Abstract
Sheep with Johne’s disease develop epithelioid macrophage microgranulomas, specific to
Keywords
The diagnosis of clinical Johne’s disease (JD) in sheep is often made postmortem with examination of gross lesions including cording or lymphangitis of the ileal serosal lymphatics, lymphadenitis, and enlargement of the mesenteric lymph nodes and corrugated granulomatous thickening of the ileal mucosa with confirmation made histopathologically. In live sheep, available diagnostic tests include fecal culture, Ziehl Neelsen stained fecal smears identifying acid fast organisms (AFO), ELISA, Map-specific polymerase chain reaction (PCR), identification of Map specific peptides, and measuring the mRNA expression of pro-inflammatory cytokines such as gamma interferon. Sheep with clinical JD have advanced ileal lesions with Map cultured from organs distant to the alimentary tract such as the peripheral lymph nodes, skeletal muscle, and liver as a result of Map bacteraemia and systemic infection. 2,4,8,9 The portal blood system facilitates Map infection of the liver, which is an organ readily accessed using routine biopsy in the live animal. Liver biopsy histopathology may provide an opportunity to identify JD in live sheep without surgery. The objectives of this study were to determine whether sheep with ileal lesions typical of Map have concurrent hepatic epithelioid macrophage microgranulomas (HEM), compare the sensitivity and specificity of liver histopathology with ELISA serology for diagnosis of ewes affected by Map, defined as those with Type 3b and 3c ileal lesions 7 (Table 1), and determine whether liver biopsy samples provide enough tissue for use as a diagnostic test.
Criteria for Classification of Lesion Types Associated With Natural Paratuberculosis Infection in Sheep. 7

Adult, Romney composite mixed breed and mixed age ewes (>3 years old) from 7 North Island New Zealand farms, with a history of clinical JD and flock size >3000 ewes, were body condition scored (BCS) with 126 ewes selected over a 2-year period with BCS ≤1.5 (scale 1–5). Vaccination against ovine JD was not practiced on these farms. A subsample of 80 ewes were blood tested for serology using ELISA (Institut Pourquier, Montpelier, France/Idexx Laboratories Inc; New Zealand Veterinary Pathology Ltd commercial diagnostic laboratory, Palmerston North). Ewes were euthanized and a simulated liver biopsy was performed using a styletted, 3-mm inner core diameter trocar (Shoof International) with the liver core fixed in 10% buffered formalin. 10 Necropsies were then performed, with samples of terminal ileum, including ileocaecal valve, mesenteric lymph node, and a 200-mm2 section of liver (1 cm × 2 cm) collected from the dorso-cranial right lobe from all ewes and fixed in 10% buffered formalin.
Histopathology
