Abstract
The evaluation of spontaneous lesions in classical inbred strains of mice has become increasingly important because genetically engineered mice (GEMs) are created on these backgrounds. Novel inbred strains—genetically diverse from classic strains—are valuable both as a new background for GEM mice and to increase the genetic variation found in laboratory mice. Newly arising spontaneous genetic alterations in commonly used strains may also lead to new and valuable mouse models of disease. This report evaluates gross and histological lesions in relatively new, classic, and rarely explored mouse inbred strains. Pathological lesions of 1273 mice from 12 inbred strains (129S1/SvW, A.CA-H2f /W, AKR/W, BALB/cW, BN/aW, C57BL/6 W, C57BL/10 W, C3H/W, C3H wad /W, CBA/W, DBA/2 W, and WOM/W) are reported. BN/aW, WOM/W, and C3H wad /W are novel inbred strains produced and maintained in the Department of Genetics and Laboratory Animal Breeding at the Center of Oncology, Warsaw, Poland. Both neoplastic and nonneoplastic lesions were examined. The prevalence of lung neoplasms was significantly higher in A.CA-H2f /W (33.3%) and BALB/cW (33.8%) mice (P < .01). The prevalence of liver neoplasms was significantly higher in the CBA/W strain (P < .01). Mammary gland neoplasms arose at a greater frequency in C3H/W mice (P < .01). The occurrence of uterine neoplasms was higher in DBA/W and 129S1/SvW mice. AKR/W and WOM/W mice developed T-cell lymphoblastic lymphoma with high frequency (110/121 [90.9%] and 159/175 [90.9%], respectively) before 1 year of age. The occurrence of nonneoplastic lesions in the kidneys of BN/aW mice was increased (P < .01).
The mouse is a useful model for common human diseases such as cancers, type 1 and type 2 diabetes, obesity, and cardiovascular and neurodegenerative disorders. The mouse models are represented by a variety of mice whose phenotypes either arose spontaneously or were specifically engineered via transgenesis.
Because most genetically engineered mice (GEM) models derive from a change in a single gene locus, they do not perfectly mimic the human population, which is heterogeneous. Most mouse models represent only a small number of mouse genetic backgrounds, which limits the mapping of genes involved in carcinogenesis and gene function as well as studies of polygenic networks and gene interactions. A community resource termed the Collaborative Cross was designed to create mice with increased genetic diversity, which will better reflect the complexity of the human population. 14,47,66 In the context of this new approach to mouse modeling, novel inbred strains—genetically diverse from classic inbred strains—appear to be valuable both for their new background for GEM mice and for their increased genetic diversity. Moreover, the possibility of new spontaneously arising genetic alterations in commonly used strains may create new mouse models of human disease.
The documentation of lesions found in inbred strains is increasingly important. The influence of genetic background on genetically engineered mouse phenotypes is apparent, and researchers should be cognizant of the phenotypes of the used strains. 5 Common lesions of background mouse strains are regarded as “background lesions.” The accurate assessment of the GEM phenotype requires separation of phenotypes due to genetic manipulation from the phenotypes of the background strain. 7 Therefore, a comprehensive knowledge of the phenotype of background strains is required, especially as inbred strains differ in their susceptibilities to particular neoplastic and nonneoplastic diseases. 8,25,44,67,73 Furthermore, substrains that arise through prolonged inbreeding within various laboratories can differ significantly from the parent strain. 26 There are abundant data on gross and histological lesions of common strains, but this information is widely dispersed. Phenomic databases such as the Mouse Phenome Database at the Jackson Laboratory, 33 Mouse Genome Informatics (MGI), 20 International Mouse Strain Resources Database (IMSR), 31 Japan Mouse/Rat Strain Resources Database (JMSR), 32 and others 11,63,77 serve as sources of consolidated mouse strain data, providing links to the references. However, the less commonly used strains are not consistently reported. Much information is available on B6C3F1 (C57BL/6 female × C3H or C3H/He male) as a result of toxicology studies. 35,43,70 Lesions of B6;129 mice, commonly used as a background strain in genetically engineered mice, are also well described. 27,74 Gross and histological lesions of strains commonly used to generate transgenic mice (C57BL/6, 129, FVB, and B6;129) have been recently reviewed. 9
There is significant background-dependent phenotypic variability in GEM strains. Phenotypes that result from the same genetic alteration can vary on different backgrounds. 29,55 It would be ideal if investigators could choose the background strain that is most appropriate for the studied disease. 17 However, for varying reasons, some strains are favored over others for mouse model production. In particular, 129 embryonic stem (ES) cells and C57BL/6 blastocysts are a favorable combination for transgenesis. The 129 ES cells are easy to modify genetically and have a very high incidence of germline transmission. Furthermore, agouti/black chimerism can be easily identified. 7,18 Interestingly, ES lines have been produced from C57BL strains, as well as F1 or F2 offspring of a cross between inbred C57BL/6 and CBA/2 or FVB/N mice. 71 BALB/c and A mice are used in ENU (N-ethyl-N-nitrosourea) mutagenesis experiments due to their fecundity, tolerance of ENU, and high rates of survival. 7 The popularity of the C57BL/6 strain results from the fact that it was chosen for the mouse genome-sequencing project and that many early GEMs were created on this background. 7
This report focuses on spontaneous lesions of aging mice in our unique inbred strains WOM/W and BN/aW, whose genomes vary significantly from the most commonly used strains (unpublished data). Furthermore, we present multiple lesions in traditional inbred strains and in a spontaneously arising variant of the C3H/W strain—C3H wad /W.
Materials and Methods
Mice
Strains
In total, 1273 mice from 12 inbred strains (129S1/SvW, A.CA-H2f /W, AKR/W, BALB/cW, BN/aW, C57BL/6W, C57BL/10W, C3H/W, C3H wad /W, CBA/W, DBA/2W, and WOM/W) originated from breeding and research colonies maintained at the Maria Sklodowska-Curie Memorial Cancer Center and Institute of Oncology in Warsaw, Poland. Inbred mouse strains are included in the register of the Committee on Standardized Nomenclature for Inbred Strains of Mice and the register of the National Academy of Sciences, Washington, DC. In the International Laboratory Code Registry, the code of our breeding center is W. 30
The origin of the examined strains and generations of inbreeding are listed in Table 1. BN/aW, WOM/W, and C3H wad /W are strains that are unique to our facility and are registered in the Jackson Mouse Database. 15,24,33,64
Origin of the Examined Strains and Generations of Inbreeding.
