Abstract
The recent application of neuroimaging techniques in veterinary neurology has led to the accurate localization of many types of intracranial lesions but has also created a clinical need, particularly with brain tumors, for a specific intraoperative diagnosis. For human brain tumors, a smear technique has been used successfully for many years to provide an extremely rapid, highly accurate intraoperative diagnosis. In similar smear preparations of intracranial lesions, obtained either by computed tomography (CT)-guided stereobiopsy or from a craniotomy, we have described distinguishing cytologic features of some primary spontaneous nervous system tumors in 80 dogs and 13 cats. A final diagnosis was confirmed by evaluation of paraffin-embedded sections from the same sample and, when appropriate, by immunocytochemical staining. Preliminary findings indicate that, in dogs and cats, this procedure is useful for rapid, accurate intraoperative diagnosis of many primary nervous system tumors. The distinguishing features of the canine and feline tumors bear a remarkably close resemblance to their human counterparts.
In veterinary neurology, the recent application of neuroimaging techniques to the brain, of both computed tomography (CT) and magnetic resonance imaging (MRI), has enhanced the clinical ability to detect and localize a wide variety of intracranial lesions. However in most cases, the current state of radiographic interpretation provides only a broad differential diagnosis for these lesions. From a clinical viewpoint, there is now a critical need for an accurate, intraoperative diagnosis of such visualized lesions. An intraoperative diagnosis of a specific nervous system tumor type can provide crucial information on operative management by the neurosurgeon and allow informed decisions immediately on intratumoral gene therapy, radiation, or chemotherapy. In people, the intraoperative cytological evaluation of smear preparations of central nervous system (CNS) tumors, supported by frozen and paraffin-embedded tissue, over many years has become a routine procedure providing a rapid, highly accurate diagnosis. 3 9 15 20 21 Cytologic profiles of smears of various types of human CNS tumors and of their histological subtypes have been well described. 4 5 15 17
Since the first report in 1930 on the application of supravitally stained smear preparations for the intraoperative diagnosis of human brain tumors, 7 improved techniques for both obtaining and evaluating smear samples have evolved. 4 15 Currently most tissue samples are obtained by either craniotomy with subsequent surgical or aspiration biopsy or, preferably, by the minimally invasive stereotactically placed needle biopsy procedure. Smear preparations are generally wet fixed in 95% alcohol and stained with hematoxylin and eosin (HE), although some institutions use toluidine blue, geimsa, or Papanicolou's stain. 15 20 Smears prepared either from a touch impression or from being air dried prior to staining are considered much less informative. 15 The method of fixation also allows for immunocytochemical staining when considered necessary. 16 While there are advantages and disadvantages for each of these techniques, probably the most important factors in optimizing diagnostic accuracy are utmost familiarity by the neuropathologist with one stain and using a wet-fixed smear preparation. 3 9 15 To date, there have been no reports on the use of this smear technique for the intraoperative diagnosis of primary feline or canine brain tumors. However, using air-dried smears obtained from either impression or needle aspiration, the cytological characteristics of two canine meningiomas and a glioma have been described. 2 8 18 With our recent development of a modified human CT-guided stereotactic biopsy system for intracranial lesions in dogs and cats 12 13 and our need for a precise clinical diagnosis for planning either conventional or experimental therapeutic approaches, we have begun evaluation of some feline and canine primary nervous system tumors using this smear technique.
Materials and Methods
Data for this study of 93 primary brain tumors was obtained from 80 dogs and 13 cats with neurological deficits that had been previously admitted to the Veterinary Medical Teaching Hospital (VMTH) at the University of California Davis (UC Davis). Using either precontrast, postcontrast, or both images acquired by CT or MRI, a variety of intracranial lesions (e.g., mass, space occupying, multifocal, or locally extensive) were visualized in these animals. Tissue samples were obtained from the lesions either by a CT-guided stereotactic brain biopsy procedure using a modified human Pelorus Mark III apparatus (n = 44) or intraoperatively at craniotomy (n = 49). 13 All smear preparations were made following procedures as previously described for humans. 1 15 Briefly, small tissue pieces (<0.5 × 0.5 × 0.5 mm) were harvested from both ends of a 3 × 1.8-mm biopsy needle tissue core sample. A smaller fragment of the tissue sample was immediately placed at one end of a standard glass microscope slide. The end of another glass slide was then placed over the tissue, and an appropriate amount of pressure was applied as the slides were gently and quickly pulled and slid apart to produce a smear. Then the smear was fixed by immediate immersion in 95% ethanol for 60 seconds. One or more slides were immediately stained with a rapid HE staining procedure taking 15 minutes, using an automated microscope slide stainer (Shandon Varistain, model 24-3, Pittsburgh, PA) and then were evaluated histologically to make a provisional diagnosis. The remaining core sample was immersion-fixed in 10% buffered formalin, routinely processed for paraffin embedding, sectioned at 5 µm, stained with HE, then evaluated for a final diagnosis. When considered appropriate, immunocytochemical staining for one or more of two neuronal marker antigens (triple neurofilament [TNF] proteins and synaptophysin [SYN]), for an astroglial cell marker antigen glial fibrillary acidic protein (GFAP), for cytokeratin expression with low (L)– and high (H)–molecular weight (MW) cytokeratin antibodies, for vimentin, for S-100 α- and β-subunits, and for the canine common leucocyte antigen CD45RA, as well as for CD11d, CD18, CD57α, and CD79α, were done on extra smear preparations and/or sections from paraffin-embedded tissue using previously described methods. 6 The final diagnosis on each case was made from both interpretation of the smear and confirmation from the paraffin-embedded material. All cases were peer reviewed by one author (AWB), who is highly experienced with interpretation of smear preparations of human nervous system tumors. To be included in this study, both adequate smear preparations and paraffin-embedded HE-stained sections from the same sample had to be available.
Results
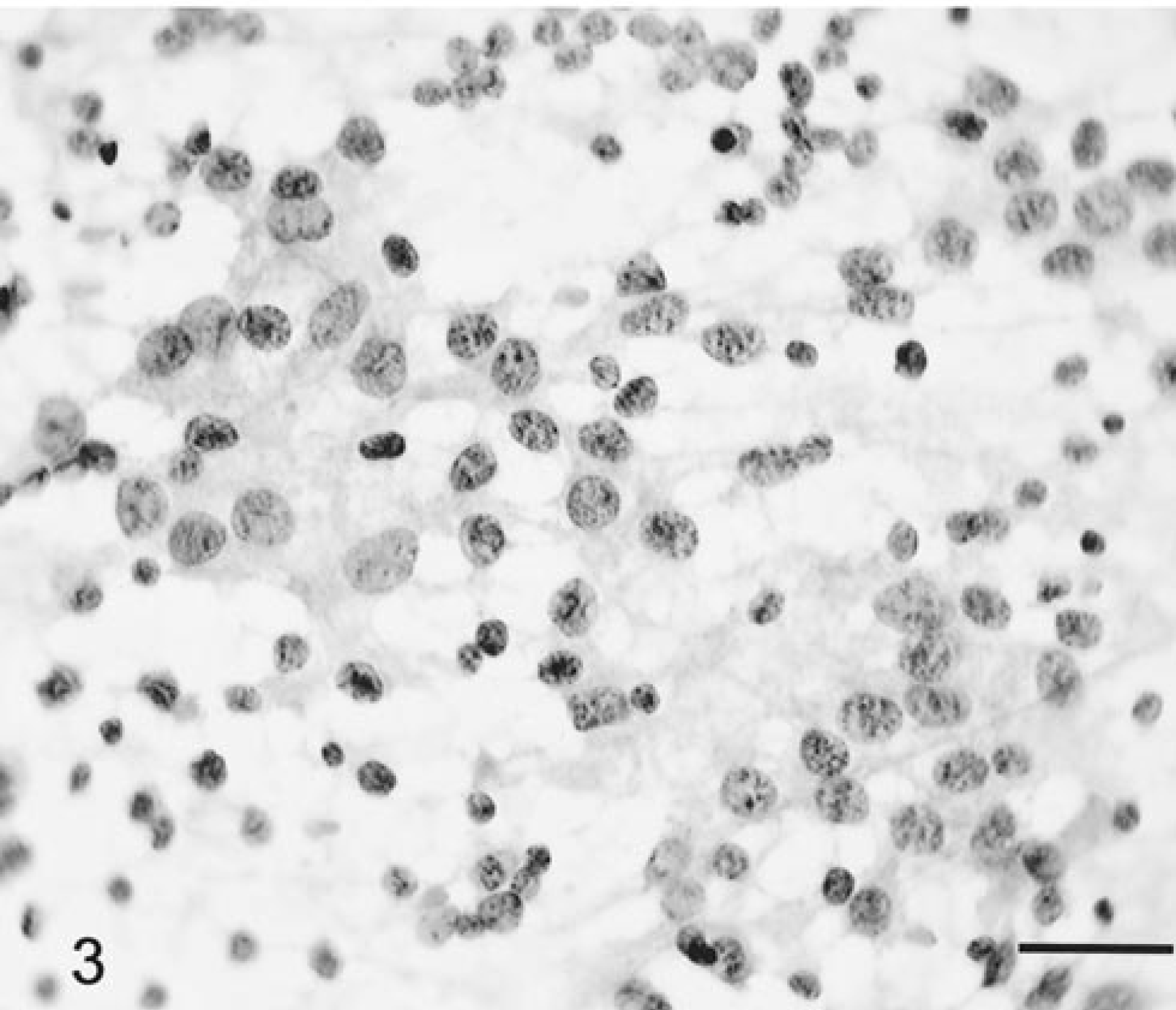
A total of 93 primary tumors of the CNS tumors were evaluated from 80 dogs and 13 cats (Table 1). The tissues were derived from a total of 44 stereobiopsies and 49 craniotomies. The number of samples derived from either stereotactic biopsy or craniotomy for each tumor type in both dogs and cats is listed in Table 1. The histologic diagnosis of the tumors included five oligodendrogliomas (five dogs, no cats), 11 astrocytomas (8, 3), two oligoastrocytomas (2, 0), one glioblastoma multiforme (1, 0), six choroid plexus tumors (6, 0), three ependymomas (2, 1), four primary CNS lymphomas (3, 1), five primary CNS histiocytic sarcomas (4, 1), two primitive neuroectodermal tumors (PNET) (2, 0), 51 meningiomas (44, 7), one granular cell tumor (1, 0), and two schwannomas (2, 0). Using formalin-fixed tissues, the 44 canine meningiomas were histologically subclassified into meningothelial, transitional, psammomatous, and secretory subtypes. 4 The purpose in thus classifying these meningiomas was to look for any possible correlation between the different meningioma subtypes and their cytologic appearance.
A list of CNS tumor types diagnosed in dogs and cats and the number of samples derived from either stereobiopsy or craniotomy for each tumor type and species.∗
n [n] = number of dogs [number of cats].
Tumor types
The major cytologic distinguishing characteristics of each of the CNS tumor types evaluated are described.
