Abstract
Brucella ovis is a bacterial pathogen present in most major sheep-producing regions of the world. The pathogen is associated with ram infertility, decreased ewe conception rates, and premature lambs. Twenty ELISA seropositive or indeterminate rams were culled from a B. ovis–positive flock, and donated to the Wyoming State Veterinary Laboratory for evaluation of infection. Tissues from each ram were collected at autopsy for additional testing, including bacterial culture, PCR, and histopathology. Of 17 seropositive rams, 11 rams were also positive by culture and PCR, and had evidence of mild histologic lesions; 1 seropositive ram was positive by culture with mild histologic lesions, but negative by PCR. Five seropositive rams were negative by culture and PCR and had no histologic lesions. Three indeterminate rams were negative by culture and by PCR and had no histologic lesions. The tissues in which B. ovis was most often detected included the epididymis, vesicular gland, and ampulla. Although this was a small study, the observation that 5 of 17 (29%) rams that were initially seropositive had no evidence of infection is interesting. A convalescent test for valuable seropositive animals prior to culling may be useful, and reproductive tissues may be evaluated postmortem if confirmatory testing is desired.
Brucella ovis is a gram-negative coccobacillus and facultative intracellular pathogen. The pathogen is associated with epididymitis and impaired fertility in rams, as well as failure to conceive, embryonic resorption, abortion, stillbirth, and weak newborn lambs in ewes. 6 The main transmission route for B. ovis is venereal. Rams are carriers of the bacteria; ewes are thought to act as mechanical carriers between rams. 13 Another route of transmission is from ram to ram, through direct contact. 5 Palpable lesions are detected in the testes and epididymides of ~35% of B. ovis–infected rams. 19 This is important during breeding soundness examination, particularly if only the rams with testicular or epididymal abnormalities are sampled for B. ovis testing. If 65% of infected rams with palpably “normal” testes or epididymides are not being sampled, bacterial infection could potentially be maintained, resulting in lowered fertility within the flock.
In addition to causing an adverse effect on the health of the flock, B. ovis can have major economic consequences, with decreased conception rates, prolonged lambing periods, reduced marketability, and loss of valuable genetics of culled rams.2,19 The estimated cost of retaining an infertile ram is US$302 per year, which takes into consideration the decreased number of ewes a ram is able to breed, decreased conception rates if ewes are successfully bred, and lambing loss attributed to infertility. 7 Thus, the economic impact of flocks infected with B. ovis may be significant. The purpose of our study was to determine if using serology as the only tool in determining B. ovis status is a reasonable approach for disease management.
The flock from which the rams in our study originated contains >10,000 primarily Rambouillet ewes, and 420 rams. All rams were in good nutritional condition and >1 y old. A subset of rams is annually tested for B. ovis, but all rams are not tested every year. This flock grazes on a mixture of private and public land, and there was opportunity for commingling with another large flock that does not routinely perform B. ovis testing. In 2014, following a decrease in lamb crop, all rams were serologically tested at the Wyoming State Veterinary Laboratory (WSVL; Laramie, WY) using an indirect enzyme-linked immunosorbent assay (iELISA); 121 of 420 (28%) rams tested either positive (113) or indeterminate (8).
Twenty rams (16 positive, 3 indeterminate, 1 not tested) were donated to the WSVL by the producer for additional testing 21 d after the initial ELISA. The rams were selected randomly, with all animals having no palpable lesions in the epididymis or testis. Upon arrival, a follow-up blood sample was taken, and a physical examination was performed. None of the rams had clinical abnormalities consistent with epididymitis. Autopsies were performed, and 12 tissues (ampulla, epididymis tail, vesicular gland, bulbourethral gland, testis, 1 medial iliac lymph node, and 1 superficial inguinal lymph node; Table 1) were evaluated for lesions consistent with B. ovis infection. Although 11 animals had abscesses in lymph nodes or lungs, no gross abnormalities were observed in the tissues sampled. Tissues and abscesses were evaluated with histopathology, bacterial culture, and PCR at the WSVL.
Comparison of results (positive/total) of 3 tests for Brucella ovis on tissues from reproductive tissues and lymph nodes of 20 rams.