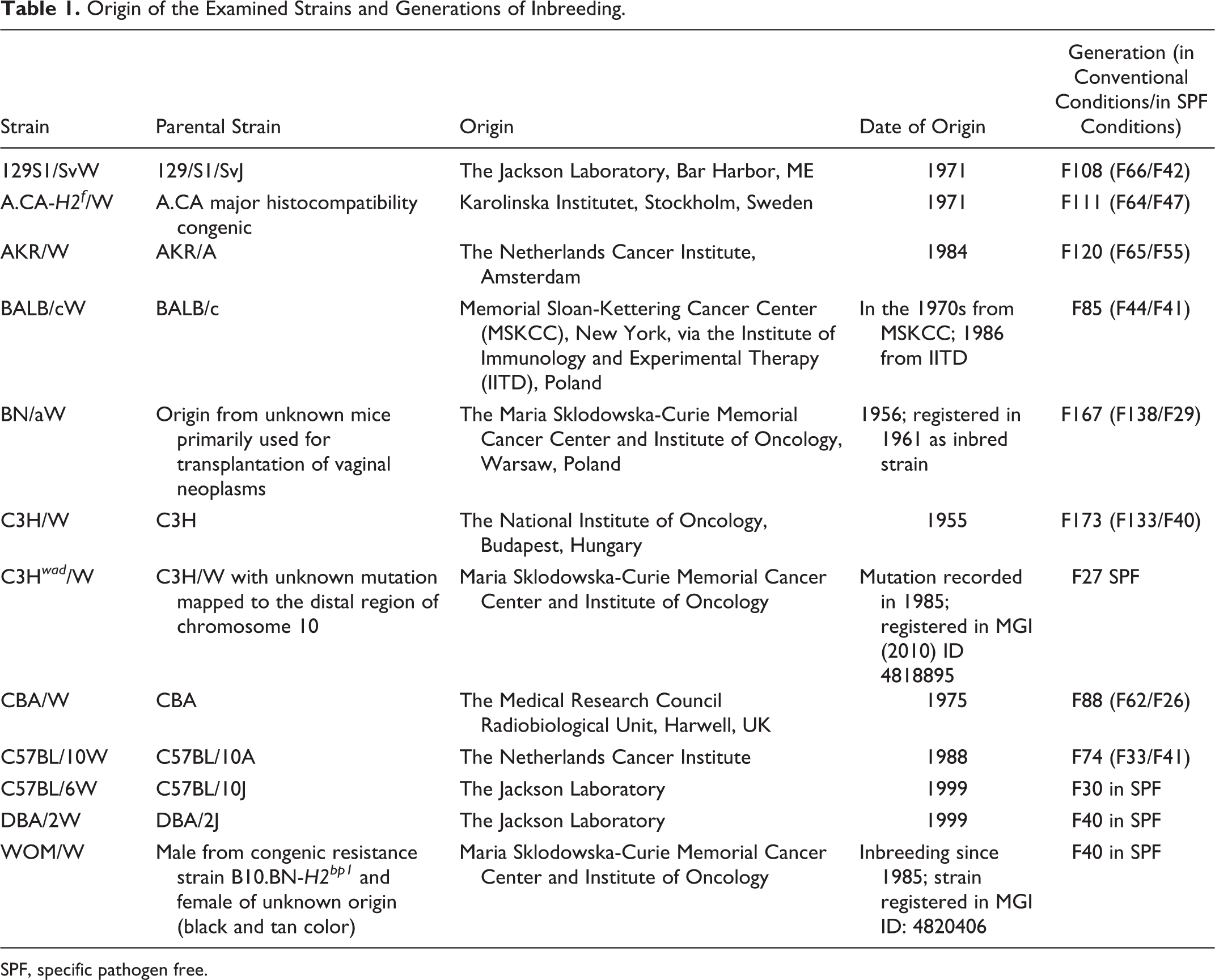
SPF, specific pathogen free.
Information about the congenic A.CA-H2f /W strain is not available in the Mouse Phenome Database at the Jackson Laboratory, and currently phenotypic data for this strain are scarce. The other strains are commonly used in biomedical research.
Inbred status
Mice of inbred strains are genetically defined using microsatellite markers. Each strain is characterized by more than 450 microsatellite markers, which are distributed across all autosomes and chromosome X and a smaller portion of chromosome Y. Mice from the studied strains are homozygous at all the examined loci. Based on the obtained microsatellite panel, a set of markers for genetic monitoring has been established. To exclude breeding contamination, every 3 generations, animals from all strains are tested with a set of selected markers that include 17 microsatellite markers and an additional marker on chromosome Y.
Husbandry and housing
The animals were housed in a barrier system under specific pathogen-free (SPF) conditions (see below). Mice were allowed ad libitum access to water and a standard pelleted diet for breeders (LABOFEED H; Feed Factory “MORAWSKI,” Kcynia, Poland). The diet contained crude protein (23%, plant origin), crude fat (4.1%, plant origin), crude fiber (4.6%), crude ash (5.5%), and nitrogen-free extracts (50.8%). The diet was supplemented with dried degreased milk and whey, vitamins, and minerals.
The food, cages with bedding, and filled water bottles were autoclaved. The cages had 400 cm2 of floor area. Since 3 to 4 males or 4 to 5 females were maintained in 1 cage, this translates into about 80 cm2 of floor area per animal. 16 Mice sharing 1 cage were of the same age and, if possible, siblings. Light cycle was 12 hours dark/12 hours light. The temperature was stabilized at 22 ± 2°C. Mice received humane care and ethical treatment, and all procedures were in accordance with the Polish Animal Protection Act No. 1002/2003 and Polish Animal Experimentation Act No. 289/2005.
Health monitoring
Health monitoring was performed based on the recommendations of the Federation of European Laboratory Animal Science Associations (FELASA). 51 Bacteriology, parasitology, and serology were performed every 3 months (except for some serological tests) on 3 randomly selected animals from each breeding room. Ectoparasites and pinworms were not detected in any examined mouse. Trichomonas muris was found in the cecum and/or colon in wet mounts but did not cause clinical disease or lesions. The trophozoites of Giardia muris were detected in the proximal part of the small intestine, and cysts were found in feces, again without clinical manifestations. Serology was performed every 3 months to identify antibodies to mouse hepatitis virus, epizootic diarrhea of infant mice virus, mouse parvovirus, Sendai virus, pneumonia virus of mice, Theiler’s encephalomyelitis virus, and minute virus of mice (MVM), as well as antibodies to bacteria Clostridium piliforme, cilia-associated respiratory bacillus, and Mycoplasma pulmonis and once yearly for detection of antibodies to ectromelia, Reo-3, mouse cytomegalovirus, adenovirus K-87/FL, and lymphocytic choriomeningitis virus. The results were generally negative, and only ∼4% were equivocal for MVM and C. piliforme (± result for one mouse in 1 control). Mice were not tested for murine norovirus and Helicobacter species.
Observation Protocol
Mice were monitored daily for neoplasm formation and/or signs of chronic progressive diseases. During physical examination, mice were specifically evaluated for inactivity, poor grooming, weight loss, respiratory disorders, pale ears, visible tumors, and palpable internal masses. Mice were maintained until near end of life (EOL) and euthanized when clinical signs suggested death within 24 hours. The criteria for EOL included nonresponsiveness to touch, cold body temperature on touch, slow or labored respiration, hunched body position, matted fur, and failure to drink and eat. 54 Mice with symptoms of chronic illness observed longer than 3 to 4 weeks were sacrificed for humane reasons. Mice were sacrificed at the age of 1100 days or older if they exhibited age-associated weakness. Very few mice (5) died naturally. The ages of mice with neoplasms and without neoplasms but suffering from nonneoplastic lesions are shown in Table 2. The age of mice on the date of sacrifice was recorded as their neoplasm age. Data were collected from 2003 to 2011.
Age of the Examined Mice in days (Mean, Median, Minimal, and Maximal Age).
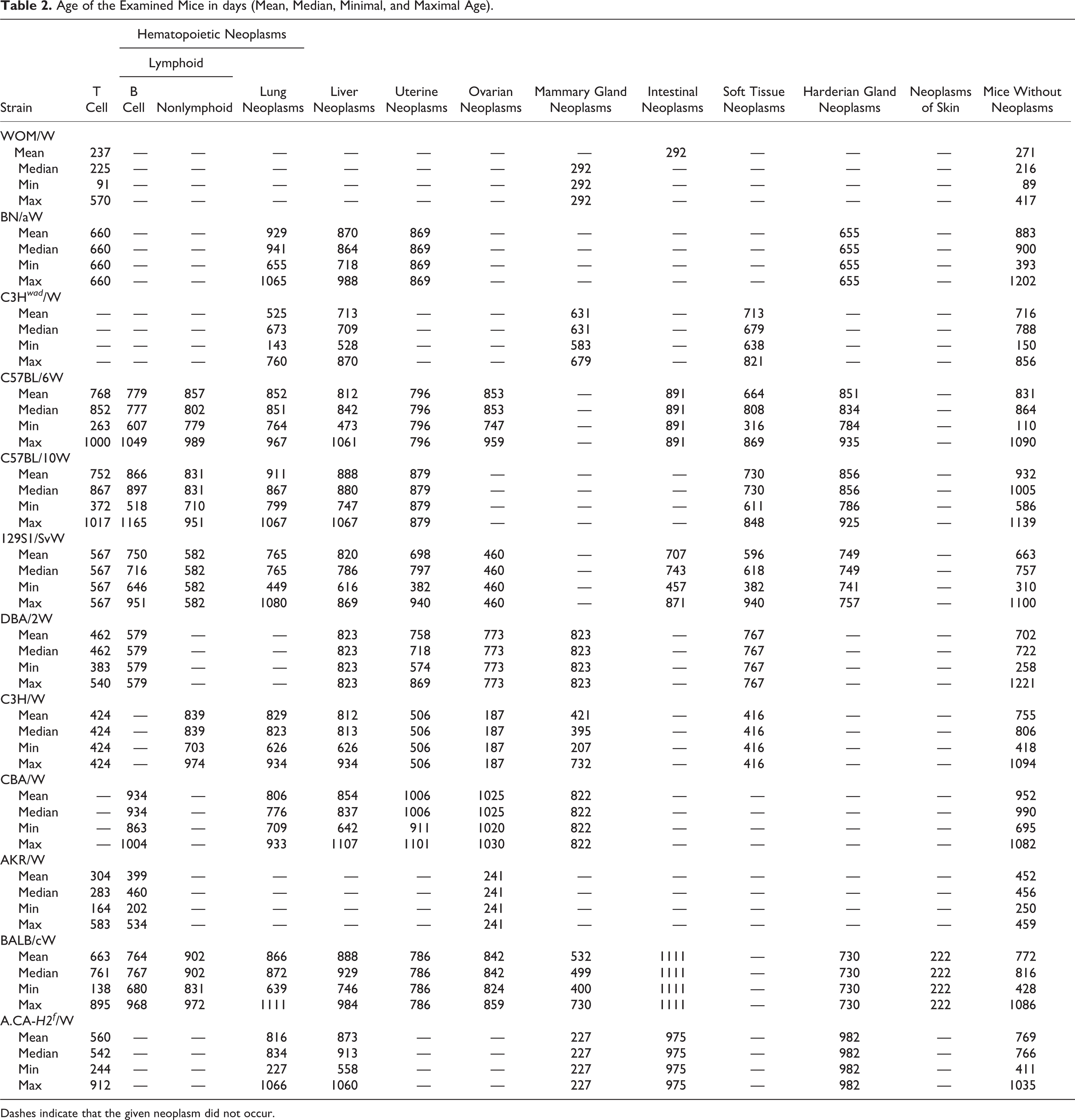
Dashes indicate that the given neoplasm did not occur.