Oligodendroglioma
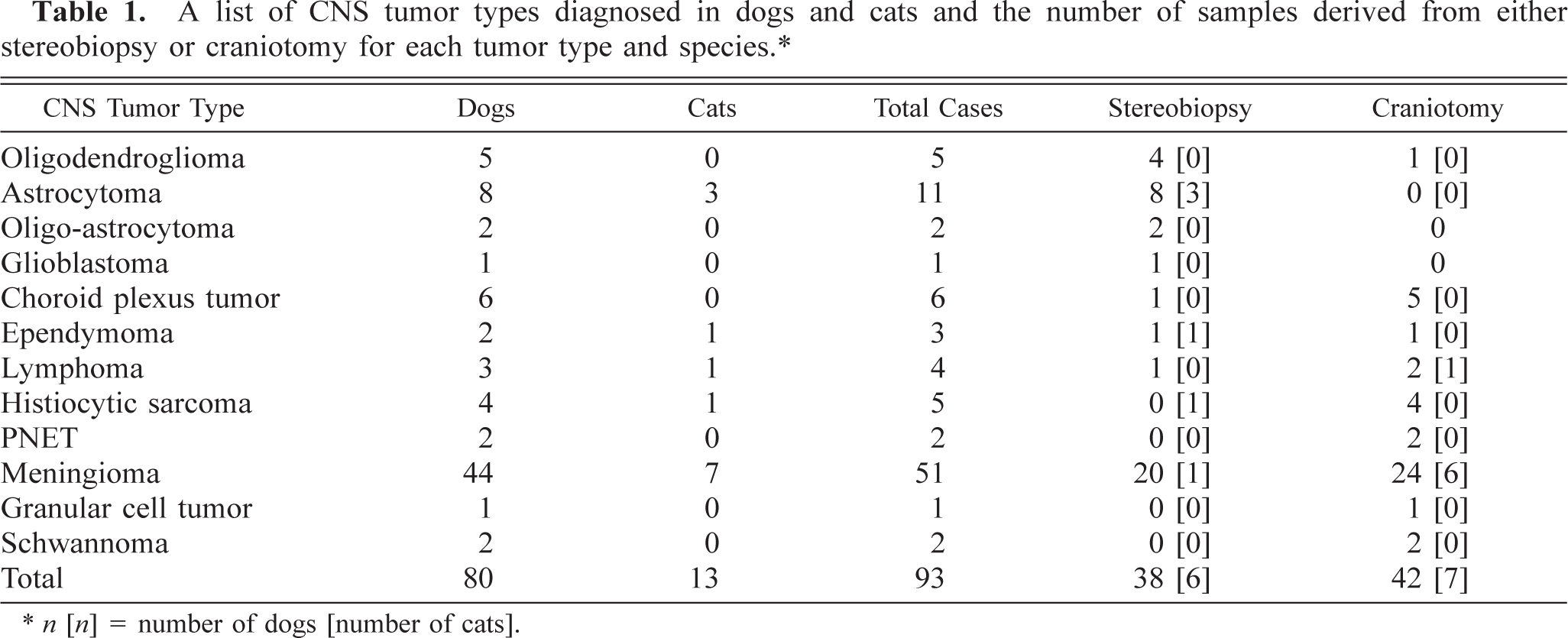
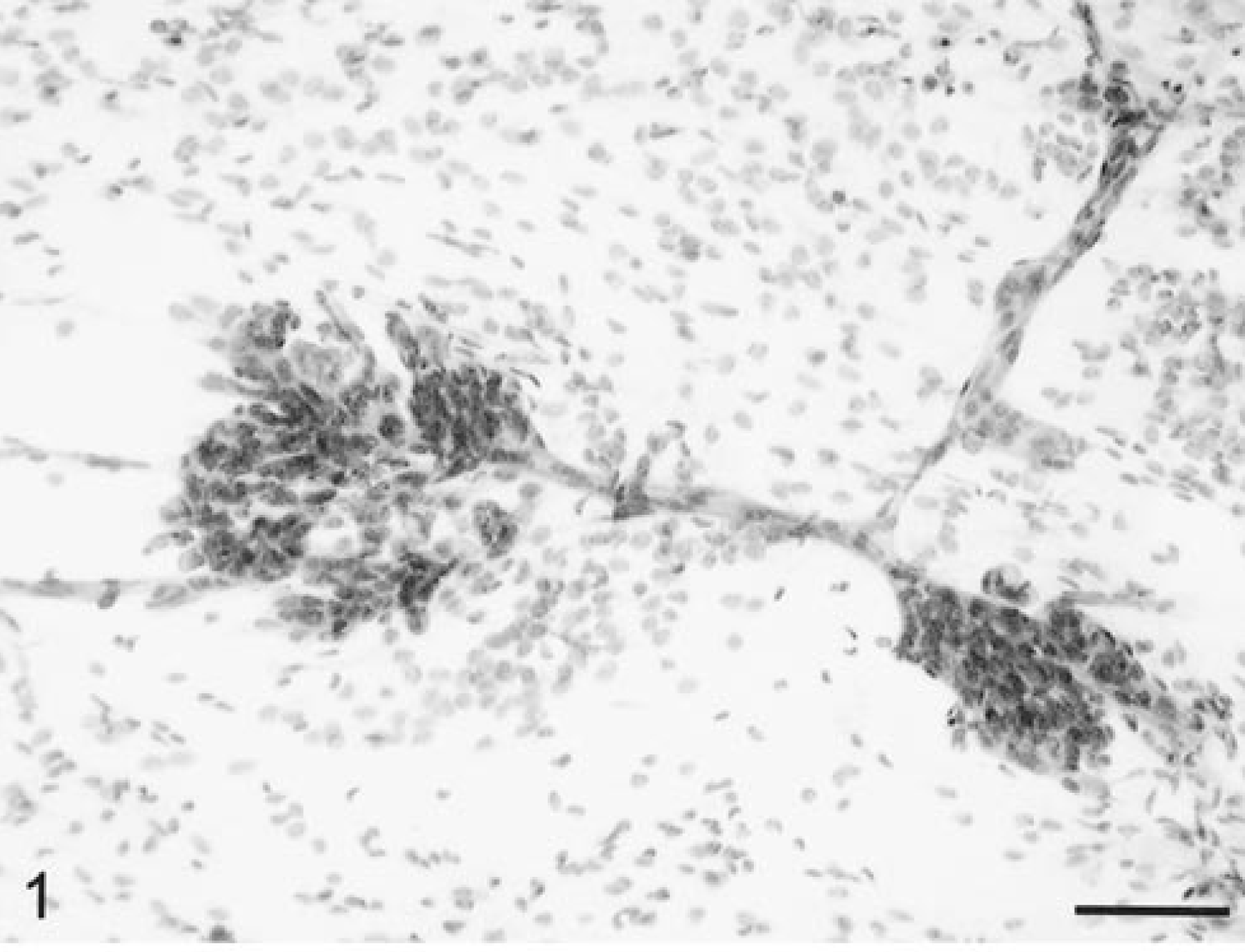
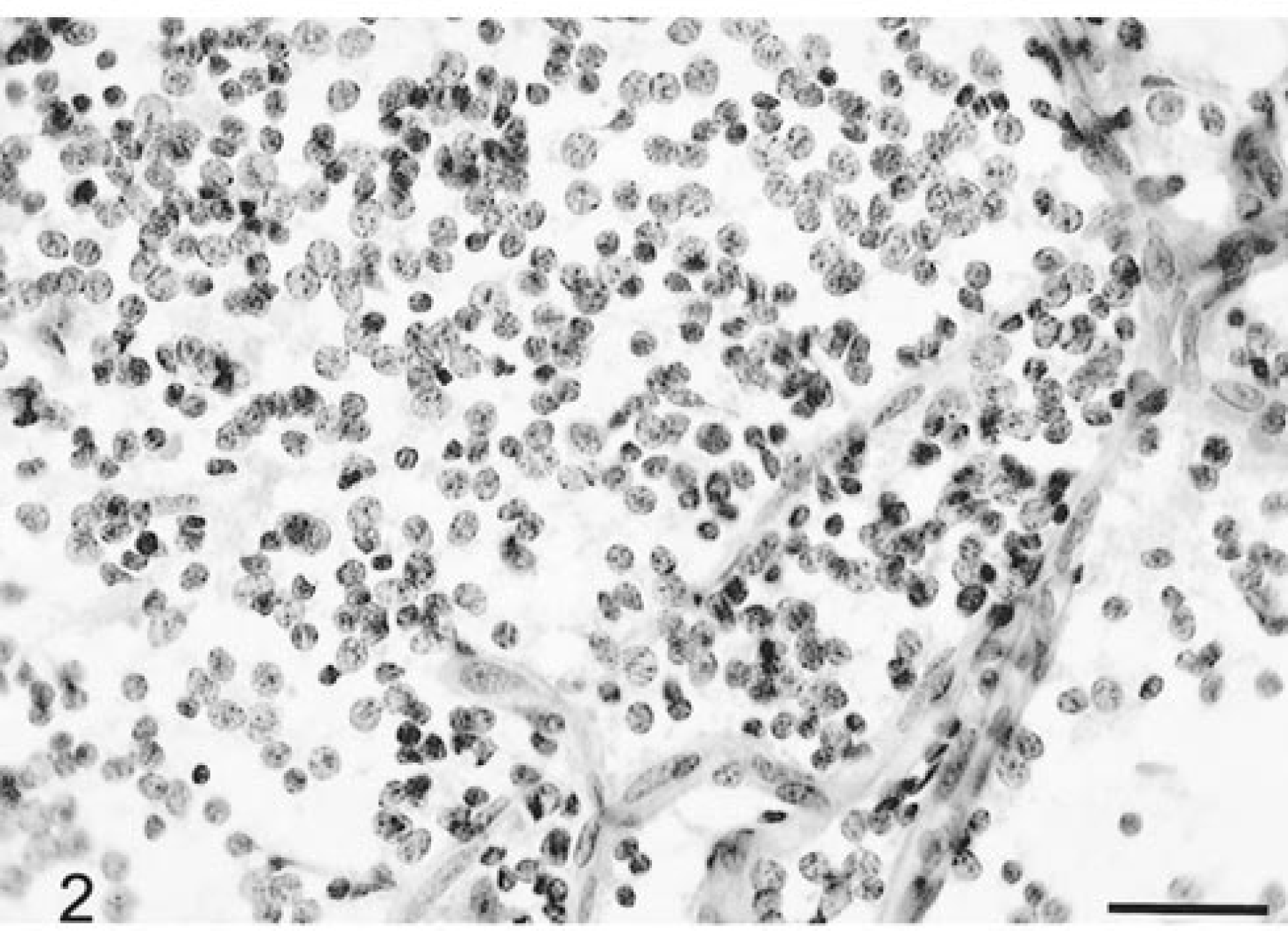
Five canine oligodendrogliomas, including three anaplastic subtypes, were evaluated. Grossly, all the tumor samples were very soft, often gelatinous to mucoid in consistency, and the smear preparations spread easily. On low-power magnification, there were large numbers of prominent, finely branching blood vessels. These often had segmentally thickened profiles due to focal proliferation of microvascular cells forming glomeruloid capillary tufts (Fig. 1). Blood vessels were very distinctive in being free of attached tumor cells. All smear preparations were relatively cellular. The relatively small cells were closely packed and were usually round, with moderate anisokaryosis (Fig. 2). Nuclei were uniformly round to ovoid and very basophilic. Nuclear chromatin was fine, granular, and evenly dispersed, whereas nucleoli were small, singular, or absent. There was minimal granular, eosinophilic cytoplasm with poorly defined borders (Fig. 2). Between cells was a finely granular, wispy eosinophilic matrix. The anaplastic oligodendrogliomas had more pleomorphic cells, some with a variable amount of prominent eosinophilic cytoplasm (Fig. 3). There were many necrotic cells, up to two mitotic figures per 40× high-power field (HPF), and a small number of reactive astrocytes.
Brain; oligodendroglioma; canine. There are branching vessels, some of which have segmental, focal glomeruloid microvascular proliferations. Marked hypercellularity with cells individually dispersed. HE. Bar = 70 µm.
Brain; oligodendroglioma; canine. Branching thin-walled capillaries and dispersed tumor cells with uniform round nuclei and relatively little cytoplasm HE. Bar = 30 µm.
Brain; anaplastic oligodendroglioma; canine. Marked nuclear pleomorphism with larger cells having abundant cytoplasm. HE. Bar = 25 µm.