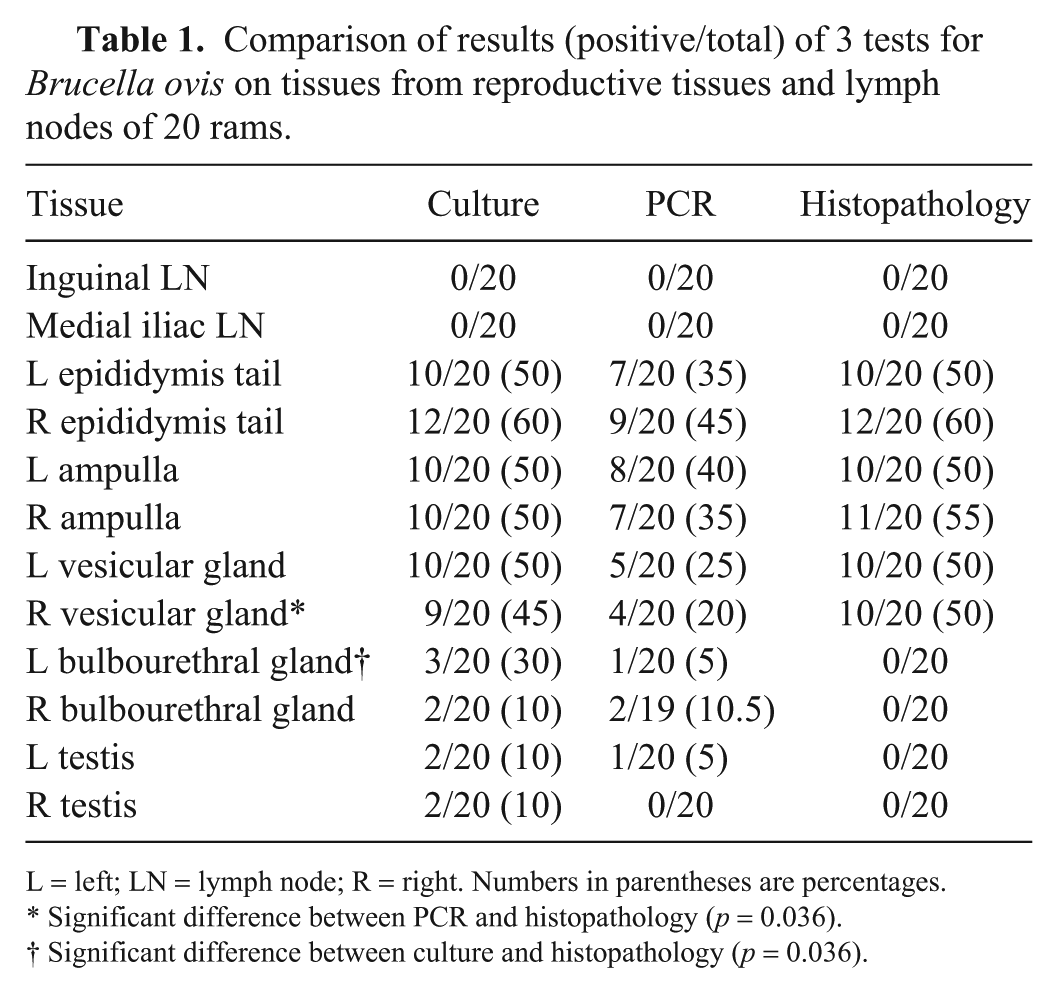
L = left; LN = lymph node; R = right. Numbers in parentheses are percentages.
Significant difference between PCR and histopathology (p = 0.036).
Significant difference between culture and histopathology (p = 0.036).
