Abstract
In an attempt to establish a primate model of chronic cadmium toxicosis, we ovariectomized cynomolgus monkeys and treated them with CdCl2 by repeated intravenous injections for 13 to 15 months. The animals showed normocytic-normochromic anemia. The cadmium treatment resulted in increases of urinary enzyme activity indicative of renal tubular degeneration. Histopathology of the kidney revealed renal proximal tubular atrophy accompanied by interstitial fibrosis. Decreased bone mineral density was evident in the trabecular and cortical zones of the lumbar vertebra and femur, with osteoid accumulation around the trabeculae and Haversian canals. Iron deposition at the mineralization front and osteoclasts hyperplasia were indicative of impairment of bone mineralization and an increase of resorption. Blood inorganic phosphorus and 1α,25(OH)2 vitamin D3 levels decreased and urinary deoxypyridinoline level increased in cadmium-treated animals. The renal and bone lesions closely resemble those of itai-itai disease patients, the most severe case of cadmium toxicosis in terms of clinical chemistry and histopathology. Thus, ovariectomized monkeys chronically exposed to cadmium can serve as a primate itai-itai disease model, which is beneficial for developing novel therapeutic methods, investigating the mechanisms of the renal and bone lesions, and establishing more clearly defined criteria for diagnosing the disease.
Keywords
Cadmium (Cd) is a ubiquitous environmental pollutant and may be toxic to the kidney and bone. 9,17 Recent studies on human exposure to the metal in industrial settings or through dietary intake and smoking in many countries have shown strong correlation with high Cd exposure in the incidences and severity of renal dysfunction and osteoporosis, not only in Cd workers and inhabitants in heavily polluted areas, but also in the general population of many countries, including Japan, Sweden, Belgium, Poland, China, and others. 1, 4 –6,13,17 –20,37,38,40
Itai-itai disease (IID; “ouch-ouch disease” in English) is one of the most morbid examples of chronic intoxication with Cd and is characterized with clinical disorders such as renal anemia, tubular nephropathy, and osteomalacic osteopenia. 32, 44,45 IID was officially designated as an environmental pollution-caused disease by the Japanese government in 1968. Since then, a total of 187 inhabitants in the area, polluted with Cd discharged from a zinc mine, have officially been recognized as IID patients, and 90% of them are postmenopausal women. 23 This prompted us to investigate Cd intoxication in ovariectomized animals to mimic the status of bone metabolism in these patients. We previously developed a murine model that resembles renal anemia, tubular nephrosis, and osteomalacic osteopenia reported in IID patients. 14, 15,24,25,30
The purpose of this study is to produce a primate model of IID. A primate model is valuable especially for elucidating the mechanism of bone lesions because it is similar to human bone metabolism. 22, 41 In our previous study with rats, 14,15,24,25,30 we performed ovariectomization (OVX) on the animals before the Cd treatment to put their bone metabolism in a postmenopausal-like condition. Furthermore, we adopted the intravenous route, rather than oral, for the Cd administration to reproduce Cd toxicosis in a short-term experiment, since Cd intake by gastrointestinal absorption in monkeys was expected to be extremely low, given that the absorption rate was 5% to 7% in humans. 11 These procedures were also applied in this experiment. In this report, we describe chronic Cd toxicosis developed in nonhuman primates (cynomolgus monkeys), which closely resembles the pathologic changes reported in IID patients.
Materials and Methods
Animals and Treatment
Ten female cynomolgus monkeys (Indonesian origin, more than 5 years old) were purchased from CLEA Japan (Kanagawa, Japan). The animals were quarantined and acclimated for about 6 months, during which they showed no abnormalities in their general health conditions. Thus, these animals were subjected to bilateral OVX under pentobarbital anesthesia.
The animals were housed individually in cages throughout the experimental period. The environmental conditions of the animal room were maintained as follows: temperature at 24°C to 28°C, relative humidity at 40% to 70%, and lighting at 12 hours. The animals were provided with 100 g of normal primate diet everyday (CMK-1, CLEA Japan) with extras such as bananas and were allowed free access to drinking water. For drinking water, tap water was passed through a 5-μm filter and irradiated with ultraviolet light. The animals were maintained according to the care and use of laboratory animals specified by the Japanese Association for Laboratory Animal Science and our institution.
Experimental Design
One month after OVX, animals were randomly assigned to the following 3 groups to give practically equal mean body weights among groups: control group (n = 4); CdCl2 group, 1.0 mg/kg (n = 3); and CdCl2 group, 2.5 mg/kg (n = 2). We assigned 4 monkeys to the control group to obtain as much background for bone morphometry as possible. Moreover, we assigned 3 monkeys to the 2.5-mg/kg group; however, 1 of them was lost by accident early in the experiment. At the initiation of Cd treatment, body weights ranged from 3.14 to 3.70 kg. CdCl2 (purity > 95%, Wako Pure Chemical Industries Ltd, Osaka, Japan) was dissolved in saline and administered intravenously at a constant dosing volume of 0.1 ml/kg via the tail vein, 3 times a week for 15 months, except for 1 animal (2.5-mg/kg group), which was sacrificed in moribund condition at month 13. The actual dosing volumes for individual animals were calculated from their body weights, determined on a weekly basis. The animals of the 2.5-mg/kg group, however, showed severe anemia in month 9; therefore, the dosing frequency was reduced to twice a week for the remainder of the treatment period.
Hematologic and Blood Chemical Examination
Blood samples were obtained from the femoral vein at the baseline and at 1-month intervals to determine changes in hematology, blood chemistry, bone metabolism markers, and vitamin D3 metabolites during the Cd treatment period. The animals were fasted 15 to 17 hours before each blood sampling.
Red blood cell count, hematocrit value, hemoglobin concentration, mean corpuscular volume, mean corpuscular hemoglobin, and mean corpuscular hemoglobin concentration were measured with an automated hematology analyzer (NE-4500, Toa Medical Electronics Co, Ltd, Tokyo, Japan) using EDTA-treated blood samples. Calcium and inorganic phosphorus were analyzed with an autoanalyzer (736-10, HITACHI Ltd, Tokyo, Japan) using plasma samples obtained by centrifugation of heparin-treated blood samples. The following items related to bone metabolism were analyzed at Mitsubishi Chemical BCL Co, Ltd (Tokyo, Japan), using plasma samples. Intact parathyroid hormone was determined by immunoradiometric assay method, serum intact osteocalcin by radioimmunoassay, serum bone-specific alkaline phosphatase by immunoradiometric assay, and serum tartaric acid-resistant acid phosphatase by enzyme-linked immunosorbent assay every 3 months. Levels of 25(OH) and 1α,25(OH)2 derivatives of vitamin D3 were measured by HPLC/CPBA and HPLC/RRA, respectively, methods using serum sample obtained on blood sampling at month 12.
Urinalysis
Fresh urine samples were collected at the baseline and at 1-month intervals to determine changes in the following urinalysis parameters. Protein and glucose were routinely analyzed by a paper test. Alkaline phosphatase, γ-glutamyl transpeptidase, leucine aminopeptidase, N-acetyl-β-D-glucosamidase, and lac-tate dehydrogenase activity were measured with an automated analyzer (COBRA II, Packard Instrument Co, Ltd, Tokyo, Japan). Urine deoxypyridinoline was measured by HPLC. Since fresh urine is necessary for the above activity measurements, enzyme activity and content were corrected by creatinine concentrations because the daily urine volume was expected to have large variation due to renal tubular lesions.
Histopathologic Examination on the Kidney, Liver, and Parathyroid
At necropsy, tissue samples of the liver, kidneys, and parathyroid were collected and fixed in 10% neutral buffered formalin solution and routinely processed for histopathologic sections of hematoxylin and eosin stain, PAS, and Masson’s trichrome stain. For immunohistochemical examination, kidney and liver sections were stained for metallothionein (MT; anti-human metallothionein antibody; DAKO Japan, Kyoto, Japan) by the avidin–biotin–peroxidase complex methods using a commercial kit (Vector Laboratories, Burlingame, California, USA). The anti-MT antibody diluted by phosphate-buffered saline (pH 7.6, 1:400) was mounted on the sections and incubated over night at 4°C, following pretreatment with 0.3% H2O2-methanol solution (30 minutes) and diluted normal serum (20 minutes) to avoid nonspecific reactions. The reaction for anti-MT antibody were visualized with 3,3′-diaminobenzidine tetrahydrochloride (DAKO Japan). Slides were counterstained with methyl green.
Mineral Density and Histomorphometric Analysis of the Bone
The lumbar vertebrae, femur, and sternum were collected from each animal and fixed in 70% ethanol solution. The bone mineral density was measured with the lumbar vertebrae and femur by peripheral quantitative computed tomography (pQCT; XCT-960A, Norland/Stratec Inc, Germany) and dual-energy X-ray absorptiometry (DCS-600, ALOKA, Tokyo, Japan). In the pQCT analysis, each lumbar vertebra was scanned to record the images of 3 cross sections at the center of the bone at 2-mm intervals. Each femur was scanned at 2-mm intervals from the endplate to record images of 10 cross sections in the trabecular region and at the center of the bone to record 3 cross sections in the cortex region.