Astrocytomas
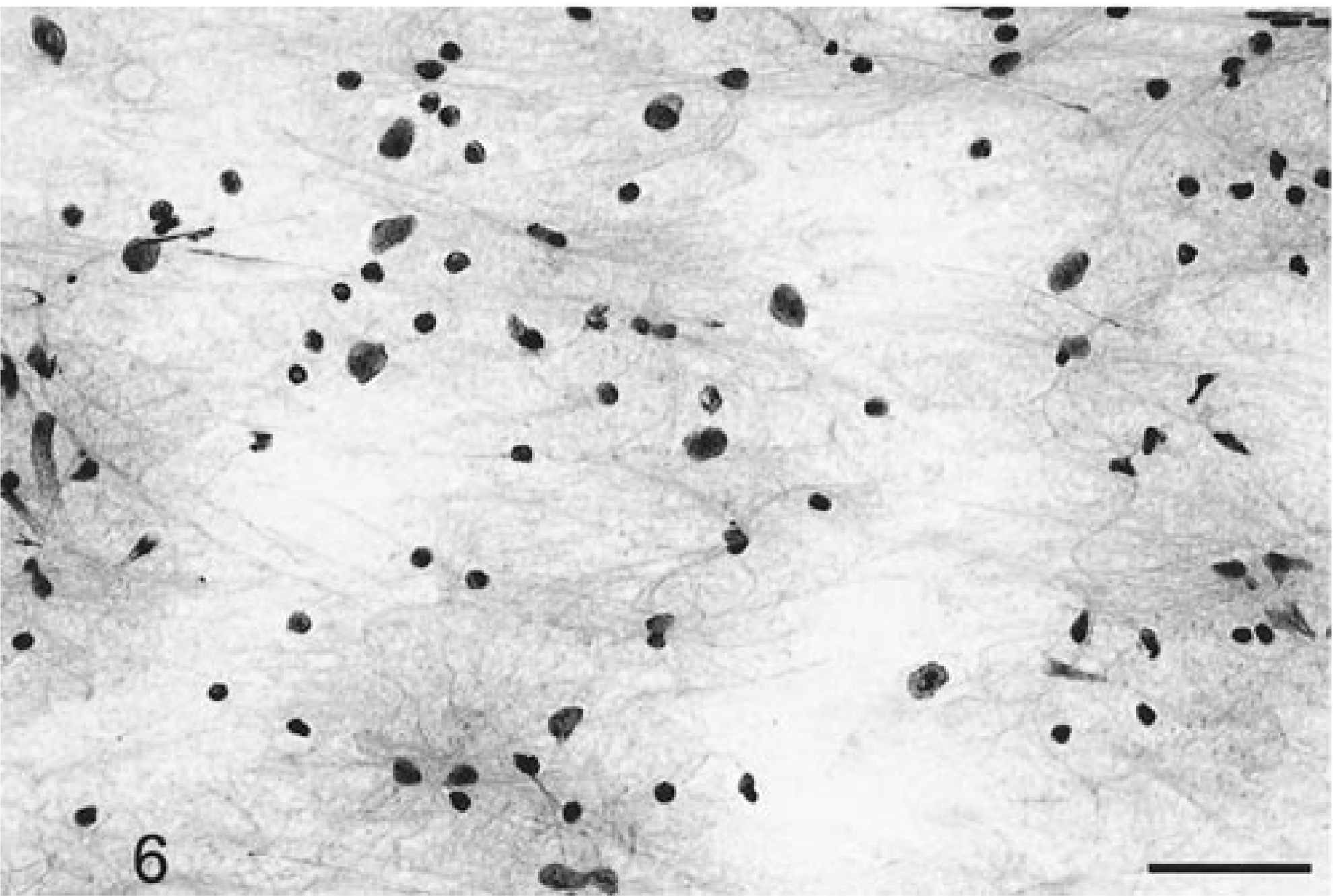
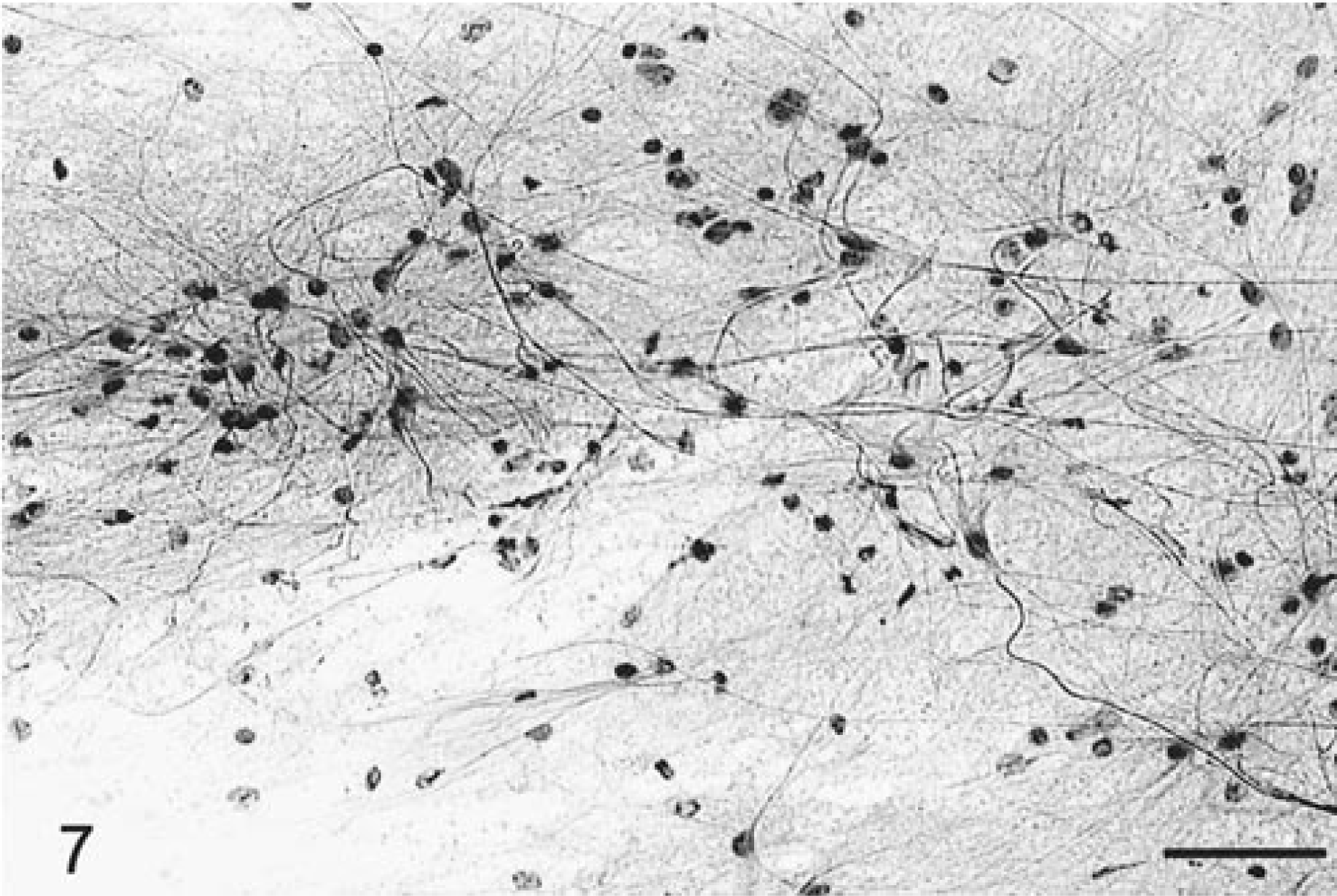
Eleven astrocytomas were evaluated from eight dogs and three cats. Most of the tumors smeared very readily and evenly. At low power, there was moderate hypercellularity (Fig. 4) compared with that of normal brain (Fig. 5). Blood vessels were thin walled, well defined, demarcated, and branching but otherwise unremarkable. Tumor cells emanated from the vessel margins and formed radiating streams of generally elongate cells with long, thin cytoplasmic processes (Fig. 4). The cells had elongate nuclei with an irregular border and coarsely stippled chromatin. The cytoplasm was usually minimal, and staining was lightly eosinophilic. There was an extensive network of randomly crisscrossing, eosinophilic, sharp fibrillary cytoplasmic processes between these tumor cells (Figs. 4, 6, 7). Sometimes these processes were extremely thick with multiple branches. The processes were best visualized by adjusting the condenser to produce a refractile light source (Fig. 7). Both the cell cytoplasm and these processes were strongly positive for GFAP and vimentin on the smear preparations. Marked nuclear atypia was often present, and up to two mitotic figures per 40× HPF were found in the more anaplastic astrocytomas (Fig. 6). Entrapped neurons were often seen. Numerous necrotic cells and rarely occasional neutrophils were admixed among the singly dispersed cells. There was sometimes a wispy, bluish staining matrix containing a finely fibrillated meshwork of processes.
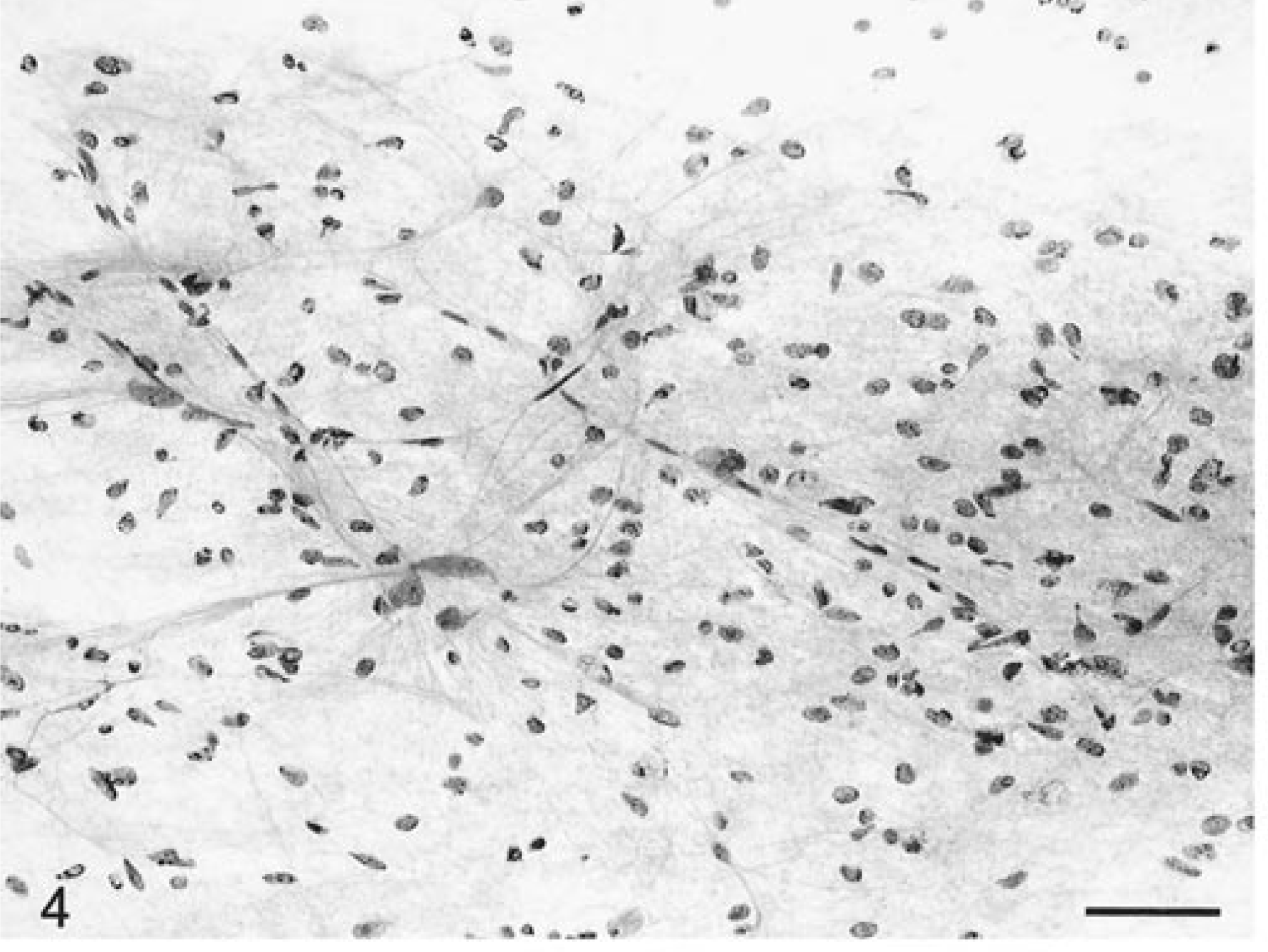
Brain; astrocytoma; canine. Branching thin-walled vessels with hypercellularity due to elongate pleomorphic tumor cells, which have fine streaming processes. HE. Bar = 52 µm.
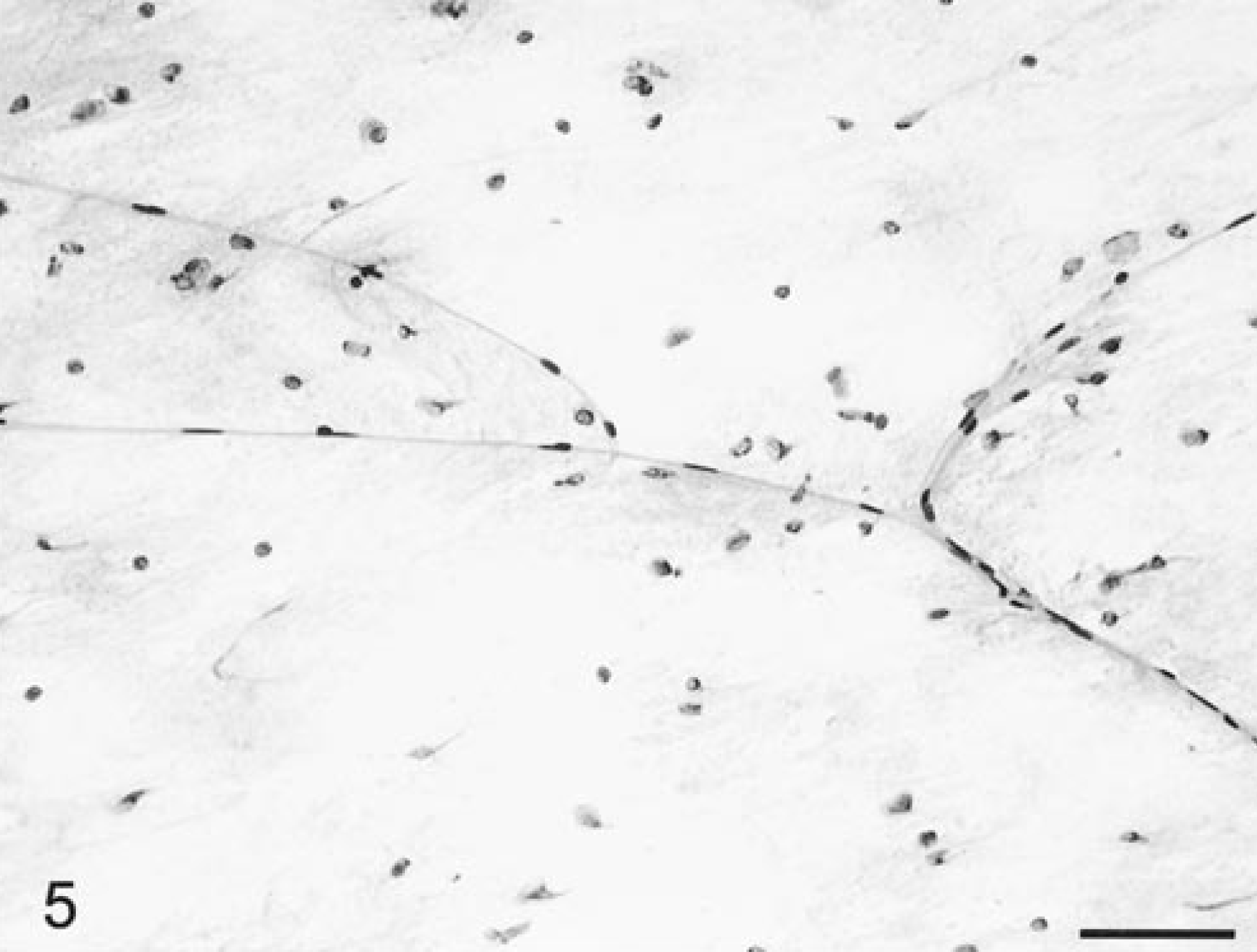
Normal brain; feline. Note cell density and thin-walled branching capillaries. HE. Bar = 53 µm.