The National Veterinary Services Laboratories (NVSL) ELISA was utilized for serology. Briefly, this ELISA uses a hot, saline water–soluble extract of the REO-198 strain prepared according to the World Organization for Animal Health (OIE; http://www.oie.int/fileadmin/Home/eng/Health_standards/tahm/2.07.08_OVINE_EPID.pdf). Controls include “high-positive” sera, “low-positive” sera, and “negative” sera. Antigen and controls were purchased from NVSL (9-ELA, 9-HP, 9-LP, 9-N), and were provided with details of the optimal working dilutions (https://www.aphis.usda.gov/animal_health/lab_info_services/downloads/AmesReagentManualCurrent.pdf). The B. ovis antigen was diluted in carbonate buffer, coated on polystyrene microtiter plates, incubated, and then blocked with 10% bovine serum albumin solution diluted 1:10 in sterilized water prior to sample addition. The serum sample was added, followed by protein G-biotin from Streptococcus spp. (KPL, Gaithersburg, MD), Vectastain conjugate (Vector Laboratories, Burlingame, CA), blue phosphatase substrate (KPL), and APStop solution (KPL). All incubations were at 37°C in a humidified chamber, followed by triplicate washes with high salt buffer, with the exception of the blue phosphatase substrate incubation step, which was performed at room temperature on a microplate shaker. The average optical density was calculated for each sample and control, with the average sample-to-positive (S/P) ratio NVSL-specified cutoff values used to determine results. A S/P ratio >0.75 was recorded as positive, <0.40 as negative, and >0.40 to <0.75 as indeterminate.
Fresh tissues collected at autopsy were frozen at −20°C, and cultured following WSVL protocol. Briefly, 1 g of each thawed tissue was added to 5 mL of tryptose broth and homogenized in a stomacher; sterile swabs were used to independently sample the homogenate and plate onto 1 Columbia blood agar and 3 modified Thayer–Martin media plates (Hardy Diagnostics, Santa Maria, CA). The plates were incubated at 37°C in 10% CO2 and read by the same technician every 24 h, up to 10 d. In most of the culture-positive tissues, B. ovis appeared ~3–5 d after incubation as small, pinpoint, dull-yellow, opaque colonies. Suspected positive isolates were confirmed using routine biochemical methods and the Biolog identification system (Biolog, Hayward, CA). Other bacteria isolated from the samples or abscesses sampled during autopsy included Trueperella pyogenes and Corynebacterium pseudotuberculosis (Table 2).
Correlation of Brucella ovis serology with culture, PCR, and histopathology in 20 rams.