The bones collected were decalcified in 10% formic acid/10% formalin, and the prepared sections were stained with hematoxylin and eosin. For determining osteoid volumes, the lumber vertebrae and femur diaphysis were treated with 0.5% cyanuric–chloride methanol solution containing 1% (0.1 M) N-methylmorpholine before decalcification (Yoshiki method). As for the lumbar vertebrae (fourth), osteoid volume (OV) and bone volume (BV) of the trabeculae and tissue volume (TV) in the whole trabecular region were measured on the section stained with the Yoshiki method by tracing the circumference using an image analyzer (IPAP, Sumika-Technos Inc, Osaka, Japan). According to the Yoshiki method, osteoid dyes strong red, whereas bone dyes light red. OV is the area of osteoid in the whole trabeculae on the section, and BV is the area of whole trabeculae composed of bone and osteoid. TV is the area of whole trabecular region containing both trabeculae and marrow space. In this regard, the ratios of BV/TV and OV/BV were calculated. Similarly, BV and OV were determined for the cortex of femur diaphysis, and TV and BV of the trabeculae were measured for the sternum (fourth) and femur distal metaphysis. Additionally, undecalcified sections of the lumber vertebrae (fourth) stained with Villanueva bone stain and Prussian blue were prepared; the iron (Fe) deposition volume (FeV) and BV of the trabeculae were also measured on the Prussian blue–stained sections by the same method.
Analysis of Tissue Cd and MT Contents in the Liver, Kidney, and Femur
Small portions of the collected livers, kidneys, and femurs were dissolved in 14-N nitric acid. Cd contents in these tissues were measured on an atomic absorption spectrophotometer. MT contents in the liver and kidney were analyzed by the Cd saturation–hemolysate method. The MT concentrations were calculated on the basis of nanomoles of MT per 1 g of wet tissue, assuming that 7 Cd atoms bind to the 1 MT molecule.
Results
Body Weight Change
Animals (n = 2) treated with 2.5 mg/kg of Cd showed gradual reduction in body weight from month 6, while animals (n = 3) treated with 1.0 mg/kg of Cd as well as those (n = 4) treated with the vehicle alone showed no significant changes in body weight throughout the treatment period. None of the animals in either Cd-treated group showed loss of appetite during the treatment period.
Hematologic and Blood Chemical Findings
Cd treatment caused normocytic and normochromic anemia (Fig. 1). Animals treated with 1.0 mg/kg of Cd showed gradual decreases in the erythroid parameters from month 6, where the maximum decrease to the initial values was 36%, 35%, and 36% for red blood cell count, hemoglobin concentration, and hematocrit value, respectively, at the end of the treatment period. Decreases in these erythroid parameters were also noted in the 2.5-mg/kg group from about month 6, but the changes were rather drastic, as the maximum decrease was seen in month 9 and was 71%, 66%, and 67% for red blood cell count, hemoglobin concentration, and hematocrit value, respectively. Slight recovery of these parameters was observed with a reduction in frequency of Cd injection. However, no apparent changes were observed in mean corpuscular volume, hemoglobin, and hemoglobin concentration. A progressive reduction of plasma inorganic phosphorus level was seen in both Cd-treated groups from month 4 (Fig. 2a), where the maximum decrease to the initial values was 74% and 81% in the 1.0- and 2.5-mg/kg groups, respectively, at the end of the treatment period. In addition, a slight decrease in the calcium level was observed in the 2.5-mg/kg group in month 11 and after (Fig. 2b).
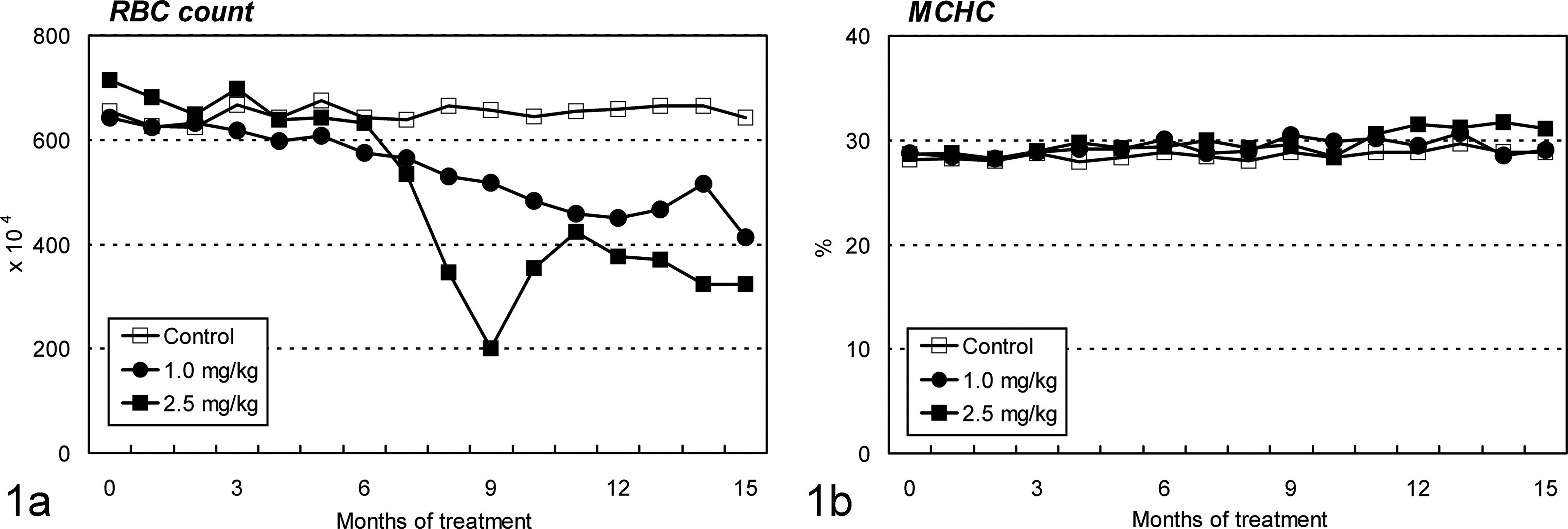
Changes in red blood cell (RBC) count and mean corpuscular hemoglobin concentration (MCHC) in ovariectomized cynomolgus monkeys during chronic intravenous treatment with cadmium. (a) Decreases in RBC are seen in both cadmium-treated groups from about month 6. (b) No apparent changes are observed in MCHC. Both graphs show that cadmium treatment causes normocytic and normochromic anemia.
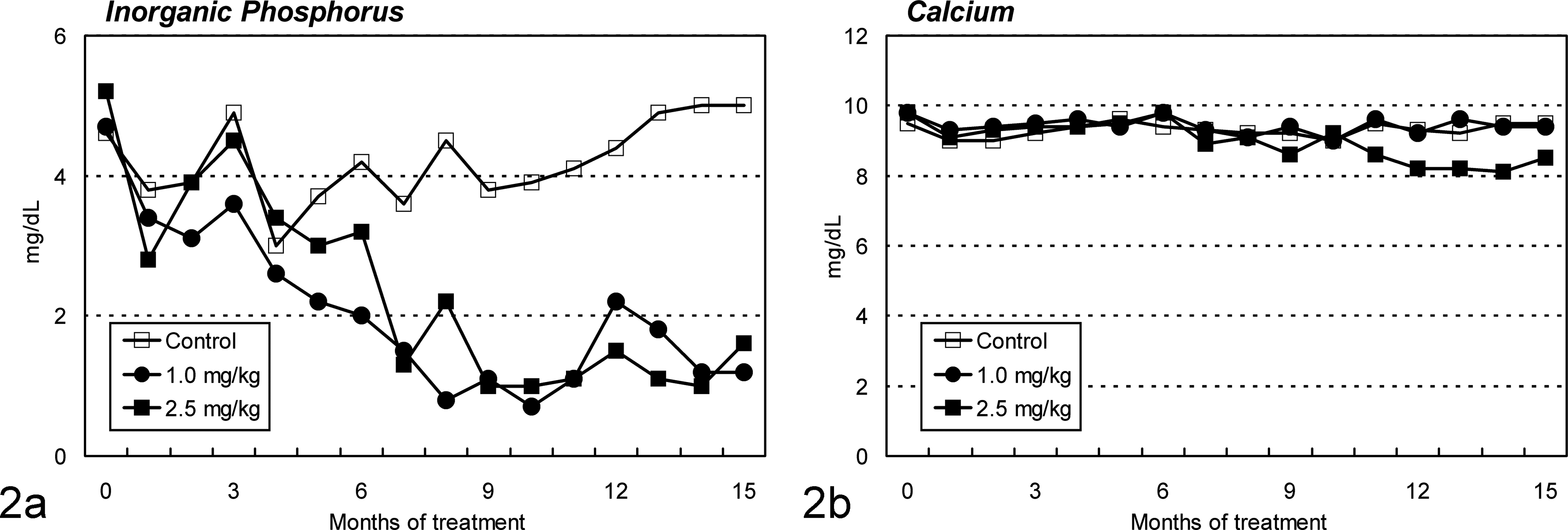
Changes in plasma inorganic phosphorus concentration and plasma calcium concentration in ovariectomized cynomolgus monkeys during chronic intravenous treatment with cadmium. (a) Decreases in inorganic phosphorus level are seen in both cadmium-treated groups from month 4. (b) Slight decreases in calcium level are observed in the 2.5-mg/kg group from about month 11.