Brain; astrocytoma; canine. Prominent nuclear pleomorphism with elongate to ovoid tumor cells with minimal cytoplasm and thin intertwining fibrillary processes. HE. Bar = 43 µm.
Brain; astrocytoma; canine. Highly refractile fibrillary processes emanating from pleomorphic tumor cells. HE. Bar = 45 µm.
Oligo-astrocytoma
The two canine oligo-astrocytomas were easy to smear and cells remained densely clustered around branching blood vessels or also dispersed into closely aggregated but separated cells. Blood vessels were numerous, highly branched with some thickening of their walls, with capillary proliferation, though glomeruloid formation was not seen. Individual cells had prominent nuclei without obvious cytoplasm, though some cells had bipolar fibrillary processes. Their nuclei were lightly basophilic, round to oblong, varied in size, and often had irregular borders. Foci of calcification were scattered throughout (Fig. 8).
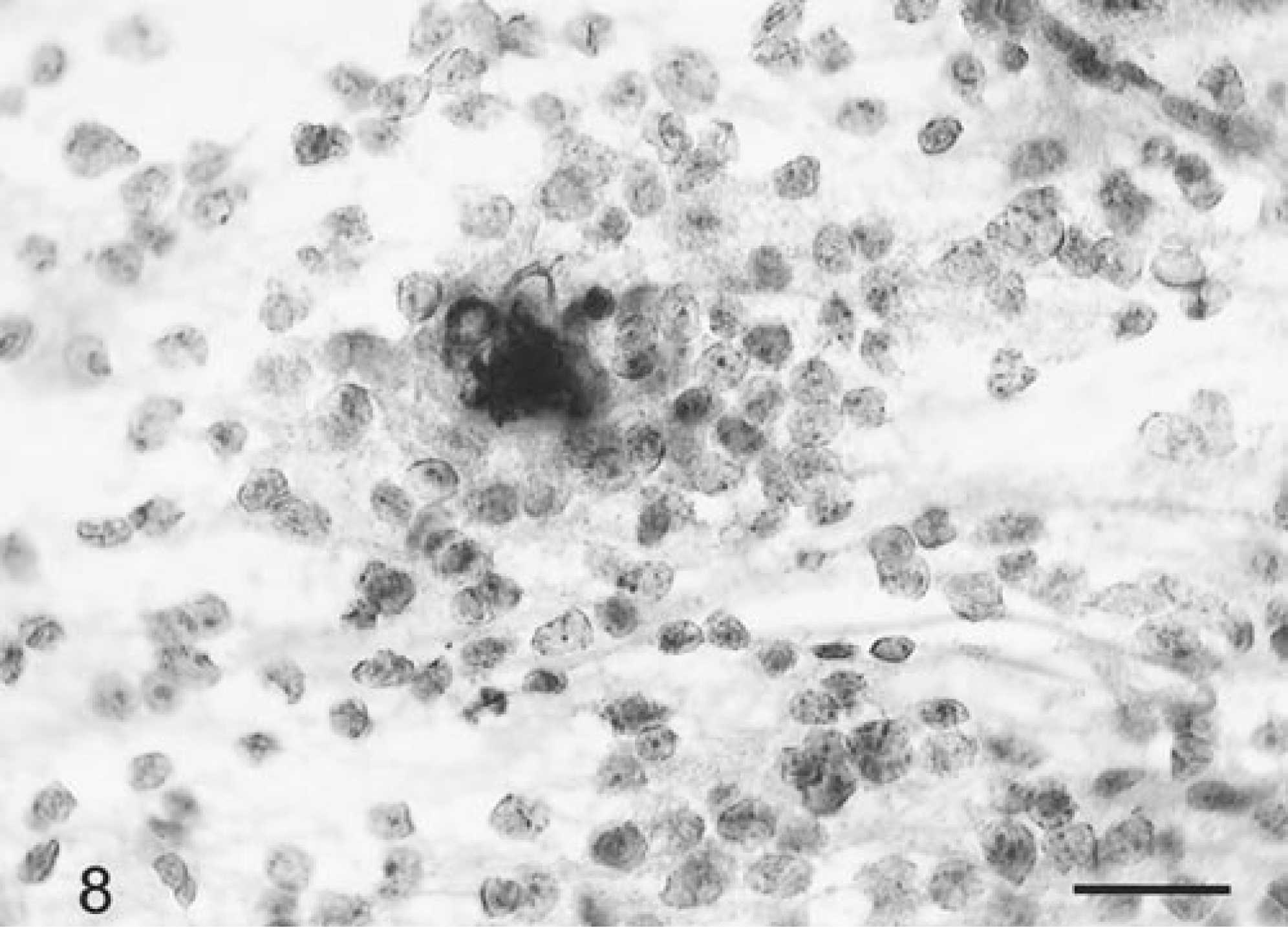
Brain; oligo-astrocytoma; canine. Focal area of calcification in an area of oligodendroglioma cell differentiation. HE. Bar = 22 µm.
Glioblastoma multiforme
The single canine glioblastoma multiforme had very prominent vascular glomeruloid formation compared to the better differentiated astrocytomas. Nuclear karyomegaly was also more prominent than in the astrocytomas, and there were up to three mitotic figures per 40× HPF. There were numerous necrotic cells and debris. Positive GFAP staining was seen in about 15% of all tumor cells.
Choroid plexus tumors
Five choroid plexus papillomas and one carcinoma, each occurring within a ventricular site in the six dogs, were examined. Grossly, all the tumors were soft and friable and spread easily with minimal pressure exerted. Cytologically, similarities with normal choroid plexus architecture were readily apparent (Fig. 9). At low power, the blood vessels were numerous and very prominent and formed multibranching papillary or medusoid-shaped fronds (Fig. 9). Between the vessels were large numbers of round to irregularly shaped cell clusters or sheets, as well as dispersed individual cells. Tumor cells were often arranged in both broad sheets of a single layer of epithelia-like cells (Fig. 10) or, less commonly, in distinctive radiating patterns similar to normal choroid plexus (Fig. 11). The vessels were very thickened and dense due both to a fibrovascular core and to variably thick layers of haphazardly but outwardly radiating, cuboidal- to oblong-shaped cells (Fig. 11). In these patterns, the cells were in a regular layer of columnar epithelium with basally oriented nuclei and prominent apical cytoplasm. The individually dispersed cells were small with round to ovoid, uniform-sized nuclei with one or more prominent nucleoli, finely granular chromatin, and a dark-staining nuclear border. The eosinophilic cytoplasm of the cell body was plentiful, round to oblong, sometimes bipolar, and elongate but did not form processes. The carcinoma had prominent anisokaryosis but otherwise was cytologically similar to the papillomas. Mitotic figures were not seen in any tumors. Some of the tumors had mineralized foci not to be confused with psammomatous bodies. None of the tumor cells stained positively for either L- or HMW cytokeratins or for GFAP, but cells in all cases were uniformly and strongly positive for vimentin.
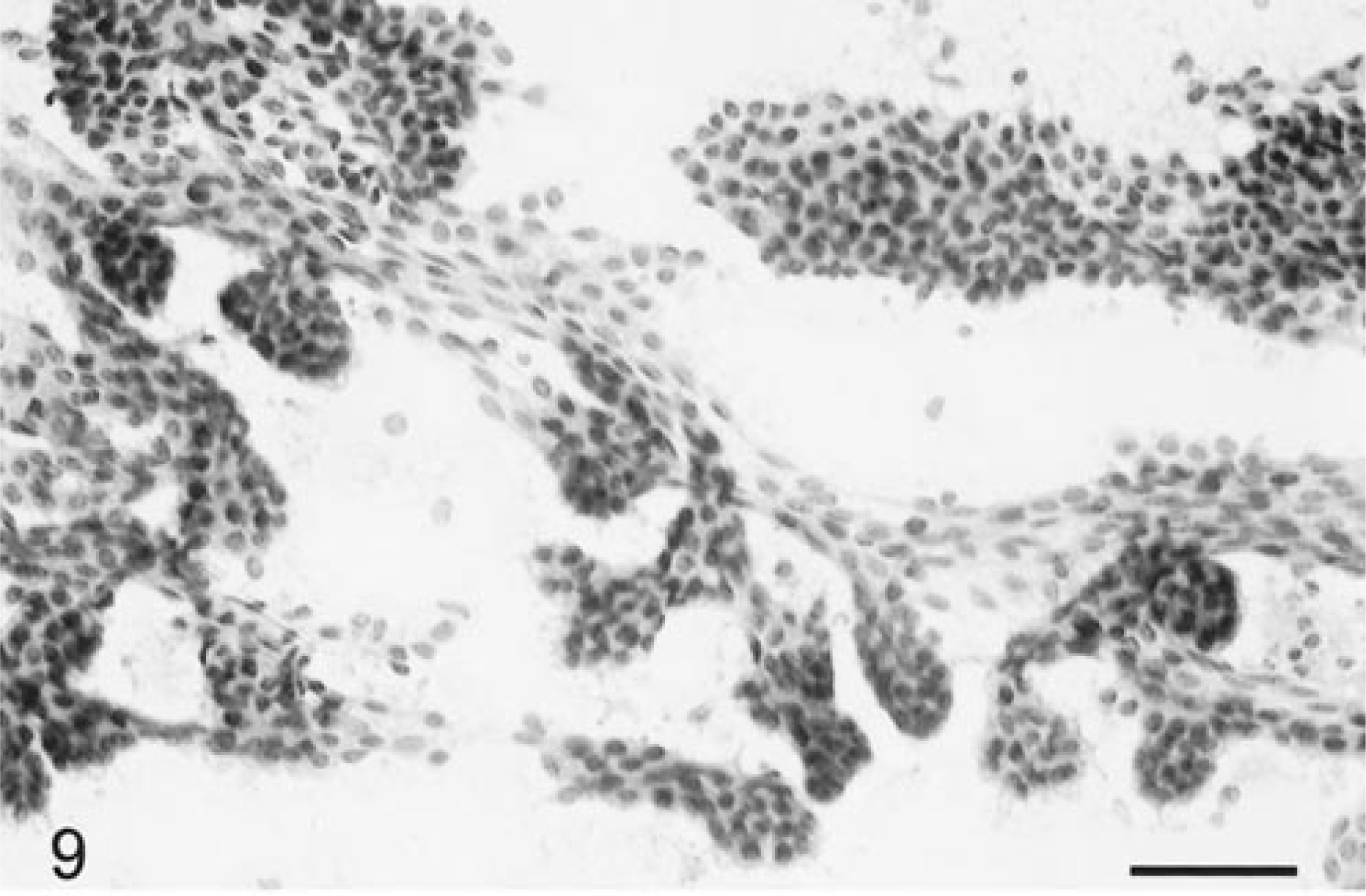
Brain; choroid plexus tumor; canine. Low magnification of the branching papilliform architecture resembling normal choroid plexus. HE. Bar = 70 µm.
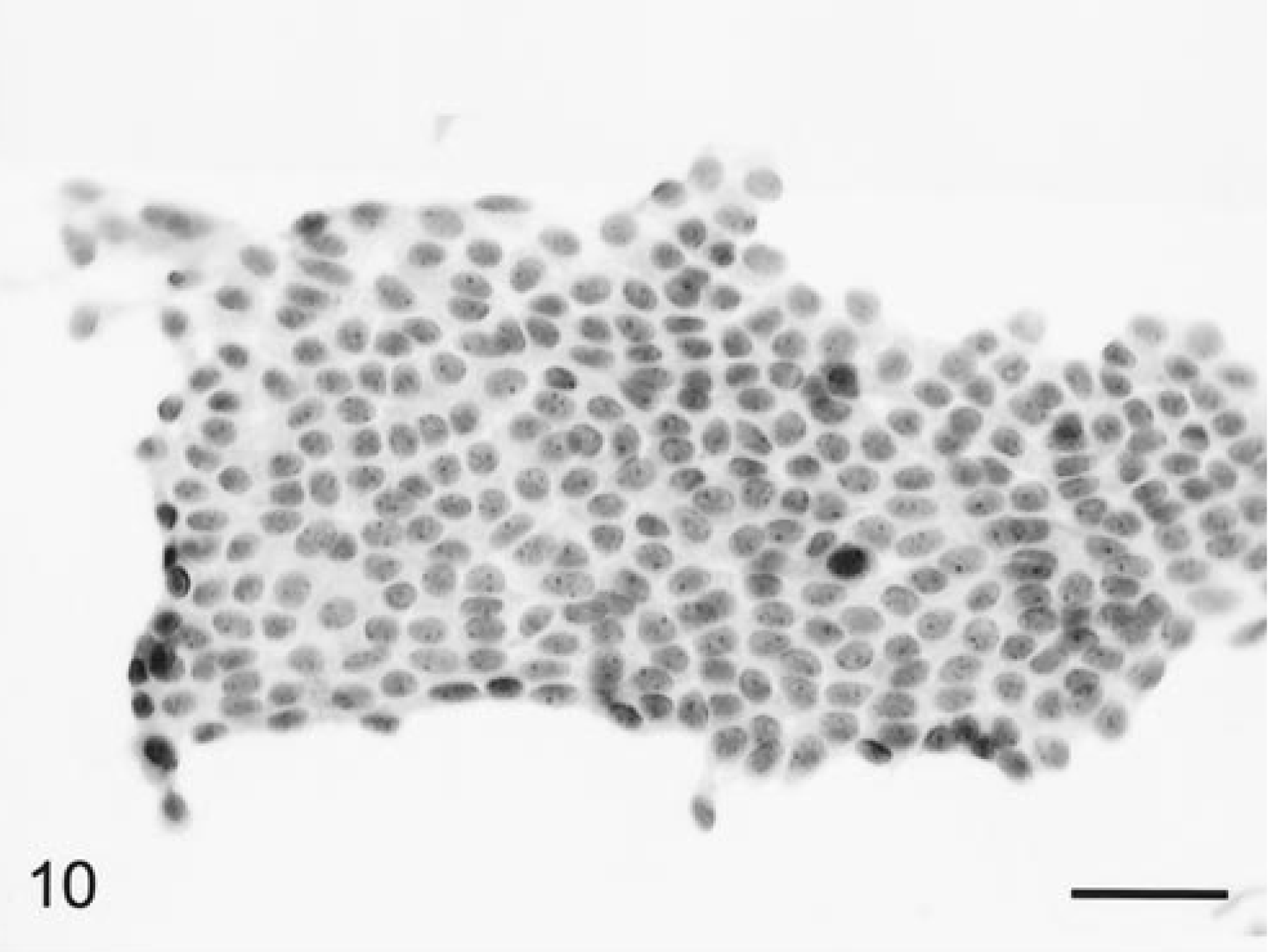
Brain; choroid plexus tumor; canine. Sheets of uniform-sized, epithelia-like cells. HE. Bar = 35 µm.
