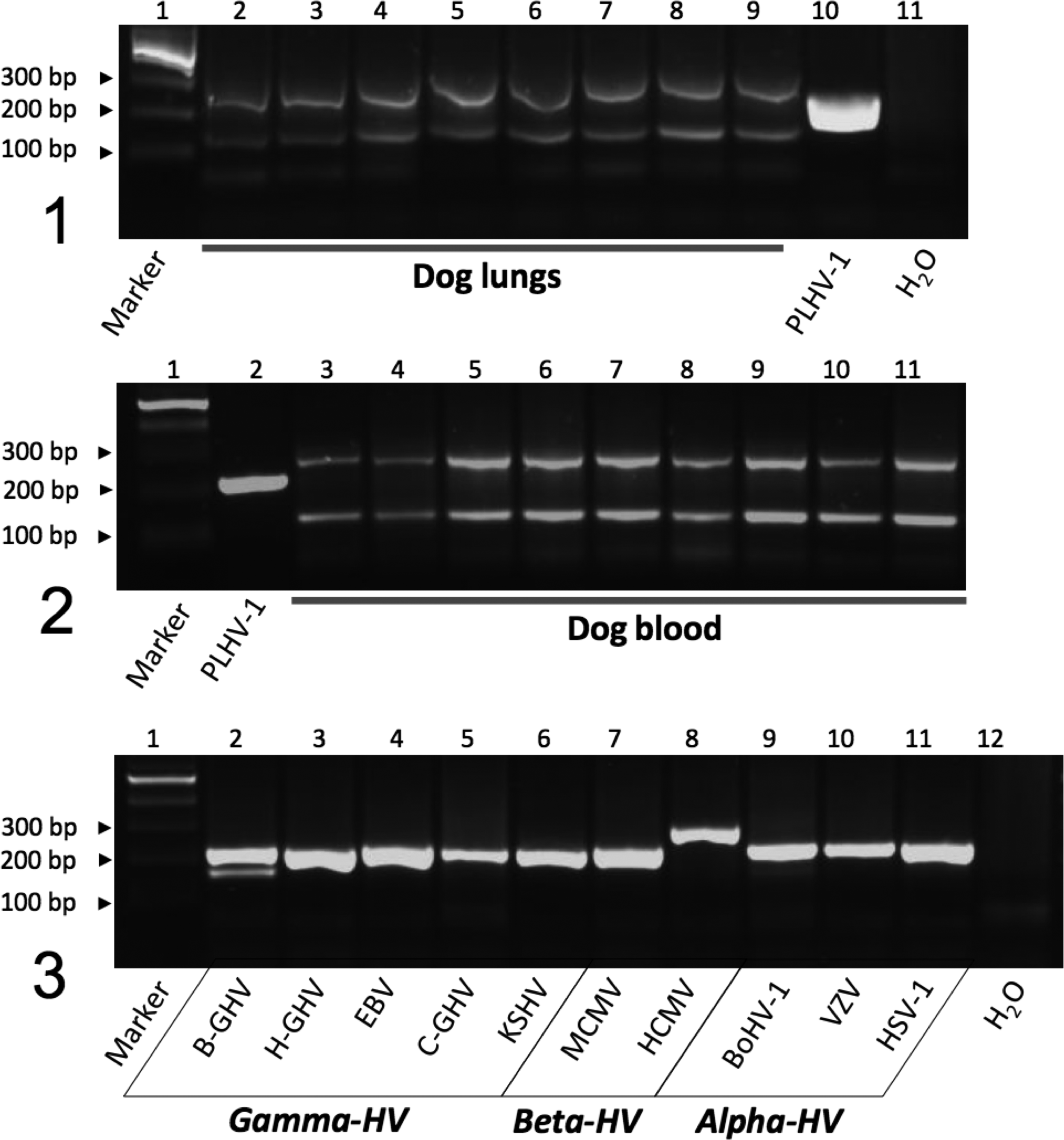
Analysis of lung (Fig. 1) and blood (Fig. 2) samples collected from West Highland White Terrier (WHWT) dogs affected with CIPF with panherpesvirus generic DPOL PCR.
In conclusion, the results of the present study suggest that an association between herpesvirus infection and CIPF is unlikely. Investigation of other causative agents potentially associated with CIPF is warranted.
Footnotes
Acknowledgements
The authors thank Cornelia Walter for excellent technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