Pathological Examination
A complete necropsy was performed on each mouse according to previous recommendations. 19,22 Organ sampling and trimming were based on the Revised Guides for Organ Sampling and Trimming in Rats and Mice. 36,48,58
All tissues (42 samples) and gross lesions from 5 mice per sex, per strain were examined. For all additional animals within a given strain, lungs, trachea with thyroid and parathyroid glands, liver with gallbladder, kidneys with adrenal glands, spleen and testes with epididymides, ovaries, uterus, vagina, pancreas, and visible lesions were sampled. Tissues were fixed in EAFS fixative containing ethanol, acetic acid, formol, and 0.9% NaCl. The intestine was opened, rinsed with 0.9% NaCl, and examined for visible neoplastic foci or enlarged Peyer’s patches or lymphoid follicles. The parts of intestine with visible lesions were recoiled on cotton swabs and fixed. If hematopoietic neoplasms were suspected, thymus, mesenteric lymph nodes, and sternum were fixed for hematoxylin and eosin (HE) and immunohistochemical staining. Blood and smears of bone marrow, obtained from femur, were stained with modified Giemsa stain, and the percentage of neoplastic hematopoietic cells was estimated. Immature granulocytes were identified using Leder stain (naphthol AS-D chloroacetate esterase reaction) on air-dried imprints or paraffin sections. 46 Congo red stain was used to identify amyloid in suspected cases.
The size of neoplasms detected in each organ was estimated as visible foci (3–4 mm), small (5–7 mm), moderate (8–10 mm), or large (>10 mm). The fixed tissues were routinely processed, embedded in paraffin, and sectioned at 4 μm. Three sections, 100 μm apart, were taken from neoplastic tissues and stained with HE.
Immunophenotyping
Flow cytometry analysis and/or immunohistochemistry were performed when a hematopoietic neoplasm was suspected. Immunophenotypic analysis was performed using flow cytometry analysis on cell suspension from thymus, mesenteric lymph nodes, each enlarged lymph node, and bone marrow. In selected cases of hematopoietic neoplasms, the correct diagnosis required complementary immunohistochemistry.
The single-cell suspension for flow cytometry analysis was obtained by mechanical dissociation of lymphoid tissues in Hank’s balanced salts followed by passage through a fine-gauge needle and filtration of cells through a 40- to 50-μm nylon mesh. The single-cell suspension (1.0 × 106/1 ml) was analyzed by FACSCalibur (Becton-Dickinson, San Jose, CA). The specific 15 monoclonal antibodies (MAbs) for flow cytometry were labeled directly with either FITC (CD90.2/CD90.1, CD3∊, CD8, CD5, CD19, CD45-[B220], CD79b, GAM [goat anti–mouse immunoglobulin], IAd, IgM) or with PE (CD4, TCRβ, RAM KAPPA [rat anti–mouse κ], IgD). The monoclonal antibody CD3∊ was used to detect both surface (sCD3∊) and intracellular (cCD3∊) antigen. The fixation and permeabilization system (Leucoperm; Serotec Ltd, Oxford, UK) was used for analysis of the intracellular antigen. All antibodies were manufactured by BD Biosciences Worldwide Immunocytometry Systems Pharmingen (San Diego, CA). Working dilution of these antibodies in flow cytometric application was 1 μg to stain 1.0 × 106 cells.
Immunohistochemical staining was performed on paraffin sections using either Avidin-Biotin-Peroxidase Complex (DAKO Denmark A/S, Glostrup, Denmark) or MOM (Mouse on Mouse immunodetection kit; Vector Laboratories, Burlingame, CA). Both methods were used according to the manufacturers’ recommendations. The applied antigen retrieval unmasking method was microwave oven heating for 10 minutes in 0.01 M citrate buffers of pH 6. The specific antibodies for immunohistochemistry were biotin conjugated: anti-IgM, CD45R-(B220), CD5, CD3∊, TCRβ, Gr-1, anti–mouse Igκ light chain, anti–mouse Ig λ1 and λ2, or pure (anti-IgD). All antibodies were manufactured by BD Biosciences Worldwide Immunocytometry Systems Pharmingen. The antibodies were titrated, and the determined working dilution was 1:50 for all antibodies except for CD45R-(B220), which was used at a concentration of 1:100.
Classification of Lesions
Neoplasms were classified according to the recommendation of the Mouse Models of Human Cancer Consortium. Pulmonary lesions were classified based on the “Classification of Proliferative Pulmonary Lesions of Mouse: Recommendations of the Mouse Models of Human Cancers Consortium” (Boston, 2001). 52 The intestinal neoplasms were classified according to “Pathology of Mouse Models of Intestinal Cancer: Consensus Report and Recommendations.” 4 Classification of mammary gland neoplasms was based on “The Mammary Pathology of Genetically Engineered Mice: The Consensus Report and Recommendations From the Annapolis Meeting” (1999). 10 Lymphoid and nonlymphoid hematopoietic neoplasms were classified according to “Bethesda Proposals for Classification of Lymphoid Neoplasms in Mice” 49 and “Bethesda Proposals for Classification of Nonlymphoid Hematopoietic Neoplasms in Mice.” 38 Lymphoma subclassification was based on a combination of histologic features, cytomorphology, and flow cytometric and immunohistochemical immunophenotyping. 13,60 Neoplasms of the other organs were classified based on classification of rodent neoplasms. 45,69 Nonneoplastic lesions were diagnosed according to the standard criteria used in the description of lesions in mouse pathology and recommended by Frith and Ward. 23
Determination of Cause of Death and Contributed Cause of Death
Cause of death (COD) and contributed COD (CCOD) were determined according to recommendations for toxicological studies. 37,42
Statistical Analysis
Statistical analysis of the occurrence of neoplastic and nonneoplastic lesions was performed using the Kruskal-Wallis test at a level of significance (P = .05), with P < .01 highly significant. The Kruskal-Wallis test was used because the original data set consisted of nominal variables and ranked variables.
Results
The observed strains varied in the prevalence and types of neoplasms; these are presented in Tables 3 and 4. The ages of mice with or without neoplasms are shown in Table 2.
Prevalence of Neoplasms in the Examined Mice.