Urinalysis and Urine Chemical Findings
In both Cd-treated groups, the marker enzyme activity of the brush border membrane of the proximal tubules increased from months 4–5, where the maximum increase to the initial values in the 1.0-mg/kg group were 12.3-fold (month 5), 2.2-fold (month 5), and 2.2-fold (month 13) for alkaline phosphatase (Fig. 3a), γ-glutamyl transpeptidase, and leucine aminopeptidase, respectively. In the 2.5-mg/kg group, the maximum increase, which was 29.2-, 3.2-, and 5.3-fold of the initial values, respectively, was seen in month 5. The marker enzyme activities in the proximal tubular epithelium became evident immediately after the increments of the marker enzyme activity of the brush border membrane, where the maximum increase in the 1.0-mg/kg group was seen in month 13, which was 18.6- and 14.6-fold for N-acetyl-β-D-glucosamidase (Fig. 3b) and lactate dehydrogenase, respectively, and that in the 2.5-mg/kg group was 47.3- and 9.6-fold, respectively. Although the onset times were almost the same in both Cd-treated groups, excretion of these marker enzymes in the urine was more severe in the high-dose group. Glucosuria and proteinuria were also noted in the 2.5-mg/kg group from months 6–7 (data not shown).
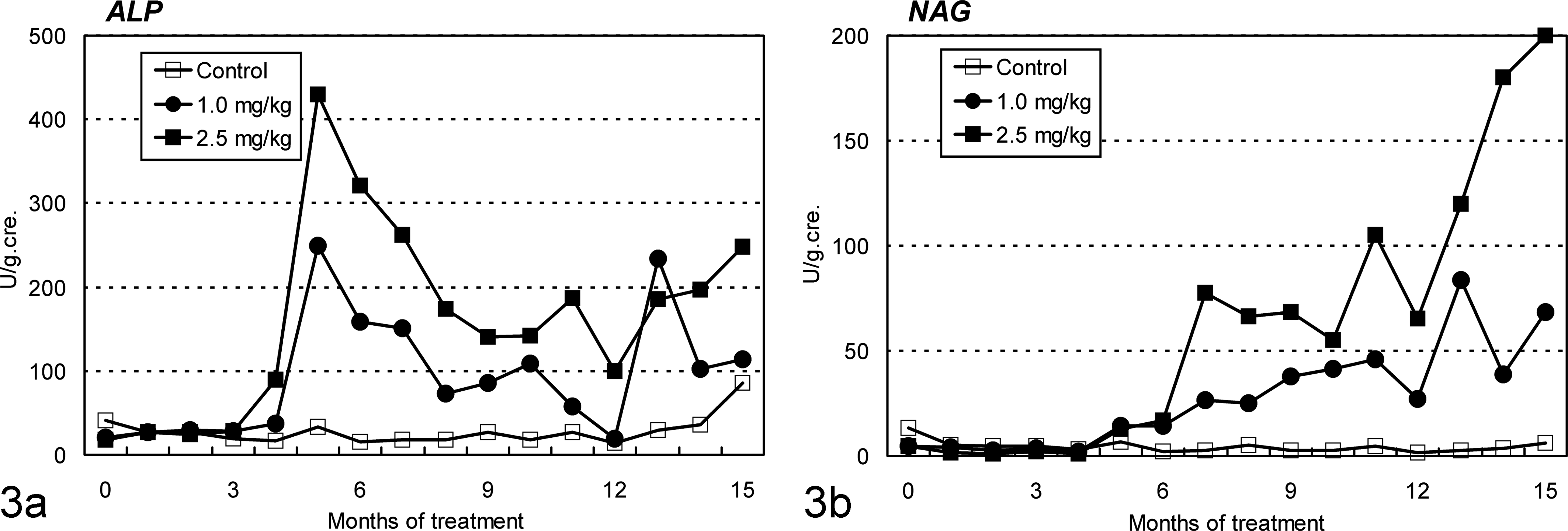
Changes in urinary alkaline phosphatase (ALP) and N-acetyl-β-D-glucosamidase (NAG) in ovariectomized cynomolgus monkeys during chronic intravenous treatment with cadmium. (a) ALP activity, a marker enzyme of the brush border membrane of proximal tubules, is detected in both cadmium-treated group from about month 4. (b) NAG activity, a marker enzyme of the proximal tubular epithelium, becomes evident soon after ALP activity is detected.
Bone Metabolism Markers in Serum and Urine
Urinary level of the metabolite of bone-specific collagen, deoxypyridinoline, markedly increased in the late stage of the Cd treatment, especially in the 2.5-mg/kg group (6.7-fold of the initial value; Fig. 4). Serum level of intact parathyroid hormone increased occasionally in some of the animals in both Cd-treated groups. No significant changes, however, were observed in serum levels of intact osteocalcin, bone-specific alkaline phosphatase, or tartaric acid-resistant acid phosphatase (data not shown).
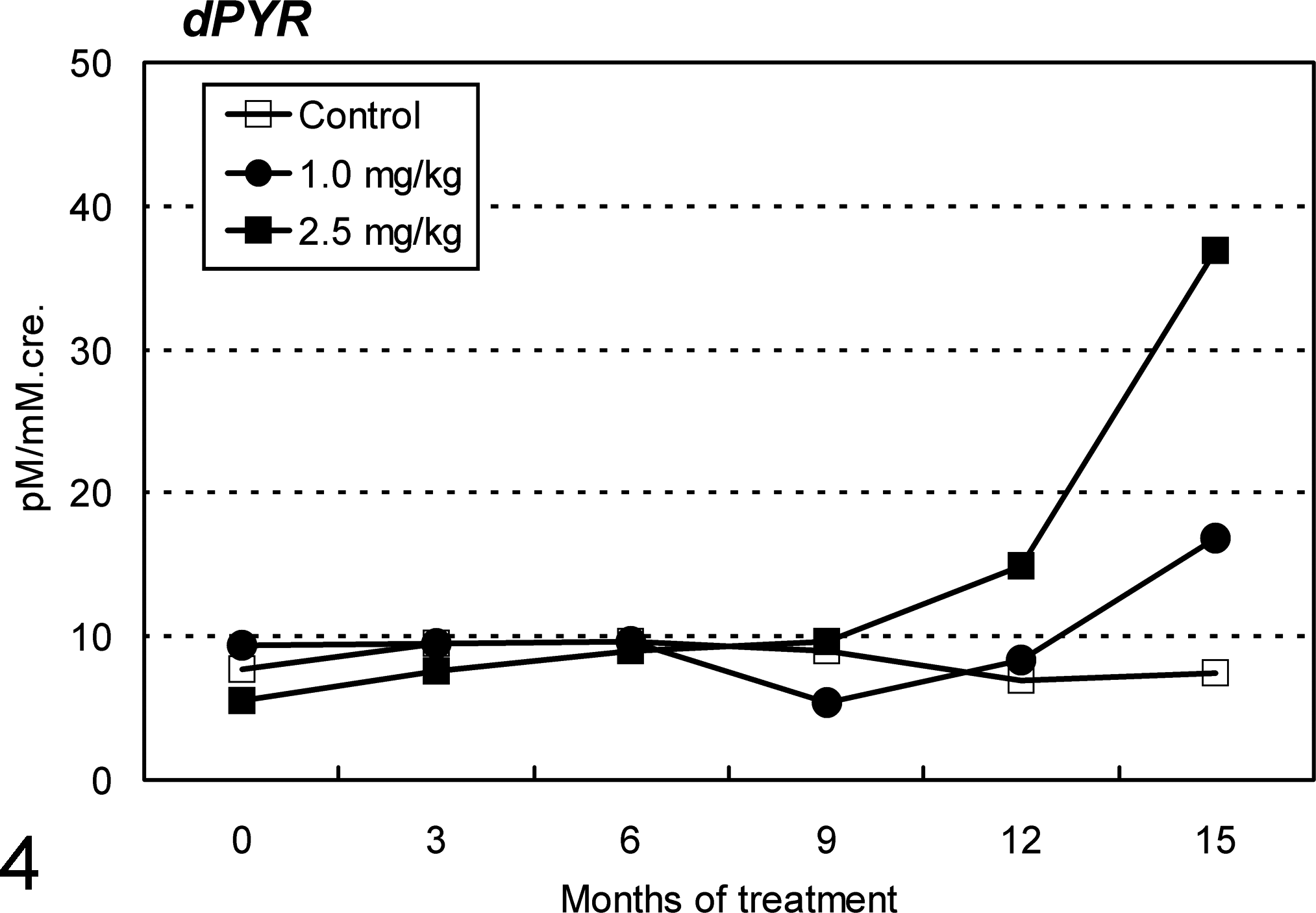
Changes in urinary deoxypyridinoline (dPYR), the metabolite of bone-specific collagen, in ovariectomized cynomolgus monkeys during chronic intravenous treatment with cadmium. Marked increases in dPYR level are seen in the late stage of cadmium treatment, especially in the 2.5-mg/kg group.
Serum 25(OH) and 1α,25(OH)2 Derivatives of Vitamin D3
Serum 1α,25(OH)2 vitamin D3 level decreased markedly in all Cd-treated animals, except 1 animal in the 1.0-mg/kg group (Fig. 5a), where the mean ratios to the control value were 76% and 22% in the 1.0- and 2.5-mg/kg groups, respectively. In contrast, the 25(OH) vitamin D3 level tended to increase in all Cd-treated animals (Fig. 5b), where the mean values in the 1.0- and 2.5-mg/kg groups were 1.6- and 1.3-fold, respectively, of the control value.