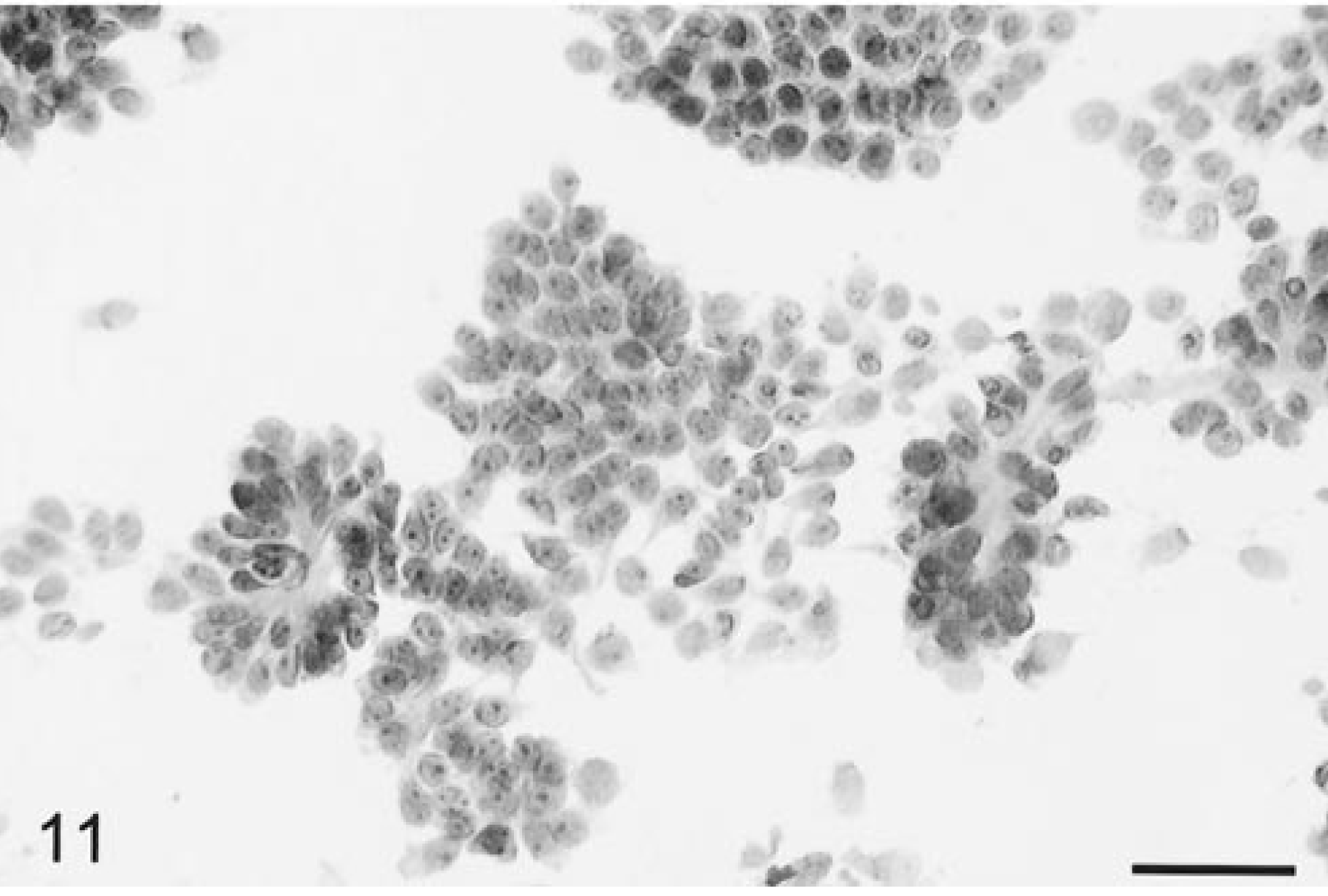
Brain; choroid plexus tumor; canine. Epithelial islands of cells and some cell clusters arranged around blood vessels. HE. Bar = 38 µm.
Ependymoma
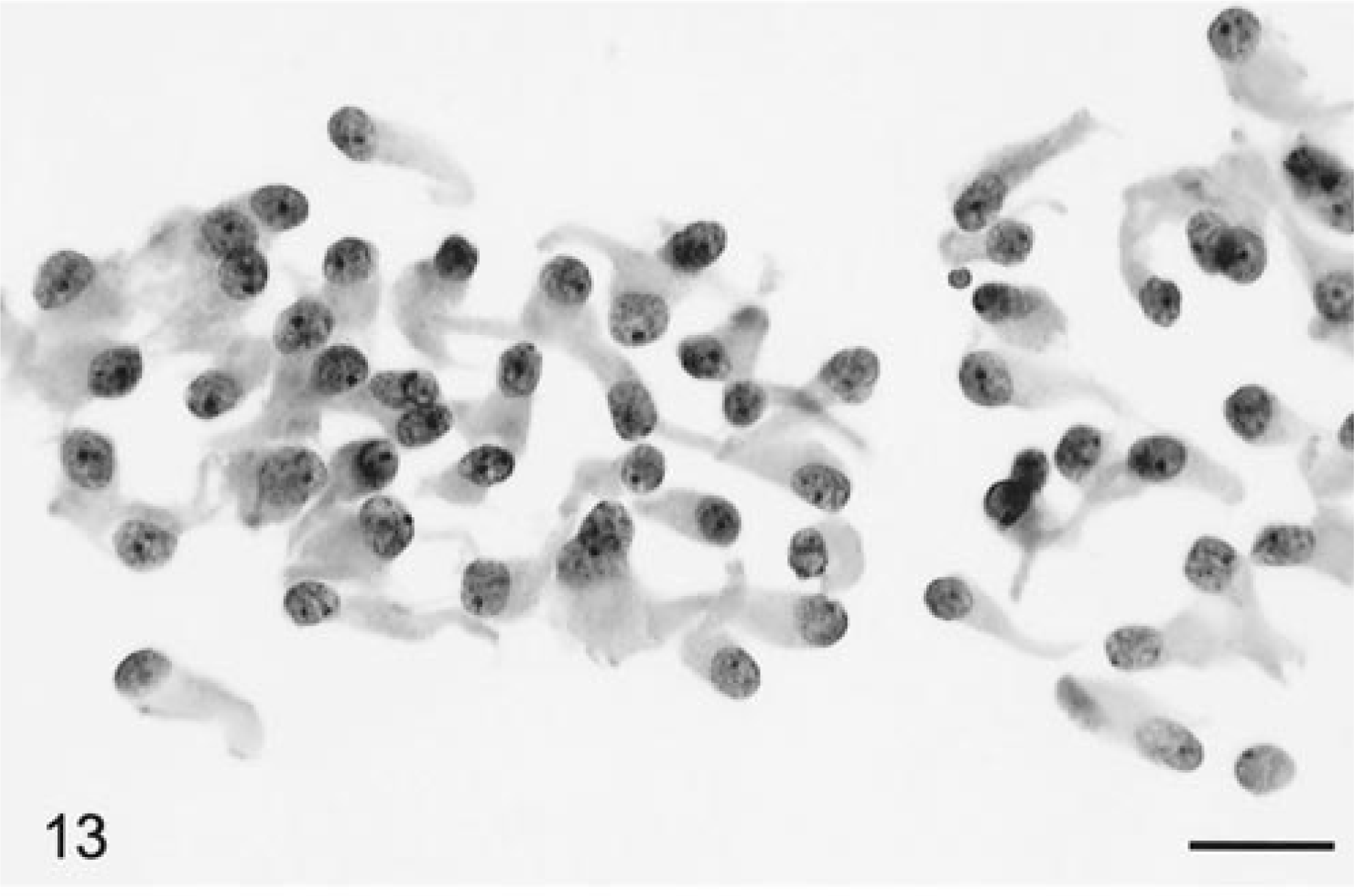
Two canine and one feline papillary ependymoma were evaluated. The tissue smeared easily. In all cases at low-power magnification, there were prominent thickened blood vessels, many of which had some branching. The blood vessels in longitudinal orientation were thickened due to perivascular palisading of layers of ependymal tumor cells (Fig. 12). The distinctly elongate cells were generally piled up several cells thick and radiating out from the vessels. Tumor cells were generally aligned perpendicularly to the vessel and with their nuclear pole oriented outward. Beside the perivascular pseudorosettes, there were sheets of single detached cells. On high-power magnification, these cells had nuclei that were round to ovoid, elongate, and sharp bordered and had a cribriform chromatin pattern with a small nucleolus. The cytoplasm was oblong, eosinophilic, and usually unipolar. Compared to the canine tumors, the feline ependymoma cells had more prominent unipolar eosinophilic cytoplasm with sharply defined borders (Fig. 13). Some perivascular cells had large karyomegalic nuclei with abundant cytoplasm (Fig. 14). Mitotic figures were very rare but necrotic cells were common. Nuclear and cytoplasmic atypia were common (Fig. 14).
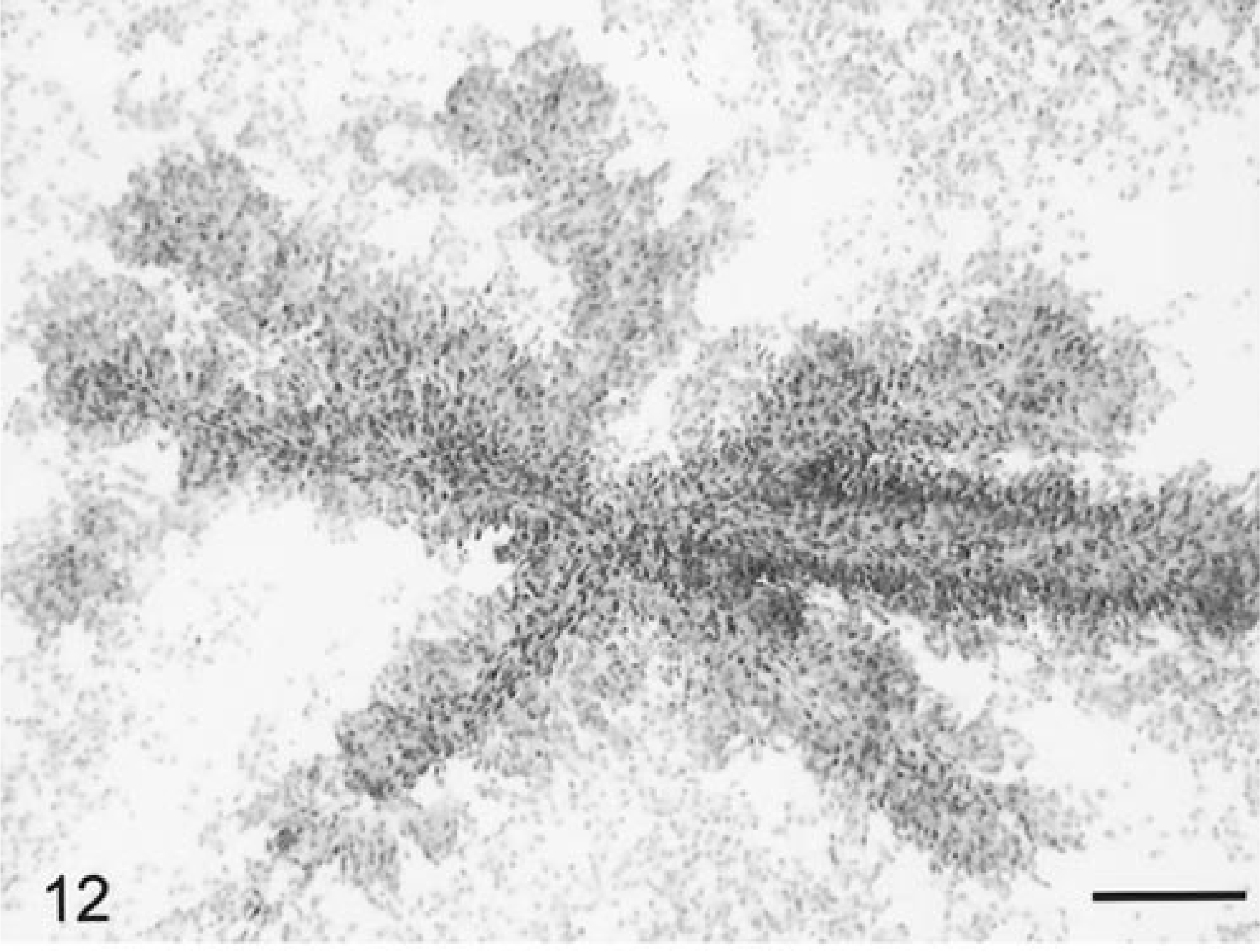
Brain; ependymoma; feline. The highly branched blood vessels are thickened and prominent due to layers of tumor cells closely attached and perpendicularly oriented to the vascular core. HE. Bar = 105 µm.