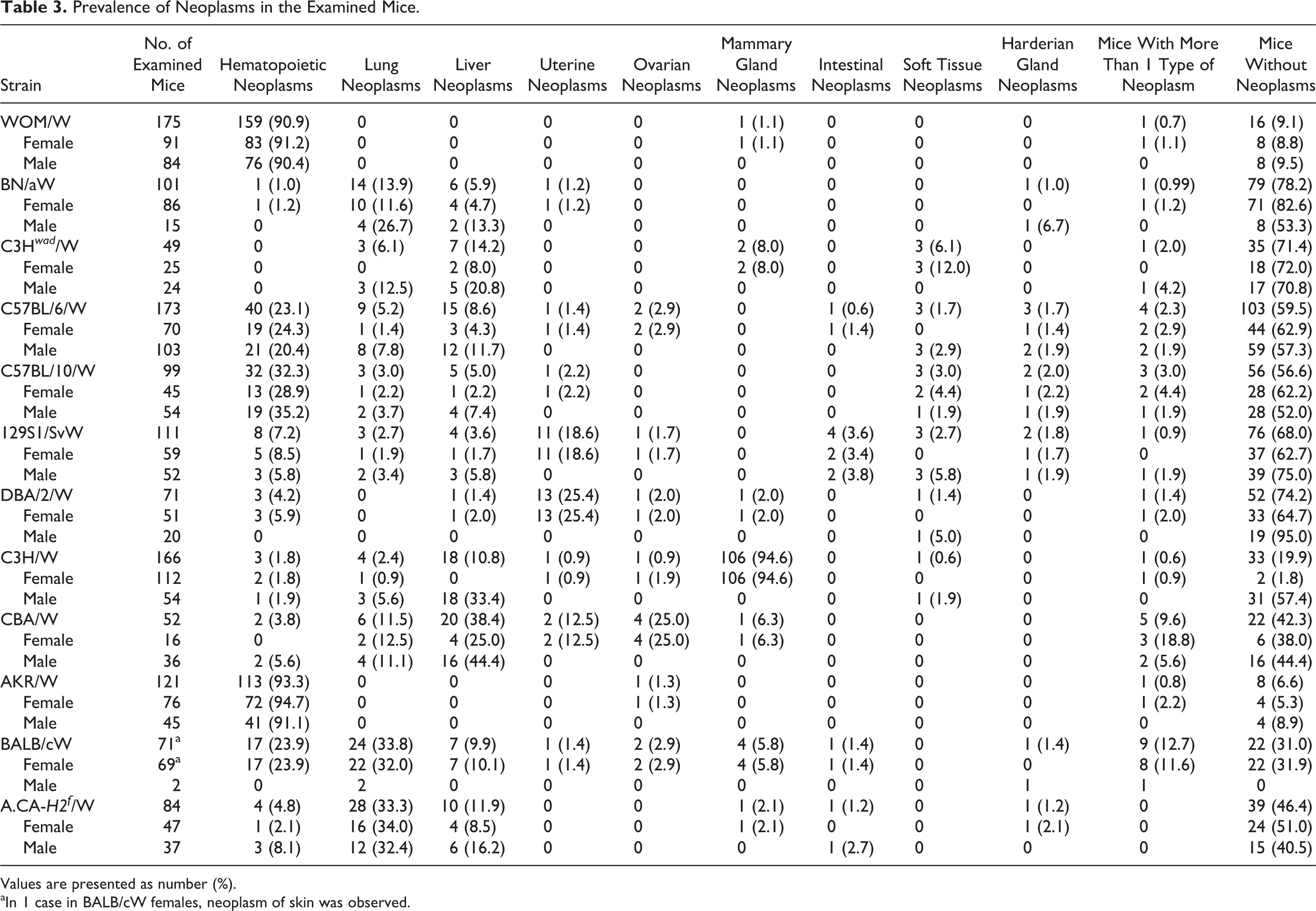
Values are presented as number (%).
aIn 1 case in BALB/cW females, neoplasm of skin was observed.
Prevalence of Types of Neoplasms in the Examined Mice (Numbers of Affected Animals).
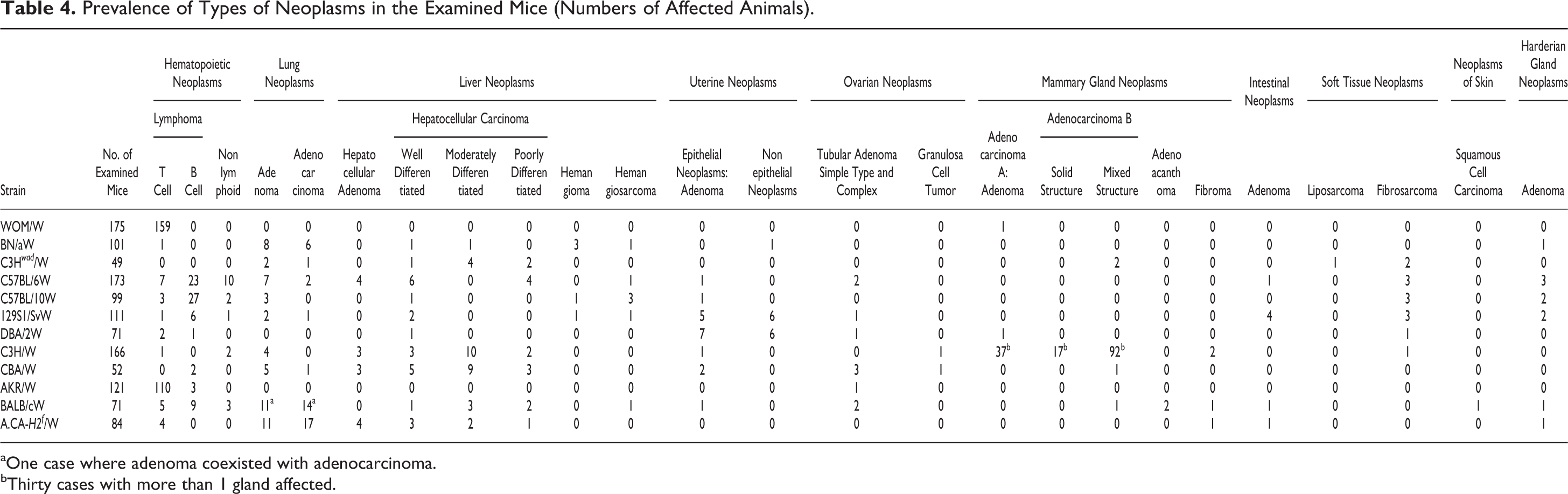
aOne case where adenoma coexisted with adenocarcinoma.
bThirty cases with more than 1 gland affected.
The most frequently observed nonneoplastic lesions are listed in Table 5. The most common nonneoplastic lesions in the examined strains were splenic congestion, reactive lymphoid hyperplasia in lymph nodes and spleen, ovarian cysts, lesions in the accessory male glands (ectasia and cystic degeneration of seminal vesicles, suppurative seminal vesiculitis, cysts and inflammation in preputial glands), otitis media, dental hypertrophy, and rectal prolapse. Nonneoplastic lesions in kidneys were observed mainly in BN/aW mice. Cardiomyopathy was found in C57BL/6W, C57BL/10W, and 129S1/SvW mice. The prevalence of histological types of neoplasms is shown in Table 4. Lymphomas were the most common neoplasia in the examined strains (364 cases/1273 mice, 28.6%). T-cell lymphomas were most commonly identified in AKR/W and WOM/W mice, and B-cell lymphoma was most common in C57BL/6W and C57BL/10W mice. Lymphoma was not observed in 49 C3H wad /W mice, and only 1 case was detected each in BN/aW and C3H/W mice. C3H/W mice had a high prevalence of neoplasms of the mammary gland.
Prevalence of Common Nonneoplastic Findings in the Examined Strains (Numbers of Affected Animals).