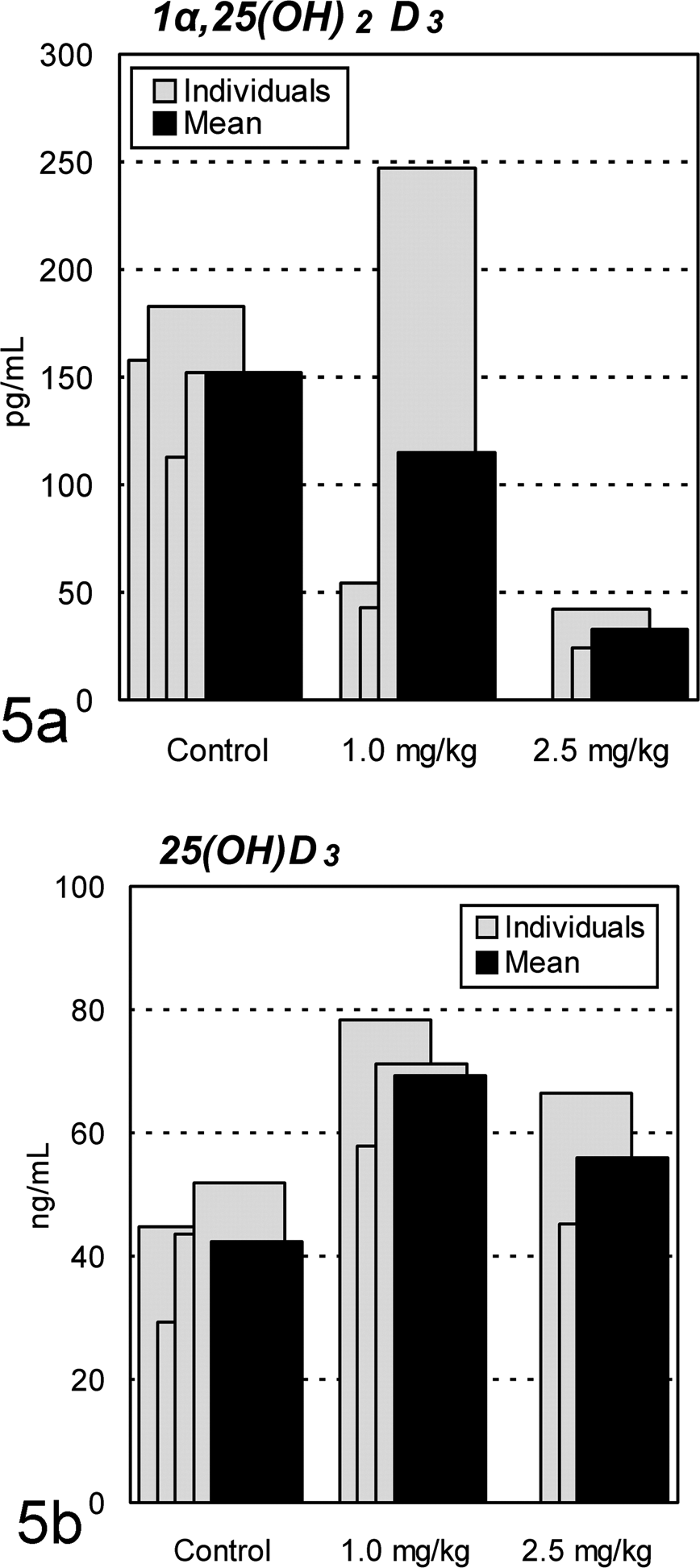
Changes in serum 1α,25(OH)2 and 25(OH) vitamin D3 in ovariectomized cynomolgus monkeys treated chronically with cadmium by intravenous injection. (a) Marked decreases in 1α,25(OH)2 vitamin D3 level are seen in all cadmium-treated animals, except 1 animal in the 1.0-mg/kg group. (b) Trend toward increased 25(OH) vitamin D3 level is seen in cadmium-treated animals.
Necropsy and Histopathologic Findings
At the necropsy, the kidneys of Cd-treated animals were slightly enlarged, pale, and firm compared to those of the controls. Histopathologically, Cd treatment caused lesions in the renal cortex. Necrosed tubular epithelial cells were observed in the proximal tubules, which frequently dropped into the lumina (Fig. 6b, c). Simultaneously, epithelial cells with enlarged nuclei appeared in the proximal tubules, suggesting epithelial regeneration (Fig. 6c). Epithelial atrophy accompanying thickening of the basement membrane was noted in the proximal tubule, and the tubules were frequently dilated (Fig. 6b, c). In the severe atrophic tubules, flat or low-columned epithelial cells were seen, and these changes diffused in all proximal tubular segments. Furthermore, diffuse interstitial fibrosis accompanying slight lymphocytic infiltration was observed in the cortex, and the interstitium was expanded by abundant collagen (Fig. 6b, c, e). No remarkable histologic abnormalities were found in the glomeruli, distal tubules, or collecting tubules. Almost all proximal tubular epithelial cells were positive for MT immunostaining in Cd-treated animals (Fig. 6g), whereas some proximal tubular epithelial cells were weakly positive in control animals (Fig. 6f). The livers or parathyroid glands were also unremarkable (Fig. 7b), except for 1 animal in the 1.0-mg/kg group, in which the parathyroid chief cells showed slight hypertrophy. In the liver, the hepatocytes were diffusely positive for MT immunostaining in Cd-treated animals (Fig. 7d), whereas a few perilobular hepatocytes were positive in control animals (Fig. 7c).
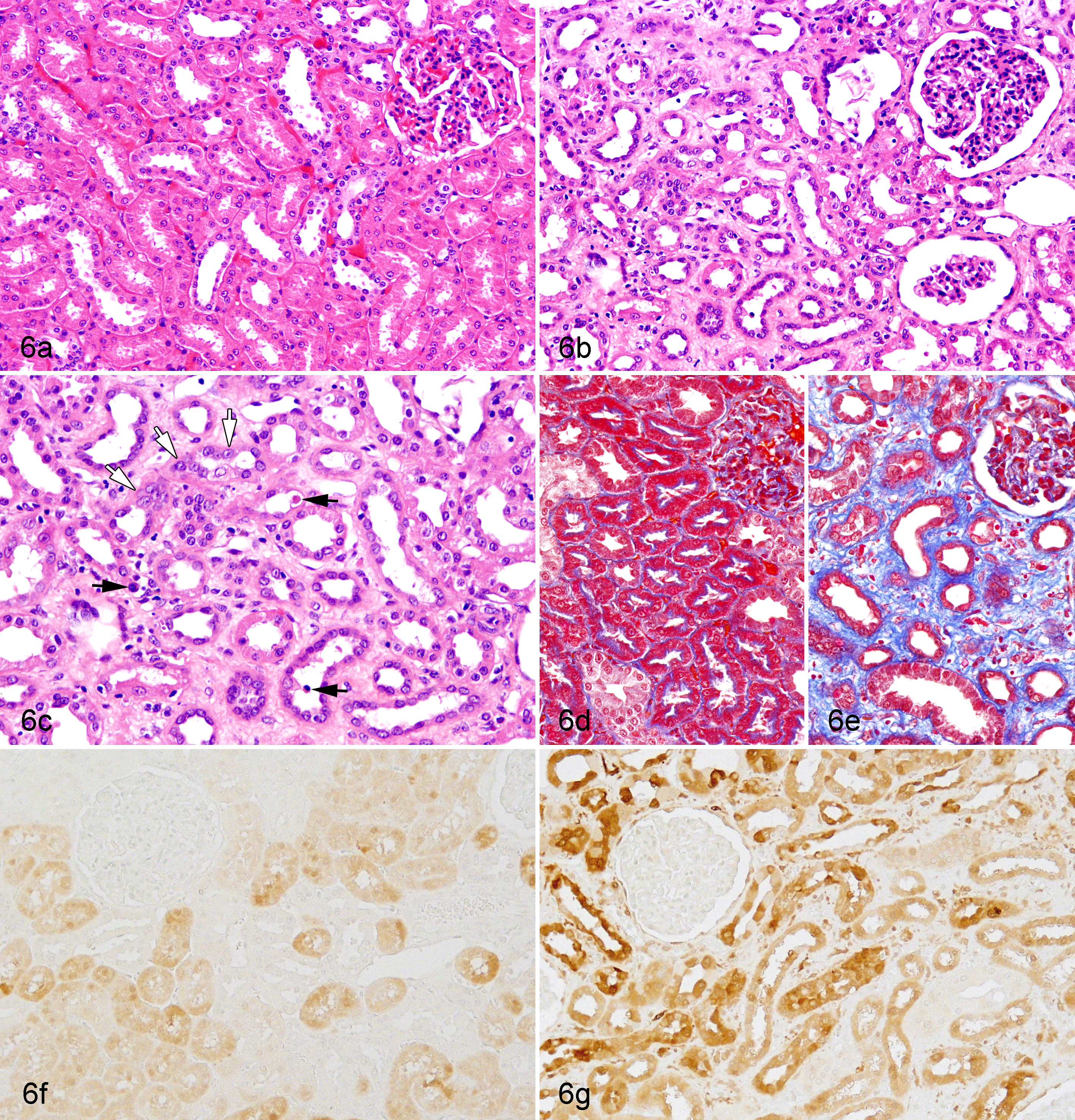
(a) Normal kidney; ovariectomized cynomolgus monkey, control animal. (b) Kidney; ovariectomized cynomolgus monkey, chronic intravenous cadmium administration, 1.0 mg/kg. (c) Higher magnification of Figure 6b. Necrotic tubular epithelial cells (black arrows) and regenerative epithelial cells with enlarged nuclei (white arrows) in the proximal tubules are observed in cadmium-treated animals. Diffuse interstitial fibrosis accompanying atrophy and/or dilatation of proximal tubules is also seen. No remarkable histologic abnormalities are found in the glomeruli or distal tubules of any animals. Hematoxylin and eosin stain. (d) Normal kidney; ovariectomized cynomolgus monkey, control animal. (e) Kidney; ovariectomized cynomolgus monkey, chronic intravenous cadmium administration, 1.0 mg/kg. The fibrotic area is stained in blue in cadmium-treated animals. Masson trichrome stain. (f) Normal kidney; ovariectomized cynomolgus monkey, control animal. (g). Kidney; ovariectomized cynomolgus monkey, chronic intravenous cadmium administration, 1.0 mg/kg. Almost all proximal tubular epithelium is positive for metallothionein in cadmium-treated animals, whereas some proximal tubular epithelial cells are weakly positive in control animals. Metallothionein immunostain and methyl green as counterstain.