Brain; ependymoma; feline. Dispersed tumor cells with elongate, plentiful, unipolar distribution of solid-staining eosinophilic cytoplasm. Nuclei are round to slightly ovoid with a single prominent nucleolus. HE. Bar = 20 µm.

Brain; ependymoma; feline. Anaplastic ependymoma with focal nuclear and cytoplasmic atypia. There are also many individual smaller sized tumor cells with a polar nuclear location. HE. Bar = 24 µm.
Primary CNS lymphoma
These four tumors (three canine and one feline) were classed tentatively as “primary” tumors of the CNS based on the criterion of the absence of any other gross or microscopic involvement of lymphoma in any extraneural organs at necropsy examination. 4 Blood vessels were prominent and highly branched. Tumor cells were individually dispersed but tightly packed, monotonously round, and uniform in size and stained slightly basophilic, and some had minimal eosinophilic staining cytoplasm (Fig. 15). Mitotic figures were up to three per 40× HPF. At the margin of lesions, parenchymal invasion occurred from perivascular clusters of tumor cells. One canine and the feline lymphoma each stained positively immunocytochemically for the canine B-lymphocyte marker CD79α, whereas the other canine lymphoma stained positively with the T-lymphocyte receptor marker CD3.
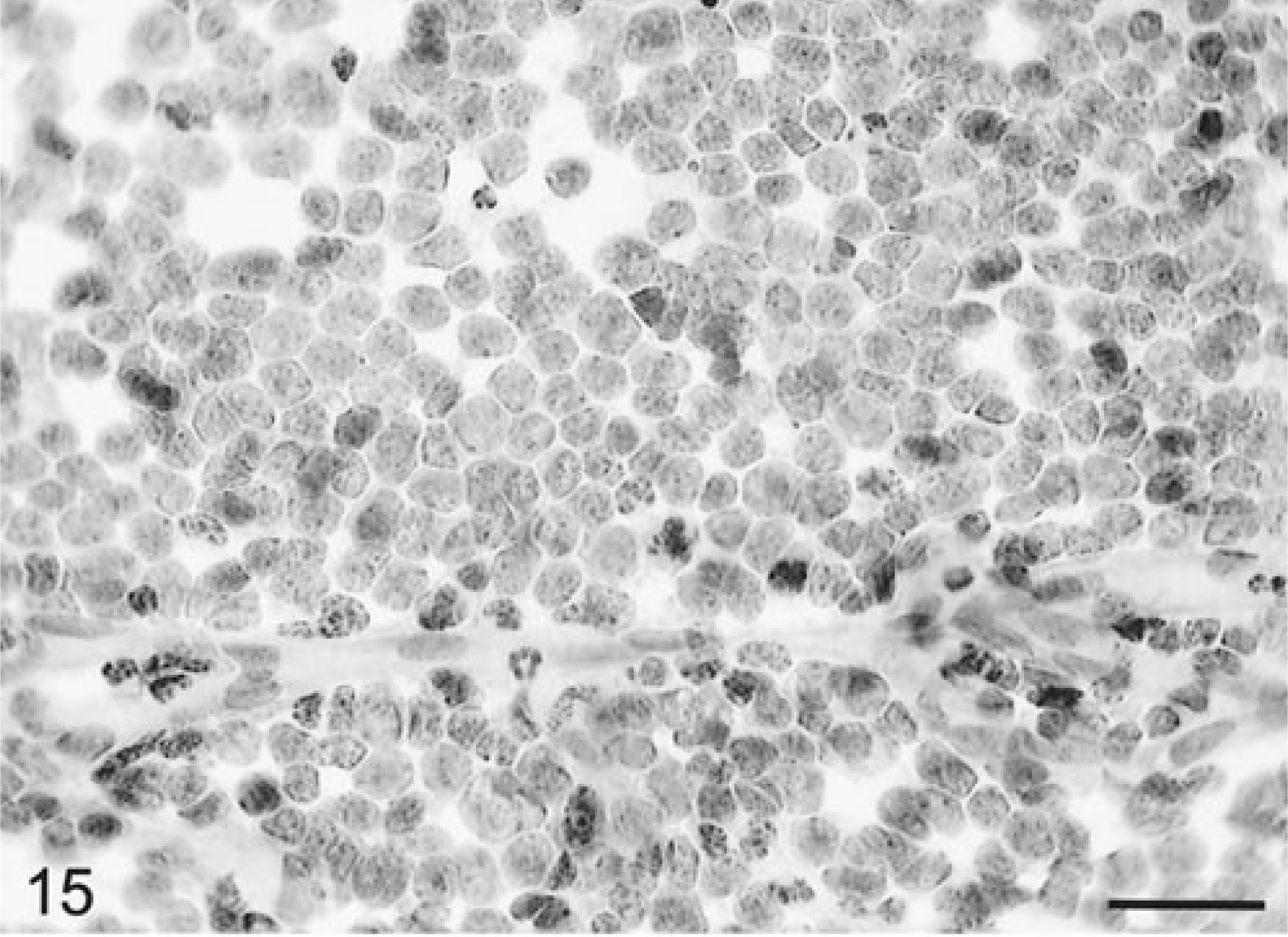
Brain; CNS lymphoma; canine. Thin-walled branching capillaries with sheets of closely packed cells with round, fairly uniform nuclei and minimal cytoplasm. There are single necrotic cells and several mitotic figures. HE. Bar = 25 µm.
Primary histiocytic sarcoma
Four canine and one feline primary CNS histiocytic sarcomas were identified. They all presented as a single lesion as visualized by MRI or CT. The criterion for their primary CNS status was the same as for the CNS lymphomas. 4 In the dog, the tissue was very cohesive and densely cellular, and most cells stayed closely compacted around blood vessels. However, there was some dispersion of individual cells. Cells were round but varied greatly in size (Fig. 16). Nuclei had one or two pinpoint prominent nucleoli and a sharp nuclear border with a variable amount of eosinophilic, granular cytoplasm. There were bi- or multinucleate giant cells whose nuclei piled up or overlapped (Fig. 16). Normal mitotic figures were numerous. One mass that readily smeared consisted of well-differentiated macrophages with less anaplasia and minimal giant cell formation and a low mitotic index. Some cells had phagocytosed nuclear debris.
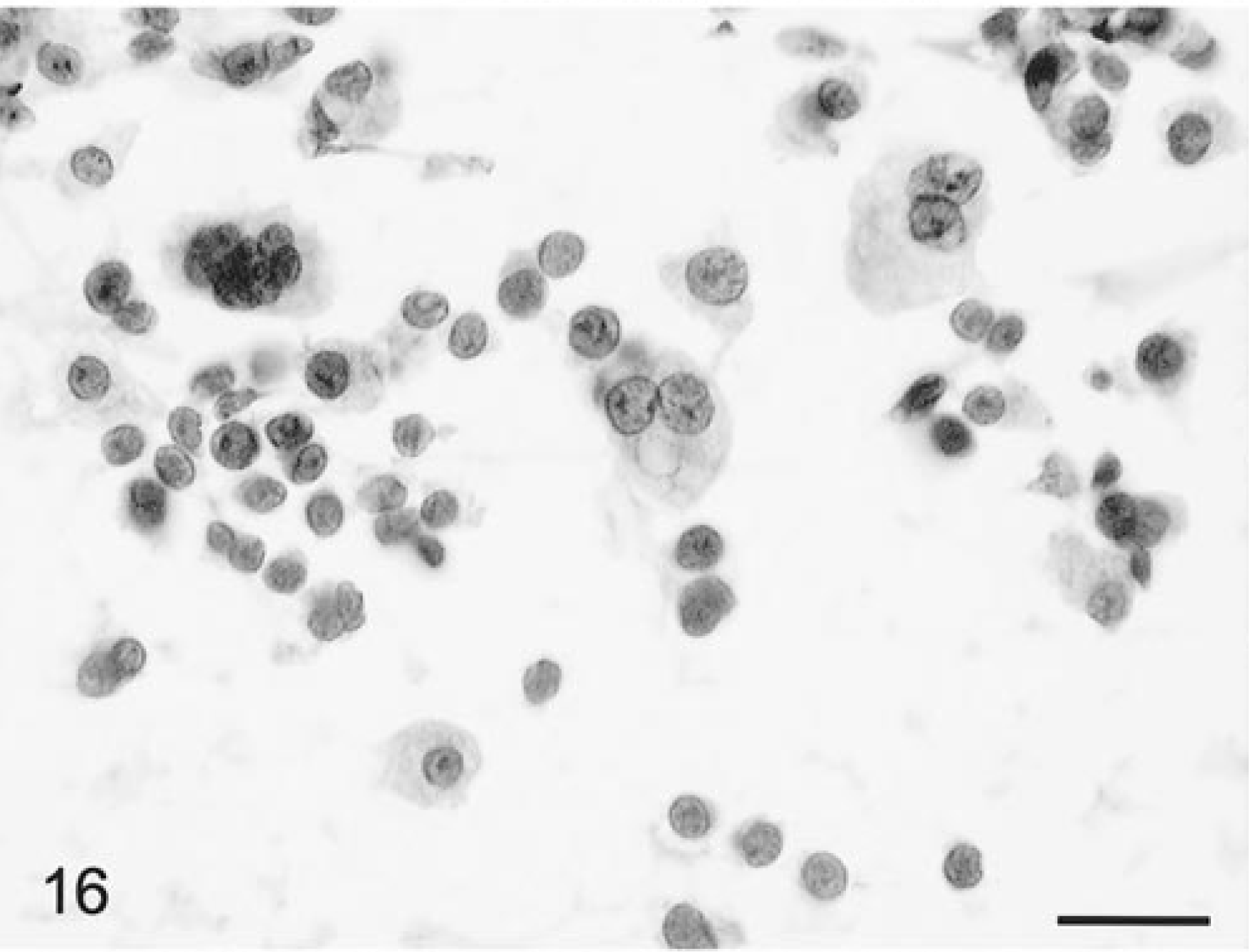
Brain; primary histiocytic sarcoma; canine. There are aggregates of macrophage-like cells with a usually round nucleus, with plentiful well-defined cytoplasm and some bi- and multinucleated cells. HE. Bar = 26 µm.
In the cat, sheets of cells were evenly dispersed and had extreme pleomorphism and anaplasia. The predominant cells had generally round to slightly elongate nuclei with dense, uniformly dispersed chromatin with sometimes a prominent nucleolus. Occasional cells had bean-shaped, indented nuclei. Some cells had giant karyomegalic nuclei, and other giant cells contained from two to multiple densely packed crenellated nuclei. Both single and multinucleated cells had phagocytosed nuclear debris. Normal and bizarrely shaped mitotic figures were very common. There was an admixture of some neutrophils and necrotic cells. The blood vessels were unremarkable and cells were readily detached from their walls. Canine tumor cells were invariably positive for CD45RA and most had strong cytoplasmic membrane staining for both CD11d and CD18.
Primitive Neuroectodermal Tumor (PNET)
The single canine infratentorial medulloblastoma was cohesive and thus difficult to smear. At low magnification, the cells remained in relatively large clusters closely adhered to blood vessels (Fig. 17). The cells were closely packed, with a large round to oblong, strongly basophilic staining nucleus and had an eccentric rim of scant and finely granular cytoplasm. Their nuclei had a sharp border and one or two fine, pinpoint nucleoli. Homer–Wright rosettes were sometimes found (Fig. 17). Thin granular, nonrefractile processes sometimes ran between cells. Cells were rarely separately dispersed and were embedded in a finely granular, eosinophilic, wispy matrix. Immunostaining of smears demonstrated that some cells had strongly positive cytoplasmic staining for SYN and TNF and also of their processes with the latter. There was also positive GFAP staining of a few interspersed cells.

Brain; medulloblastoma; canine. Cells are densely packed; nuclei elongate to ovoid and deeply basophilic staining, Homer–Wright rosette formation (arrow). HE. Bar = 19 µm.