For histopathology, fixed tissue sections of ileum, mesenteric lymph node, liver, and liver biopsy cores were processed routinely and examined for the presence of granulomatous inflammation and AFO. One section per tissue was cut at 3 μm and stained with haematoxylin and eosin (HE) and Ziehl Neelsen (ZN). For liver biopsy cores, 24 serial longitudinal sections at 3 μm were taken. Alternate sections were discarded and the remainder serially mounted onto 4 glass slides (3 sections per slide) with 6 sections stained using either HE or ZN. Each biopsy section measured approximately 3 mm × 12 mm, with 6 sections providing approximately 200 mm2 of liver for examination, equivalent to the size of the single section examined from the necropsy liver sample. Ewes were classified retrospectively as either not histologically affected (not infected or latently infected) or histologically affected with Type 1, 2, 3a, 3b, or 3c ileal lesions (Table 1). 7
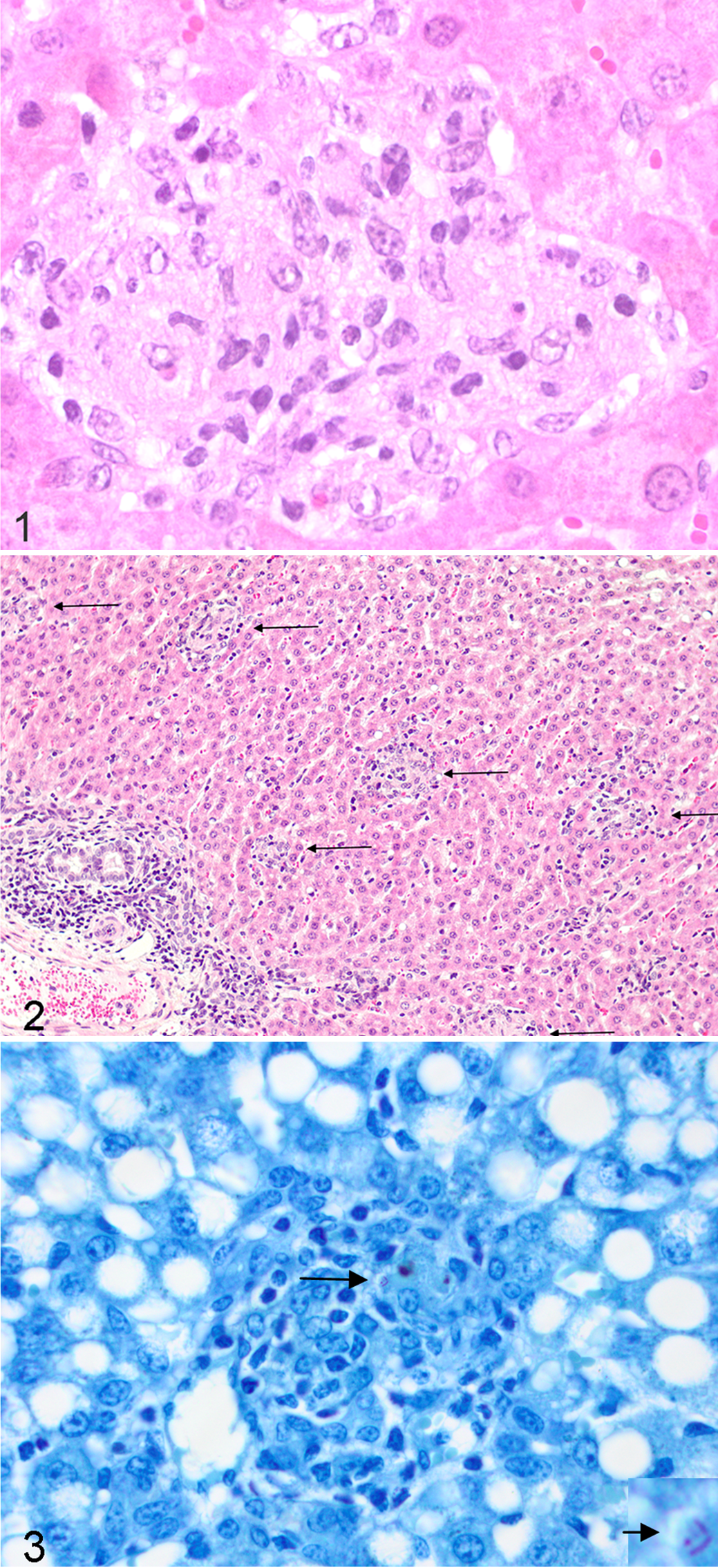
Livers were examined for the presence of spherical HEM consisting of epithelioid macrophages with clear and large nuclei, prominent nucleoli, with lightly staining HE foamy vacuolated cytoplasm, with or without acid-fast organisms with some lymphocytes present (Fig. 1). Histopathlogy was performed blinded to the identity of the ewe.
Statistical Analysis
The sensitivity and specificity of liver biopsy histopathology and ELISA as diagnostic tests for ewes with ileal lesions and JD were determined as described by Altman and Bland. 1 The level of agreement between the presence of HEM in liver tissue and either the ileal lesion score or ELISA positivity were determined using Cohen’s kappa coefficient. 3
Results
Data on ileal and liver lesions, AFOs, and ELISA results are presented in Table 2. Sixty (47.6%) ewes had ileum histopathology typical of that caused by Map including 51 classified with Type 3b (
Number of Ewes Sampled With Ileum and Liver (biopsy and section) Histopathology, Acid Fast Organism (AFO), and ELISA Serology.
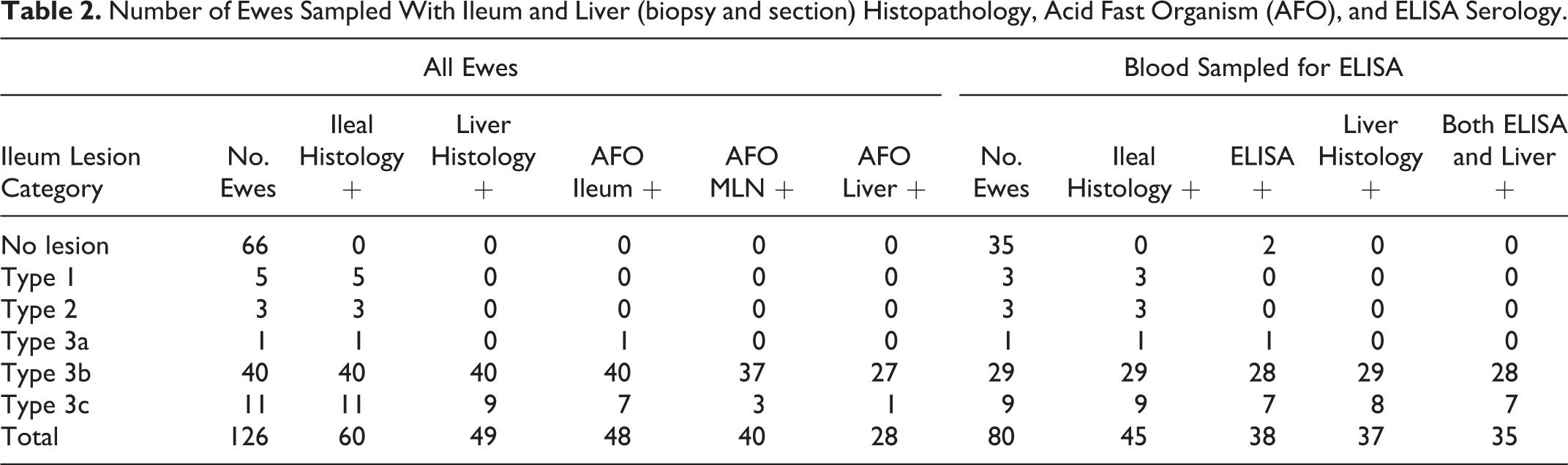
Hepatic epithelioid macrophage microgranulomas were observed only in ewes with Type 3b (100%,
Of the 80 blood tested subsample of ewes, 45 had ileum histopathology comprising 3 Type 1 and Type 2, 1 Type 3a, 29 Type 3b, and 9 Type 3c lesions. ELISA was positive in 28 of 29 Type 3b and 7 of 9 with Type 3c lesions, 2 of 35 ewes without Map ileal lesions, and the ewe with the Type 3a ileal lesions. Hepatic epithelioid macrophage microgranulomas were present in all ewes with Type 3b, 8 of 9 with Type 3c lesions, none with Type 1, 2, or 3a ileal lesions or ewes without Map ileal lesions. Neither liver histology nor ELISA were positive in ewes with Type 1 or Type 2 ileal lesions, and HEM were found in all ELISA positive ewes with Type 3b and 3c ileal leasions (Table 2).