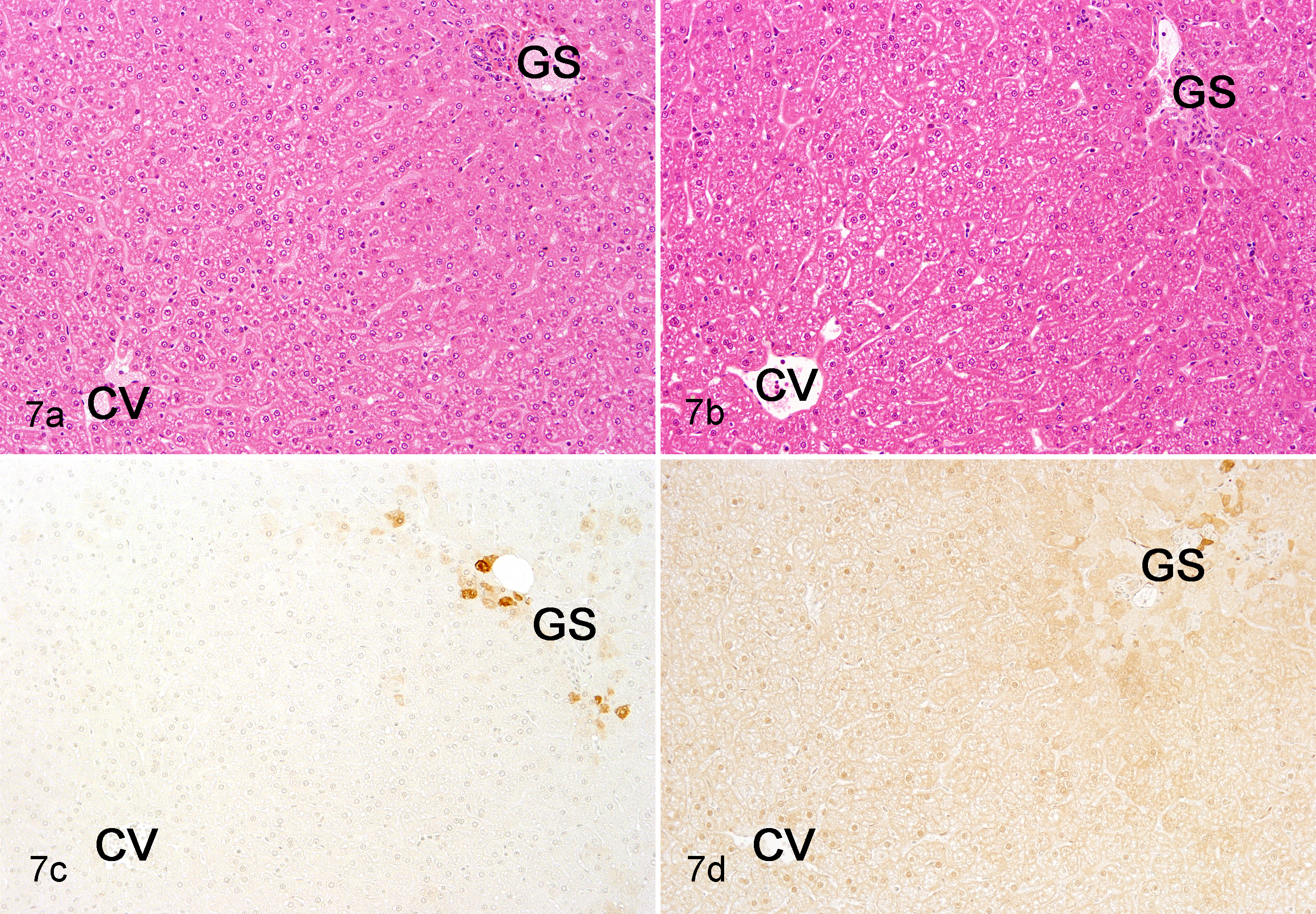
(a) Normal liver; ovariectomized cynomolgus monkey, control animal. (b) Liver; ovariectomized cynomolgus monkey, chronic intravenous cadmium administration, 1.0 mg/kg. No remarkable histologic abnormalities are found in cadmium-treated animals. Hematoxylin and eosin stain. (c) Normal liver; ovariectomized cynomolgus monkey, control animal. (d) Liver; ovariectomized cynomolgus monkey, chronic intravenous cadmium administration, 1.0 mg/kg. The hepatocytes are lightly and diffusely positive for metallothionein immunostaining in cadmium-treated animals, whereas a few perilobular hepatocytes are positive in control animals. Metallothionein immunostain and methyl green as counterstain. CV, central vein; GS, Glisson’s sheath.
Mineral Density and Histomorphometry of the Bone
A pQCT analysis of the lumbar vertebrae revealed decreased bone mineral density with thin cortex of the femurs (Fig. 8). Bone mineral density decreased in the lumbar vertebra by 26% and 20% and in the femurs by 27% and 22%, respectively, in the 1.0- and 2.5-mg/kg groups when determined by dual-energy X-ray absorptiometry (data not shown).
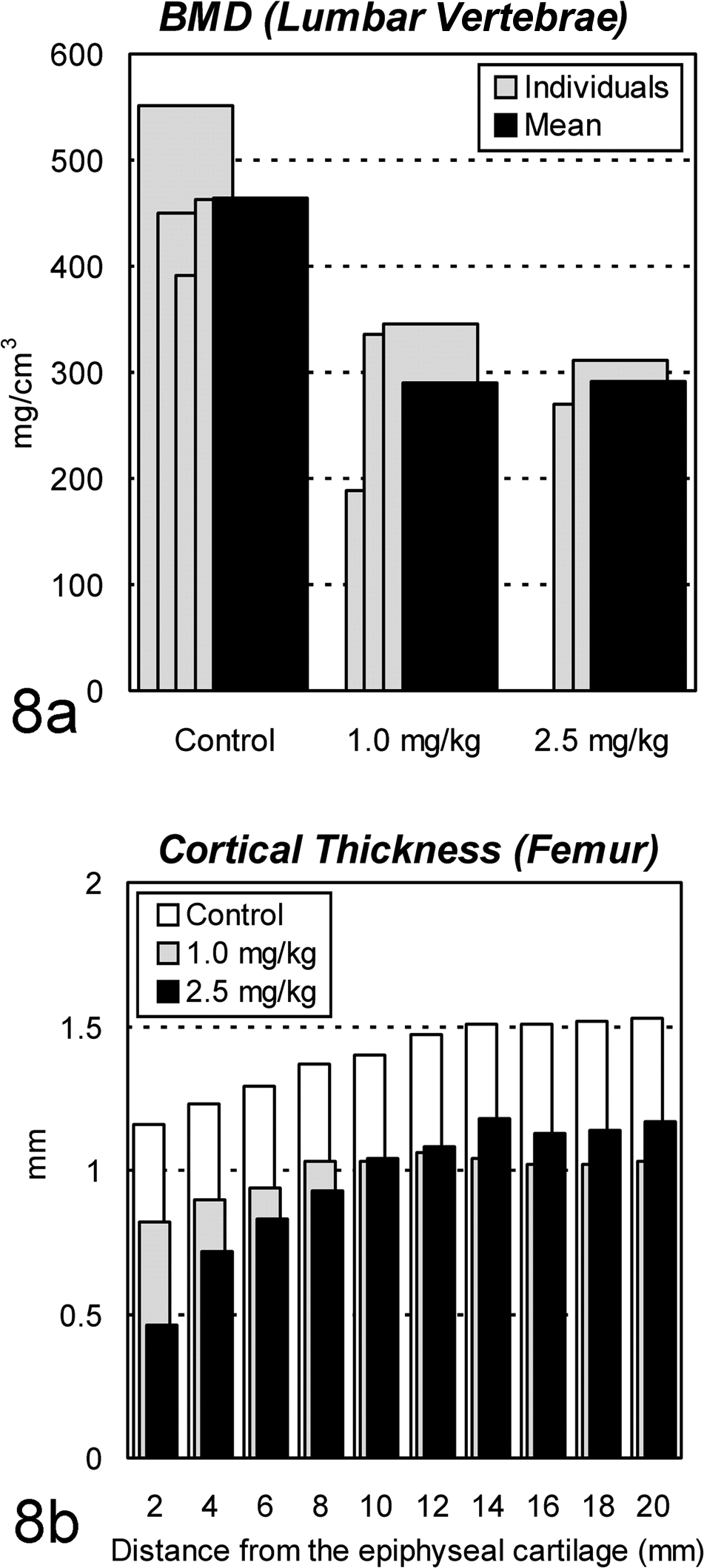
Changes in bone mineral density (BMD) and cortical thickness by peripheral quantitative computed tomography in ovariectomized cynomolgus monkeys treated chronically with cadmium by intravenous injection. (a) Decreases in BMD are seen at the center of the fourth lumbar vertebra in each cadmium-treated animal. (b) Decreases in cortical thickness are seen in the metaphysis of the femur in both cadmium-treated groups.