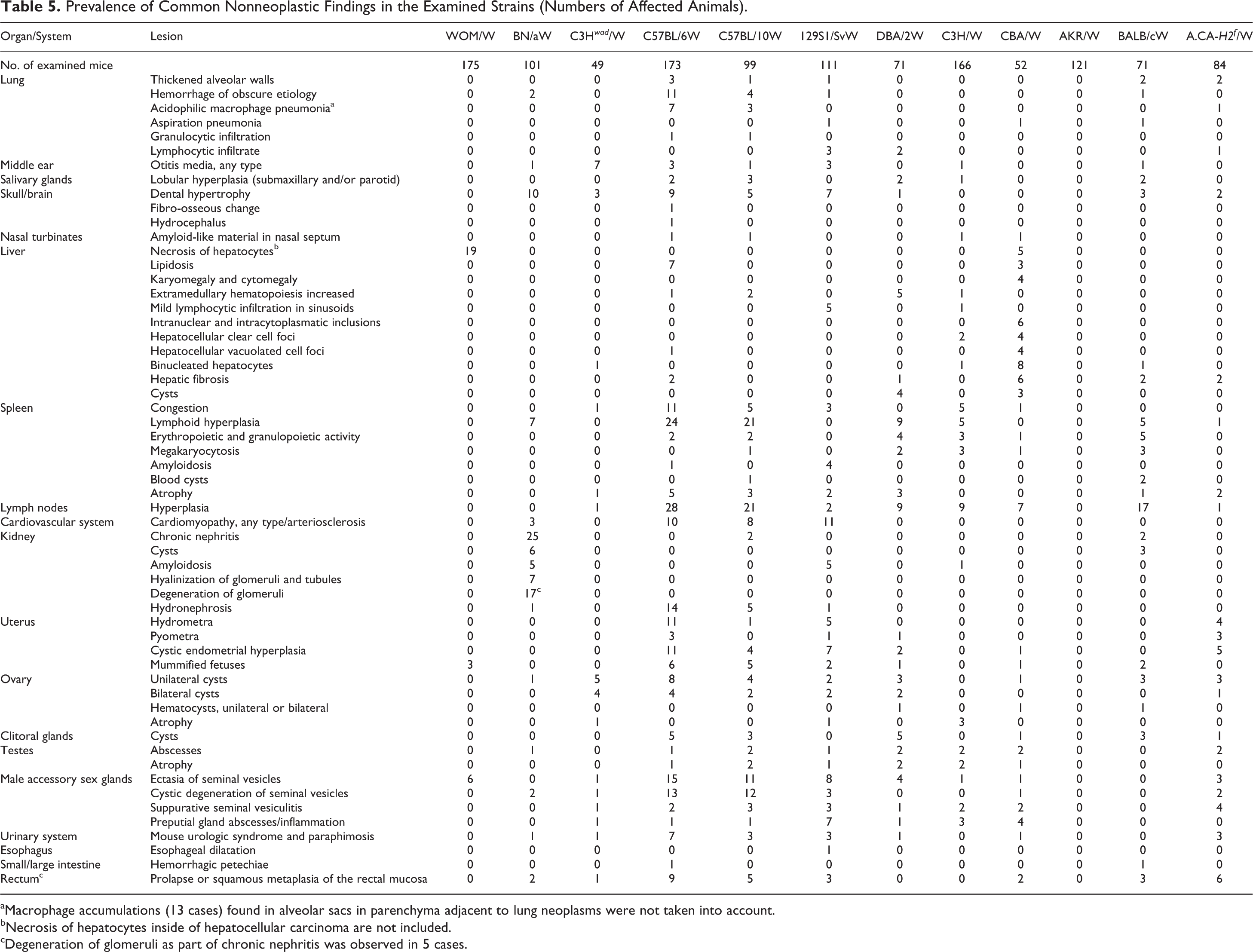
aMacrophage accumulations (13 cases) found in alveolar sacs in parenchyma adjacent to lung neoplasms were not taken into account.
bNecrosis of hepatocytes inside of hepatocellular carcinoma are not included.
cDegeneration of glomeruli as part of chronic nephritis was observed in 5 cases.
Neoplastic and Nonneoplastic Lesions in the 3 Unique Strains: WOM/W, BN/aW, and C3H wad /W
WOM/W mice commonly developed T-cell lymphoblastic lymphoma/leukemias (159/175, 90.9%). Necropsy typically identified an enlarged and nodular thymus with variable splenomegaly and lymphadenomegaly. Histopathologic findings included lymphoblastic infiltration into the liver, lung, and kidneys. In addition, gut-associated lymphoid tissue was involved in 13.8% of lymphoma cases, testes in 20.1%, and pericardium invasion in 11.3%. Neoplastic cells were found in the bone marrow in 137 of 159 cases (86.1%) and in the peripheral blood in 69 of 159 cases (43.4%).
T-cell lymphomas were positive for CD90.2 and cytoplasmic cCD3∊. They were poorly positive for CD5 and negative for CD45-(B220), CD79b, GAM, IAd, IgD, and IgM. Neoplastic T cells with coexpression of CD4 and CD8 were in the thymus and in peripheral tissues such as lymph nodes and bone marrow in 33 of 159 cases (20.8%). The population of T cells negative for CD4, CD8, and surface sCD3∊ (but cCD3∊+) was dominant in the thymus and in peripheral tissues in 78 of 159 cases (49.0%). Abnormal T cells with immunophenotypes CD4+CD8– or CD4–CD8+ in the thymus and peripheral tissues were noted in 11 (6.9%) and 8 (5.0%) cases, respectively. The immunophenotype of neoplastic T cells derived from the thymus and/or lymph nodes was consistent with the immunophenotype of T cells from the affected bone marrow in the same animals.
Other types of neoplasms were not observed, except for 1 case of mammary gland neoplasm (adenocarcinoma type A). That neoplasm was found in a female also diagnosed with T-cell lymphoma. Nonneoplastic lesions were rare in tissues other than the liver in WOM/W mice (see Table 5). Hepatocellular necrosis was interpreted to be the result of infiltrating neoplastic T cells in 19 cases. This strain also had 6 cases of seminal vesicular ectasia (common in aged male mice) without hyperplastic epithelium, and mummified fetuses were detected in 3 found-dead females.
BN/aW mice developed neoplasms in 22 of 101 cases (21.8%). Lung neoplasms were most common (14/101, 13.9%); they were more frequent in males (26.7%) than in females (11.6%). Lung neoplasms were solitary or multiple. The neoplasms were diagnosed as adenomas or adenocarcinomas. The size of adenomas was 3 to 4 mm; adenocarcinomas were greater than 4 mm with irregular nodular growth. Liver neoplasms, identified in 6 of 101 (5.9%), were observed more frequently in males (13.3%) than in females (4.7%). Hepatic vascular neoplasms (hemangiomas and hemangiosarcomas) were more common than hepatocellular carcinomas (see Table 4). Hematopoietic neoplasms, uterine neoplasms, and Harderian gland neoplasms occurred in single cases (see Tables 3, 4).
Nonneoplastic lesions in the kidney were common in BN/aW mice (see Table 5). Sex predilection was not evident (56/101, 55.4%; females 55.9%, males 53.3%). The lesions included chronic interstitial nephritis, glomerular amyloidosis, tubular ectasia, hyaline and proteinaceous casts in tubules, and cortical cysts (see Table 5 and Figs. 1–4).