The sensitivity and specificity for liver histopathology (section or biopsy) for predicting the presence of Type 3b and 3c ileal lesions was 96% (95% confidence interval [CI], 0.87-0.99) and 100% (95% CI, 0.95-1), respectively, and for ELISA, 92% (95% CI, 0.79-0.97) and 93% (95% CI, 0.81-0.98), respectively. Almost perfect level of agreement was shown between ileum and liver histopathology (Cohen’s kappa = 0.82,
Discussion
No HEM or hepatic AFO were found in Type 1, 2, or 3a ewes or in ewes without ileal Map lesions, suggesting that portal vein Map bacteraemia may not have occurred. In contrast, bacteraemia and disseminated Map infection appears to have occurred in ewes with Type 3b and 3c ileal lesions, with HEM found in 96% of cases. In these ewes, AFO did occur in HEM (<10/granuloma) although not every granuloma revealed AFO in stained slides (Fig. 3).
Infrequently, other forms of microgranulomas without AFO were observed in the liver sections from Type 1, 2, 3a, 3b, and 3c ewes including ones dominated by neutrophils or lymphocytes, with or without eosinophil infiltration, the etiology of which is unknown although they may be as a result of possible migrating parasite larvae.
Despite the availability of diagnostic tests to identify clinically affected sheep, there are currently none that reliably identify sheep with subclinical JD or where early Map bacteraemia has occurred. Blood culture has little application for diagnosis in early Map infections. 2 ELISA has a wide range for sensitivity and specificity, being 16% to 85% and 95% to 100%, respectively. 6 It also appears that liver histopathology was unable to identify sheep subclinically affected by Map (Type 1, 2, or 3a ileal lesions). However, previous longitudinal studies where lambs have been orally dosed with high numbers of Map have found young animals with Map infection disseminated to tissues outside the alimentary tract including bacteraemia prior to the onset of clinical disease. 2,4,5 In those studies, where hepatic lesions were described, no conclusions were made as to their significance. 4 Nevertheless, they did demonstrate the occurrence of systemic and disseminated Map infection in young animals and are evidence that Map bacteraemia and histopathological involvement of the liver is not just limited to adult sheep with Type 3b and Type 3c ileal lesions. Hepatic epithelioid macrophage microgranuloma lesions do occur in young sheep subclinically affected by Map. We therefore hypothesize that in sheep destined to develop clinical JD, an early Map bacteraemia occurs and liver biopsy histopathology may provide a surrogate measure of the progression of ileal lesions in lambs and indirectly describe the relationship between the development of HEM and clinical JD.
The principal conclusions from this study are that there is a high level of agreement between HEM and Type 3b and 3c ileal lesions in ewes, that liver biopsy histopathology in sheep has high sensitivity and specificity for identifying ewes with Type 3b and Type 3c ileal lesions and that liver biopsy cores provide adequate tissue for this diagnostic test in live animals. Liver biopsy histopathology is a potential research diagnostic model, and while this study has provided a test of concept, further research is now needed to fully validate the diagnostic sensitivity and specificity for sheep with Types 1, 2, and 3a ileal lesions. Further longitudinal study is also needed to establish a timeline for the formation of liver granulomas after oral Map exposure and whether lambs destined to eventually develop clinical JD can be identified in the early subclinical stages using this technique.
Footnotes
Acknowledgements
We would like to thank Dr Geoff de Lisle from AgResearch, National Centre for Biosecurity and Infectious Disease, Wallaceville, Upper Hutt, New Zealand, for text editing, and the Lewis Fitch Veterinary Research Fund, Massey University, Palmerston North, New Zealand for contributing toward funding this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