In Cd-treated animals, from Yoshiki- and Villanueva bone staining, osteoid appeared to increase around the trabecular zone of the lumbar vertebra and around the Haversian canals of the femur (Fig. 9a–d). On histomorphometric analysis, OV in these animals was found to be 9% to 44% of the trabecular BV in lumbar vertebrae and 0.2% to 6.3% of the cortical BV in diaphyses of femurs (Fig. 10). The mean values of OV/BV in Cd-treated animals were 12 to 15 and 5 to 12 times greater than the mean of the control group, respectively. The ratio of BV/TV was also markedly decreased in the lumber vertebrae, metaphyses of femurs, and sternum (Fig. 11), indicating bone mass reduction as well as thinning of the cortex. Microscopically, osteoclasts hyperplasia was noted with the lumbar vertebra and femur. Prussian blue staining of undecalcified sections revealed clear deposition lines of Fe at the mineralization fronts of lumbar vertebrae of Cd-treated animals (Fig. 9e–f). In image analysis, the deposition volume ratios (FeV/BV) in the 1.0-mg/kg group were 6- to 10-fold higher than those of the controls, although mean values of the areas were almost comparable between the 2.5-mg/kg and control groups (data not shown).
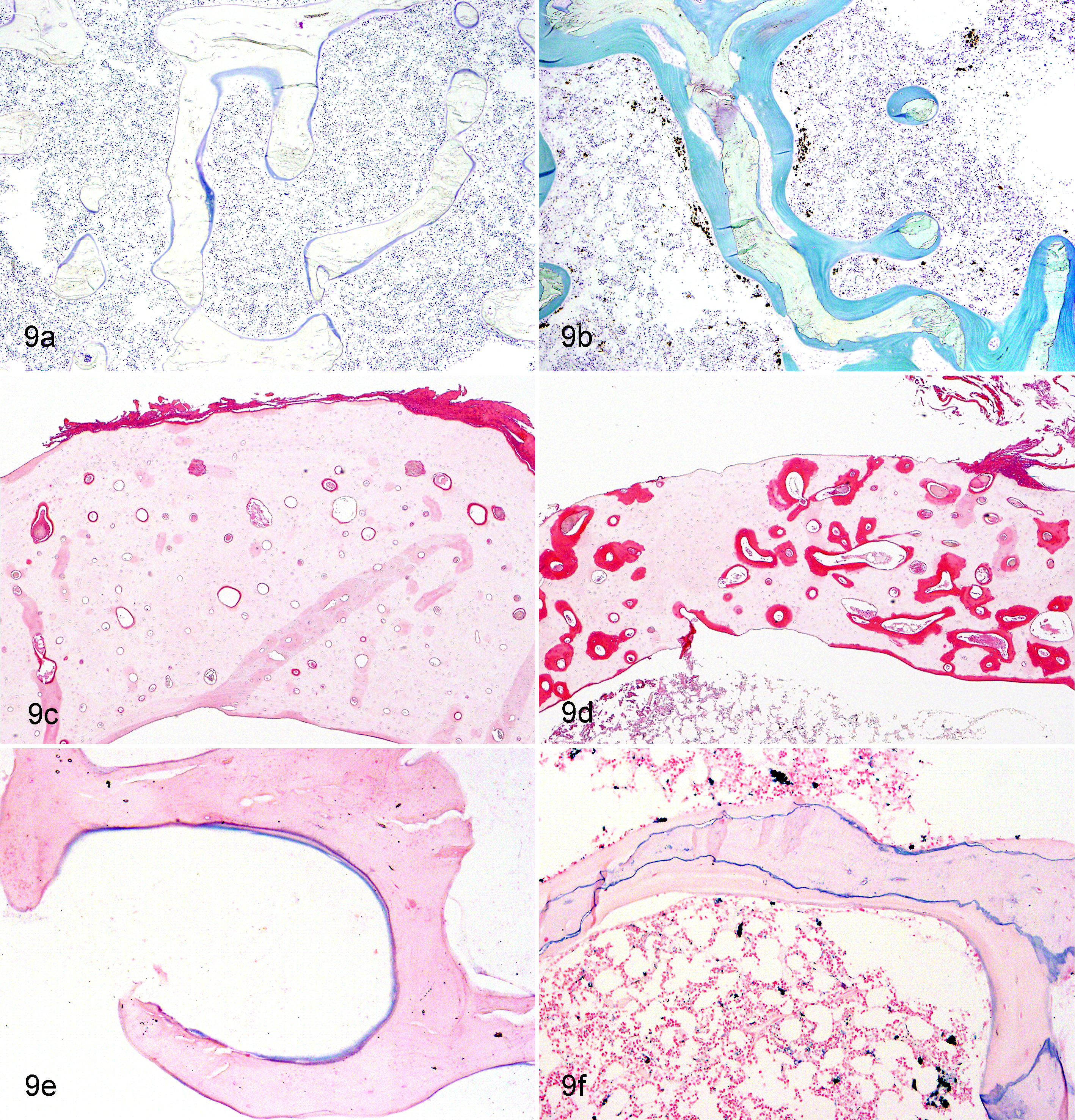
(a) Normal lumbar vertebra; ovariectomized cynomolgus monkey, control animal. (b) Lumbar vertebra; ovariectomized cynomolgus monkey, chronic intravenous cadmium administration, 1.0 mg/kg. Osteoid (stained in blue) appears to increase around the trabecular bone of the lumbar vertebra in cadmium-treated animals. Villanueva bone stain. (c) Normal femoral cortex; ovariectomized cynomolgus monkey, control animal. (d) Femoral cortex; ovariectomized cynomolgus monkey, chronic intravenous cadmium administration, 1.0 mg/kg. Osteoid (intensely stained by eosin) appears to increase around the Haversian canals in cadmium-treated animals. Yoshiki method. (e) Normal lumbar vertebra; ovariectomized cynomolgus monkey, control animal. (f) Lumbar vertebra; ovariectomized cynomolgus monkey, chronic intravenous cadmium administration, 1.0 mg/kg. Clear deposition lines of iron (stained in blue) are seen at the mineralization fronts of lumbar vertebrae of cadmium-treated animals. Prussian blue stain.
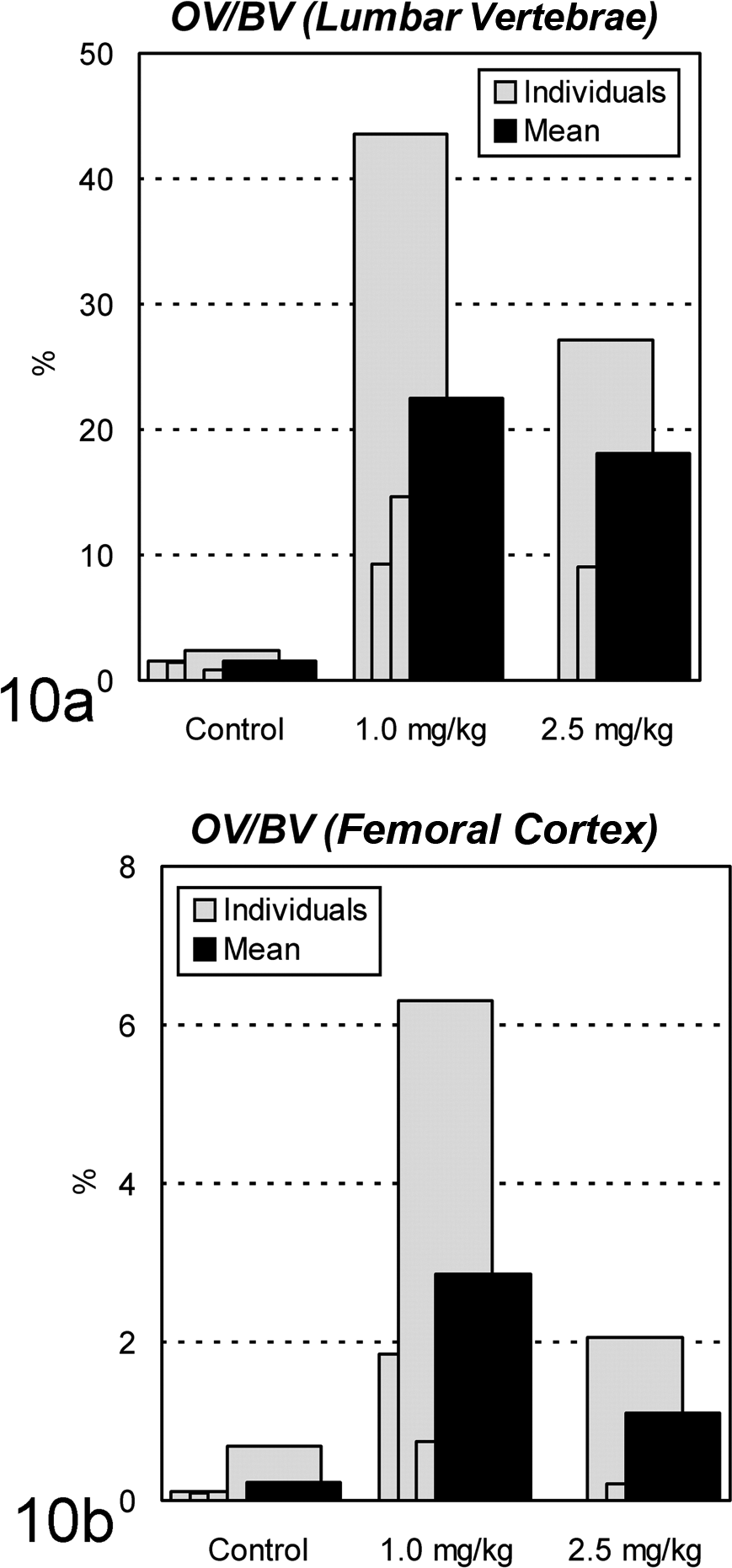
The ratio of osteoid volume/bone volume (OV/BV) by histomorphometric analysis in ovariectomized cynomolgus monkeys treated with cadmium by intravenous injection. On the (a) lumbar vertebrae and (b) femoral cortex, the mean ratio in the treated groups is 12 to 15 and 5 to 12 times greater than that in the control group, respectively.