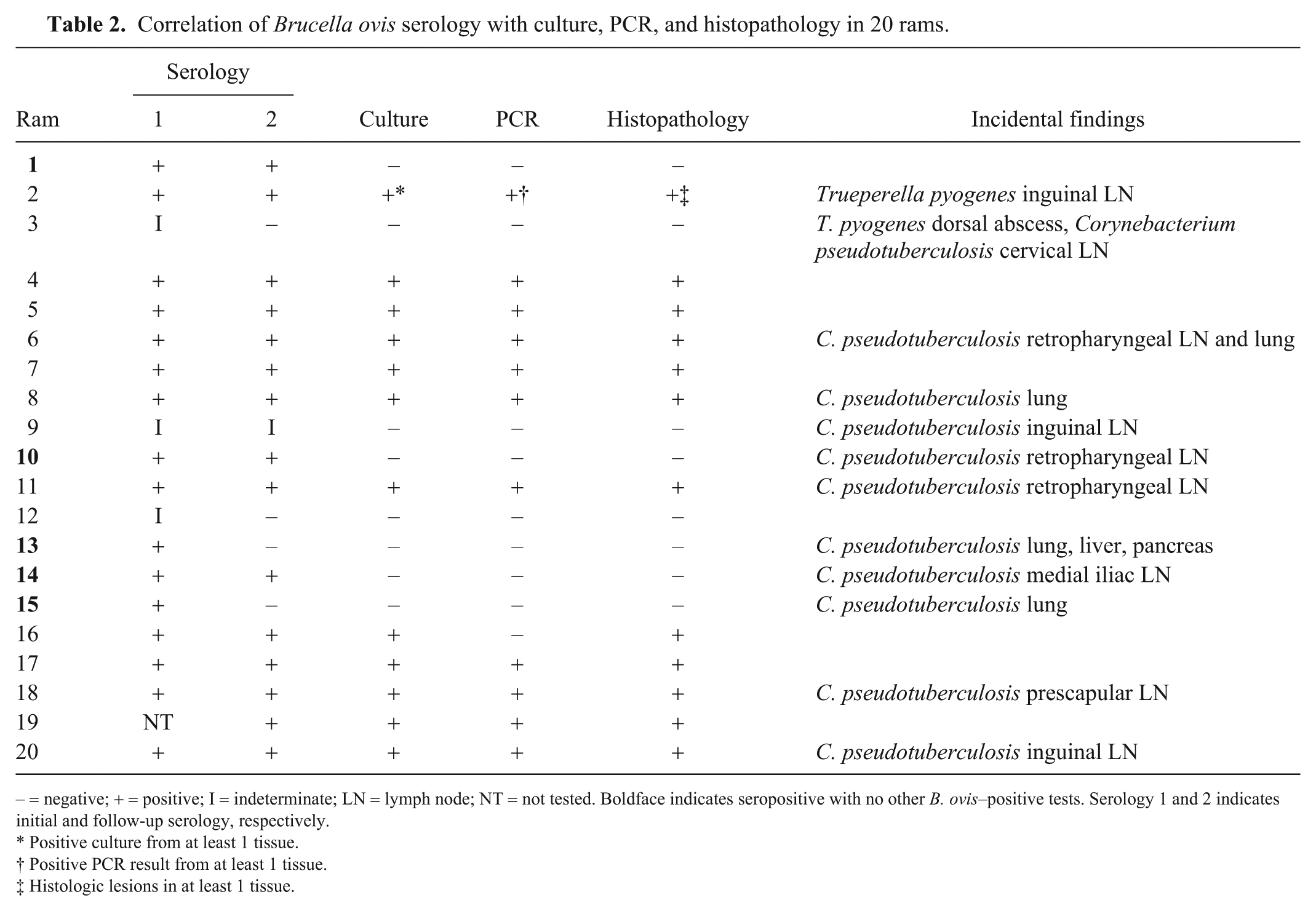
− = negative; + = positive; I = indeterminate; LN = lymph node; NT = not tested. Boldface indicates seropositive with no other B. ovis–positive tests. Serology 1 and 2 indicates initial and follow-up serology, respectively.
Positive culture from at least 1 tissue.
Positive PCR result from at least 1 tissue.
Histologic lesions in at least 1 tissue.
DNA was extracted from tissues (DNeasy blood and tissue kit, Qiagen, Germantown, MD), and PCR was performed in a 25-µL reaction using the protocol for IS711 B. ovis–specific primers. 4 The positive control was DNA extracted from a pure B. ovis culture, and negative controls included DNA from a pure culture of B. abortus RB51 strain, and water. A subset of PCR amplicons was sequenced to ensure that the expected target was being amplified.
Tissue samples collected at autopsy were fixed in 10% formalin, processed routinely, stained with hematoxylin and eosin, and evaluated by one pathologist for evidence of inflammation.
B. ovis was most often detected in the epididymis, vesicular gland, and ampulla by both culture and PCR and then correlated with the histologic findings (Table 1). There was only one tissue, the left bulbourethral gland, in which there were significantly more rams identified by culture than by their histologic lesions (Prism 2-way ANOVA multiple comparisons, 3 vs. 0, p = 0.036). For one other tissue, the right vesicular gland, there were significantly more rams identified by PCR than by histologic lesions (4 vs. 10, p = 0.036).
All 10 of the culture-positive rams had histologic lesions in the ampulla and vesicular gland. Moderate-to-severe lymphocytic–plasmacytic ampullitis and seminal vesiculitis were observed, with occasional intra-tubular neutrophilic vesiculitis in some cases. Although no inflammation was observed in the ampulla or vesicular glands of culture-negative rams, mild ampullitis has been reported in normal rams. 11 The head and tail of the epididymis showed signs of lymphocytic–plasmacytic epididymitis, with occasional neutrophils and macrophages mixed with the sperm in the tubules. In a few rams, there was mild lymphocytic–plasmacytic orchitis. The lymph nodes that were collected for histopathology had various degrees of reactive lymphoid hyperplasia and sinus plasmacytosis. This was most likely the result of chronic antigenic stimulation from B. ovis; however, other causes of lymphoid stimulation cannot be ruled out. No B. ovis lesions of the type typical of chronic and/or severe ovine brucellosis2,11 were observed.