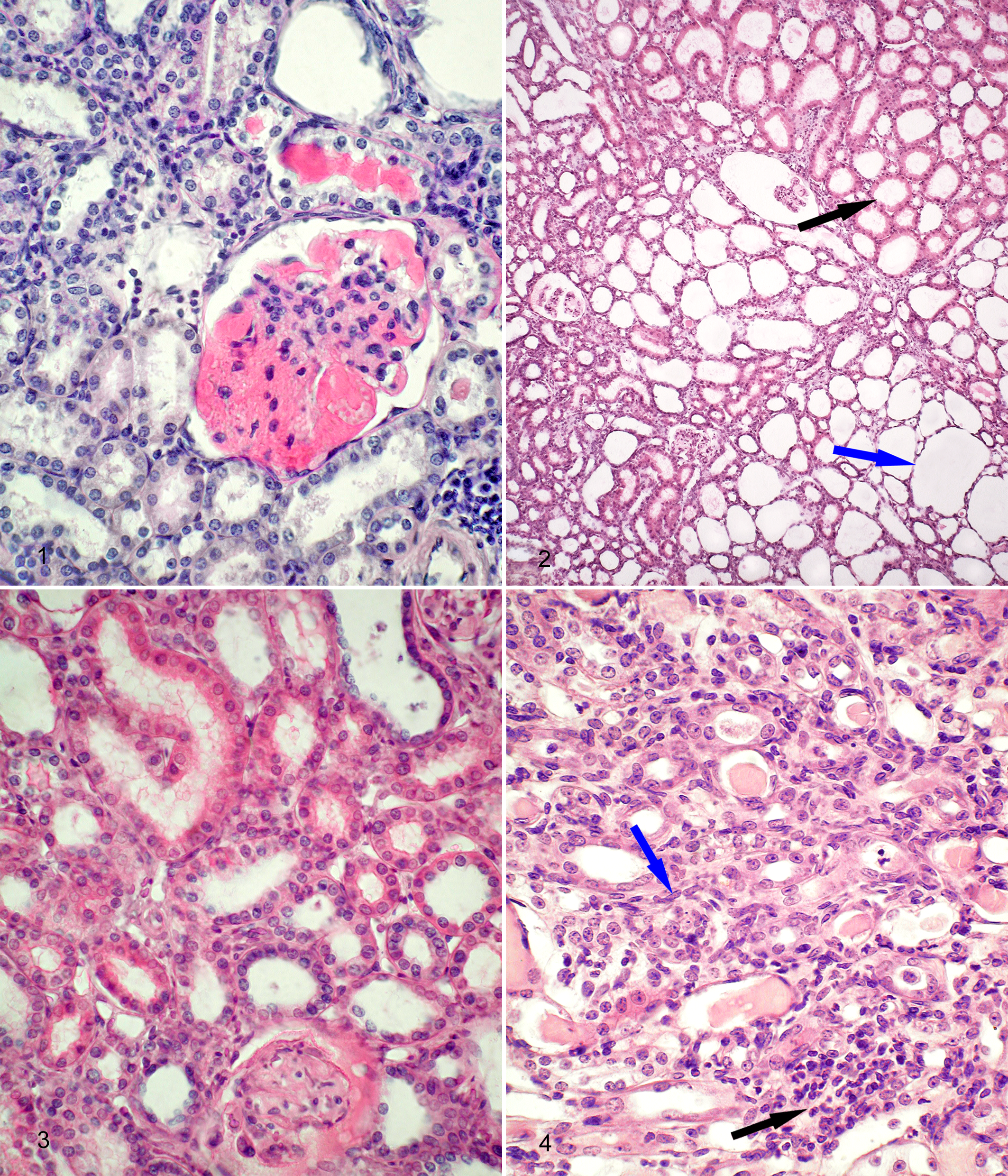
The lesions of interstitial renal disease were usually limited to the cortex and manifested as foci of mononuclear cells (predominantly lymphocytes) and local tubular dilation. Tubular dilation destroyed tubular architecture and in advanced stages resulted in formation of cortical cysts. Marked dilation of Bowman’s capsules was often observed. Glomeruli varied from normal to sclerotic/nonfunctional depending on the stage of interstitial nephritis. Renal cysts were typically bilateral, although 1 kidney was usually more affected. Nephropathy in BN/aW mice was described as mild or severe. Other nonneoplastic lesions included reactive lymphoid hyperplasia in the spleen and/or lymph nodes, dental hypertrophy, and rectal prolapse (see Table 5).
The C3H wad /W strain arose spontaneously from the C3H/W strain in the 1980s. These mice have an unmapped mutation on the distal chromosome 10 (see Table 1). 24 C3H wad /W mice developed hepatocellular carcinomas with a frequency of 14.2% (7/49). Lung neoplasms occurred in 3 of 49 mice (6.1%), neoplasms of the mammary gland in 2 of 25 females (8%), and soft tissue neoplasms in 3 of 49 mice (6.1%) (see Tables 3, 4). The soft tissue neoplasms consisted of 2 fibrosarcomas and 1 liposarcoma. The subcutaneous fibrosarcomas were medium-size neoplasms arising on the dorsum. The liposarcoma was a large (12 mm diameter) retroperitoneal mass. Common nonneoplastic lesions in C3H wad /W mice included ovarian cysts (9/25, 36%), otitis media in males (7/24, 29.1%), lesions in accessory male glands, and mouse urologic syndrome with paraphimosis (see Table 5).
Neoplastic and Nonneoplastic Lesions in Common Inbred Strains
C57BL/6W mice frequently developed hematopoietic neoplasms (40/173, 23.1%), which were typically follicular B-cell lymphomas (FBLs). These lymphomas arose in the spleen and/or mesenteric lymph nodes and infiltrated the liver; less than 10% also involved Peyer’s patches. Neither the bone marrow nor the blood had evidence of neoplastic cells. These FBLs were CD45R-(B220)+, CD19+, CD79b+, IAd+, GAM+, and Igκ light chain expression (RAM KAPPA+) by fluorescence-activated cell sorting. T-cell markers CD90.2–, CD4–, CD8–, sCD3∊–, cCD3∊–, and TCRβ– were interpreted as negative, although a small number of T cells were always detected in mesenteric lymph nodes. Immunohistochemistry showed expression of CD45R-(B220), IgM and/or IgD, and Igκ, while λ light chain expression was not noted.
Liver neoplasms (15/173, 8.6%), diagnosed as hepatocellular carcinomas or adenomas, were the second most common neoplasms in C57BL/6W mice; they were more common in males than in females (11.7% and 4.3%, respectively). Pulmonary neoplasms were also relatively common (9/173, 5.2%); most were bronchioloalveolar adenomas (7/9). A male predilection was evident (7.8% males, 1.4% females). Other sporadic neoplasms included soft tissue neoplasms (1.7%), Harderian gland neoplasms (1.7%), ovarian neoplasms (2.9%), uterine neoplasms (1.4%), and intestinal neoplasms (0.6%) (see Tables 3, 4).
The most common nonneoplastic lesion was lymphoid hyperplasia (13.9%) in the spleen and/or lymph nodes. The architecture of the spleen remained normal, but increased numbers of enlarged and coalescing lymphoid follicles were identified. In lymph nodes, follicular hyperplasia was restricted to the cortex; it did not extend into the capsule. Splenic and lymph nodal follicles were cytologically polymorphous. Because lymphoid hyperplasia and lymphoma can be part of a continuum, the clonality of lymphocyte population was assessed. Polyclonal lymphocyte populations were diagnosed as hyperplasia, and monoclonal populations were called lymphoma.
Other nonneoplastic changes included cystic endometrial hyperplasia, ovarian cysts, imperforate vaginas with associated hydrometra or pyometra, hydronephrosis, lesions in male accessory sex glands and cysts in clitoral glands in females, acidophilic macrophage pneumonia, cardiomyopathy, hepatic lipidosis (in males only), dental hypertrophy, and rectal prolapse (see Table 5).
The lesions observed in C57BL/10W mice were similar to lesions in C57BL/6W mice, although hydronephrosis and hydrometra occurred less frequently in C57BL/10W mice, while lipidosis in liver was not found in C57BL/10W mice (see Tables 3–5).
129S1/SvW mice developed uterine neoplasms most frequently (11/59 females, 18.6%). Five were adenomas, 5 were endometrial stromal sarcomas, and 1 was a leiomyosarcoma. Hematopoietic neoplasms (7.2%) were chiefly B-cell lymphomas. Intestinal neoplasms (3.6%) developed in the small intestine and were diagnosed as adenomas. Lung neoplasms occurred in 2.7% of examined mice. Liver neoplasms (3.6%) consisted of both hepatocellular carcinomas and vascular neoplasms. Soft tissue neoplasms developed in 2.7% of examined mice and neoplasms of Harderian glands in 1.8% (2/111) of mice (see Tables 3, 4). Male bias was observed in lung and liver neoplasms, and conversely, female bias was found in lymphoma occurrence. Nonneoplastic lesions that contributed to death included atriomegaly and dilation of the caudal vena cava and renal amyloidosis. Nonneoplastic lesions considered to be incidental included splenic amyloidosis, hydrometra, cystic endometrial hyperplasia, lymphocytic infiltrates within the liver, thyroid and periovarian fat, dental hypertrophy, rectal prolapse, and lesions in male accessory sex glands (see Table 5).
DBA/2W female mice developed uterine neoplasms in 13 of 51 cases (25.4%); 7 were diagnosed as adenomas, and 6 were endometrial stromal sarcomas. Lymphomas (4.2%), liver neoplasms (1.4%), ovarian neoplasms (2.0%), mammary gland neoplasms (2.0%), and neoplasms of soft tissue (1.4%) were diagnosed sporadically (see Tables 3, 4). The observed nonneoplastic changes included hepatic cysts, ovarian cysts, proliferative lesions of the hematopoietic system, clitoral gland cysts, and lesions in testes and male accessory sex glands (see Table 5).
C3H/W mice are carriers of the exogenous milk-transmitted mouse mammary tumor virus (MMTV). Neoplasms developed in mammary glands in 106 of 112 females (94.6%) at age ∼400 days (median, 395 days; min, 207 days; and max, 732 days). The occurrence of neoplasms in all mammary glands was similar.