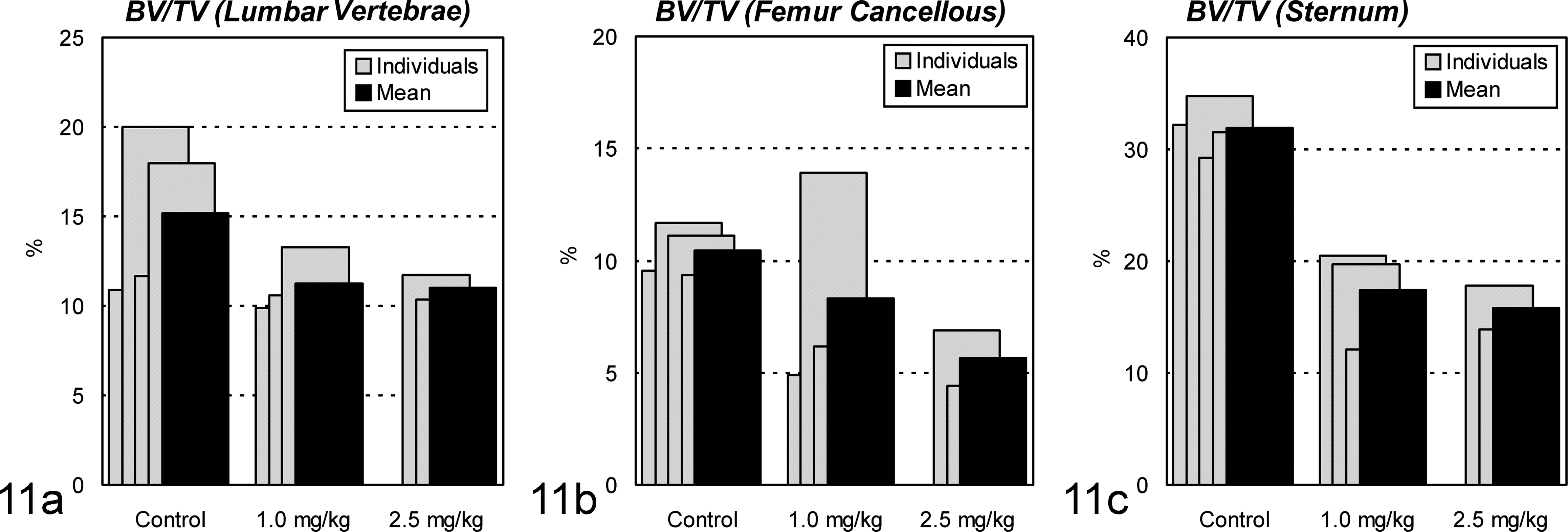
The ratio of bone volume/tissue volume (BV/TV) by histomorphometric analysis in ovariectomized cynomolgus monkeys treated with cadmium by intravenous injection. On the (a) lumbar vertebrae, (b) femur cancellous, and (c) sternum, the mean ratio in the treated groups is decreased.
Tissue Concentrations of Cd and MT
In all Cd-treated animals, Cd accumulated in the kidney, liver, and bone in large amounts (Fig. 12). The concentrations in the kidney were about 500 μg/g of wet tissue for both the 1.0- and 2.5-mg/kg groups, which were extremely higher than the critical concentration in the kidney, 20 μg/g of wet tissue, to cause renal damage in humans and rats. 10, 42 As for MT, remarkable accumulation was also observed in the kidney and liver (Fig. 13). This was consistent with the histopathologic observation with MT-immunostaining described above, demonstrating remarkable induction of MT in the renal proximal tubules and hepatocytes.
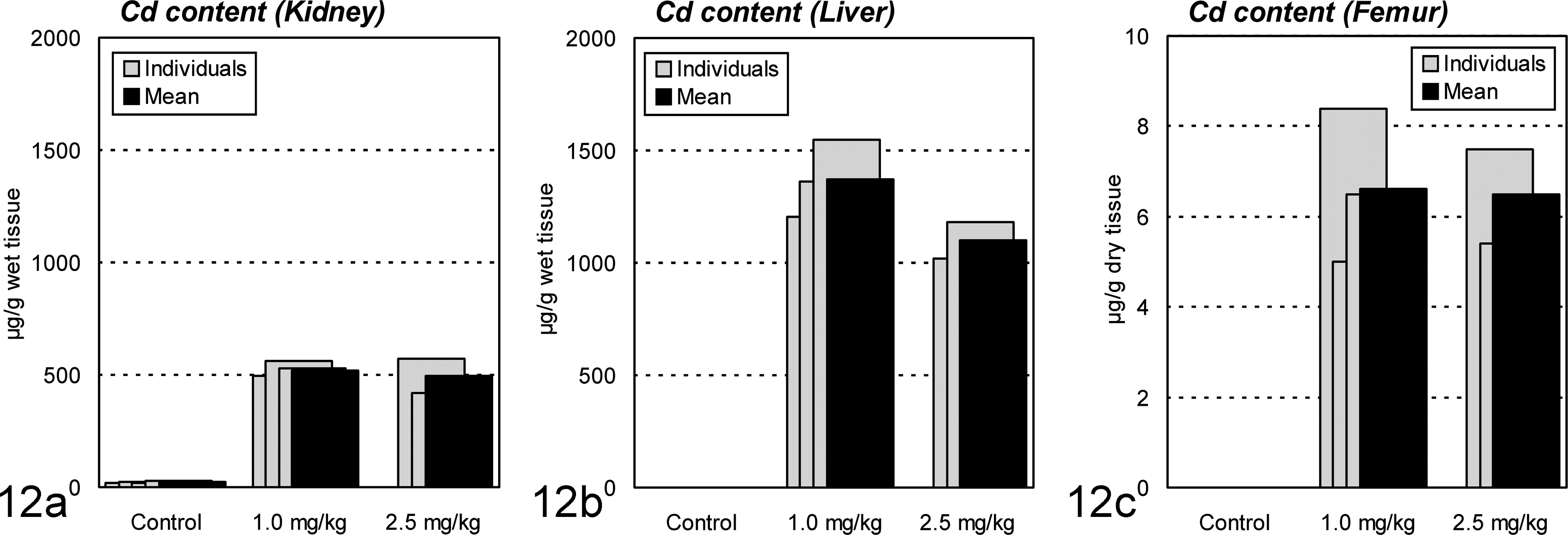
Cadmium (Cd) contents in the kidney, liver, and femur of ovariectomized cynomolgus monkeys treated with cadmium by intravenous injection. The concentrations in the (a) kidney are about 500 μg/g of wet tissue for both of the 1.0- and 2.5-mg/kg groups, which are much higher than the concentration in the control kidney, 20 μg/g of wet tissue. Similar remarkable accumulation is observed in the (b) liver and (c) femur of treated animals.