A single canine supratentorial PNET was evaluated. The tumor smeared easily and had blood vessels that were very prominent with their accentuated branching. The densely packed basophilic staining cells formed clusters and had irregular to elongate nuclei, finely granular chromatin, and minimal cytoplasm. There was a background gliofibrillary meshwork in some areas. The cells seemed to infiltrate from multiple clusters around blood vessels into the adjacent neuropil.
Meningioma
Smear preparations of meningiomas from 44 dogs and seven cats were evaluated. Although the feline meningiomas were histologically similar, in the canine tumors there was a wide diversity of histologic subtypes, including meningothelial, transitional, secretory, and psammomatous patterns. 4 With the exception of the meningothelial subtype, however, there were generally mixed patterns of these histologic subtypes throughout the tumors, and rarely did any one pattern occur in pure form. Generally, tumor tissue smeared readily except from some of the transitional subtypes, which were more cohesive and difficult to spread. At low magnification, tumor cells were broken up into many clusters or cohesive cell aggregates, as well as separate into individual cells (Fig. 18). Generally, blood vessels were not prominent and tumor cells usually were not closely adherent to their walls. A few tumors did have very thickened walls with multiple branching blood vessels. Meningioma cells had round to slightly elongate, uniform-sized nuclei with a small prominent nucleolus with diffuse punctate chromatin and a well-defined nuclear border (Figs. 19, 20). Rarely there were intranuclear cytoplasmic evaginations, but these were plentiful in some individual tumors of the meningothelial subtype (Fig. 20). More elongate cells sometimes had a central bar or fold through the longitudinal axis of their nucleus (Fig. 20). The eosinophilic, granular, wispy to solid cytoplasm varied in amount, was round to elongate when present, and often had a polar location. Surprisingly, the tumor cells had very similar and distinctive nuclear morphology despite differences in their histologic subtypes. Mitotic figures were extremely rare. Some tumors had marked cellular anaplasia or nuclear atypia. Transitional subtypes always had distinctive whorls (Fig. 19); the meningothelial types broke up into distinctive cell clusters, and the psammomatous subtypes had multiple psammoma bodies (Fig. 21). Neutrophils were usually found in those tumors that had histologic foci of necrosis and/or neutrophil accumulation (Fig. 22). Tumor cells were generally strongly and uniformly positively staining for vimentin, and clusters of cells in some tumors were variably positive for L- and HMW cytokeratins. The feline tumors smeared less readily and tended to have more clumping. Cells were more elongate, and there were often linear patterns of tissue calcification and necrosis.

Brain; meningioma; canine. These tumors, irrespective of histologic subtype, tend to disperse into clumps associated with vessels, though there is some individual dispersion. HE. Bar = 83 µm.
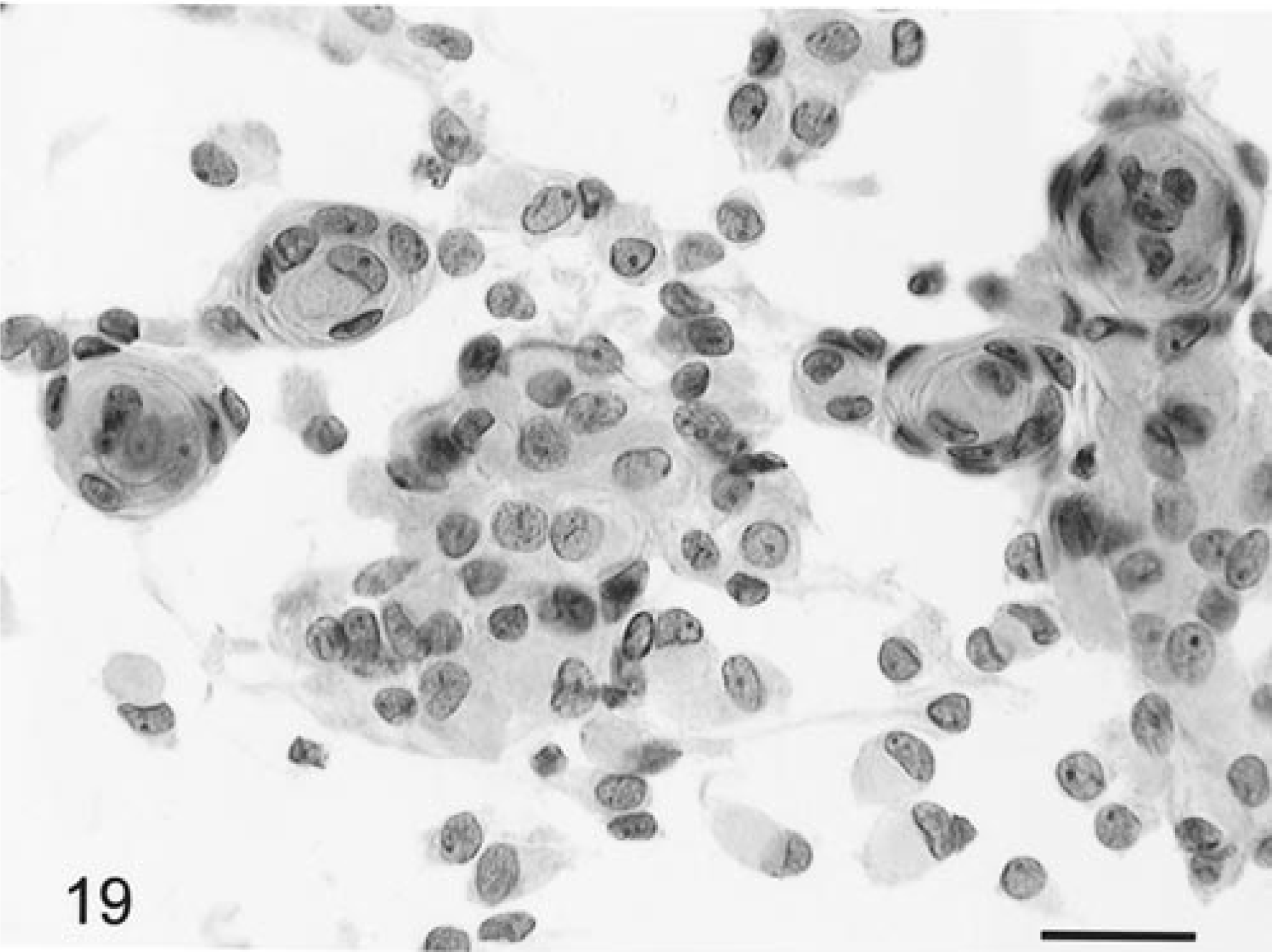
Brain; meningioma, transitional subtype; canine. There are islands of cells as well as the distinctive whorl formation. Nuclei are round, ovoid, or irregular with a prominent nucleolus and with abundant deep-staining cytoplasm. HE. Bar = 14 µm.

Brain; meningioma; syncytial subtype; canine. Cells have a more wispy cytoplasm. There is longitudinal folding within some nuclei. Several cells have an intranuclear cytoplasmic evagination. HE. Bar = 10 µm.
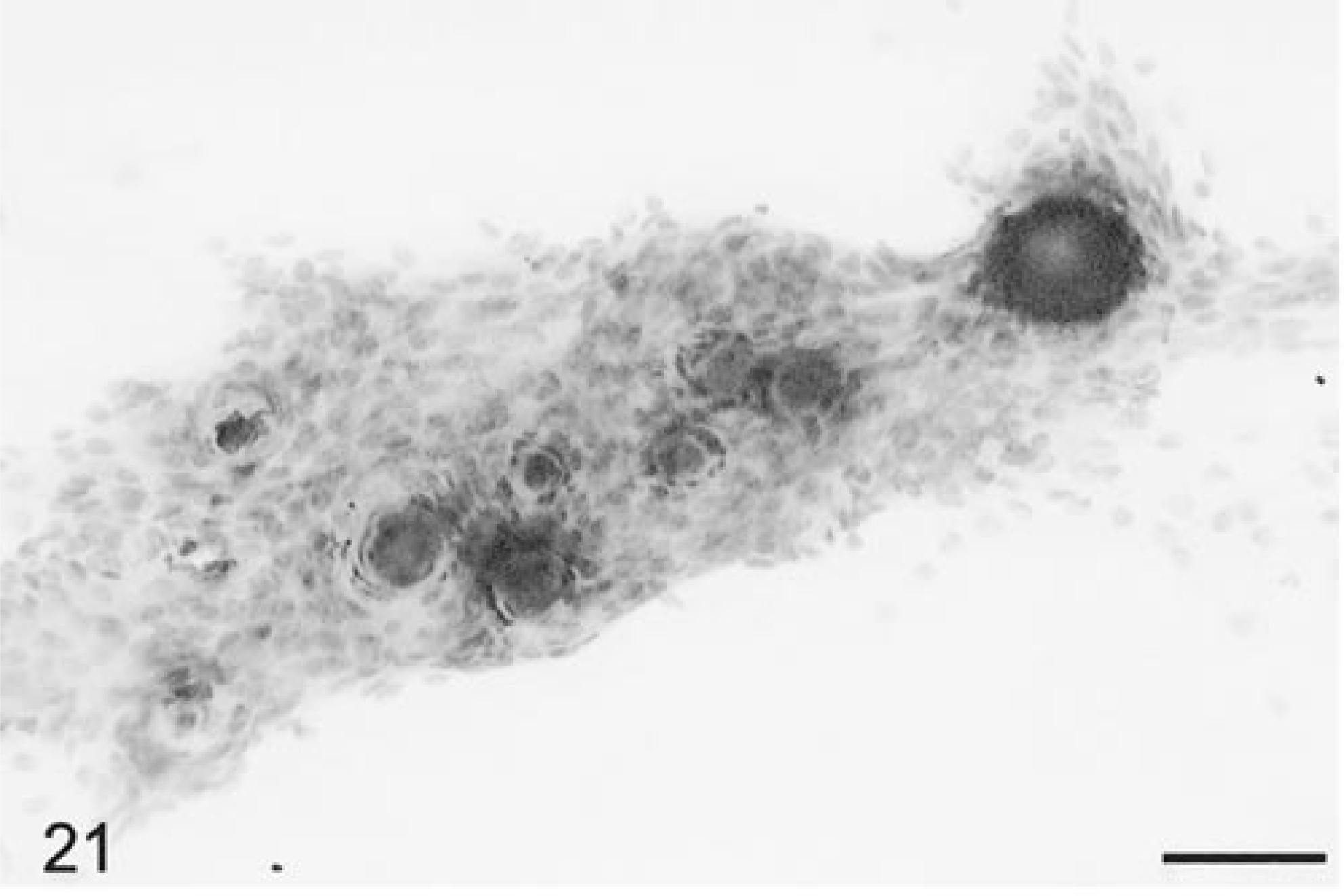
Brain; meningioma, psammomatous type; canine. Densely packed cluster of cells with numerous psammomatous bodies. HE. Bar = 60 µm.
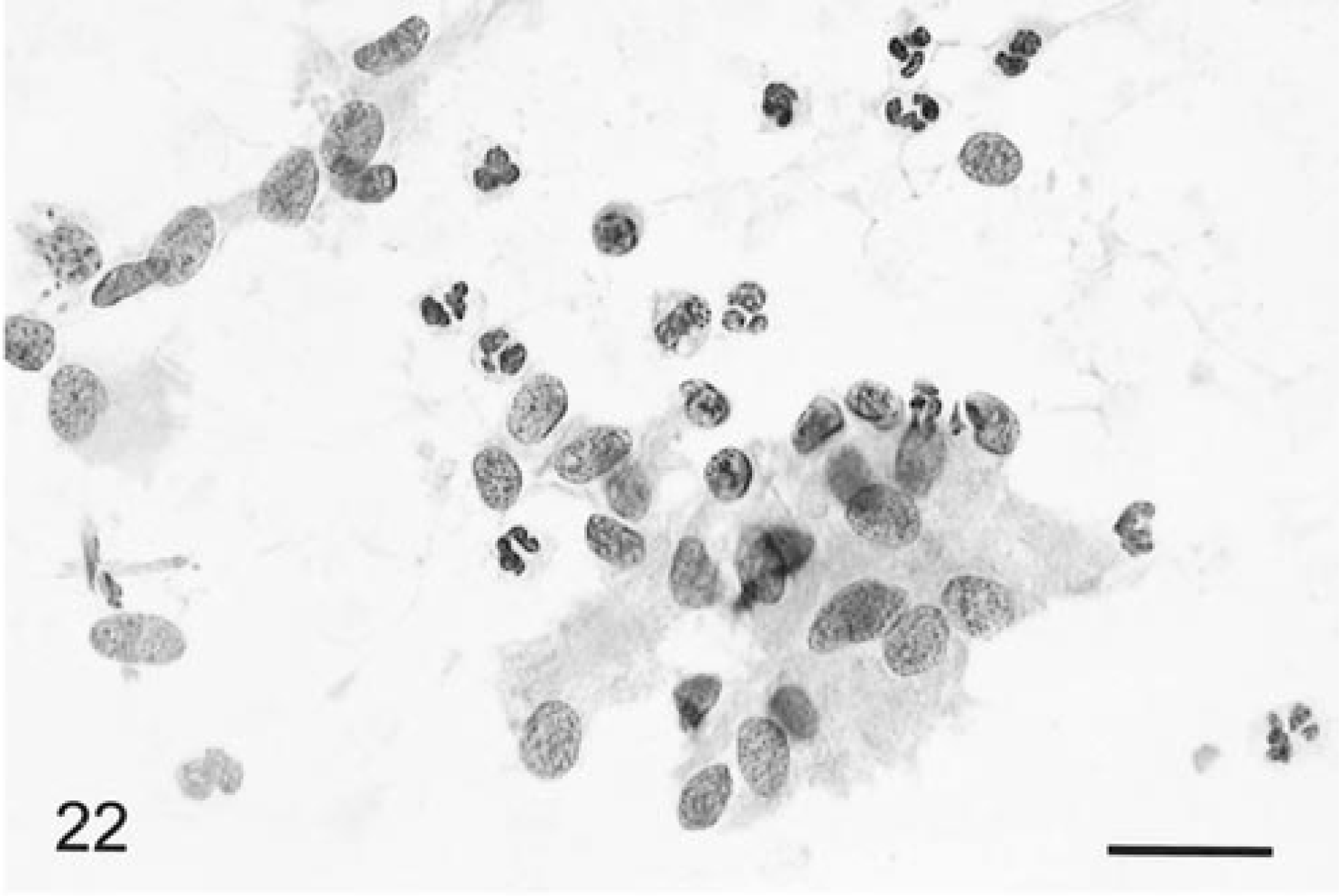
Brain; meningioma, meningothelial subtype; canine. An aggregate of tumor cells with numerous intermingled neutrophils. The histologic specimen had multiple islands of necrosis and also foci of neutrophil accumulation. HE. Bar = 16 µm.