Mammary gland neoplasms were classified as adenoma (adenocarcinoma A in the Dunn classification) or as adenocarcinoma (adenocarcinoma B in the Dunn classification) (see Table 4). Liver neoplasms (18/166, 10.8%) occurred only in males; 15 were hepatocellular carcinomas, and 3 were adenomas. Lung neoplasms (2.4%) and hematopoietic neoplasms (1.8%) developed rarely, and others were observed only sporadically (see Tables 3, 4). Nonneoplastic lesions were uncommon in C3H/W mice. The observed lesions included splenic congestion, reactive lymphoid hyperplasia, ovarian atrophy, and lesions in male accessory sex glands (see Table 5).
CBA/W mice often developed liver neoplasms (20/52, 38.4%), and hepatocellular carcinomas were more common (17 cases) than hepatocellular adenomas (3 cases). Males were more susceptible than females (44.4% and 25.0%, respectively). Lung neoplasms (6/52, 11.5%) were primarily adenomas; no sex predisposition was observed. Ovarian neoplasms (tubular adenomas) were identified in 4 of 16 females (25%). Hematopoietic, uterine, and mammary gland neoplasms were uncommon (see Tables 3, 4).
Nonneoplastic changes in the liver coexisted with neoplasms or were found separately. They consisted of hepatocyte degeneration, intranuclear and cytoplasmic inclusions, karyomegaly and cytomegaly, the presence of binucleated hepatocytes (more than 20% of hepatocytes), hepatocellular foci of cellular alterations, lipidosis, and hepatic cysts. Several other common nonneoplastic lesions were observed, including cystic endometrial hyperplasia, ovarian cysts, reactive hyperplasia in lymph nodes, and lesions in testes and male accessory sex glands (see Table 5).
AKR/W mice developed hematopoietic neoplasms (113/121, 93.3%), which were typically T-cell lymphoblastic lymphomas (110 cases) and rarely follicular B-cell lymphomas (3 cases). Sex predilection was not observed. T-cell lymphoblastic lymphomas developed in thymus and neoplastic cells infiltrated into the spleen, lymph nodes, liver, and kidneys. Bone marrow was involved in 93 of 110 cases (84.5%) and peripheral blood in 60 of 110 cases (54.5%). Nonneoplastic changes were not identified in this strain.
BALB/cW female mice developed a wide spectrum of neoplasms. Lung neoplasms predominated (24/71, 33.8%) and were diagnosed as adenoma (11) and adenocarcinoma (14); in 1 case, adenoma coexisted with adenocarcinoma. Hematopoietic neoplasms (17/71, 23.9%) were identified as B-cell lymphomas (12.7%), T-cell lymphomas (7.0%), and nonlymphoid hematopoietic neoplasms (4.2%). Liver neoplasms occurred in 9.9% (7/71) of mice and were diagnosed as hepatocellular carcinoma (6) and hemangiosarcoma (1). Mammary gland neoplasms (5.8%) were diagnosed as adenoacanthoma or adenocarcinoma (see Tables 3, 4). The other neoplasms (uterine, ovarian, intestinal, Harderian gland, and skin) occurred sporadically (see Tables 3, 4). The most common nonneoplastic lesions were reactive lymphoid hyperplasia in lymph nodes and spleen, ovarian cysts, renal cysts, dental hypertrophy, and rectal prolapse (see Table 5).
Data from BALB/cW males are scant, since BALB/cW males are highly aggressive and difficult to maintain for a long period during the observation. The data obtained from the observation of BALB/cW males cannot be interpreted. The previous observation (1980–1985) of BALB/cW males showed a prevalence of lung neoplasms (15/140, 36.4%), liver neoplasms (3/140, 2.1%), and hematopoietic neoplasms (2/140, 1.4%).
A.CA-H2f /W mice frequently developed lung neoplasms (28/84, 33.3%); large adenocarcinomas predominated (17/28) (see Table 4). Liver neoplasms (10/84, 11.9%) developed more frequently in males than in females (16.2% and 8.5%, respectively). Hematopoietic neoplasms (4.8%) were diagnosed as T-cell lymphoblastic lymphomas. Other neoplasms were uncommon (see Tables 3, 4).
Common nonneoplastic lesions included hydrometra, pyometra without a determined cause, cystic endometrial hyperplasia, ovarian cysts, and rectal prolapse. In males, suppurative seminal vesiculitis and mouse urologic syndrome with paraphimosis were observed (see Table 5).
Statistical Results
The prevalence of lung neoplasms was significantly higher in BALB/cW (33.8%) and A.CA -H2 f /W (33.3%) mice than in other strains (P < .05). There was no statistical difference between these 2 strains. The prevalence of liver neoplasms was the greatest in CBA/W mice (38.4%, P < .01). C3H/W mice, irrespective of sex, have a lower prevalence of liver neoplasms (10.8%) than CBA/W mice. However, if only males are studied, the prevalence of liver neoplasms (33.3%) is similar to CBA/W mice. The examined strains did not significantly differ in the prevalence of uterine neoplasms, although the highest occurrence was observed in DBA/2W and 129S1/SvW mice. The prevalence of neoplasms of the mammary glands was significantly higher in C3H/W mice (P < .01), which carry exogenous MMTV.
The frequency of T-cell lymphomas was significantly higher in WOM/W (90.9%) and AKR/W mice (90.9%) than in other strains (P < .01). No significant differences were found between these 2 strains. The occurrence of B-cell lymphomas in C57BL/10W mice (27.3%) was significantly higher than in other strains (P < .05). The occurrence of B-cell lymphomas in C57BL/6W mice (13.3%) was higher than in the other strains, but the difference was not statistically significant. No statistically significant differences among strains were found in the occurrence of nonlymphoid hematopoietic neoplasms. There was no significant difference in the frequency of other types of neoplasms between strains. The prevalence of nonneoplastic lesions in kidneys of BN/aW mice (55.4%) was significantly higher than in the other strains (P < .01).
Discussion
The gross and histological evaluation of aged mice provides important information about strain-specific disease predispositions. 56,61
In this article, we have shared pathological data for aged mice of common and new strains to supplement knowledge about strain-specific disease predispositions.
First, we have described neoplastic and nonneoplastic lesions in aged mice from 12 inbred strains. These data are important for differentiating strain-specific lesions in animal models from those due to gene modification. We identified the COD for 1236 mice; in 37 mice, the cause of death was not unequivocally identified. The cause of death was generally attributed to a single disease in the case of neoplasms. Chronic degenerative nonneoplastic lesions were considered the COD if a single disease was observed and the impact of that lesion was evident. Aging mice usually showed a complex set of lesions that contributed to death. The causes of nonneoplastic lesions remain unknown in most cases. However, Helicobacter infection should be taken into consideration in cases of rectal prolapses and lymphocytic infiltration in liver sinusoids, necrosis of hepatocytes, fibrosis, and cytomegaly in liver. 2,76
Second, we have described new strains (WOM/W, BN/aW), which can be considered new mouse models of T-cell lymphoma and age-related nephropathy, respectively. Moreover, we described the type and diversity of mouse diseases that these strains develop as they age.