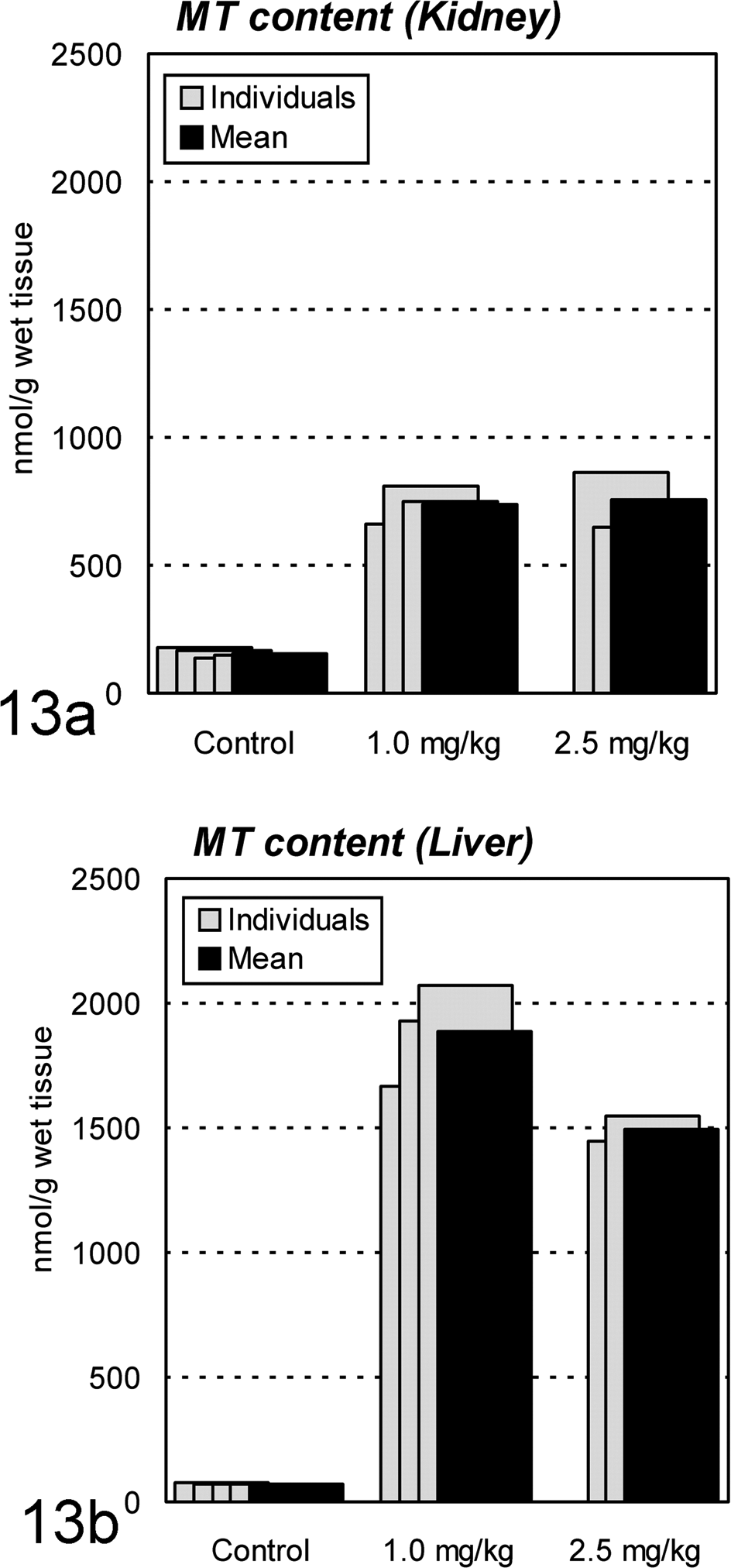
Metallothionein (MT) contents in the kidney and liver of ovariectomized cynomolgus monkeys treated with cadmium by intravenous injection. Remarkable accumulation is observed in both (a) kidney and (b) liver.
Discussion
For the past 3 decades, several attempts 26, 31,35,36 have been made to reproduce an IID model in monkeys. Dietary treatment (100 g/day containing 3, 30, 300 ppm of Cd) for 55 weeks caused denaturation, destruction, and regeneration of the epithelial cells in the renal proximal tubules in male rhesus monkeys, without causing any changes in the osseous tissues. 35 Kawashima et al 26 and Masaoka et al 31 also reported that no skeletal abnormality was noted in 6- or 9-year dietary Cd treatment to male cynomolgus or rhesus monkeys. However, in this study, we succeeded in producing a primate model of chronic Cd toxicosis with simultaneous occurrence of renal and bone lesions, which closely resembled IID in clinical and pathologic features. Between our study and the previous studies cited above, there were 2 major differences: (1) gender and related pretreatment (OVX) of monkeys used and (2) administration route of Cd. As mentioned, 90% of IID patients are postmenopausal women, 23 and OVX, which accelerates bone resorption, is a conventional procedure for reproducing an osteoporosis animal model. 21 We injected Cd directly into the veins, which prevents the problem of low or uncertain absorption from the gastrointestinal tract, 11 and the animals may not be the exact model of Cd toxicosis in humans that ingest Cd through the mucosal membranes of the respiratory and digestive tract in the forms of Cd-contaminated dust, vapor, water, and food. 12 Cd that is passed through the membranes of the digestive and respiratory tracts will be transferred via blood stream and accumulate in various organs, particularly in the liver and kidneys. Cd in the human body is usually combined with MT. Chronic effects of Cd are caused by the MT-bound or free Cd released from the organs, and total amounts of Cd held in the liver and kidneys parallel the magnitudes of Cd toxicosis. 39 In our animal experiment, Cd injected into the veins also transferred and accumulated mainly in the liver and kidneys, bound with MT, and caused the chronic toxic effects largely identical to those of humans. As mentioned above, this primate model is beneficial to investigate the unsolved problems of chronic Cd toxicosis of humans: pathologic mechanisms of Cd-induced nephropathy and osteopathy, novel therapy for patients, and appropriate criteria for the diagnosis of the disease.
Hypophosphatemia, enzymurias indicating the impairments of tubular epithelial cells, and nephropathy with diffuse tubular atrophy and interstitial fibrosis were identical between our monkeys and IID patients. 45 Glomerular lesions have been described in human cases, but the kidneys of murine 24 and present primate models lacked glomerular changes. Metallothionein staining of the models suggested that Cd concentrated exclusively in the tubular epithelium. Therefore, we suspect that the primary target of Cd is the proximal tubular epithelium and that varying degrees of hyalinization or sclerosis of the glomeruli are secondary to the advanced tubulo-interstitial lesion of the kidneys in human IID.
The anemia in IID patients is normocytic normochromic nephrogenous anemia. 16 We reported in rats that nephrogenous anemia of poor erythropoietin response substituted Fe deficiency anemia with an advancement of renal damages after 6 to 12 months of Cd treatment. 14 The animals showed normocytic normochromic anemia from the sixth month, 2 months later than the emergence of enzymuria, indicating renal tubular damage. Major sources of erythropoietin are the peritubular capillary endothelial cells and tubular epithelial cells of the kidney. 21 Therefore, the anemia of the Cd-treated monkeys may also be nephrogenous, like that of its human counterpart, although erythropoietin was not measured in this study.
Cd-treated monkeys exhibited severe osteomalacic osteopenia with active bone resorption by osteoclasts, continuous hypophosphatemia, and an increase in d-PYR excretion into urine. These changes were consistent with those of IID, in which bone lesions have been considered to be a complication of osteomalacia due to impairment of mineralization and osteoporosis due to an advance in bone resorption. 32 The magnitudes of bone lesions observed in our monkeys were compatible with those of IID patients of moderate with those of IID patients of moderate degree, though osteomalacic osteopenia of such extent has not previously been reported in animal models.
The mechanism of Cd osteopathy is rather controversial. In general, it is considered that the osteopathy is triggered by a renal dysfunction (indirect mechanism) 2, 27,28,34 or caused by direct effects of Cd on bone tissues (direct mechanism). 29, 33,43 Our primate model, having osteomalacic osteopenia, clinically showed hypophosphatemia and decreased serum levels of 1α,25(OH)2 vitamin D3. Both changes were also noticed in IID patients or inhabitants in Cd-polluted area. 2, 28,34 1α,25(OH)2 vitamin D3 is produced by hydroxylation at position C1 of 25(OH) vitamin D3 in the renal proximal tubules. Damages of proximal tubular epithelial cell cause the impairments of inorganic phosphorus reabsorption and conversion of 25(OH) vitamin D3 to 1α,25(OH)2 vitamin D3 at the tubules, resulting in osteomalacic osteopenia of the bone. Therefore, renal dysfunction might contribute the development of bone lesions in our primate model.
Cd, Al, and Fe ions could be adsorbed onto and incorporated into crystals of calcium hydroxyapatite and increase osteoid in bone tissue. 3, 7,8 In IID patients, Al, Fe, and Cd deposition was demonstrated at the osteoid–bone interface (mineralization front). 32, 33 In our previous study, 15 deposition of Cd and Fe in bones was noted in a rat model of chronic Cd toxicosis. In the present experiment, Prussian blue–stained undecalcified sections of bones revealed Fe deposition at the mineralization front of Cd-treated monkeys. These results suggest that metals such as Fe and Cd directly inhibit calcium incorporation into the osteoid at the mineralization front.
Malnutrition, vitamin D deficiency, impaired absorption of calcium at the intestinal mucosa, and multiparous birth have been raised by some researchers as potential causes or cofactors of IID. In this study, the animals given 1.0 mg/kg of Cd showed the hallmarks of IID without commitment of any of the factors mentioned above. In conclusion, we have examined chronic Cd toxicosis in OVX monkeys and found that renal and bone lesions developed in the animals. These lesions closely resembled those reported in IID patients in terms of clinical chemistry and histopathology. Thus, OVX monkeys chronically exposed to Cd can serve as a primate IID model.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared receipt of the following financial support for the research, authorship, and/or publication of this article. This study was supported financially in part by the Japanese Enviromental Agency.