Granular cell tumor
This tumor spread relatively easily despite a granular cohesive gross texture. The cells were readily dispersed into sheets of individual cells, although the unevenly sized cells tended to form groups. There were also clumps of cells aggregated around innocuous blood vessels. The cells had an eccentrically and often peripherally located round nucleus, which had a single prominent nucleolus and sharp nuclear border (Fig. 23). Occasional binucleate cells were found. The abundant cytoplasm was comprised of densely packed, refractile, strongly eosinophilic staining granules and bounded by a round sharp cytoplasmic membrane (Fig. 23 insert). There was marked variation in the amount of cytoplasm within cells. Some nuclei had cytoplasmic evaginations. There were also occasional neutrophils and mononuclear cells. Cells were negative for S-100 α- and β-subunits, GFAP, CD45RA, and the various canine macrophage antigen markers.
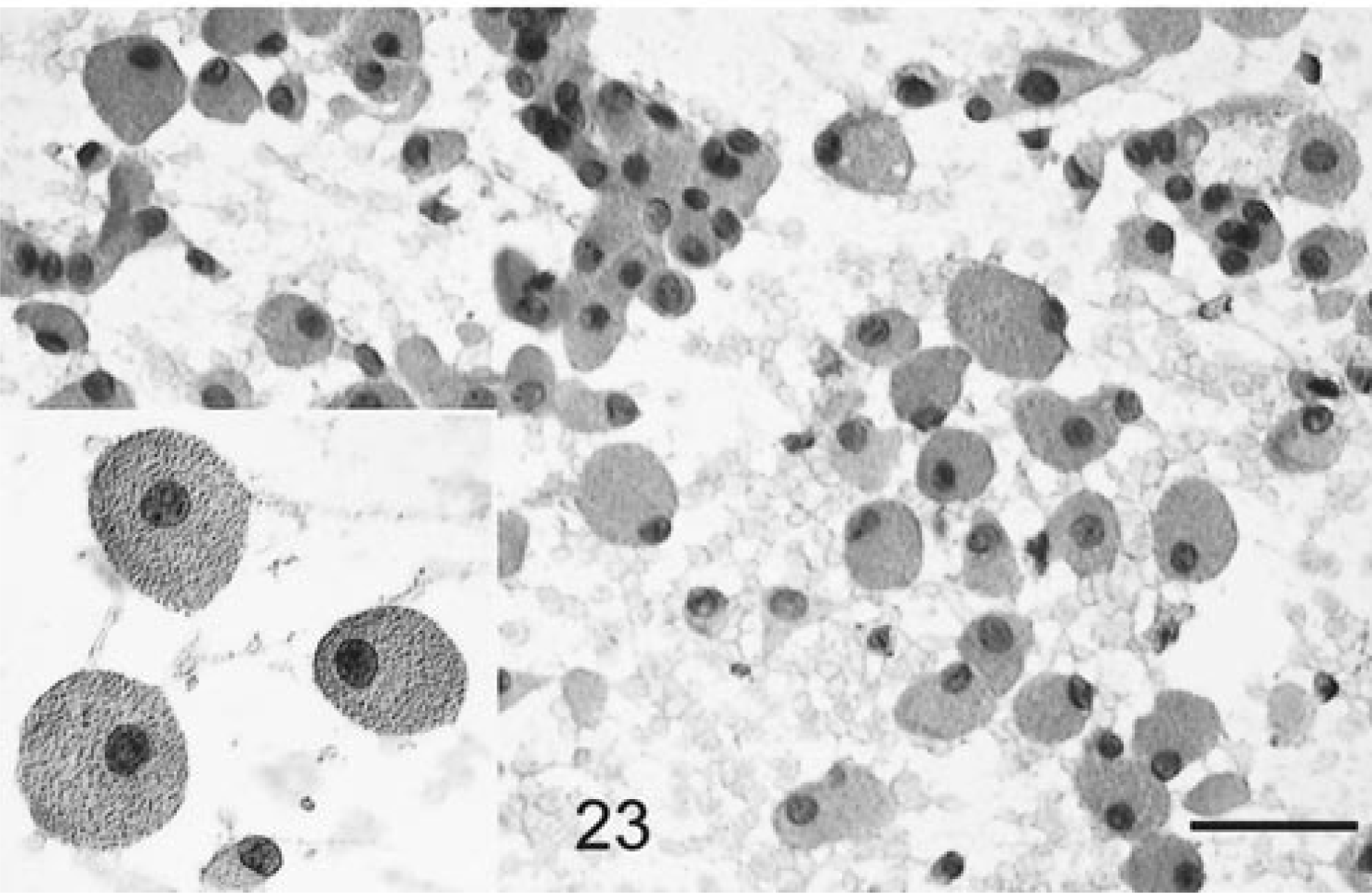
Brain; granular cell tumor, canine. Large, individually dispersed or clumped round cells with an eccentric nucleus, single nucleolus, and abundant granular cytoplasm. HE. Bar = 21 µm. Insert: Distinctive cytoplasm packed with large eosinophilic granules.
Schwannoma
Two canine schwannomas were identified. The tissue samples were both very firm and extremely difficult to smear. At low-power examination, the tissue was very cellular, did not disperse on the slide, and remained as a dense, cohesive cellular mass with ragged margins. There was inevitably some nuclear streaming reflecting the crushing artifact from the pressure applied to produce a smear. The cells were very dense and uniformly spindle shaped (Fig. 24). On high-power magnification, the nuclei were round to ovoid with fine, granular chromatin evenly dispersed throughout the nucleus. Nucleoli were small or inapparent. The cytoplasm was eosinophilic and granular without defined borders. A few myelinated axons emanated from the periphery of the tissue mass (Fig. 24).

Trigeminal nerve; schwannoma; canine. The cells are extremely cohesive, but at the edges of the tissue tumor, cells are elongate, closely packed, with no distinctive cytoplasm. Several myelinated nerve fibers are detected emanating from within the tumor (arrows). HE. Bar = 25 µm.
Discussion
The results of this study suggest that smear preparations of the various major categories of brain tumors each have distinctive cytologic profiles and that this technique, as with human CNS tumors, can provide an accurate, rapid intraoperative diagnosis. The distinguishing cytologic features of the canine and feline nervous system tumors examined bear a close resemblance to their human counterparts. Although the specificity, sensitivity, and diagnostic accuracy of this procedure were not addressed in this report, 10 the overall diagnostic accuracy from samples obtained by both craniotomy and stereobiopsy was about 80%. This already compares very favorably with the 69–94% accuracy claimed in some large human series. 1 9 11 14 20 21
The major advantages of this system for intraoperative diagnosis are the speed, the ease of preparation, the technical simplicity and need for minimal technical equipment, the high degree of cytologic resolution compared to frozen preparations, the low cost, and the very small sample size required. 9 15 Limitations of the system are that some tumors are so tough and coherent (e.g., schwannomas, fibrillary astrocytomas, and some meningiomas) that adequate smear preparations are difficult to make and can lead to obscuring iatrogenic, mechanically induced crush artifacts. In such instances, a definitive diagnosis may be deferred until stained paraffin-embedded sections are available. Second, the smear preparations give excellent cytologic detail, but these differ markedly from the familiar and conventional histologic appearance of HE-stained paraffin-embedded tissue. Considerable experience is thus required for their correct interpretation, as well as an intimate knowledge of appearance of normal brain tissue in different sites (e.g., from cerebellar or cerebral cortex or white matter). Third, there are sampling errors inherent in the small size of the sample, which can be further compounded by the stereotactic needle placement with respect to the lesion site. Tissue from necrotic areas within tumors or adjacent to the tumor can lead to an erroneous or nondiagnostic interpretation. In all cases, the neurological findings and the nature and type of radiological changes must be considered in coming to a final diagnosis. Evidence from human studies indicates that smear cytology is a useful, safe, and reliable technique for an intraoperative diagnosis, as long as the limitations of the technique are clearly understood by both the neurosurgeon and neuropathologist. 15 Optimal application of this technique requires close collaboration between the radiologist, neurosurgeon, and neuropathologist. 9 15 19
A number of pitfalls await the inexperienced neuropathologist, and most come under the category of reactive changes at the periphery of a tumor. With limited experience, there is usually a tendency to overinterpret lesions. The most common nonspecific reaction is reactive astrocytosis, and this change must be differentiated from low-grade astrocytomas. Such astrocytosis can occur at the periphery of neoplastic, inflammatory, degenerative, or vascular lesions. Reactive astrocytosis can become a major diagnostic problem, particularly if there is a perilesional placement of the stereotactic biopsy site. 4 17 19 If a smear has both fibrillary and gemistocytic astrocytes and is not hypercellular, it is more likely to be reactive. Inflammation and macrophages are unlikely with a tumor, and necrosis does not always suggest a tumor. There must also be consideration of the site from which the tissue was removed (e.g., white or grey matter, cerebellar cortex, etc.) since each has different cytologic architecture. 4 9 15 17
The most common artifact is from air drying, arising when the slide is not immediately immersed in alcohol after smearing and resulting in poorly stained, swollen, or distorted nuclei. A common error is to examine the quality of the smear preparation prior to fixing in alcohol; this only enhances the air-drying artifact. Crush artifacts with cell distortion and nuclear streaming occur with inappropriate pressure applied for the particular tumor type. An appropriately small tissue sample must be selected, otherwise the cell density will be too thick and the smear inadequate for cytologic detail.
The differential diagnosis of each of these tumor categories presents different problems of interpretation. The low-grade astrocytomas must be differentiated from reactive fibrillary and/or gemistocytic astrocytosis as described above. Staining positivity for GFAP, however, was not consistent in our smear preparations but was much more uniform on the formalin-fixed, paraffin-embedded tissue, suggesting the difference was due to differing sensitivity of the antibody to alcohol or formalin fixation. Oligodendrogliomas can be distinguished from astrocytomas by the cell processes, the refractile fibrillary cell processes, and the gliofibrillary matrix of the latter. Oligo-astrocytomas present a greater challenge, and there is an admixture of both cell types. Canine oligodendrogliomas also have consistent and very distinctive microvascular cell proliferation, forming glomeruloid tufts. With papillary ependymomas, the differential diagnosis includes a choroid plexus tumor. The former tend to have a gliovascular core with small lumenally oriented cells distinct from the sheets of epithelial cells and epithelial cells arranged around branching frond-like collagenous vascular cores and rosette-like epithelial islands in the latter. Also, smears of ependymomas have had strong positive uniform staining for GFAP. Psammomatous and transitional meningiomas could be readily distinguished by psammoma bodies and the classical concentric cell whorl formation, respectively. However, irrespective of the meningioma histologic subtype, the nuclear morphology remained distinctive and consistent in most of these tumors. Some meningiomas had a few intranuclear cytoplasmic evaginations, but they were not as consistent or as frequent as in human meningiomas. 4 The PNETs had to be distinguished from primary lymphomas, and the diagnosis was based mainly on the pleomorphic nuclei in the former, together with their mode of cell clustering and pattern of neuropil invasion. Positive immunocytochemical staining for either T- or B-lymphocyte markers on the canine and feline lymphomas and neuronal differentiation in some cells on the PNETs as detected by immunostaining was also helpful. 4 16
Footnotes
Acknowledgements
We thank Mr. S. Maslowski and Ms. D. K. Naydan for their superb technical skills with the neuroimaging and immunocytochemistry, respectively; Drs. W. Berry, P. J. Dickinson, G. D. Kortz, and B. K. Sturges for their clinical support of the cases; and Dr. W. Vernau for his helpful comments. This work was supported in part by a grant from the Paul and Borgie Petersen Foundation.