Direct culture identified B. ovis in a higher proportion of rams for every tissue in comparison with PCR, except for the right bulbourethral gland, inguinal lymph node, and medial iliac lymph node, in which culture and PCR had similar results (Table 1). However, the difference in detection between culture and PCR was not statistically significant based on ANOVA. No B. ovis was detected in inguinal and medial lymph nodes from any of the 20 rams, suggesting that these are not tissues routinely associated with B. ovis infection. Interestingly, culture detected B. ovis in 12 of 20 rams, whereas PCR was positive in 11 of 20 rams. One possible reason for this result may be the result of sampling a small amount of tissue for DNA extraction with subsequent PCR, particularly if B. ovis is present in low numbers in focal areas of each tissue. Thus, if postmortem PCR is performed, one recommendation would be to sample multiple tissues (epididymis, ampulla, and vesicular gland) and to use duplicate or triplicate samples for each tissue.
In the initial cohort of rams, 3 were indeterminate, 1 was not tested, and 16 were seropositive. Of those that were seropositive, 2 were seronegative on the follow-up test. Additionally, 3 that tested seropositive at both times had no evidence of infection (Table 2). All 5 rams that were either seronegative or indeterminate on the follow-up serologic test were also negative by culture and PCR, and did not have any histologic lesions suggestive of B. ovis infection. The observation that 5 of 16 (31%) seropositive rams had no evidence of an established infection is important, and corroborates a similar earlier observation. 10 In the earlier study, a subset of rams with culture-negative semen were positive by different serologic tests, including gel diffusion and complement fixation. 10 The implication is that retesting serum 2–4 wk following a positive serologic test may be of value, and may prevent the culling of valuable animals. In our sample set, if the seropositives had been retested, then 2 animals may not have been culled unnecessarily. Sensitivity and specificity of the iELISA used in our study have been reported to be high (Se = 96.3%, Sp = 99.6%). 12 One explanation of false-positive results is cross-reaction with other bacteria from genera Agrobacterium, Francisella, Ochrobactrum, Phyllobacterium, Rhizobium, or other gram-negative bacteria with similar outer membrane proteins.9,15 In our study, none of the cross-reactive bacteria were identified by bacterial culture, but 12 of 20 rams were positive for C. pseudotuberculosis or T. pyogenes, which are commonly associated with abscesses in otherwise healthy sheep.3,8
If the follow-up serology results are compared to the other testing performed, then 16 of 19 (84%) samples were in concordance regarding serology and culture or PCR. There were 3 of 19 (16%) discordant rams that tested positive by serology, but negative on both culture and PCR, suggesting exposure to B. ovis without an established infection. One indeterminate sample did not have any evidence of infection. However, indeterminate results are difficult to classify and many producers will cull indeterminate animals. It is important to note that 2 of 3 animals that initially tested as indeterminate, tested seronegative on follow-up serology. Rams experimentally infected with B. ovis have detectable antibodies by ELISA on day 11, and the levels continue to rise until day 41, but decrease to low levels by day 63. 1 This would support the recommendation of retesting animals that are seropositive or indeterminate within 2–4 wk, because these rams could be mounting a detectable immune response during the initial sample collection.16,18 Retesting could include the use of another serologic test such as complement fixation or a repeat ELISA and/or PCR on semen for detection of the organism.14,16
Serologic testing has an important role to play as a screening tool for determining flock exposure to B. ovis. Producers may improve economic returns by testing their flocks and culling B. ovis–positive rams.17,18 However, as our study has shown, a positive serologic test result does not indicate that a specific animal is infected, and not all infected rams will exhibit clinical signs of epididymitis. Thus, additional testing should be considered prior to culling a seropositive animal from the flock. From our study, the recommendation for antemortem testing would be a follow-up serologic test, and/or PCR on a semen sample, for detection of B. ovis. The recommendation for postmortem testing would be culture and/or PCR on reproductive tissues (epididymis, ampulla, and vesicular gland).
Footnotes
Acknowledgements
We thank the producer for their generous donation of animals for the purpose of this study. We also thank the WVSL laboratory staff for assisting us with sample collection and testing. We also thank undergraduate students Sarah Myers, Emma Rovani, and Megan Weisensee for contributing to the PCR testing of the tissue samples.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This project was supported by the Kelly Palm Memorial Externship and the Wyoming State Veterinary Laboratory.