The first novel strain (WOM/W) is unique in its consistent development of T-cell lymphomas (90.9%) at the young age of ∼230 days (median, 225 days; min, 91 days; max, 570 days). Importantly, the genetic background of WOM/W mice is completely different from that of AKR/W mice (M. Gajewska, unpublished data), a classic strain prone to develop T-cell lymphoblastic lymphomas (60%–90%) at less than 1 year of age. 20 AKR mice contain endogenous ecotropic murine leukemia virus (Akv) that is involved in causing a high prevalence of lethal T-cell lymphoblastic lymphomas, primarily developing in the thymus. 28,40 WOM/W mice may be considered a novel strain with a high prevalence of T-cell lymphoblastic lymphomas similar to the AKR, C58, NOD-SCID/SCID, SCID, and HRS/J strains. 8,72
Animal models of lymphoma/leukemia are widely used to study the mechanisms of cancer development and progression as well as to assess the efficacy of new therapies. The Hematopathology Subcommittee of Mouse Models of Human Cancer Consortium (MMHCC) developed criteria to classify mouse lymphoid neoplasms and indicated their relation to diseases in humans. 6,49,62 T-cell lymphoblastic lymphoma/leukemia is recognized as a mouse counterpart of that disease in humans. 41 Moreover, published data on lymphoma in mice indicate that the specific background strain greatly influences the prevalence and type of lymphomas. 6
Our second new strain (BN/aW) has a high prevalence of renal disease and may be a valuable alternative model to investigate chronic renal disease. Nephropathy has previously been described in C57BL/6, 129, and B6C3F1 hybrid mice. 9,75,78 Spontaneous interstitial nephritis has been reported in CBA/CaH-kdkd mice, which are homozygous at kd on chromosome 10. 50,59 These mice are very susceptible to spontaneous interstitial nephritis and die between 5 and 7 months with end-stage renal failure. However, CBA/CaH-kdkd mice are poor breeders and produce only 1 or 2 litters. The prevalence of nephropathy in C57BL/6 varies by report, from rather low (6%–15%) 3,8,9 to 100% with mild to moderate glomerulonephritis. 79 Chronic nephropathy has been noted in the 129S4/SvJae strain, but it was not considered the COD. 8 B6C3F1 hybrid mice are preferred for toxicology studies, but they have a high prevalence of nephropathy. In B6C3F1 mice, nonneoplastic renal lesions include mononuclear cell infiltrates (29.8%), tubular mineralization (11.3%), interstitial nephritis (6.8%), tubular epithelial vacuolization (4.5%), tubular epithelial degeneration/regeneration (2.5%), and cortical cysts (1.3%). 12 A GEM with chronic interstitial nephritis is the [Tg(Cga-LHB/CGB)94Jhn/J] mouse, which expresses a human luteinizing hormone chimera targeted to mouse pituitary gonadotropes. In addition to the expected pathologic ovarian changes, these mice develop chronic interstitial nephritis. 57 In total, 55.4% of BN/aW mice died after 30 months as a result of nephropathy, which included chronic interstitial nephritis (24.8%), cortical cysts (5.9%), hyalinization of tubules (6.9%), degeneration of glomeruli (16.8%), amyloidosis (5%), and hydronephrosis (1%) (see Table 5). Moreover, BN/aW mice are long lived (median >850 days), develop few neoplasms, and are relatively good breeders (average, 4 litters), increasing their utility as a model of human disease.
Third, we have compared the lesions found in mice from our strains with data obtained from a literature survey. We tabulated the most prominent similarities and differences in morbidity of mice from our substrains and compared them with previously published reports (see Table 6). The traditional inbred strains maintained in our animal facility have been genetically isolated from their parental colonies for more than 20 generations and thus are defined as substrains, although their genetic differences from parental strains have not been evaluated.
Prevalence of Lesions Found in the Observed Strains in Comparison With Data From Literature.
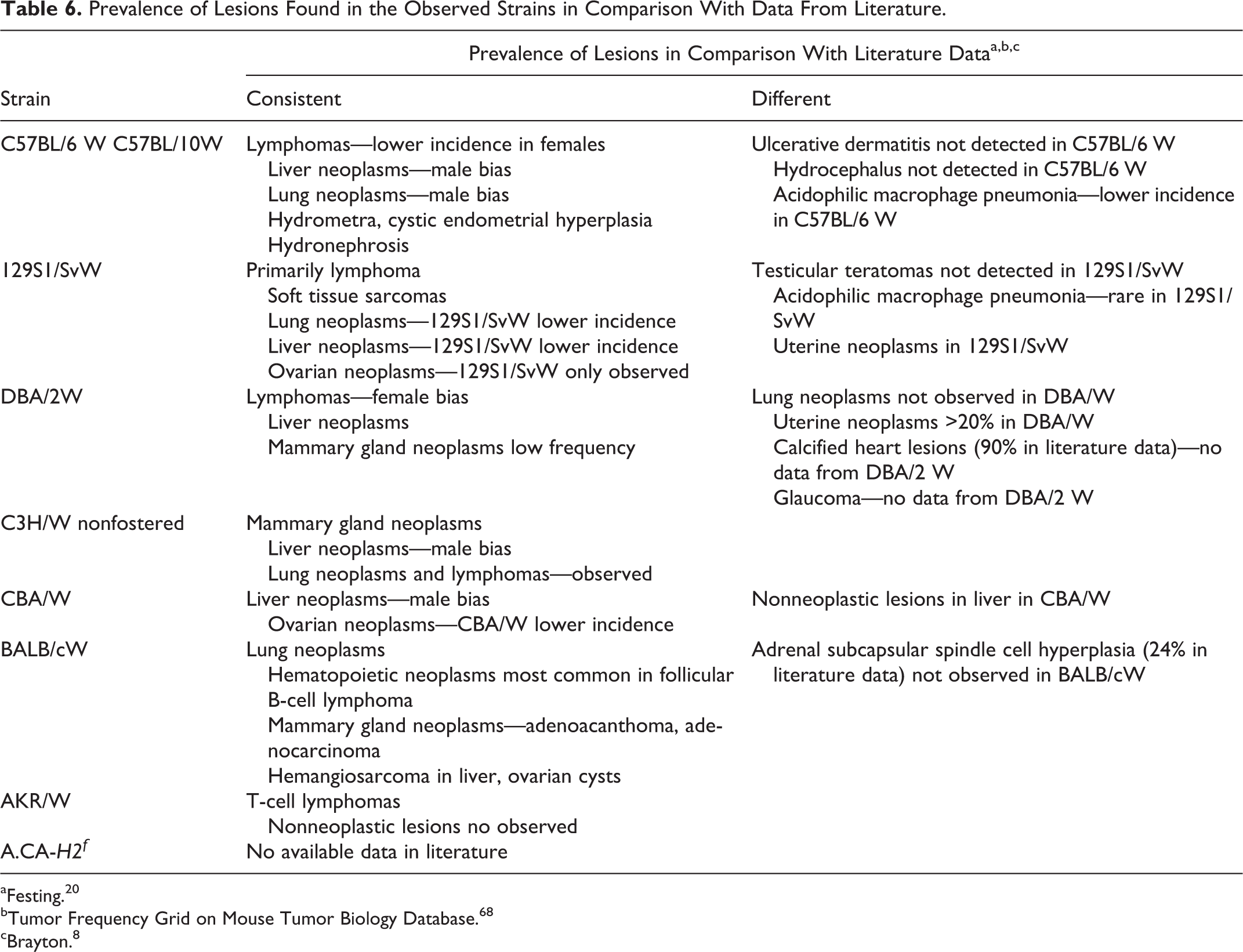
aFesting. 20
bTumor Frequency Grid on Mouse Tumor Biology Database. 68
cBrayton. 8
Our substrains differ from parental strains in their susceptibilities to neoplasms but generally do not differ with regard to the type of neoplasms they develop. Two strains, DBA/2W and 129S1/SvW, differ most notably from their parental strains in the prevalence of uterine neoplams (increased occurrence in our colony). Another difference is noted in male 129S1/SvW mice, which had no evidence of testicular neoplasms, in contrast to what is seen in the parental strain.
The 129 mice are known for their very high prevalence of testicular teratomas. 1 However, at least 16 recognized substrains of 129 exist. 53 According to the revision of 129 strain nomenclature presented by Festing et al, 21 129 is followed by letters P, S, T, and X. P stands for derivation from parental stock, S indicates the origin from the line with the Steel allele on the Kitl gene, T means that a line carries or used to carry the Ter (teratoma) gene, and X indicates lines genetically contaminated at The Jackson Laboratory around 1978 by an unknown strain. These lines differ from other 129 substrains at around 25% of single sequence length polymorphic markers and are designated 129cX/Sv. 65 The prevalence of testicular teratomas is high in the 129/terSv substrain (30%) and low (1%–3%) in 129P parental and many other substrains. 33 More than 15 genes, including Ter (having the strongest effect), Kitl(Sl), Ay, and Trp53, are involved in susceptibility to testicular teratomas. 34,39
The data obtained from this study provide an overview of the types and variety of lesions observed in aging mice of multiple strains. As expected, the examined strains differ in susceptibility to neoplasia as well as prevalence and severity of nonneoplastic lesions. The data presented here add to the growing literature on age- and strain-related lesions in mouse strains commonly used in research and the generation of GEM. This report also shows that substrains are often unique from parental strains and that no assumptions should be made when developing animal models of disease.
Footnotes
Acknowledgements
We thank the medical pathologist Elwira Bakula-Zalewska from the Department of Pathology of the Maria Sklodowska-Curie Memorial Cancer Center and Institute of Oncology in Warsaw for revision of several diagnoses.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
